Notes
Article history
The research reported in this issue of the journal was funded by the HTA programme as project number 13/103/02. The contractual start date was in April 2014. The draft report began editorial review in November 2019 and was accepted for publication in August 2021. The authors have been wholly responsible for all data collection, analysis and interpretation, and for writing up their work. The HTA editors and publisher have tried to ensure the accuracy of the authors’ report and would like to thank the reviewers for their constructive comments on the draft document. However, they do not accept liability for damages or losses arising from material published in this report.
Permissions
Copyright statement
Copyright © 2022 Griffin et al. This work was produced by Griffin et al. under the terms of a commissioning contract issued by the Secretary of State for Health and Social Care. This is an Open Access publication distributed under the terms of the Creative Commons Attribution CC BY 4.0 licence, which permits unrestricted use, distribution, reproduction and adaption in any medium and for any purpose provided that it is properly attributed. See: https://creativecommons.org/licenses/by/4.0/. For attribution the title, original author(s), the publication source – NIHR Journals Library, and the DOI of the publication must be cited.
2022 Griffin et al.
Chapter 1 Introduction
Further reading on this trial is available in the trial protocol by Griffin et al. ,1 trial feasibility reports by Griffin et al. 2,3 and Wall,4 trial non-operative intervention report by Wall et al. 5 and trial results article by Griffin et al. 6
Background
Until recently, there was little understanding of the causes of hip pain in young adults. A proportion of young adults with hip pain have established osteoarthritis, inflammatory arthritis, avascular necrosis, fractures or childhood hip disease, but the majority have no specific diagnosis. Over the last decade, there has been increasing recognition of femoroacetabular impingement (FAI) syndrome, which seems to account for a large proportion of the previously undiagnosed cases of hip pain in young adults. 7,8 Subtle deformities in the shape of the hip (ball and socket joint) combine to cause impingement between the femoral head (ball) or neck and the anterior rim of the acetabulum (socket), most often in flexion and internal rotation. 7,9 Excess contact force leads to damage to the acetabular labrum (fibrocartilage rim of the socket) and the adjacent acetabular cartilage surface. 7 FAI seems to be associated with progressive articular degeneration of the acetabulum and may account for a significant proportion of so-called idiopathic osteoarthritis, although this remains unproven. 9 The shape abnormalities of the hip joint are typically divided into the following three categories:9
-
cam-type impingement (in which the femoral head is oval rather than round, or there is prominent bone on the femoral neck)
-
pincer-type impingement (in which the rim of the acetabulum is too prominent in one or more areas of its circumference)
-
mixed-type hip impingement (which is a combination of cam and pincer types).
Surgery can be performed to improve bone shapes to prevent impingement between the femoral neck and rim of the acetabulum. In the case of cam-type FAI syndrome, this usually involves the removal of bone at the femoral head–neck junction. In the case of pincer-type FAI syndrome, it may involve the removal of bone at the rim of the acetabulum. At the same time as bony shape improvement, any soft tissue damage to the cartilage or labrum as a result of the FAI syndrome is debrided, repaired or reconstructed. Surgery can be undertaken using either keyhole (arthroscopic) surgery or more traditional open surgery to access the hip joint and correct the hip shape abnormalities associated with FAI syndrome.
Surgery for FAI syndrome has evolved more quickly than our understanding of the epidemiology or natural history of the condition, and is becoming an established treatment. 10–12 The risks of complications from open surgery are greater than those for arthroscopic surgery, and current evidence suggests that the outcomes of arthroscopic treatment for the symptoms of FAI syndrome are comparable to open surgery. 13,14 Consequently, hip arthroscopy for FAI syndrome is a rapidly growing new cost pressure for health providers. 15 However, a Cochrane review highlighted the absence of randomised controlled trials (RCTs) comparing FAI surgery with conservative care, such as physiotherapist-led exercise. 16
Multicentre RCTs are acknowledged to be the best design for evaluating the effectiveness of health-care interventions, as they provide robust evidence. 17,18 However, there are often major challenges in performing RCTs of surgical technologies19 and there were concerns that a RCT of hip arthroscopy in FAI syndrome might not be feasible.
Summary of a feasibility and pilot trial
A feasibility and pilot study commissioned by the Health Technology Assessment programme (reference 10/41/02) was completed. 2,3 It comprised (1) a pre-pilot phase, including patient and clinician surveys and interviews, and a systematic review of non-operative care; (2) a workload survey of hip arthroscopy for FAI; (3) development of best conventional care and arthroscopic surgery protocols; (4) a pilot RCT to measure recruitment rate; and (5) an integrated programme of qualitative research to understand and optimise recruitment. 2,3
The feasibility study followed the commissioning brief and specifically addressed the following parameters to inform the design of the proposed full-scale RCT.
Define eligibility criteria
The eligibility criteria were initially designed in collaboration with the Multicenter Arthroscopy of the Hip Outcomes Research Network (MAHORN), which is an academic group of highly experienced hip arthroscopists within the International Society for Hip Arthroscopy (Moffat, UK) (URL: www.isha.net). These criteria were then discussed with a further sample of 14 UK specialist hip surgeons with experience of treating patients with FAI syndrome. In individual interviews, a variety of clinical scenarios were presented and the surgeons were asked to describe decision-making for treatment. Minor modifications were made to the eligibility criteria. These criteria were then tested during the recruitment of real patients during the pilot RCT and were found to be easy to apply, with little disagreement among clinicians.
Define a protocol for hip arthroscopy for FAI syndrome
A draft protocol for arthroscopic treatment of FAI was developed in a consensus conference with MAHORN members. This draft was circulated among the sample of 14 UK hip surgeons for feedback. After editing, it was recirculated and approved by all. The protocol was then tested in 21 participants randomised to surgery in the pilot trial. We also developed a method to measure fidelity by intraoperative photographs and postoperative magnetic resonance imaging (MRI), which was assessed by a panel of independent international experts. We showed that this approach was acceptable to surgeons and demonstrated complete adherence to protocol in six of seven operations at the first panel conference.
Define a protocol for best conservative care (comparator)
We performed a systematic review of non-operative care for FAI. 20 This revealed little evidence of a standard for best conservative care, even though many NHS commissioners describe the failure of conservative care as a prerequisite for surgery. 21 There was some evidence that physiotherapy-led non-operative care is most frequently used. 20 This is complemented by established theory and evidence supporting treatment effects for physiotherapy in other painful musculoskeletal conditions, including osteoarthritis and back pain. 22,23
We used a combination of consensus methods (e.g. Delphi and nominal group techniques) among physiotherapists to agree a protocol for ‘best conservative care’. 5 We advertised to relevant networks of the Chartered Society of Physiotherapy (CSP) (London, UK) through their interactive communication system (interactiveCSP) and in the Frontline magazine24 (a twice-monthly magazine posted to 52,000 CSP members in the UK). These advertisements invited physiotherapists to help develop a consensus for a best conservative care treatment protocol for FAI syndrome. Electronic invitations were also sent to physiotherapists in the USA and Australia who were known to us through previous collaborative work on FAI syndrome. To encourage a process of ‘snowball sampling’ within the international community, these therapists were encouraged to invite colleagues with experience and interest in managing FAI syndrome to join in the consensus process.
We developed a physiotherapy-led four-component protocol to be delivered over 12 weeks, with a minimum of six one-to-one treatment sessions. 5 The protocol included (1) a detailed patient assessment; (2) education and advice about FAI syndrome; (3) help with pain relief, including hip joint steroid injections if required; and (4) an exercise programme that has the key features of individualisation, supervision and progression. We used a patient focus group to choose the most acceptable name for this protocol of best conservative care. The group made it clear that we should express that this was a coherent and valid alternative to surgery and different from the physiotherapy likely to have been received already, and recommended the name personalised hip therapy (PHT).
In the development of PHT, we struck a balance between the need for a meaningful comparator for hip arthroscopy, the need to ensure that PHT is different from previous physiotherapy that FAI syndrome patients’ may have experienced and the need for PHT to be deliverable in the NHS outside a trial. UK physiotherapists and patients felt that PHT was ‘best’ in that not all patients currently receive such a comprehensive package, but ‘conventional’ in that all its elements are widely used and the package is deliverable within usual constraints in the NHS. We tested the protocol and a logbook approach to assessing fidelity in 21 participants randomised to PHT in the pilot trial. The protocol was acceptable to patients and physiotherapists, and we demonstrated complete adherence in seven of the first eight participants.
Define willingness of centres and patients to be recruited to a randomised controlled trial
We performed a survey of all orthopaedic surgery departments in NHS hospital trusts in the UK. Clinical directors of those departments reported that 120 consultant surgeons were treating FAI syndrome. We contacted all 120 surgeons who reported having performed 2399 operations for FAI in 2011/12. A total of 1908 operations were performed by hip arthroscopy and 491 operations were open surgery. 3 Thirty-four hospital trusts had a workload of 20 or more hip arthroscopies for FAI syndrome in 1 year. We interviewed 18 of the highest-volume surgeons to explore their views about a trial comparing hip arthroscopy with best conservative care in patients with FAI syndrome. One surgeon felt that he could not participate in a trial because he was certain that surgery worked, five surgeons had a bias towards surgery but recognised the need for a trial and were prepared to randomise patients, and 12 surgeons expressed equipoise and were keen to take part in a trial.
We purposively sampled 18 patients who had been treated for FAI syndrome. Fourteen of these patients had received arthroscopic surgery and five had received physical therapy and steroid injections (one patient had both). These patients had a semistructured interview with a qualitative researcher who had not been involved in their care to explore their experiences of diagnosis and treatment, and their views on the proposed trial. The majority of the patients were young and physically active. Symptoms of FAI syndrome had affected their work, recreation and day-to-day activities, and many reported a great sense of relief when a diagnosis was made. Patients said that both surgical and conservative care would be acceptable. The majority of patients saw surgery as the solution for a condition that they perceived as mainly caused by abnormal bone shapes. On the other hand, non-operative care was perceived as attractive if it might be successful and could avoid the risks of surgery. Some patients commented that they had not been offered a non-operative option and saw this as a positive addition to available treatments. Patients were enthusiastic about research in this field, and about being involved, but had reservations about some of the language involved, for instance ‘trial’, ‘random’ and ‘50 : 50 chance’ implied a lack of personalised care. All of these patients said that they would have been prepared to take part in a RCT as long as the treatment options and uncertainty around them had been fully explained, the treatment they received had been personalised for them and they were assured that their care would be continued whatever happened in the research.
Our findings in these in-depth interviews were broadly consistent with Palmer et al. ’s25 questionnaire survey of 30 surgeons who performed FAI surgery and 31 patients with a diagnosis of FAI syndrome. In Palmer et al. ’s25 study, 71% of surgeons and 90% of patients felt that a trial of this question was appropriate.
We concluded that surgeons in most centres in the UK that perform hip arthroscopy for FAI syndrome, and their patients, would be willing to be included in a RCT.
Understand and optimise recruitment
An important objective of the pilot trial was to explore likely issues in recruitment and to develop optimum procedures for the full trial.
We interviewed all principal investigators (PIs) and research associates (RAs) during the pilot trial to ensure that the study was being described and recruitment procedures were being followed in accordance with the study protocol, and to identify when they were not. We developed training packages to correct common problems. We identified structural features associated with successful recruitment, such as running targeted clinics, having a dedicated RA in attendance and ensuring that referred patients arrived with expectations of receiving treatment for FAI syndrome, rather than being told they had been referred for surgery. This learning was shared across all sites.
We recorded and analysed 87 diagnostic and recruitment consultations with 60 new patients during the pilot trial. We identified where improvements could be made in presenting trial information and in engaging patients to consider participation, guided by our previous work. 26,27 The analysis was targeted at the recruitment levels at specific sites, with individual confidential feedback for recruiters on good practice and areas for improvement, and with anonymised findings being fed back to all sites.
Common difficulties with recruitment that were identified included poorly balanced presentations of treatment options (where surgery was presented at greater length and more favourably than PHT), graphic descriptions of surgery that may have put patients off that option or discouraged participation, presenting trial information in an order that was confusing for patients and surgeons going beyond their protocol brief to explain the trial, rather than referring patients to the trial recruiter for this information. Analysis of the consultations led to the development of a six-step model for the presentation of trial information to optimise recruitment. 3,28
Estimate recruitment rate
Ten clinical centres participated in the pilot trial and nine opened to recruitment within 6 months. At one site, local research and development (R&D) approval was delayed until just before the end of the pilot and so no one was recruited.
Of the 144 potentially eligible patients with hip problems identified at the pre-clinic screening of referral letters, 60 met the inclusion criteria after assessment and were approached for randomisation. The most frequent reasons for exclusion were a diagnosis other than FAI (53/84) and a judgement that the patient would not benefit from arthroscopic surgery (21/84). Forty-two patients (70% of those eligible) consented to take part in the pilot RCT. Among those who declined (n = 18), the most common reasons were a preference for surgery (n = 11) and a preference not to have surgery (n = 3). The mean duration and recruitment rate across all sites was 4.5 months and one patient per centre per month, respectively. The lead site recruited for the longest period (9.3 months) and recruited the largest number of patients (2.1 patients per month).
Selection of appropriate outcome measures
A variety of outcome measures have been used to study patients with FAI syndrome. Some, such as the Western Ontario and McMaster Universities Arthritis Index (WOMAC®) and the Harris Hip Score, were intended for older patients with symptoms of severe arthritis and are most suitable to measure the effect of hip replacement surgery. 29,30 These measures tend to exhibit ceiling effects and are not sensitive to change after treatment in patients with FAI syndrome. 30,31
The Non-Arthritic Hip Score is a self-administered instrument to measure hip-related pain and function in younger patients without arthritis. The score is valid compared with other measures of hip performance, is internally consistent and is reproducible. 32 However, it is not patient derived, raising concern that it may not measure what is most important to patients.
The International Hip Outcome Tool-33 (iHOT-33) is a patient-derived hip-specific patient-reported instrument that measures health-related quality of life in young, active patients with hip disorders. It was developed by a large international collaboration of patients and clinicians led by MAHORN over 5 years. It comprises 33 items, each measured on a visual analogue scale (VAS), to assess functional limitations, sports activities, and job-related and emotional concerns. Importantly, these items were generated and refined by patients, reflecting their most important concerns. The instrument generates a single score in the range of 0 to 100. People with no hip complaints usually score ≥ 95. A diverse international population of younger adults with a variety of hip pathologies had a mean score of 66, with a standard deviation (SD) of 19.3 (Damian R Griffin, University of Warwick, 2021, personal communication).
The iHOT-33 has been validated for use in patients with FAI syndrome and is sensitive to change after treatment. The minimum clinically important difference (MCID) has been determined using an anchor and distribution-based approach in a group of 27 young active patients who were independent of the development population. Clinical change was determined using a global rating scale that asked patients whether their hip condition had improved, had deteriorated or had not changed since the previous assessment, using a single VAS. The MCID was 6.1 points. 33,34
The iHOT-33 and EQ-5D-5L have been adopted as the principal outcome measures by the UK Non-Arthroplasty Hip Registry. This registry is led by the British Hip Society (London, UK) and its use in all patients having arthroscopic FAI surgery is required by the National Institute for Health and Care Excellence (NICE). 15
In our pilot study, we tested the Non-Arthritic Hip Score and iHOT-33 as potential primary outcome measures, and found both to be easy to use and acceptable to patients. The extensive patient involvement in item generation, the availability of an independently determined MCID and the use of iHOT-33 as the principal outcome measure for the UK Non-Arthroplasty Hip Registry led us to choose iHOT-33 as the most appropriate primary outcome measure for a full trial.
Develop and test trial procedures
Protocols, eligibility criteria, patient information material and case report forms (CRFs) were designed for the pilot RCT and were available for the full trial. We interviewed 18 patients who had been treated for FAI syndrome to develop patient information sheets. These patient information sheets were scrutinised by a panel of expert patients with FAI syndrome who helped to improve the content and presentation so that they addressed patients’ key concerns and information needs, and provided explanations with appropriate language and detail. Twenty-eight clinicians, including surgeons, physicians and physiotherapists, also contributed to developing these procedures and documents.
Research Ethics Committee and national R&D approvals were granted for the pilot trial promptly and without any significant concerns. The majority of the recruitment sites were then able to complete local approval within 1 month of our site initiation visits. Typical causes for a delay to approval were identified within the first few sites, allowing these to be addressed in subsequent sites at a much earlier stage. This may help considerably to ensure that further sites in a full trial can obtain local R&D approval more quickly.
Conclusion of the feasibility study and pilot trial
We showed that a robust RCT of hip arthroscopy compared with best conservative care for patients with FAI syndrome was feasible, that patients and clinicians were willing to participate, that we were able to obtain ethics and R&D approval at multiple sites, and that the trial procedures we developed worked well. The pilot trial recruited successfully (70% recruitment rate) to the protocol that will be used for the full trial and these patients were, therefore, included in the full trial analysis.
Relevance of project
Young adults with hip pain are now often aware of the diagnosis of FAI syndrome. There are many descriptions in scientific literature, popular press and on the internet, but there is an overwhelming focus on the benefits of surgery, with little regard to other treatments. 7,20 With limited evidence of effectiveness and a significant increase in the cost of arthroscopic surgery (with an NHS tariff for hip arthroscopy of £5200), a number of NHS care commissioners have begun to limit the funding for this procedure. In some areas, hip arthroscopy is not commissioned at all and in others, only patients who have failed to respond to non-operative treatments are allowed access to arthroscopic surgery. 21 Provision of non-operative alternatives to surgery for FAI syndrome is inconsistent, and the evidence and guidance for this conservative care is weak. 20 PHT is a credible physiotherapy-led ‘best conventional care’ protocol for FAI syndrome, developed for the pilot trial through clinical consensus informed by existing literature. 5 This trial will establish the best treatment for patients with FAI syndrome, taking into account clinical effectiveness, costs and risks. This will allow clinicians within the NHS to offer treatment for FAI syndrome that is in patients’ best interests. Establishing the comparative cost-effectiveness of arthroscopy and PHT will help NHS commissioners to make funding decisions based on robust evidence and to avoid the current situation of unjustified variation in provision.
Null hypothesis
Our null hypothesis was that there is no difference in the iHOT-33 questionnaire score 12 months following randomisation between adults diagnosed with FAI syndrome treated with arthroscopic hip surgery and adults diagnosed with FAI syndrome treated with best conservative care.
Objectives
The primary objective was to measure the clinical effectiveness of hip arthroscopy compared with best conservative care for patients with FAI syndrome, assessed by patient-reported hip-specific quality of life after 1 year.
The secondary objectives were to:
-
compare differences in general health status and in health-related quality of life after 12 months between treatment groups
-
compare, in a longitudinal analysis, the pattern of clinical change over 36 months
-
compare patient satisfaction with treatment and outcome after 1 year
-
compare the number and severity of adverse events (AEs) after treatment
-
compare the need for further procedures up to 3 years after initial treatment
-
compare the cost-effectiveness of hip arthroscopy for FAI with best conservative care, within the trial and for a patient’s lifetime
-
develop and report processes to optimise recruitment in a RCT or surgery compared with non-operative care
-
measure the fidelity of delivery of interventions.
Chapter 2 Methods
This trial was conducted in accordance with the Medical Research Council’s Good Clinical Practice principles and guidelines, the Declaration of Helsinki,35 Warwick Clinical Trials Unit (WCTU) (Coventry, UK) standard operating procedures (SOPs), relevant UK legislation and the trial protocol. Ethics approval was granted on 1 May 2014 (reference 14/WM/0124) by the Edgbaston Research Ethics Committee (current approved protocol version 4.0, 18 August 2017). The trial was registered as ISRCTN64081839. This project was funded by the National Institute for Health Research Health Technology Assessment programme (feasibility and pilot trial grant number 10/41/02, full trial grant number 13/103/02).
Trial design
We conducted a multicentre, pragmatic, assessor-blinded parallel-arm 1 : 1 RCT of hip arthroscopy compared with conservative care for FAI syndrome, assessing patient pain, function, general health, quality of life, satisfaction and cost-effectiveness. There was an integrated qualitative recruitment intervention (QRI) that included interviews with recruiters and patients, and observations of recruitment appointments, to ensure that patients had the opportunity to fully consider participation in the trial. 28
We hypothesised that arthroscopic surgery is superior to conservative care at 12 months for self-reported hip pain and function for patients with FAI syndrome. The trial was conducted on consenting patients treated in the NHS. Hospitals participating in the FASHIoN trial had an organised hip arthroscopy service that treated at least 20 patients with arthroscopic surgery for FAI syndrome per year.
Participants
We recruited a cohort of typical patients with FAI syndrome deemed suitable for arthroscopic surgery. This cohort included patients who may have already received a course of physiotherapy.
Inclusion criteria
-
Age ≥ 16 years (with no upper age limit).
-
Symptoms of hip pain (including clicking, catching or giving way).
-
Radiographic evidence of pincer- and/or cam-type FAI morphology on plain radiographs and cross-sectional imaging, defined as:
-
The treating surgeon believes the patient would benefit from arthroscopic FAI surgery.
-
The patient is able to give written informed consent and to participate fully in the interventions and follow-up procedures.
Exclusion criteria
-
Evidence of pre-existing osteoarthritis, defined as Tönnis grade > 138 or a > 2-mm loss of superior joint space width on an anteroposterior pelvic radiograph. 39
-
Previous significant hip pathology, such as Perthes’ disease, slipped upper femoral epiphysis or avascular necrosis.
-
Previous hip injury, such as acetabular fracture, hip dislocation or femoral neck fracture.
-
Previous shape-changing surgery (open or arthroscopic) in the hip being considered for treatment.
Screening and recruitment
Participants were recruited from among the patients presenting to young adult hip clinics in each of the centres. Patients who complained of hip pain and who did not already have a diagnosis of hip osteoarthritis were identified as potential participants by screening referral letters to collaborating surgeons. Research nurses/associates kept accurate screening logs to identify whether or not these potential participants met the eligibility criteria. Prior to their appointment, these patients were approached to seek consent for recording of their clinic consultations.
Surgeons assessed patients as usual, taking a history, examining the patient and performing appropriate imaging investigations. Patients in whom a diagnosis of FAI syndrome was made and who met the eligibility criteria received a description of the condition from their surgeon and an explanation that there were two possible treatments: (1) an operation or (2) a package of PHT. Patients were given patient information about FAI syndrome and the trial. Patients were then invited to a trial information consultation to discuss what action they would like to take.
Patients attended a trial information consultation with a trained clinical researcher. Information was again provided about FAI, FAI’s possible treatments and the trial. Patients were given an opportunity to ask questions. Patients were then invited to give their consent to become participants in the trial. Patients who wanted to take more time to consider were given an opportunity to do so. Patients who agreed to take part completed baseline questionnaires at this consultation.
Consent
Written informed consent was obtained by a researcher delegated and trained by the research team. In general, patients had at least 1 month from initial consultation to the day of surgery or start of PHT so that there was sufficient time for patients to consider taking part in the trial.
Qualitative research intervention
To optimise recruitment and informed consent, trained qualitative researchers listened to recordings of the surgeons’ and RA/nurses’ trial information consultations to identify communication patterns that facilitated or hindered patient recruitment. 28 In-depth interviews with the recruiters were undertaken to identify clear obstacles and hidden challenges to recruitment, including the influence of patient preferences and equipoise. 40 Research teams were interviewed to identify clinician equipoise, patient pathways from eligibility to consent and staff training needs at each participating site. 28 Findings were fed back to the chief investigator and Trial Management Group (TMG) so that practice could be reviewed and any necessary changes (including additional training) implemented. The number of eligible patients and the percentages of these patients who were approached and consented to randomisation were monitored at each site.
This research was linked to the Quintet programme of research within the Medical Research Council ConDuCT-II (Collaboration and innovation in Difficult and Complex randomised controlled Trials II) Trial Methodology Hub (Bristol, UK).
Randomisation
Participants were randomised, in a 1 : 1 ratio, to arthroscopic surgery or PHT using a computer-generated sequence. Allocation was made by the research nurse/associate via a centralised telephone randomisation service provided remotely by WCTU. Allocation concealment was ensured, as the randomisation programme did not release the randomisation code until the patient had been recruited into the trial. Research nurses/associates who recruited participants ensured that they were referred for the allocated intervention.
Sequence generation
To improve the baseline balance between intervention group samples, a minimisation (adaptive stratified sampling) algorithm was implemented using study site and impingement type (i.e. cam, pincer or mixed) as factors.
Blinding
The patients could not be blind to their treatment. The treating surgeons were not blind to the treatment, but took no part in outcome assessment for the trial. The functional outcome data were collected and entered onto the trial central database via postal questionnaire by a research assistant who was blind to the treatment allocation. The statistical analysis was also performed blind.
Post randomisation withdrawals
Participants could withdraw from the trial treatment and/or the whole trial at any time without prejudice. If a participant decided to change from the treatment to which they were allocated, they were followed up and data collected as per the protocol until the end of the trial. However, every effort was made to minimise crossovers from both intervention arms. It was made clear to study participants and clinicians that it was important for the integrity of the trial that everyone followed their allocated treatment. For those participants who decided to move to the other intervention arm, the numbers, direction and reasons for moving were recorded and reported in line with CONSORT (Consolidated Standards of Reporting Trials) guidance. The QRI investigated how and why participants made their decision. The QRI team provided training for physiotherapists and surgeons so that they were equipped to answer patients’ questions about the trial during treatment. During the pilot trial, we found that this reduced the risk of participants losing confidence in the trial and breaching protocol.
Interventions
The two interventions commenced as soon as possible after randomisation. We recorded the dates of randomisation and the start of allocated treatment. As this was a pragmatic trial, participants were not prohibited from undergoing any additional/concomitant care.
Arthroscopic surgery
An operative protocol was established during and implemented in the pilot trial. The agreed protocol was typical of the surgical techniques used by the majority of surgeons around the world, and representative of those used in the UK. The surgeons delivering the intervention were all NHS consultants.
Preoperative protocol
Patients underwent routine preoperative care, which included an assessment of their general health and suitability for a general anaesthetic.
Perioperative protocol
Arthroscopic hip surgery was performed under general anaesthesia with the patient in a lateral or supine position. Arthroscopic portals were established in the central and peripheral compartment under radiographic guidance and in accordance with the surgeon’s usual practice. Shape abnormalities and consequent labral and cartilage pathology was treated. Bony resections at the acetabular rim and the head–neck junction were assessed by intraoperative image intensifier radiograph and/or satisfactory impingement free range of movement of the hip.
Postoperative protocol
Patients were allowed home when they could walk safely with crutches (usually within 24 hours). On discharge, all patients were referred to outpatient physiotherapy services for a course of rehabilitation, as per usual care for that surgeon. We did not specify a protocol for this postoperative physiotherapy, but recorded it using a treatment log. Postoperative physiotherapists were distinct from those providing PHT to avoid contamination between groups. Patients also had a postoperative MRI, which included a proton density volume acquisition sequence (for MRI protocol see Appendix 1).
Fidelity assessment
To ensure the fidelity of the surgery and to identify participants for a secondary analysis, a panel of international experts reviewed operation notes, intraoperative images and postoperative MRI scan to assess whether or not adequate surgery was undertaken (see Appendix 2). This panel included Mark Philippon (USA; then chairperson of the Research Committee of the International Society for Hip Arthroscopy), Martin Beck (Switzerland; one of the investigators credited with developing the early understanding of FAI), John O’Donnell (Australia; past president of the International Society of Hip Arthroscopy) and Professor Charles Hutchinson (UK; an expert in musculoskeletal radiology). The fidelity assessment process was tested in the pilot trial. The panel rated each surgical case as satisfactory, borderline satisfactory and unsatisfactory (see Appendix 1).
Personalised hip therapy
Personalised hip therapy was a package of physiotherapy-led best conservative care for FAI syndrome. 5 It was developed during the feasibility study and ‘road-tested’ during the pilot trial. 3 Although the name for this intervention was new, the care being offered represented a consensus of what physiotherapists, physicians and surgeons in the NHS provided and regard as ‘best conventional care’. PHT was delivered by at least one qualified physiotherapist at each site. To prevent contamination of the treatment groups, the physiotherapists who delivered PHT were distinct from those who delivered postoperative physiotherapy.
Training physiotherapists
Personalised hip therapy physiotherapists were trained in a FASHIoN PHT workshop and supported by the physiotherapy lead and research facilitator (NF and JS).
We developed and tested the 1-day workshop during our pilot trial. Following the initial PHT workshops during the feasibility study, the remaining workshops were delivered through the recruitment period from November 2014 to March 2016. The workshops included lectures, presentations, discussion of real cases and working through PHT progressions. A PHT manual and exercise sheets (for patients) were provided to all the PHT physiotherapists (see Appendix 1). Ongoing training and support was provided by the physiotherapy research facilitator and this included an initial site visit and monitoring visits. The purpose of the initial visit was to ensure that the treating physiotherapist fully understood the detail of PHT. The first visit was scheduled to occur after the first patients were randomised to PHT and before they had started treatment. Monitoring visits provided opportunities for further training and to conduct a source verification audit (see Fidelity assessment). Although PHT offered a framework to deliver best conservative care, the treatment was not a fully standardised regime. Physiotherapists were trained and encouraged to tailor their treatment to each patient, focusing on deficiencies identified in their assessment and based on the patients’ progression.
Pre treatment
Participants received a PHT information pack (see Appendix 1) that described what to expect during the course of their treatment. The first core component of PHT was an assessment of pain, function and range of hip motion.
Treatment
Personalised hip therapy had three further core components: (1) an exercise programme that had the key features of individualisation, progression and supervision; (2) education; and (3) help with pain relief (which may have included one X-ray or ultrasound-guided intra-articular steroid injection if pain prevented performance of the exercise programme). The intervention was delivered over a minimum of 12 weeks, with a minimum of six patient contacts. Some of the patient contacts were permissible using either telephone/e-mail for whom geographical distance prevented all contacts being carried out face to face. The number and frequency of the treatment sessions was at the discretion of the physiotherapist and was informed by the patients’ deficiencies and progression.
Post treatment
Typically, PHT was delivered over a minimum of 12 weeks. However, in situations in which the patient needed additional review, support or guidance, further sessions with the physiotherapists were permitted up to a maximum of 10 sessions over 6 months.
Fidelity assessment
To assess the accuracy of the PHT CRFs a source verification audit was undertaken to compare the physiotherapists’ hospital notes and the PHT CRF. Source verification was undertaken at each site and with 10% of cases sampled. The CRFs were graded as either a satisfactory or unsatisfactory reflection of the hospital notes. The source verification was undertaken by the physiotherapy research facilitator (JS). The findings of the source verification audit were fed back to the fidelity assessment panel.
The PHT CRFs were assessed to determine the fidelity of each intervention and to identify participants for a secondary analysis. This assessment was completed by the panel that developed the protocol for PHT, including Nadine Foster (Senior Academic Research Physiotherapist), Ivor Hughes and David Robinson (UK; Extended Scope Musculoskeletal Physiotherapists) and Peter DH Wall (Academic Orthopaedic Surgeon).
Treatment was rated as satisfactory, borderline or unsatisfactory. The panel assessed whether or not a sufficient number of treatments had occurred (at least six sessions in 12 weeks, but fewer than 10 sessions in 6 months), whether or not the treatment included all four core components of PHT and whether or not the exercise programme was individualised, supervised and progressive.
Treatment crossover
Crossover of participants between interventions can be problematic in trials of this nature. To minimise this, care was taken prior to enrolment in the trial to ensure that potential participants:
-
were willing to receive either intervention
-
understood that both treatments were thought to provide benefit
-
were willing to remain with their allocation for 12 months
-
understood that both interventions may take 6 months to improve symptoms. 39,41
In instances where patients were not satisfied with how their treatment was progressing prior to reaching the primary outcome, they were able to have a further consultation with their treating surgeon where they were treated in their best interests.
Outcomes
Baseline data were collected from participants once consent was obtained and prior to randomisation. Follow-up questionnaires were administered centrally by a data clerk via post. If participants failed to respond, they were contacted via telephone, e-mail or via their next of kin, where necessary.
Primary outcome
The primary outcome was hip pain, function and hip-related quality of life measured using the iHOT-33 at 12 months following randomisation. The iHOT-33 is a validated hip-specific patient-reported outcome tool that measures health-related quality of life in young, active patients with hip disorders. 31 The iHOT-33 consists of the following domains: symptoms and functional limitations, sports and recreational activities, job-related concerns and social, emotional and lifestyle concerns.
We chose it following our feasibility and pilot study, as it is more sensitive to change than other hip outcome tools, it does not show evidence of floor or ceiling effects in patients undergoing hip arthroscopy and patients were involved extensively in item generation and, therefore, we can be confident that it measures what is most important to patients. The iHOT-33 has an independently determined MCID. The iHOT-33 is also used as the principal outcome measure for the UK Non-Arthroplasty Hip Registry, which is mandated for arthroscopic FAI surgery by NICE. 15,31
Secondary outcome measures
Health-related quality of life: EuroQol-5 Dimensions, five-level version
The EQ-5D-5L is a validated measure of health-related quality of life, consisting of a five-dimension health status classification system and a separate VAS. EQ-5D-5L is applicable to a wide range of health conditions and treatments, and provides a simple descriptive profile and a single index value for health status. 42 Responses were converted into health utility scores using established algorithms. 43
General health: Short Form questionnaire-12 items
The Short Form questionnaire-12 items (SF-12) is a validated and widely-used health-related quality-of-life measure that is used for hip conditions and treatments. 44 SF-12 is able to produce the physical and mental component scales originally developed from the Short Form questionnaire-36 items with considerable accuracy, but with far less respondent burden. 45 Responses were converted into health utility scores using established algorithms. 46
Patient satisfaction
Patient satisfaction was measured using questions that our team (NF) had used in previous trials with musculoskeletal pain patients. 47 We measured two distinct dimensions of satisfaction in all participants during follow-up: (1) ‘overall, how satisfied are you with the treatment you received?’ and (2) ‘overall, how satisfied are you with the results of your treatment?’ Responses were on a five-point Likert scale.
Qualitative assessment of outcome
We conducted in-depth one-to-one interviews with a purposively selected sample of 25–30 participants in each of the trial groups. These samples included older and younger, male and female, more and less active, and more and less satisfied participants recruited at different trial sites. The qualitative interviews supplement the quantitative outcomes. Interviews explored experiences of the trial processes, the treatments and the consequences of treatment to participants’ lives, health and well-being.
Adverse events
We recorded the number and type of AEs up to 12 months. Any AEs were reported on the appropriate CRF and returned to WCTU. Any serious adverse events (SAEs) were faxed to WCTU, within 24 hours of the local investigator becoming aware, where the chief investigator determined causality and expectedness. SAEs deemed unexpected and related to the trial were reported to the Research Ethics Committee within 15 days.
Resource utilisation
Information on health-care resource use was collected by incorporating questions within the patient follow-up questionnaires. We confirmed the feasibility and acceptability of this approach in our pilot trial. In addition, patient self-reported information on service use has been shown to be accurate in terms of the intensity of use of different services. 48
Need for further procedures
We recorded any further treatments performed in both groups, such as hip arthroscopy, open hip preservation surgery, hip replacement or additional ‘out-of-trial’ physiotherapy. We ascertained the need for further procedures by questionnaire at 2 and 3 years. In addition, we also propose a 5- and 10-year no-cost ascertainment of hip replacement by linkage to the UK National Joint Registry and Hospital Episode Statistic databases.
Follow-up
The follow-up schedule is outlined in Table 1. The primary outcome was collected 12 months following randomisation.
| Time point | Data collection |
|---|---|
| Baseline | Demographics, physical activity (UCLA Activity Scale),49 iHOT-33, SF-12, EQ-5D-5L, preoperative imaging and economics questionnaire |
| Intervention | Operation notes and photographs or PHT log, complications records 6 weeks post start of intervention and postoperative MRI (surgery intervention only) |
| 6 months | iHOT-33, SF-12, EQ-5D-5L, resource utilisation and AEs |
| 12 months (primary outcome) | iHOT-33, SF-12, EQ-5D-5L, patient satisfaction, resource utilisation and AEs |
| 2 years | iHOT-33, EQ-5D-5L and further procedures questionnaire |
| 3 years | iHOT-33, EQ-5D-5L and further procedures questionnaire |
| 5 and 10 years | Linkage to the National Joint Registry and Hospital Episode Statistics to identify need for hip replacement |
Adverse event management
Adverse events are defined as any untoward medical occurrence in a clinical trial patient that do not necessarily have a causal relationship with the treatment. All AEs were listed on the appropriate CRF and returned to the FASHIoN trial central office.
Serious adverse events are defined as any untoward and unexpected medical occurrences that:
-
result in death
-
are life-threatening
-
require hospitalisation or prolongation of existing inpatients’ hospitalisation
-
result in persistent or significant disability or incapacity
-
are a congenital anomaly or birth defect
-
are important medical conditions that, although not included in the above, may require medical or surgical intervention to prevent any of the outcomes listed above.
All SAEs were entered onto the reporting form and faxed to WCTU within 24 hours of the investigator becoming aware of them. Once received, causality and expectedness was confirmed by the chief investigator. The Research Ethics Committee were notified, within 15 days, of SAEs that were deemed to be unexpected and related to the trial. All such events were reported to the Trial Steering Committee (TSC) and Data Monitoring Committee (DMC) at their next meeting.
Serious adverse events that were expected as part of both interventions are listed in Risks and Benefits below. All participants who experienced SAEs were followed up as per protocol until the end of the study period.
Risks and benefits
Both interventions were thought to provide benefit in patients with FAI syndrome. The short-term risks of the study related to the two interventions. These risks are described below and informed the expected SAEs.
Arthroscopic surgery
Hip arthroscopy requires a general anaesthetic. The risk of complications from hip arthroscopy is about 1–2% and these include the following:
-
Infection, which is thought to occur in less than 1 in 1000 patients.
-
Bleeding, possibly causing bruising or a local haematoma.
-
Traction-related complications. (To perform hip arthroscopy, traction is required to separate the hip joint surfaces. Sometimes after the procedure, the pressure from the traction can cause some numbness in the leg, but the numbness usually resolves within a few hours or days.)
-
Osteonecrosis. (During surgery, the blood supply to the hip joint could be damaged; however, there are no reported cases of osteonecrosis following arthroscopic FAI surgery.)
-
Femoral neck fractures. (This is also a very rare complication and would require a further procedure to fix the fracture.)
Personalised hip therapy
There are some small risks with pain medications and joint injection. However, the main risk is muscle soreness and transient increases in pain from the exercises that were undertaken.
Statistical analysis
The primary analysis was the difference, at 12 months, in hip-related quality of life (using the iHOT-33) between the two treatment groups, blinded, on an intention-to-treat basis and presented as the mean difference between the trial groups with a 95% confidence interval (CI). The iHOT-33 data were assumed to be normally distributed after appropriate variance-stabilising transformation.
The minimisation randomisation procedure should have ensured treatment group balance across recruiting sites. We had no reason to expect that clustering effects would be important for this study, but the possibility of such effects was explored as part of the analysis. We planned to account for clustering by generalising a conventional linear (fixed-effects) regression approach to a mixed-effects modelling approach where patients are naturally grouped by recruiting sites (random effects) and, if amenable to analysis, also by physiotherapist and surgeon. This model formally incorporated terms that allowed for possible heterogeneity in responses for patients due to the recruiting centre, in addition to the fixed effects of the treatment groups and patient characteristics that may prove to be important moderators of treatment effect, such as age, sex and FAI type. The analysis was conducted using specialist mixed-effects modelling functions available in the software packages Stata® release 14 (StataCorp LP, College Station, TX, USA) and R (The R Foundation for Statistical Computing, Vienna, Austria). All tests were two sided and were considered to provide evidence for a statistically significant difference if p-values were < 0.05 (i.e. a 5% significance level).
Secondary analyses was performed using the above strategy for other approximately normally distributed outcome measures, including iHOT-33 at 6 months, SF-12 (and computed subscales) and EQ-5D-5L. Differences in dichotomous outcome variables, such as AEs, complications related to the trial interventions and the need for further procedures, were compared between groups using chi-squared tests (or Fisher’s exact test) and mixed-effects logistic regression analysis, adjusting for the stratifying variables, with differences between trial intervention groups quantified as odds ratios (and 95% CIs). The temporal patterns of AEs were presented graphically and, where appropriate, a time-to-event analysis (Kaplan–Meier survival analysis) to assess the overall risk and risk within individual classes of AEs. Ordinal scores for patient satisfaction were compared between intervention groups using proportional odds logistic regression analysis, assuming that the estimated intervention effect between any pair of categories is equivalent.
Our inferences were drawn from the intention-to-treat analysis. We performed two exploratory secondary analyses. One exploratory analysis compared patients who received surgery with those who received conservative care. A second exploratory analysis compared patients randomised to surgery and PHT and who received treatment deemed to be of a high fidelity by the respective review panels. We performed a subgroup analysis by FAI type because it was possible that treatment effect is moderated by type. We anticipated that adequate steps were taken to prevent crossovers from being a major issue in this study. Therefore, we expected the main intention-to-treat analysis to provide definitive results. An independent DMC monitored crossovers and adherence to treatment and advised on appropriate modifications to the statistical analysis plan as the full progressed.
The feasibility and pilot studies2,3 were designed explicitly to assess feasibility and measure recruitment rates, and not to estimate treatment effectiveness. Data from the pilot were pooled with data from the full trial, and analysed together.
Sample size
The development work for iHOT-33 reported a mean iHOT-33 score of 66 (SD 19.3) in a heterogeneous population with a variety of hip pathologies. The baseline iHOT-33 data from our pilot trial suggests that the target population of patients being considered for hip arthroscopy for FAI in the UK have lower scores with less variability than the heterogeneous population, with a mean score of 33 (SD 16).
During our feasibility study, we estimated the likely effect size of hip arthroscopy compared with best conventional care for FAI to be 0.5. The MCID for iHOT-33 in this population is 6.1 points. Our sample size calculation is, therefore, based on a SD of 16 and a MCID of 6.1 (i.e. a standardised effect difference between groups at 12 months of 0.38).
Table 2 shows the expected sample size for scenarios with 80% and 90% power to detect an effect of this size, at a 5% significance level, assuming an approximately normal distribution of the iHOT-33 score. Table 2 also shows sample sizes for small to moderate (0.32) and moderate (0.47) effect differences, which are broadly consistent with other pragmatic RCTs measuring clinical effectiveness.
| SD | Power | Standardised effect difference | |
|---|---|---|---|
| 80% | 90% | ||
| 13.3 | 144 | 192 | 0.47 |
| 16.0 | 218 | 292 | 0.38 |
| 19.3 | 316 | 422 | 0.32 |
A systematic review of observational studies50 reported effect sizes of hip arthroscopy for FAI of between 0.67 and 2.95 up to 5 years after surgery, but these are likely to be overestimates of the real effect we might measure in this trial. These observational studies were uncontrolled studies, and we anticipate that our best conventional care protocol will provide some benefit.
We have, therefore, adopted a conservative approach, seeking to demonstrate an effect difference between groups equal to the MCID. We proposed to recruit sufficient patients to be able to analyse 292 patients at the 12-month follow-up. Allowing for 15% loss to follow-up, we aimed to recruit a sample of 344 participants (i.e. 172 participants in each group). This would provide 90% power to detect a difference of 6.1 iHOT-33 points, if that is the true difference.
Analysis plan
A full statistical analysis plan was developed and approved by the trial statistician(s) and the chief investigator. This plan was also reviewed by the DMC once finalised, in line with the SOPs at WCTU.
Software
All routine interim data reports and final statistical analyses were conducted using Stata 14. A bespoke secure database was created by the programing team at WCTU to enter, store and maintain all trial data and monitor them for accuracy and integrity. A secure Open Database Connectivity data link was used to obtain data when necessary, and data export was restricted to only those members of the trial team who required access for analysis purposes.
Data validation
A FASHIoN data monitoring plan was developed at the outset of the study. The plan covered all aspects of data collection, including data entry, receipt, storage, checking, security and transfer.
Monitoring of data collection was also conducted by the independent DMC, which received regular reports on data quality and completeness as part of its ongoing support to the study. Prior to the final analysis, data were checked for outliers and missing data. Outcome data were validated using defined score ranges for each measure. Any queries were reported to the trial co-ordinator who liaised with the relevant recruiting centre, if appropriate. All subsequent changes to the data were recorded in accordance with the relevant SOP and the FASHIoN data management plan.
Missing data
Data were not available because of withdrawal of patients, lack of completion of individual data items and loss to follow-up. Reasons for missing data were ascertained and reported as far as possible. Any patterns of missing data were carefully considered, including, in particular, whether or not data could be treated as missing completely at random. No formal statistical testing was planned to assess missing data, but model assumptions were checked and patterns explored. If judged appropriate, missing data in the primary outcome (iHOT-33) were imputed using an imputation procedure in Stata (from the mi set of commands). Any imputed data were on an individual item level, as opposed to an overall score level. Reasons for ineligibility, non-compliance, withdrawal or other protocol violations and deviations are stated, and any patterns summarised, in Chapter 4.
Interim analyses
There were no pre-planned interim data analysis for the FASHIoN study, and the study sample size and design were powered only for the final analysis.
Exploratory analysis
A post hoc unplanned exploratory analysis was undertaken to investigate the effect of the timing of treatment on the primary outcome, an issue that was not identified prior to study design and conception. The most appropriate approach was to include an additional binary covariate in the model, which indicated whether treatment was early (< 12 weeks) or late (> 12 weeks) and assess whether or not the inclusion of this covariate improved the model fit and had an impact on the size and interpretation of the treatment effect.
Economic evaluation
Overview
A prospective within-trial cost–utility analysis was conducted to estimate the cost-effectiveness of arthroscopic surgery compared with PHT as treatment options for FAI syndrome. Costs were expressed in GBP (2016 price year) and health outcomes in quality-adjusted life-years (QALYs). The base-case analysis was based on the intention-to-treat population and conducted from the perspective of UK NHS and Personal Social Services. The time horizon covered the period from randomisation to end of follow-up at 12 months post randomisation. Costs and outcomes were not discounted because of the short 1-year time horizon adopted for this within-trial evaluation. Sensitivity and subgroup analyses were conducted to investigate the likely impact of alternative data inputs and assumptions on cost-effectiveness, and identify subgroups most likely to benefit from treatment. Findings are reported in accordance with the CHEERS (Consolidated Health Economic Evaluation Reporting Standards) guidelines. 51
Measurement of resource use and costs
Data were collected on (1) resource use and costs associated with delivery of the interventions, (2) health and social care service use during the 12 months of follow-up and (3) broader societal resource use and costs (e.g. private medical costs and lost productivity costs, such as lost income over the 12 months of follow-up).
Cost of personalised hip therapy
Personalised hip therapy was delivered to trial participants primarily by experienced physiotherapists (grade 7 and above) within NHS hospital outpatient clinics. The number and duration of PHT sessions attended were recorded for all patients who received this intervention. The unit cost of a band 7 hospital physiotherapist (including qualifications and overheads) was obtained from the Personal Social Services Research Unit (PSSRU) Unit Costs of Health and Social Care 201652 and was £55 per hour. Unit costs were multiplied by duration of physiotherapy contact (in minutes) and summed across sessions attended to give total treatment costs per patient. Indirect costs associated with delivery of the intervention, such as use of the treatment room facility, administrative support and overheads, are taken into account in PSSRU unit cost calculations and, therefore, separate costs for these were not included in our estimate of PHT costs.
Cost of surgery
A micro-costing exercise was undertaken to estimate resource use and costs associated with delivery of arthroscopic surgery for FAI. Resource use data were collected for a subsample of trial participants who had received the surgery using a specially designed costing questionnaire that captured the following items:
-
duration of surgery
-
post-surgical inpatient length of stay
-
number, specialty and grade of clinical staff involved in the surgical procedure
-
quantity and type of disposable arthroscopic equipment and/or implants used.
Surgery time was defined from start of anaesthesia to time patient left the operating room on completion of surgery. Inpatient length of stay was counted as 1 day if the patient was admitted and discharged on the same day, 2 days if the patient was discharged the next day and so on, which is in line with NHS reference costing methodology. 53 Anaesthetic drugs and associated consumables, such as syringes and needles, were collected separately during a sample of operations and assumed to be the same for all patients who had the surgery.
Total cost of surgery was calculated for each patient by summing across the following five categories: (1) staff time, (2) theatre use in hours, (3) disposal surgical equipment, (4) anaesthetic drugs and disposables and (5) post-surgery inpatient bed-days. Operating room/theatre running costs were estimated based on data published by Information Services Division (Edinburgh, UK). 54 The Scottish data reported total number of theatre hours used and total allocated costs across NHS hospitals in Scotland for the 2015–16 financial year. Allocated costs are defined to include expenditure on non-clinical staff, property and equipment maintenance, domestics and cleaning, utilities, fittings and capital expenditure, and excluded clinical staff costs. 55 The hourly running cost of an operating room/theatre was obtained by dividing the total allocated costs per year by the total theatre time (in hours) per year.
Unit costs of clinical staff time were obtained from the PSSRU Unit Costs of Health and Social Care 201652 compendium. As stated above, these unit costs already factor in direct cost of staff salaries and employer oncosts and training costs, as well revenue and capital overheads, administrative support, office space and work-related travel. The cost of disposal surgical equipment and implants were primarily obtained from the 2016 online edition of the NHS supply chain catalogue. 56 When cost data were not available from the NHS catalogue, procurement department unit costs from the University Hospital Coventry and Warwickshire (Coventry, UK) were applied (Felix Achana, University of Warwick, 2012, personal communication). Cost of anaesthetic drugs were obtained from the prescription costs analysis database. 53
Resource use during follow-up
Health and social care service use were collected from trial participants for the 3-month period prior to randomisation (to establish baseline data) and the 1-year period post randomisation. Resource use data were collected at three assessment points (i.e. baseline and 6 and 12 months post randomisation) and included:
-
details of hospital inpatient and day case admissions
-
details of outpatient and accident and emergency attendances
-
primary/community care encounters
-
use of personal social care services (e.g. Meals on Wheels, laundry services and social care contacts)
-
prescribed and over-the-counter medication use
-
supplied or self-purchased walking aids, such as crutches and walking sticks, and adaptations to home or work environments
-
any other additional costs incurred by patients and their families as a result of their hip pain, including private medical costs and out-of-pocket expenditures (e.g. travel costs by patients and family members), child care costs and lost income.
Resource inputs were valued by attaching unit costs derived from national compendia to resource inputs.
Hospital-based services included inpatient admissions, day care, outpatient and accident and emergency attendances, and diagnostic tests and scans. Unit costs for these services were obtained primarily from the 2015/16 NHS reference costs main schedules. 57 Per diem costs were calculated for each inpatient admission as a weighted average of Healthcare Resource Group (HRG) codes of related procedures and/or clinical conditions. For example, the average cost per day for inpatient stay in an orthopaedic ward with procedures carried out on the hip/leg was calculated as a sum total of the weighted average of lower limb orthopaedics (trauma) HRG codes divided by average length of stay across elective and non-elective inpatient services.
Primary and community health and social care services included face-to-face or telephone contacts and/or home visits by a general practice doctor, practice nurse, community physiotherapy or other community health or social care professionals. Consultation costs were derived from the PSSRU Unit Costs of Health and Social Care 201652 compendium.
The cost of private physiotherapy and other private medical costs were obtained from online sources and referenced appropriately in the unit cost tables.
The cost of prescribed medication was obtained primarily from the prescription cost analysis database53 and electronic searches of the British National Formulary (BNF) 2016 edition. 58 Typical dosage and duration of treatment reported in the BNF for each medication were used in calculating quantity of individual preparations if the daily dose and/or duration of the course of medication were not reported. The quantity of over-the-counter medicines were rounded to the nearest pack and unit costs obtained from online sources.
The cost of walking aids and adaptations were either provided by the patients themselves (if self-purchased) or taken from the NHS supply chain catalogue56 if supplied by a health provider during the trial follow-up period. It was assumed that walking aids, such as crutches, sticks, grab rails, dressing aids and specially adapted shoes, were supplied as part of treatment if the cost of purchase were not provided by trial participants.
Patient-level costs were generated for each resource variable by multiplying the quantity reported by the respective unit cost weighted by duration of contact, when appropriate. Summary statistics were generated for resource use variables by treatment allocation and assessment point. Between-treatment group differences in resource use and costs at each assessment point were compared using the two-sample t-test. Statistical significance was assessed at the 5% significance level. Standard errors (SEs) are reported for treatment group means and bootstrap 95% CIs for the between-group differences in mean resource use and cost estimates.
Measurement of outcomes
The health-related quality of life of trial participants was assessed at baseline and at 6 and 12 months post randomisation using the EuroQol-5 Dimensions, three-level version (EQ-5D-3L) in the feasibility study, the EQ-5D-5L in the main trial and the SF-12 in both feasibility and main trial samples. 59–61 Responses to each health dimension were categorised as optimal or suboptimal with respect to function, with optimal level of function indicating no impairment (e.g. ‘no problem’ on the EQ-5D-3L dimensions) and suboptimal indicating any functional impairment. Between-group differences in optimal and suboptimal level of function for each health dimension were compared for each outcome measure using chi-squared tests.
The responses to each health-related quality-of-life instrument were converted into health-related quality-of-life weights (also referred to as utility weights) using established algorithms for each instrument. Utility values were generated using the UK value set for the EQ-5D-3L, the interim crosswalk value set for mapping from the EQ-5D-5L to the EQ-5D-3L, the newly published EQ-5D-5L tariffs for the EQ-5D-5L and the Short Form questionnaire-6 Dimensions (SF-6D) tariff based on SF-12 responses. 46,62–64
Quality-adjusted life-years were generated for each patient using the area under the baseline-adjusted utility curve, assuming linear interpolation between the three utility measurements. QALYs were generated for patients in the feasibility sample using utilities derived from EQ-5D-3L and SF-6D tariffs and for those in the main study sample using the EQ-5D-5L crosswalk tariff, the new UK EQ-5D-5L tariff and the SF-6D tarrif. 46,62–64 Health utility values and QALYs accrued over the 12-month follow-up were summarised by treatment group and assessment point and presented as means and associated SEs. Between-group differences were compared using the two-sample t-test, similar to the summary analyses of resource inputs and costs.
Cost-effectiveness analysis methods
Missing data
Multiple imputation by chain equations implemented through the MICE package in R was used to handle missing costs and health utility data at each assessment point. Multiple imputation avoids problems associated complete-case analyses, is consistent with good practice and requires data to be missing at random only. 65 Appropriateness of this missing-at-random assumption was assessed by comparing the characteristics of patients with and without missing costs and health-related quality-of-life data at each follow-up time point. Imputations were generated separately by treatment group, as recommended by Faria et al.,66 using the predictive mean matching method, which has the advantage of preserving non-linear relationships and correlations between variables within the data. Twenty imputed data sets were generated and the analyses were fitted to each imputed data set. The results from the 20 data sets were then combined using Rubin’s rules. The imputation, analysis and pooling of results steps were performed simultaneously within the MICE package. The imputed data were used to inform the base case and all subgroup and sensitivity analyses, with the exception of one sensitivity analysis, which was conducted using only complete data.
Base-case cost-effectiveness analysis
The base case took the form of an intention-to-treat analysis conducted from a UK health and social service perspective. Health outcomes were expressed in QALYs using utilities generated from the EQ-5D-3L (for feasibility study participants) and the EQ-5D-5L to EQ-5D-3L crosswalk tariff (for the main trial participants). Total costs accrued over 12 months of follow-up were calculated for each patient by summing the delivery costs of the intervention(s) received (irrespective of treatment allocation) and a sum total of follow-up costs reported at the 6- and 12-month assessment points relevant to the perspective of interest. For example, if a patient allocated to the surgery arm of the trial had PHT rather than surgery, then the treatment costs assigned would be the costs associated with delivery of the PHT intervention.
The cost of PHT was calculated by multiplying the unit cost of physiotherapy with the duration of contact (in minutes) and summed across all sessions attended. The cost of surgery was obtained from the micro-costing exercise carried out to estimate resource use and costs associated with the delivery of hip arthroscopy. Patients who had surgery were assigned treatment costs simulated from a normal distribution, with mean and variance estimates obtained from the surgery costing exercise. To avoid double counting treatment costs, self-reports of outpatient physiotherapy attendance (for treatment of lower limb problems) during follow-up were excluded from the total cost calculations for those in the PHT group (as these would have been included in the estimation of PHT costs). Similarly, self-reports of orthopaedic inpatient admissions (for the category ‘your hip/leg’) by those who had the surgery were excluded if one admission episode was reported during follow-up. When more than one orthopaedic inpatient stay was reported during follow-up, then the first admission episode was excluded in the total cost calculations and the remainder countered as repeat admissions.
Broader societal costs were also calculated (and used in sensitivity analyses) by adding to the health and social care costs, private medical costs and relevant indirect costs, such as lost income and purchase of specialised equipment.
Two seemingly unrelated normal error regressions were fitted to the data using the systems fit implementation in R. These regressions were used to simultaneously estimate incremental costs and benefits of surgery compared with PHT while accounting for correlation between the two. The regressions controlled for treatment allocation, age, sex, recruitment site, type of impingement, baseline costs (regression equation for costs only) and baseline health-related quality of life (regression equation for outcomes). The incremental cost-effectiveness ratio (ICER) was calculated by dividing the between-group difference in adjusted mean total costs by the difference in adjusted mean QALYs. The cost-effectiveness of hip arthroscopy was determined by comparing the ICER value with cost-effectiveness thresholds of £20,000 and £30,000 per QALY gained, in accordance with NICE recommendations,67 and to the recent empirical £13,000 per QALY estimate suggested by Claxton et al. 68 The incremental net (monetary) benefit of the surgery compared with PHT was calculated for a range of cost-effectiveness thresholds. Net benefit values reflect the opportunity cost of (or the benefits forgone from) adopting a new treatment when resources could be put to use elsewhere. A positive net benefit would suggest that, on average, the new treatment provides net gain compared with the alternative, and can be considered cost-effective at the given cost-effectiveness threshold.
Uncertainty around the mean cost-effectiveness estimates was characterised through a Monte Carlo method. 69 This involved simulating 1000 replicates of the ICER from a joint distribution of the incremental costs and QALYs and plotting the simulated ICERs on the cost-effectiveness plane. Cost-effectiveness acceptability curves were also plotted to give graphical displays of the probability that surgery is cost-effective across a wide range of cost-effectiveness thresholds.
Sensitivity analyses
Sensitivity analyses were conducted to investigate aspects of study design and data collection for which alternative methods existed, but where there was uncertainty regarding which method or approach was best. For example, the cost of surgery was estimated based on data from a subsample of patients who had the surgery in the study. Surgery costs could also be obtained through the HRG case-mix method. Other sensitivity analyses included broadening the perspective of the analysis to capture wider societal costs and their impact on relative cost-effectiveness of the interventions. A list of all sensitivity analyses carried out are presented in Table 3.
| Sensitivity analysis | Description of changes to base case considered in sensitivity analysis |
|---|---|
| 1 | Unadjusted analysis |
| 2 | Complete-case analysis |
| 3 | Per protocol sample 1: restricted analysis to patients who received allocated treatment |
| 4 | Per protocol sample 2: restricted analysis to patients whose surgery or PHT was deemed to be of good quality, as assessed by clinical panel |
| 5 | Altering the cost of surgery from £3042 (estimate from the micro-costing) to £2680 based on HRG code HT15Z (Minor Hip Procedures for Trauma, elective long stay) |
| 6 | Altering the cost of surgery from £3042 (estimate from the micro-costing) to £5811 based on HRG code HT12A (Very Major Hip Procedures for Trauma with CC Score 12+, elective long stay) |
| 7 | Adopting a societal perspective that includes both direct health and social care costs and broader societal costs |
| 8 | Use QALYs generated using the SF-6D utility algorithm |
Subgroup analyses
Heterogeneity in cost-effectiveness estimates was explored through the following subgroup analyses:
-
recruitment period (feasibility vs. main trial samples)
-
type of impingement (cam vs. mixed/pincer)
-
sex (female vs. male).
Longer-term modelling
Given the known limitations of within-trial economic evaluations, the study protocol had allowed for long-term economic modelling to be conducted if the within-trial economic evaluation suggested surgery to be clinically effective and likely to be a cost-effective treatment for FAI. 70 The model would have estimated the long-term (i.e. lifetime) costs and consequences of surgery and assessed whether or not any short-term benefits are sustained over the medium to long term.
Research Ethics Committee approval
The trial obtained approval from the Nation Research Ethics Committee West Midlands – Edgbaston (14/WM/0124) on 1 May 2014 and has been registered with the International Standard Randomised Controlled Trial Number ISRCTN64081839.
Trial Management Group
The TMG oversaw the study and included a multidisciplinary team of clinicians and researchers who had considerable expertise in all aspects of design, running, quality assurance and analysis of the trial. The TMG team met monthly to assess the study progress. The TMG comprised:
-
Professor Damian Griffin (chief investigator, trauma and orthopaedic surgeon)
-
Mrs Rachel Hobson (study manager)
-
Ms Jaclyn Brown (senior project manager)
-
Dr Nick Parsons (statistics)
-
Mr James Griffin (statistics)
-
Professor Stavros Petrou (health economics)
-
Mr Felix Achana (health economics)
-
Professor Nadine Foster (PHT lead)
-
Mr Peter Wall (co-applicant, orthopaedic registrar)
-
Dr Marcus Jepson (qualitative research)
-
Dr Alba Realpe (qualitative research fellow)
-
Mr Edward Dickenson (surgical research fellow)
-
Professor Charles Hutchinson (co-applicant, imaging)
-
Joanna Smith (physiotherapy research fellow)
-
Siobhan Stevens (clinical trial administrator).
Trial Steering Committee
A TSC with an independent chairperson and a ‘lay’ representative was set up. Meetings were held at regular intervals determined by need, but no less than once per year.
The remit of the TSC was to:
-
monitor and supervise the progress of the trial towards its interim and overall objectives
-
review, at regular intervals, relevant information from other sources
-
consider the recommendations of the DMC
-
inform the funding body on the progress of the trial.
The TSC comprised:
-
Professor Ashley Blom (chairperson)
-
Alan Girling (independent member)
-
Mr Richard Villar (independent member)
-
Professor Damian Griffin (chief investigator)
-
Mrs Rachel Hobson (study manager)
-
Dr Nick Parsons (statistician)
-
James Griffin (trial statistician)
-
Mr Jeremy Fry (lay representative)
-
Mr David Ralph (lay representative)
-
Mrs Ceri Jones (research network representative)
-
Mr Matthew Gane (sponsor representative).
Data Monitoring Committee
All data collected in this trial were entered into a secure trial database held at WCTU. All data collected were anonymised after the collection of baseline demographic data and all participants were given a unique trial number. Identifiable participant data were held in a locked filing cabinet and coded with a trial participant number to tag identifiable data to the outcome data. The WCTU quality assurance manager undertook audits of trial records in accordance with WCTU SOPs.
A DMC was established and comprised members who were independent of the sponsor and who did not have competing interests. The DMC reviewed trial progress, interim data and safety aspects of the trial. The DMC also reviewed the statistical analysis plan. Any recommendations were fed back to the TSC by the DMC chairperson. Outcomes were not analysed until all primary outcome data were collected.
The DMC comprised:
-
Professor Lee Shepstone (chairperson)
-
Professor Simon Donell
-
Dr Nicholas Mohtadi.
Patient and public involvement
Patients were heavily involved in the development of the trial protocol during the feasibility and pilot studies. Patient groups helped generate the patient information sheet and a patient representative sat on the TSC. Patient representatives helped to inform the trial dissemination plan, including website information, Plain English Summary, a video [URL: https://warwickorthopaedics.org/hip/2018/ukfashion.php (accessed 27 September 2021)] and a dissemination event. Additional patients were invited to attend the trial dissemination event and contributed to extensive discussion. Patients contributed to this report and are listed as authors.
Chapter 3 Qualitative research to improve recruitment and to assess outcomes
This chapter reports an integrated qualitative research intervention conducted in parallel with the main trial. The intervention was based on the Quintet recruitment intervention. 71 This research followed up the work we undertook during the pilot trial and expanded the scope to cover all participating sites in the full trial. 28
In the following sections, we report the two phases of the QRI. The first phase aimed at understanding recruitment to a trial as it happened. The second phase was the implementation of action plans developed on the basis of the first phase results. A third section of the chapter contains an evaluation of the QRI, comprising a report on the achieved recruitment rates and the results of a survey of FASHIoN recruiters after the recruitment period finished.
Understanding recruitment as it happened
Optimal procedures for site set up and recruiter training were identified at the end of the internal pilot. The TMG used these procedures to guide site teams in setting up their site for participation in the full trial. The details are described below.
Randomised controlled trial set up
We identified system features associated with successful recruitment, including:
-
having a dedicated RA in attendance at clinics
-
dividing recruitment information between the recruitment surgeon and the RA
-
ensuring that referred patients arrived with expectations of receiving treatment for FAI, not necessarily having surgery
-
running specific clinics for the target population.
During site initiation visits, the TMG and site research teams discussed how feasible these activities were at their site. Staff at most sites organised clinics that allowed RAs to be present and share the screening and approaching tasks with the recruiting surgeon. Referral letters and information about their diagnosis were modified to manage patient expectations. However, not many sites were able to host specific clinics for the target population.
Recruiter training
Analysis of pilot recruitment consultations pioneered in previous work72–76 led to the development of a six-step model (Figure 1). The objectives of the model were to improve informed consent and encourage participation in the FASHIoN trial. 3 This model guided the development of a recruiter training programme, which is explained in Action plans to promote informed consent and improve recruitment.
FIGURE 1.
Six-step model for recruitment to the FASHIoN trial.
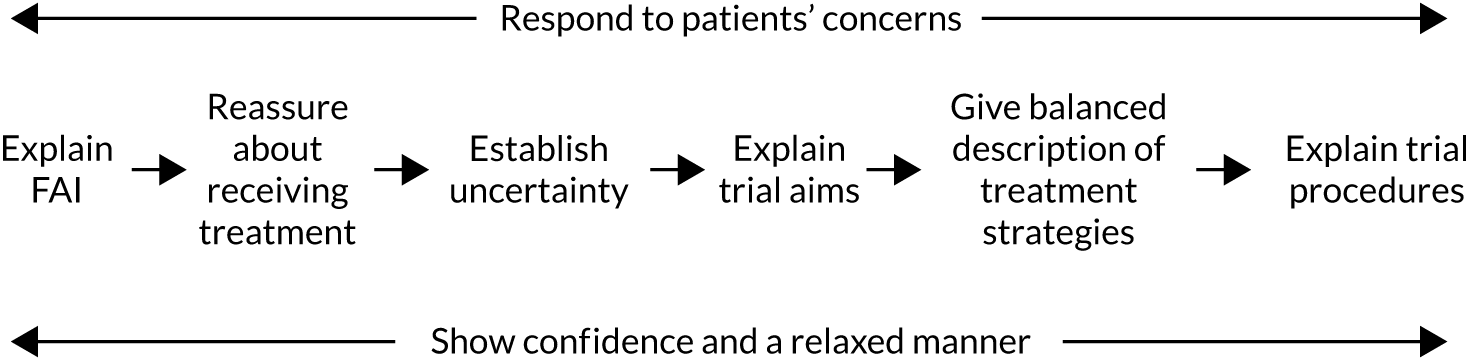
After sites started recruitment, the QRI team used various methods to describe recruitment as it happened. The QRI team contributed to monitoring and supporting recruitment by providing feedback on their findings to the chief investigator and TMG, which provided the basis for a plan of action to improve it. 71 These methods were:
-
mapping eligibility and recruitment pathways
-
interviewing clinicians and RAs responsible for recruitment
-
analysing audio-recorded recruitment appointments.
The QRI methods were not necessarily employed sequentially. The ethnographic nature of the QRI meant that the research was moulded to fit the needs of the project and was completed when theoretical saturation was reached (i.e. new data collection did not materially add to the findings). Observation of investigator meetings was carried out informally as one member of the QRI team (AR) worked closely with the TMG.
Qualitative research data were collected at 20 participating sites that recruited patients for the RCT. Two sites were excluded from this report, as they had only started recruitment 2 months before the end of the trial. In the following sections, we will report the data sets, analytic approach and results of each of the three QRI components.
Mapping of eligibility and recruitment pathways
A comprehensive logging process of potential RCT participants through screening and eligibility was put in place to ensure compliance with the CONSORT checklist. These data were made available to the TMG and qualitative researcher (AR) on a monthly basis. This information was complemented with information from weekly trial co-ordination meetings. Trial staff often shared with the QRI team information obtained during their individual contact with participant sites recruiters, such RAs, PIs or R&D personnel.
We also obtained information about recruitment pathways from in-depth, semistructured, face-to-face and telephone interviews with site research staff. Soon after recruitment started at sites, we invited PIs and RAs recruiting patients to the FASHIoN trial to an interview. We targeted new staff at pilot sites and research teams at sites new to the trial. More details about these interviews are provided in Interviews with clinicians and research associates responsible for recruitment.
Aggregated data from these sources were used to develop site-specific flow charts of the most likely patient pathways. These pathways were assessed for their complexity and compliance with the protocol-planned patient pathway (Figure 2). We aimed at identifying variations between centres, finding steps where patients could be ‘lost’ to the RCT and, when possible, working with site research teams to modify the pathway to facilitate recruitment.
FIGURE 2.
Recruitment pathway in accordance with the protocol: the FASHIoN trial.
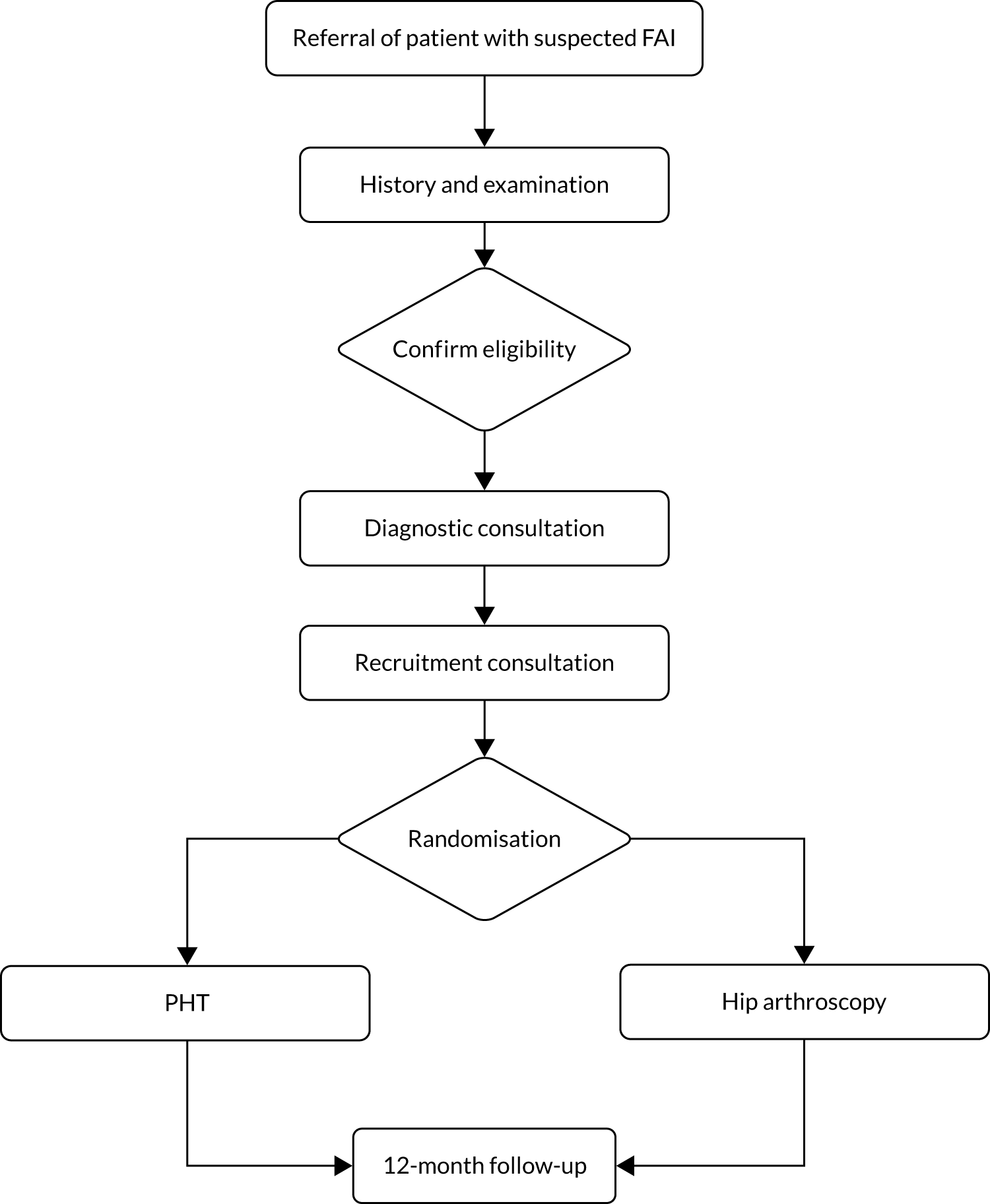
Findings
Patient pathways from eligibility to recruitment varied across participating sites, which was reflected in different numbers of patients screened, approached and consented to be trial participants. Differences between sites are expected in a pragmatic trial; however, these differences were relevant to trial recruitment success. Therefore, we decided to compare regular sites and sites that struggled to recruit. Struggling sites were defined as sites that had a recruitment rate (i.e. the number of eligible patients recruited per centre per month) smaller than the target rate of 0.5 patients recruited per month. Table 4 highlights contrasting aspects of the recruitment pathway between the two site categories.
| Site | Characteristic |
|---|---|
| Regular site | Accurate screening logs (e.g. databases included potential participants that need follow-up at a later date) |
| Follow-up of patients who had gone for further tests and procedures | |
| Collaborative decision-making process for eligibility | |
| An ‘active’ RA helping clinics with eligibility, approach and follow-up | |
| Struggling site | Multiple contacts with other clinicians before referral |
| Complex cases | |
| Inconsistent screening logs (e.g. registering only patients who were eligible or all patients in clinics independently of diagnosis) | |
| No accurate follow-up (e.g. previous patients added as new in screening logs) | |
| Surgeon-only decision-making for eligibility | |
| A ‘passive’ RA who waits for surgeons’ directions |
Patients were referred from other orthopaedic units and musculoskeletal triage systems. Most patients had a multitude of contacts with different professionals and very rarely came directly from general practitioners (GPs). In fact, struggling sites reported receiving a number of ‘complex cases’. These cases were generally referrals from other orthopaedic surgeons who had advised patients to have hip arthroscopy. This type of referral concerned recruiting surgeons, in particular, because patients had waited a long time to receive surgery, for example one PI said:
The patient’s mind has already been made up by people they’ve seen before me and generally by the nature of the condition, they’ve often seen a lot of people before they get to my clinic.
PI site 21
Adding to long waiting times, often suspected FAI patients did not have the correct imaging at the first appointment with the consultant. It was not possible for orthopaedic surgeons to confirm patients’ eligibility to participate in the FASHIoN trial and usually further tests had to be ordered. This meant that approaching patients about the trial had to be postponed until their diagnosis was confirmed. In addition, some patients had to be treated or, at least, examined for concurrent symptoms, as explained by one RA:
We have had a few [patients with] FAI but they have got trochanteric bursitis, or we have got ones with the back pain, so they first have to investigate that, and then they come back.
RA site 8
Although potential participants had been identified during screening and recorded on the screening log, some of these prospective participants were not then approached at follow-up clinics. The TMG published a list of patients waiting for FAI to be confirmed and asked RAs to find out their final diagnosis. This step in the recruitment pathway helped to identify a few eligible patients, among many others, who did not fulfil the eligibility criteria.
Other misunderstandings and errors in screening logs were made in the first 6 months of recruitment. For example, although a high-volume centre was registering every patient attending an orthopaedic clinic, independently of their diagnosis, other sites reported only patients who agreed to become FASHIoN trial participants. To avoid further errors, the screening logs were redesigned to facilitate a standard registration of eligible and approached patients.
In relation to the decision about who to approach, some regular sites’ research teams collaborated in the decision process, whereas struggling sites relied on the recruiting surgeon’s decision alone. The following quotations exemplified this contrast. The first quotation is from a site that recruited well, the second from one that was less successful:
So prior to the clinic on that Monday, I go through all the notes of the patients and have a look at their referral letters to see, because some of the referral letters, the GP or whoever else has referred them, may suggest that they suspect that there’s an impingement. So if there are any of those, I flag that up to [the surgeon] and I see if they’ve had an MRI done.
RA site 17
[The surgeon] is quite happy to discuss all the patients at the beginning of the morning to make sure we’ve found the right ones, you know, [the surgeon] checks them all through.
RA site 21
Research associates in regular sites had an active role in detecting potential participants and confirming their eligibility with recruiting surgeons. During the induction, RAs were trained to identify potential patients based on the referral letters by the trial clinical research fellow (EJD). Surgeons and RAs would look at their clinic list together and decide who to approach. Teams took ownership of the trial and often talked in collective terms about their activities, as exemplified in the quotations above. These teams created a reliable recruitment system that was repeated consistently.
In contrast, recruiting surgeons at struggling sites usually had the responsibility to check patient eligibility and introduce the study to potential participants. Surgeons would then instruct RAs to contact the potential trial participant at a later date. RAs often contacted patients by telephone. Occasionally, RAs would be contacted on the same day and would be able to talk to the patient. Sites often did not have a reliable recruitment system or a settled recruiting team. The following quotations illustrate these difficulties:
What tends to happen is [recruiting surgeon] sees them in clinic, he has a chat with them. He then e-mails me and says ‘I’ve seen Mr Smith, can you contact him? He is appropriate for the study’.
RA site 3
So he’ll e-mail me . . . He’ll introduce the study to the patients and he’s got information sheets that he passes on then I then approach the patient by phone call or letter.
RA site 14
Research associates who become proficient and confident at identifying potential participants had a pivotal role in organising recruitment. Their actions reduced the impact of contacts with other professionals (e.g. registrars) who interfered in the recruitment path:
The registrars will discuss [potential participants] with [consultant surgeon] first and I always make sure they have. I never take that for granted, because [consultant surgeon] is who has to sign the actual form.
RA site 10
Some recruiting surgeons would ask RAs to be present while they talked to patients about the trial, whereas other surgeons would spend time talking to patients alone first and then ask if they would like to talk to the RA. We recorded reluctance from surgeons to share recruitment responsibilities, as illustrated in this quotation:
I’m struggling with this trial in that you have to stop at a point to hand over to the research nurse.
PI site 11
We observed that recruiting surgeons contributed to promote patient equipoise when they worked with RAs to have a separate consultation with patients. A RA from a site that recruited consistently said:
The surgeon presents the [interventions] both as equally beneficial to the patient.
RA, site 12
Finally, often RAs at regular sites embraced the use of the six-step model enthusiastically, whereas in struggling sites the model was perceived as less useful. This is a quotation from an RA in relation to the model:
And I don’t read [the model] out word for word but it kind of makes sure you’re doing things in the right order and with the right stresses and, and going the right way. So yes, I find [the model] quite useful.
RA, site 2
Interviewing clinicians and research associates responsible for recruitment
In-depth, semistructured, face-to-face and telephone interviews were conducted with PIs and RAs recruiting patients to the FASHIoN trial. Staff at new participating sites and those new staff at pilot sites were invited to take part. Interviewees were encouraged to express their own views about the RCT and any recruitment challenges expected or experienced. The interview topic guide covered:
-
personal views about the evidence supporting the trial and their equipoise
-
the patient pathway from eligibility to recruitment
-
how interviewees feel the protocol fits their clinical settings
-
interviewees’ views on recording consultations with patients
-
any adjustments interviewees thought were needed to the trial procedures.
The recordings were transcribed and analysed thematically by Alba Realpe, using techniques of constant comparison and case study approaches. Themes were compared, looking for shared or disparate views among research team members. Coding was carried out using the qualitative data analysis software NVivo (QSR International, Warrington, UK). Detailed descriptive accounts of the themes and cases were produced. The initial coding was checked by other QRI researcher (MJ), with inconsistencies resolved by discussion. The cases were presented to the chief investigator and TMG, and were used to plan specific actions and support for participating sites.
Findings
Data interpretation focused on learning about recruitment difficulties that sites experienced, with the purpose of finding solutions in collaboration with the TMG. Reported difficulties coincided with those reported in past research,40 which was used to classify findings in three axial themes: (1) logistic difficulties, (2) recruitment experience and skills and (3) engagement with the trial.
Logistic difficulties
Sites readily reported logistic issues that had an impact on their recruitment performance. This category is related to the detailed organisation and implementation of the recruitment process at the participating site. The most frequent issues in this category are reported as follows.
Infrequent scheduled clinics
Annual leave, academic breaks, different site locations and bank holidays had an effect on the number of patients available to surgeons because clinics were held on specific days of the week (e.g. Mondays). For example, one RA said that ‘we’ve been struggling getting going because there’s been holidays’ (RA site 19). More struggling sites (n = 6) reported this issue than regular sites (n = 2).
Clinic cancellation impact on future recruitment
When clinics were cancelled, waiting lists became longer than expected and recruitment to the trial stopped. For example, in one site clinics were held without the knowledge of research support staff and in another site recruitment was postponed to stop clinics over-running.
Low staffing levels imposed pressures on principal investigators
Low staffing levels was a relevant issue for senior consultants who relied on registrars and junior doctors in teaching hospitals:
[The surgeon] has also had problems with not having registrars or fellows to help in the clinic.
RA site 8
Consequently, RA attendance to clinics became a key factor for recruitment success because they were able to take on research activities, such as screening logs and trial-specific paperwork.
Lack of young hip-only clinics
Finding eligible patients in generic orthopaedic clinics was difficult because clinics included revision surgery or other lower limb conditions. To overcome this limitation, site staff rescheduled new patients into young hip-only clinics. However, not all sites had systems that allowed such flexibility. Indeed, struggling sites had fewer young hip-only clinics than regular sites.
Lack of a private room to talk to potential participants
This issue was often reported as a major recruitment difficulty because, in accordance with the TMG recommendations and protocol, patients ideally should be approached soon after talking to their surgeon. Finding a private room to talk to patients had logistic implications for teams running busy clinics. For example, one RA described a situation in a clinic:
I had to hang around outside a room waiting for a patient to come out.
RA site 21
Another RA felt that she had to battle with colleagues and management until she was ‘able to have a room to myself and talk to patients’ (RA site 12).
Concerns with processes after trial consent
Site teams reported a few cases (n = 3) of patients deciding to opt out of the trial after randomisation. There were also difficulties with long waiting times for having the hip surgery and postoperative MRI.
Recruitment experience and skills
Recruitment experience and skills refers to the site teams’ reported barriers to recruitment derived from their experience approaching patients to this trial, such as patient responses to the trial and how to respond to them. The main issues in this category were as follows.
Uneven levels of experience conducting randomised controlled trials in orthopaedics
Having experience conducting RCTs in orthopaedics helped teams to have recruitment success in this particular trial. Seven regular site teams and one struggling site team reported having carried out RCTs before. However, of the eight research-naive teams that took part, four achieved their recruiting targets.
Principal investigator’s lack of knowledge of the non-surgical arm
Eight PIs reported having difficulties explaining PHT to patients. The PIs did not know the physiotherapy protocol and one surgeon expressed some doubts about offering this treatment:
We don’t know what a normal physiotherapy would be for this condition.
PI site 11
This issue was relevant, as five of the eight PIs were at sites that struggled to recruit.
Strong patient preferences for surgery
Site teams reported the issue of patients preferring surgery. On the one hand, struggling sites found that patients were less inclined to take part in the trial because they had already had physiotherapy or were in physical occupations (e.g. police, fire fighter). It was reported that these patients thought that they required a more invasive treatment (e.g. surgery). For example, a recruiter explains:
We get quite a lot of dancers so I’ve seen one or two dancers who’ve come and they just want to get back to dancing, and they feel that surgery will just resolve it all.
RA site 14
On the other hand, regular site teams cited patients preferring not to be in the trial because of specific personal circumstances (e.g. moving abroad) or information bias from referral surgeons. Although the issue of having previous physiotherapy was also brought up in these sites, it was often successfully challenged.
Lack of opportunities to practise recruitment skills
Recruiters reported requiring practise to develop an effective approach, but lack of, or only sporadic, potential participants made this practise difficult to maintain.
Having to consent to audio-record consultations
Recruiters mentioned being uncomfortable about asking for consent to record recruitment consultations. In part, this was because of the extra logistics involved, such as having a working audio-recorder ready, but mainly recruiters felt uncomfortable for approaching potential trial participants before they discussed their diagnosis with the treating surgeon.
Having to explain randomisation to patients
Recruiters thought that patients often found randomisation unacceptable. For some RAs, the problem was that patients preferred not to have their treatment selected at random. For other RAs, the difficulty was based on misunderstandings:
[After explaining about randomisation] the next following question is, ‘so which do I get to pick?’ [laughs].
RA site 12
Engagement with the trial
Engagement with the TMG set apart regular and struggling sites. Although most regular sites were approached to take part in the FASHIoN trial and, therefore, communication between the main site research team and site teams was established early on, other site teams submitted a request through the National Institute for Health Research portfolio and, therefore, staff at struggling sites in this group expected to receive most of the support from their local R&D group. As a consequence, their communication with the main site research team was scarce.
Overall, recruiters expressed positive views of the FASHIoN trial, independent of how well their site team recruited, as illustrated by the following quotation:
I’ve found FASHIoN, you know, a particularly nice project to work on. It seems to be very well set up and organised and a very supportive team, which has been really good.
RA site 21
Recruiters also remarked on the type of patients and their reactions to the trial:
It’s a really interesting patient group being, sort of, bit younger to what we normally look at with hips because I work on another hip trial which is mainly looking at the hip fractures . . . this has been lovely to be able to chat to a younger group of patients, sort of who are a similar age to myself as well which is always interesting.
RA site 16
Recruiters noticed patients’ positive reaction to information-sharing about their condition:
Because for some of them it’s the first time they would have had information, printed information in front of them, that really tells them about the condition. So they appreciate that.
RA site 20
Similarly, recruiters reported patients being pleased with having an extra consultation with the research nurse and an opportunity to ask further questions:
They know that we’re not rushing them; that they can ask anything.
RA site 2
Yeah, more in the men than the women and sort of then explaining why we leave it to the research nurses to discuss the trials rather than the surgeon. And they like that and they think that is a good way of doing things. I haven’t had any sort of bad comments about me coming in to discuss the trial and not [recruiting surgeon] and I haven’t had any comments made towards them saying we don’t know which works best.
RA site 16
Positive patient reactions to the trial contributed to easing recruiters’ concerns about offering this trial to patients, especially in relation to having physiotherapy instead of surgery. Clinical equipoise is discussed below.
Views on equipoise
Most recruiting surgeons expressed their agreement about the lack of strong scientific evidence in favour or against hip arthroscopy. Surgeons supported the conduct of a RCT in this area.
Being surgeons, most recruiters preferred surgical intervention for FAI syndrome, but reported that they were able to ‘suspend’ their clinical judgement. Some recruiters seemed rather comfortable offering the trial to patients:
I feel [FAI] patients don’t get enough physiotherapy.
PI site 20
I’ve got no qualms about offering [patients] [PHT] at the start of the study.
PI site 21
Other recruiters (two PIs and eight RAs) expressed concerns because they believed that those in the surgical arm were worse off:
If the physio[therapy] didn’t work you’ve always got the option of the arthroscopy, but then if you opt for the arthroscopy, OK they can say there’s still the physio afterwards but then you’ve put yourself through all that trauma.
RA site 11
Finally, other recruiters assumed a laissez-faire attitude to patient trial participation decisions:
I mean basically I have got my own electronic [list], which has got their phone numbers and address and everything on it and then as I work through they either become consented, which one lady has, or they drop out as not interested.
RA site 3
Different levels of equipoise were observed at regular and struggling sites. Most revealing was the lack of surgeon engagement in struggling sites:
I feel the consultants probably need to be more on board with it and sell it a bit more. So that’s been challenging because we’re new to research . . . I’m not usually a very pushy person but I feel that I have to do that to the patient as well as a consultant, in a nice way, you know.
RA site 14
Analysing audio-recorded recruitment appointments
The third and last component of the QRI (i.e. analysing the audio-recordings of recruitment appointments) had the following objectives:
-
to identify communication practices that encourage or hinder patient participation into the trial
-
to use this information to develop training materials and support for research teams’ recruitment efforts.
During the pilot, we hypothesised that using a six-step model of recruitment encouraged participation in this trial. Therefore, the six-step model was the benchmark for the analysis of communication practices in the full trial. We assessed whether or not recruiters used the six-step on their recruitment consultations and, if so, whether or not this use influenced conversion of patients into trial participants.
Procedure
Participating sites were asked to produce as many recordings as possible of two pivotal appointments in the recruitment to this trial. The first appointment to be recorded, named the ‘diagnostic consultation’, was the appointment when patients were told about their diagnosis. We advised recruiting surgeons to present the trial using the first four steps of the recruitment model (i.e. explain the condition, reassure about receiving treatment, introduce uncertainty and explain study purpose), to answer patient questions and to invite patients to talk to RAs about the trial.
The second appointment was the ‘recruitment consultation’, defined as the appointment at which randomisation was discussed. The training of RAs emphasised using the six-step model to structure information given to patients about the trial during the recruitment appointment.
The TMG explained and promoted the QRI at site initiation visits. All materials for recording consultations (i.e. patient information sheets, consent forms for audio-recording, digital audio recorders and instructions for the operation of the recorder and the naming and transferring of data to the QRI team) were given to a main contact within the site team, generally the RA assigned to the trial.
Prior to their consultation with the surgeon, RAs gave patients who could potentially become trial participants at clinics the patient information sheet for the audio-recording of consultations to read. If patients agreed to become trial participants, they were asked to sign a consent form. RAs then proceeded to record the following appointments as per protocol. The recordings were then anonymised and sent securely to the QRI researcher for analysis.
A coding manual was developed and applied directly to the audio-recordings (not transcripts) using NVivo software. After the analysis, the QRI researchers decided what confidential feedback would be given to the recruiters. Teams were assured that the feedback to them was going to be confidential and positive (not critical). Other issues were fed back to the RCT chief investigator/TMG, or were used anonymously in training programmes.
Analytic approach
The sample was purposively selected to include patients who agreed and did not agree to take part in the FASHIoN trial. Good-quality recordings of recruitment consultations (and diagnostic consultations, when available) were analysed with a modified version of the Quanti-Qualitative Appointment Timing method that has been pioneered in previous studies. 75
Using the Quanti-Qualitative Appointment Timing approach, we determined the percentage of time recruiters spent in each of the six steps of the recruitment model and the sequence in which they were used by recording when a recruiter used a step for the first time. We then evaluated appointment content for unbalances and untoward emphases that may upset patient equipoise using focused conversation analysis. In addition, patient questions, concerns and preferences were coded and frequencies tabulated.
Two coders tested the reliability of the coding system (i.e. inter-rater reliability). Consultations of six individual patients were coded and Cohen’s kappas calculated. After reliability checks were satisfactory, we applied the coding to the remaining consultations.
Findings
We received 153 audio-recordings corresponding to 129 individual patients from 11 sites between September 2014 and March 2016. Table 5 contains the characteristics of the audio-recordings, patients and clinicians in this sample. Patient and recruiter characteristics did not differ from those of the trial participants as a whole.
| Audio-recording characteristic | Total |
|---|---|
| Collected, n | 153 |
| Selected for analysis, n | 107 |
| Excluded, n | 46 |
| Diagnostic and recruitment consultation pairs, n | 34 |
| Recruitment consultation by surgeon, n | 13 |
| Recruitment consultation by RA, n | 26 |
| Individual patients, n | 78 |
| Males, n | 42 |
| Females, n | 36 |
| Average age (years) | 34.8 |
| Individual recruiters, n | |
| RAs (nursing or physiotherapy background) | 18 |
| Orthopaedic surgeons | 12 |
| Average number of recordings submitted | 3 |
Recordings selected for analysis comprised a combination of patients who agreed to take part in the trial (n = 44) and those who declined (n = 29). The average length of recruitment consultations was 16 minutes 50 seconds and the average length of diagnostic consultation was 7 minutes 57 seconds. The minimum number of recordings per site was two and the maximum 22. Other recordings were excluded because they were incomplete (n = 17), patients in the recordings were not eligible (n = 12), patient diagnosis was uncertain (n = 8) or the recording included more than one recruiter (n = 9).
Coding reliability
The QRI researcher and an independent qualitative researcher achieved substantial agreement (κ = 0.67) in the coding of a subsample of six consultations. Both the QRI researcher and the independent qualitative reached almost perfect agreement when identifying statements explaining details of hip arthroscopy, PHT and randomisation (κ > 0.8). Coders disagreed more often (κ < 0.4) when identifying statements of reassurance (step 2) and uncertainty (step 3), in part, because reassurance was seldom used and uncertainty often appeared paired with study purpose (step 4).
Diagnostic consultations
We received 34 diagnostic consultations and in 22 of these consultations patients became trial participants. The remaining 12 participants declined participation. We observed two different approaches to diagnostic consultations. Some surgeons spent time going through trial information with patients, whereas others made a brief intervention and handed over the potential participant to the RAs.
Figure 3 shows the distribution of the time spent on different elements of the consultation, comparing consultations in which the patient agreed to participate with those in which they did not. Recruiting surgeons spent more time framing the decision using the first four steps of the model in trial participant consultations than in non-participant consultations. This was also the case in relation to answering patient questions and providing information about PHT, hip arthroscopy and RCT procedures. These recruiters spent less time dealing with patient concerns and preferences than their colleagues in non-participant consultations.
FIGURE 3.
Percentage of time dedicated to specific recruitment tasks in diagnostic consultations.
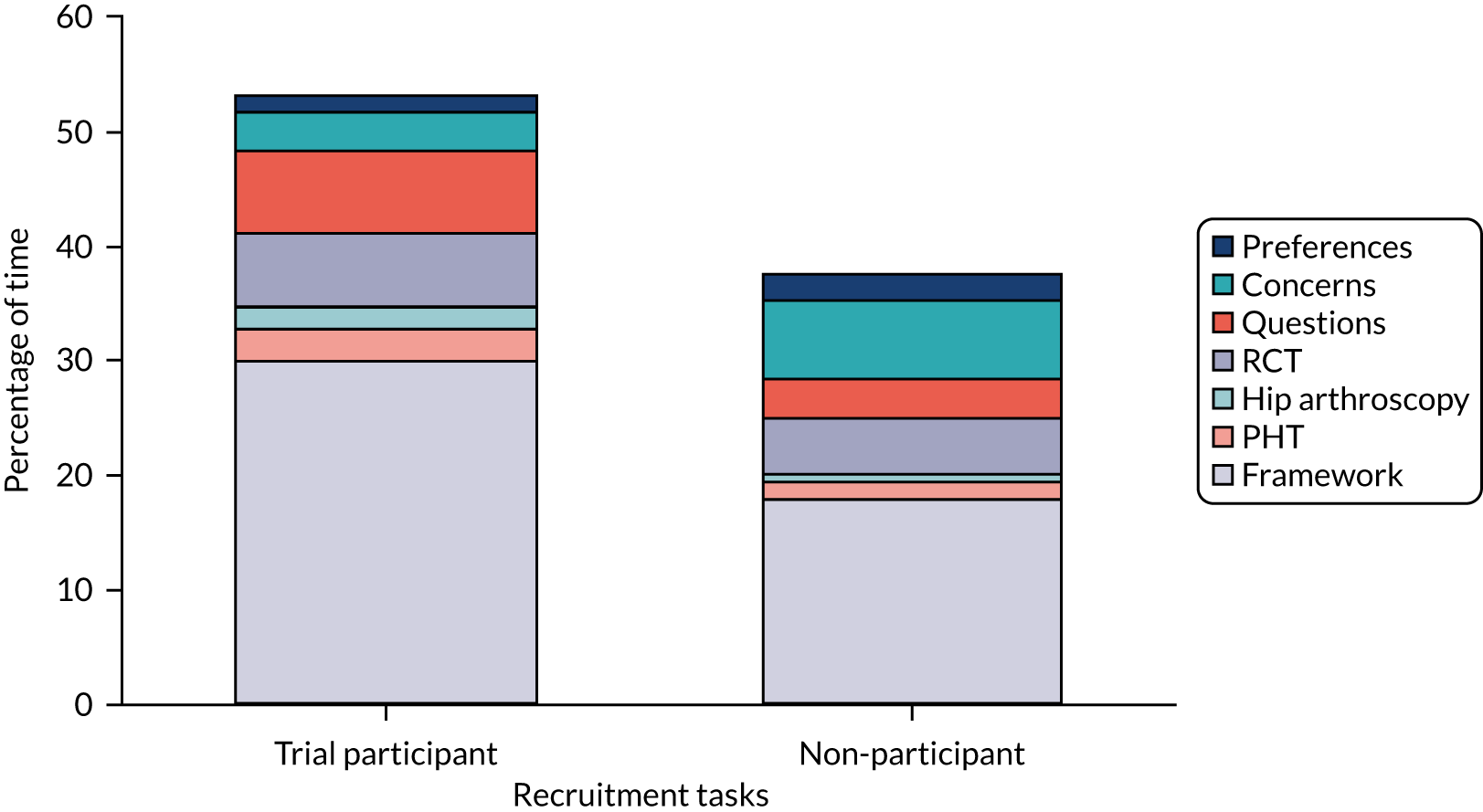
In relation to the sequence in which information was given, we observed that surgeons used the first four steps before explaining study procedures in 16 consultations and in 10 of these of consultations patients became trial participants. When the sequence was reversed, four out of five patients took part in the study. Surgeons did not mention randomisation in 13 consultations and in eight of these consultations patients became trial participants.
These results suggested that surgeons managed to convert patients into participants more often when they framed the decision of trial participation, answered patient questions about their medical condition and treatment alternatives, and did not go into much detail about study procedures or randomisation.
Recruitment consultations
Our results indicate that the majority of recruiters used the model to structure their consultations, as per training. On average, 72.8% (SD 16.5%) of the consultation time was spent introducing information based on the six-step model. The remaining time was spent taking medical history, giving instructions and chit-chat.
Figure 4 shows time distributions of recruitment consultations. More time was spent talking about study procedures and the non-surgical arm in trial participant consultations than in non-participant consultations. Furthermore, patients appeared to have fewer questions, concerns and preferences in the former than in the latter.
FIGURE 4.
Percentage of time dedicated to specific recruitment tasks in recruitment consultations.
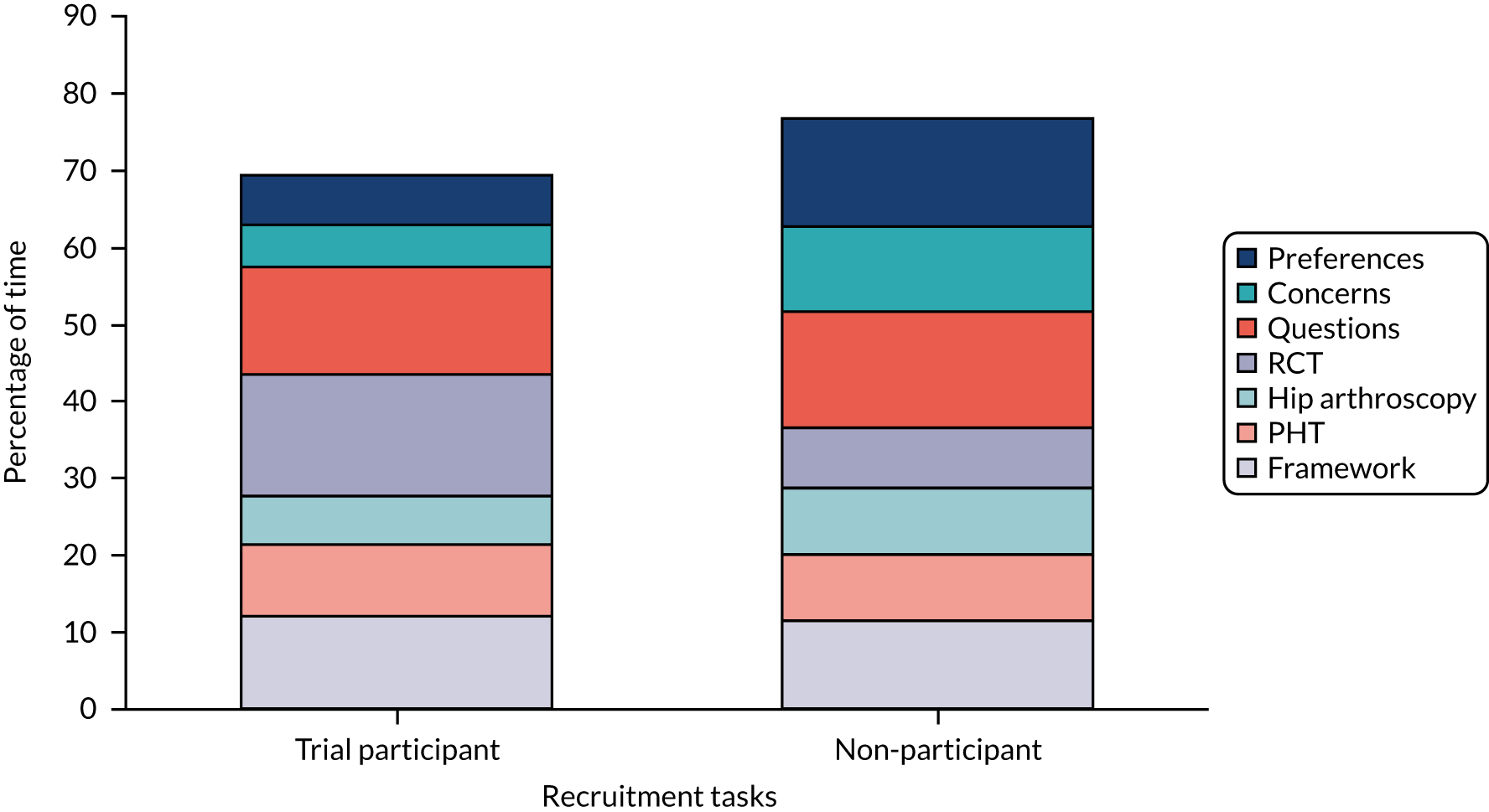
Most recruiters (n = 47, 64%) used the information delivery sequence suggested by the model. Recruiters framed patient decisions using the first four steps of the model and then proceeded to introduce information about randomisation. Recruiters did not use this framework in 20 cases (27%). However, independent of the chosen sequence, 50% of approached patients became trial participants.
In relation to the content of recruitment consultations, we observed little variation on topics because recruiters were provided with extensive training. The content of consultations became highly standardised in a script available to recruiters in cue cards (see Training site teams on recruitment strategies and trial-specific information).
Our observations and recruiters’ reports indicated that recruitment to this trial depended on two pivotal aspects of recruitment: (1) recruiters needed to make an argument in favour of PHT and (2) recruiters needed to describe randomisation in a way that was acceptable to potential participants.
Making an argument in favour of PHT was especially challenging when patients have had previous hip-specific physiotherapy. Recruiters succeeded at promoting patient equipoise when they knew details about PHT and were equipped to answer patient questions about this treatment arm. Recruiters used their knowledge to dispel misconceptions or calibrate treatment expectations by given details about how a treatment may affect the person at an individual level.
The following extract is an example of how this strategy worked. The recruiter offers an interpretation of patient preference and confronts this assumption based on her own clinical experience, which the patient accepts:
Yeah and it’s funny maybe I don’t get that when I – and I find like picking up my son if I go like that hhh I don’t have any power, I don’t have any – is that sort of thing
[Inaudible]
But I think . . .
The thing is, if you just must think, because if you think about the surgery as mechanical . . . just taking the bump away and then it’s away . . . but I also work in the clinic follow-up of people post surgery
Hmmm
And I did find that some of the high-level sports people it’s taking us a year to a year and a half to actually get them – and some people you don’t, some people are always off, so you do get worse off so ehm . . . after surgery it’s not all just straightforward
No, no, I understand.
Contrast between previous physiotherapy and the new PHT arm could not always be achieved, either because recruiters used statements that were too general or irrelevant to patient needs, or simply because they did not know how different these approaches really were. The following extract exemplifies this type of exchange. The patient declares that he has already had physiotherapy and the recruiter attempts an open question but changes it for a closed one. The patient rejects the assumption and emphasises the difference. The recruiter moves to explain general aspects of PHT, but the patient rejects this. The recruiter persists but gives up at the end:
I[ve already said] I had months of physio already though
Ahh yeah What kind of phys[io] is that through your GP?
No it was through the army
OK
Private physio
Right ahh . . . So what the physio is doing ahh is . . . the physio is
a personalised hip therapy programme so you’d come to the hospital and see Mr [surgeon]’s specialist physio who deals with just hips, nothing else ehm which is where you get the personalised structure from ahh and what she would do with you is work with you to build up your muscles ehm and your hip [flexes]
I’ve already been doing [that]
And everything around . . . yeah ehm look at how ehm where your pelvis is positioned? So if you tilt your pelvis slightly forward, slightly back to get you in a good postural positioning [laughs] I’m preaching to a convert here, ain’t I?
Describing randomisation in a way that was acceptable to patients often challenged recruiters’ equipoise. At times, conversations became awkward, hindering recruitment. The following extract illustrates this effect:
I’d still do your study but I go the surgical route
That’s a – that’s another proble(h)m ehmm . . . to make it a fair test ahh . . . we . . . randomly allocated . . . your . . . option . . . so I don’t get a say in what you get, you don’t get a say and Mr [surgeon] doesn’t get a say
[Sniggers]
So leave it to chance of which option you’d be given
I can’t do that because I want to go back
I know
I’m in [timescale]
Have they said what sort of, what sort of
6 months to a year from when I get my discharge papers
That’s your window of getting back in?
No that’s how long I’ve gotta wait 6 months to a year
OK
Before I can reapply
OK
So I want to get my fitness back.
Laughter in the patient response can be interpreted as responding to the awkward discussion about the surgeon not having a say, which the recruiter framed as ‘another problem’. The patient continues to be concerned about the timescale of their treatment and uses this reason to opt out of the trial.
Finally, we noticed that step 2 of the model (i.e. reassurance about receiving treatment) was seldom used by recruiters. Only 23 of the 73 recordings contained reassuring-type statements and most of the patients from these recordings became participants (74%). Recruiters expressed concerns about using reassuring-type statements, as they may have been construed as undue influence or coercion. Nevertheless, it can be argued these statements were ethically valid because the FASHIoN trial was a superiority trial and, therefore, it was assumed that both treatments were effective. An example of a reassuring statement is as follows:
[Surgeons] know that both [treatments] work, treatment, trial or whatever, you’re going to get treatment anyway, so physio and surgery, they both work.
RA site 4
Patient questions, concerns and preferences
Box 1 shows a sample of the list of common patient questions, expressions of concern and preferences that we collected during our analysis. Patient questions were more often recorded in trial participant consultations, whereas there were more expressions of concerns and preferences in non-participant consultations. This trend was expected, as patients in the latter group may not have been in equipoise.
How did I get FAI?
Could it get much worse if left untreated?
How long does it take to recover from hip arthroscopy?
How many times would I have to come here [for PHT]?
How does treatment get decided?
Patient concerns
I’m confused, the other doctor showed me a tear.
I’m just in absolute agony at all times.
If it is my abnormal hip shape, what difference physio is going to make?
I don’t like the idea of the computer choosing which way.
HR is working to possibly getting me back to work because I don’t get sick pay.
Patient preferences
I’m intrigued by both [treatments] really.
I need to be in control of this [decision].
I’m sick of having [physio]therapy.
Physiotherapy is more suitable for me right now.
HR, human resources.
Our observations of the recruitment process as it happened informed decisions about what support and training the QRI team and TMG could offer to site teams. These activities are described below.
Action plans to promote informed consent and improve recruitment
The QRI team worked closely with the FASHIoN team, creating action plans as soon as information from Phase I was available. The plans were implemented by the trial team and the QRI researcher assigned to the trial. This method is consistent with the QRI described by Donovan et al. 71
The results of Phase I indicated that sites that recruited well and those that struggled confronted similar challenges and, therefore, the trial team sought to standardise procedures as much as possible. Action plans focused on the following three objectives:
-
improving screening of eligible patients
-
facilitating recruitment site teams’ participation and engagement
-
training site teams on recruitment strategies and trial-specific information.
Improving screening of eligible patients
A simplified screening form was introduced to correct screening misunderstandings and to obtain accurate estimates of patient recruitment. RAs received detailed instructions on how to fill out the new screening logs.
Research associates were asked to submit screening data and upcoming recruitment clinic dates on monthly basis, including whether or not a RA would be available to attend clinics. This information was reported during the monthly TMG meetings.
Reports to the TMG included an approach percentage and a conversion percentage for each site. The approach percentage was the percentage of eligible patients who had been asked to take part in the trial. The conversion percentage was based on the number of approached patients who became trial participants. These two percentages allowed the trial team to identify where potential problems in the flow of patients may be. For example, two sites recruited 0.6 patients per month; however, the sites differed on their approach and conversion percentages. One site had a low approach percentage because patients travelled from Ireland to receive treatment and, therefore, these patients were ineligible to participate in the trial. The other site had a nearly 100% approach, but it did not convert patients to participants. There was not much that could be done with the composition of clinics in the first site, but the second team was offered further training on talking to patients about the trial.
The majority of participating site teams responded positively to these changes and RAs developed their own systems to report their recruitment numbers regularly. Screened patients almost doubled between September and January (n = 352) and the following 4 months after these changes were implemented (n = 681). The number of approaches and recruitment conversions increased slightly from 27 to 30 at the same sites.
Facilitating recruiting sites’ participation and engagement
Information collected through interviews and screening logs for each site was organised into cases. Each case detailed team structure and number of patients screened, eligible, approached and recruited, as well as a description of any particular difficulties at logistic, engagement or skill level. Cases were discussed with the chief investigator and TMG in weekly meetings. A summary was presented and strategies to address particular issues discussed during the TMG monthly meetings.
Logistics/organisational difficulties
-
Contact R&D group to guarantee time allocation for a research assistant to attend clinics regularly.
-
Request screening logs and clinic dates monthly from sites through an Microsoft Excel® datasheet (Microsoft Corporation, Redmond, WA, USA).
-
Provide further training on eligibility criteria.
-
Alert PIs and other management personnel about delays in setting up a site.
-
Deploy clinical fellows to cover clinics when RAs are not available.
Engagement with the trial
-
Regular contact between trial co-ordinator and RAs.
-
Regular telephone contact with PIs led by the clinical research fellow and the chief investigator.
-
Monthly e-mail newsletters with recruitment rates and other news about the trial.
-
Token incentives for successful recruiters, such as chocolate boxes and coffee vouchers.
-
Branded stationery with trial logo [e.g. water bottles, pens, Post-it® slips (3M, Saint Paul, MN, USA) and mugs].
Generic and trial-specific recruitment skill level was an important aspect that required optimisation across sites. Improving recruiters’ skills became a main target of the recruitment intervention, as explained in the next section.
Training site teams on recruitment strategies and trial-specific information
A comprehensive training programme was designed to support site teams in optimising their recruitment opportunities. Different activities were organised from the start to the end of the recruitment period. These activities responded to observations of common barriers and good practice across sites. A list of activities in chronological order is presented below.
Recruiters’ workshop
At the beginning of the full trial, a half-day training event was offered to PIs and RAs from the 13 sites that were opened to recruit. The workshop aim was to equip site teams with knowledge and techniques that they could adapt to recruitment processes at their sites. The CI and trial team presented key aspects of conducting the trial, including a detailed description of the protocol, eligibility criteria, treatment specifics and study procedures. The QRI team taught the six-step model and illustrated how to deal with common issues using case examples. Research teams had the opportunity to ask questions and discuss issues raised. Trial teams received the presentation materials and a manual that contained examples of good recruitment practice collected during the pilot trial.
Six-step model manual
A document explaining the six-step model for recruitment to the FASHIoN trial was distributed with the study file. The document contained a description of each step and provided examples extracted from the pilot recordings modelling good practice. The manual was an abbreviated version of the published article that RAs could consult for easy recall. 28
Training the trainer
The clinical research fellow of the study (EJD), who was an orthopaedic surgeon in training, was responsible for delivering RA training on this specific trial. The QRI team worked closely with the clinical fellow, who received specific and intensive training at the beginning of the trial. His recruitment consultations were recorded and analysed, and feedback was discussed at these early stages. The clinical fellow also shadowed the chief investigator when approaching potential participants at clinics in the main site. The clinical fellow became an accomplished recruiter and was involved in all training events that were conducted during the full trial. He offered support through telephone and e-mail contact to RAs at participating sites.
One-to-one training
The clinical research fellow met in person with RAs at either the first site recruitment clinic or soon after. The purpose of this meeting was to discuss the recruitment strategy and build confidence in approaching patients. RAs had the opportunity to shadow the trainer or receive instant feedback from his observations. The clinical fellow and RA would discuss the strengths and weakness of the approach and further training contacts were planned, if required. When a face-to-face meeting could not be arranged, the clinical research fellow offered to role play over the telephone before the RA started recruitment.
Feedback on audio-recordings
Research associates who submitted recordings received individual written feedback from the QRI researcher. The feedback focused on issues related to phrases or vocabulary that could hinder recruitment. Discussions between RAs and researchers were positive, building on strengths, addressing misunderstanding and avoiding criticisms. This feedback offer was optional and not every RA received it.
Teleconferences
Two 1-hour teleconferences with RAs were conducted at the mid-point in the recruitment period. The calls aimed at increasing awareness of common recruitment issues and problem-solving by sharing experiences with each other. In the first teleconference, common patient questions and preference statements found on the QRI analysis of audio-recordings were used to facilitate discussion. The second teleconference focused on increasing RAs’ knowledge of the trial treatments and, therefore, their ability to answer questions. Most of the RAs attended these teleconferences.
Teleconference summary leaflet
After the first teleconference, a guide was distributed to sites, which contained the most common patient questions, concerns and preference statements and suggestions to address them.
Newsletter supplement
A supplement to the newsletter, containing a summary of the main characteristics of the trial treatments, was distributed to teams following the second teleconference. The content was created and checked by the clinical team running the trial.
Note cards
Pocket-sized note cards containing key trial information messages organised according to the six-step model of recruitment were created. These cards were the result of the extensive analysis of recruitment consultations and were introduced as a memory aid for RAs. The note cards were distributed in the last few months of recruitment.
Evaluation of the qualitative recruitment intervention
The evaluation of the QRI comprised two data sets. The first data set was the overall trajectory of recruitment rates after action plans from the QRI were implemented. The second data set was the results of a survey of the RAs who participated in recruiter training activities. The survey asked RAs what work well in the trial and what could have been improved for future trials.
Recruitment rates overview
The FASHIoN RCT aimed to recruit 344 participants. Recruitment was planned to take place across 25 centres over a 20-month period. Centres would be open and recruiting 6 months after recruitment started. Forty-two patients had already been recruited in the internal pilot. The main RCT, therefore, needed to recruit an additional 302 patients.
Recruitment targets were set, assuming centres would comply with this schedule. Figure 5 shows the original recruitment target line. However, there were considerable delays in opening centres, which had an impact on recruitment targets. An amended recruitment target line was estimated based on the actual numbers of centres open per month for the main trial. The recruitment target of 302 patients was achieved in 22 months and included 23 centres.
FIGURE 5.
Target vs. actual cumulative recruitment: the FASHIoN trial.
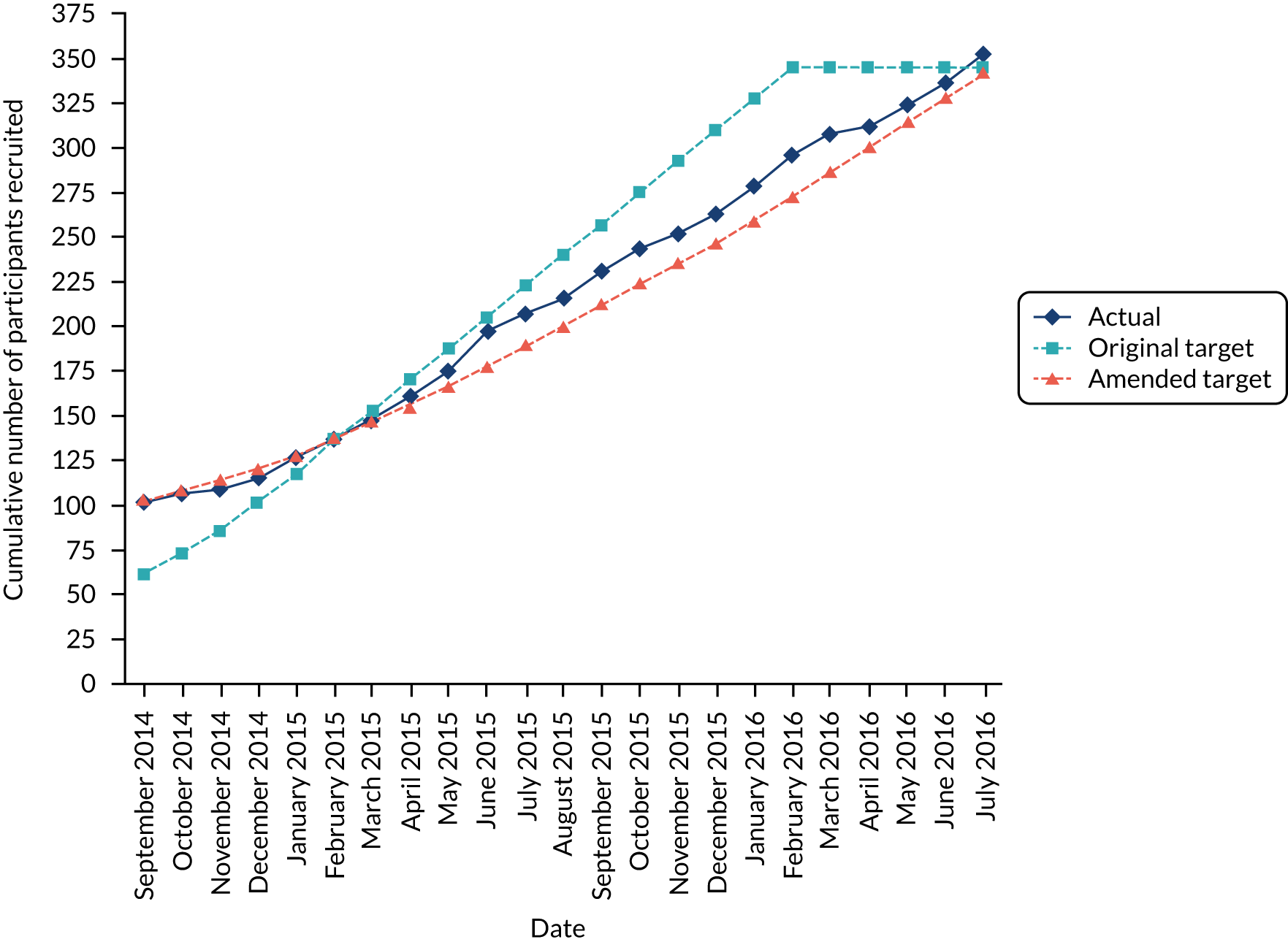
Recruitment figures roughly followed the amended target line until March 2015, after which the actual recruitment figures started to exceed the amended target. Recruitment rates steadily remained above the amended target for the remainder of the recruitment period.
Research associates’ survey on recruiter training activities
We conducted an online survey of FASHIoN trial RAs at the end of the recruitment period. The main aim of this survey was to gather views of a diverse group of recruiters about the strengths and weakness of recruitment training and trial conduct. We also sought advice about how to improve the design of future orthopaedic trials.
Procedure
We sent an invitation with a link to an online survey to the e-mail addresses of all RAs in the FASHIoN site delegation logs. Questions focused on three aspects of the FASHIoN trial:
-
recruitment training activities
-
study procedures (e.g. trial manager visits and attending clinics)
-
study documentation (e.g. trial file, patient information sheet and CRFs).
Research associates scored each trial aspect for its usefulness or quality on a five-point Likert scale. RAs were also asked to comment on what they would keep or remove from the FASHIoN trial activities. RAs compared the FASHIoN trial with other surgical and orthopaedic trials that they were working on and, finally, they provided details of role and experience. We sent the questionnaire 4 weeks after the recruitment period had finished. The survey ran for 6 weeks and two reminders were sent to those who had not replied.
Answers were aggregated and presented in tables organised by the online platform. A summary of the main findings is presented below.
Findings
We sent the invitation to 38 individual e-mails and received replies from 27 RAs: the response rate was 71%. Non-respondents were mainly staff who had a brief involvement with the trial, had covered annual leave, were RA managers or had left their role. We are confident that those who replied were directly involved in talking to potential participants in this trial.
The majority of RAs had a clinical background (only three RAs did not). There were 18 nurses and five physiotherapists. Experience as a recruiter to research studies varied. Forty-eight per cent (n = 13) of recruiters had < 2 years in the role, whereas others had been recruiting for up to 10 years. Seven of the most experienced recruiters were senior research nurses and managed other research nurses. More than half (66%) of recruiters had recruited for other orthopaedic trials and spent most of their working time on this role. RAs were involved with a range of studies (from 2 to > 20). Eight (29%) recruiters were working on the FASHIoN trial exclusively.
Recruitment training activities
We asked recruiters how useful they found each of the seven training activities described in Phase II. We obtained many ‘not applicable’ responses recorded in activities that happened once. The results are as follows.
One-to-one training
One-to-one training refers to meetings between the clinical research fellow and RAs at the first recruitment visit. Seventy-seven per cent of RAs rated this training as overwhelmingly useful and only six RAs said that it was not applicable. In the comments, one RA wrote:
It is good to receive one-to-one training. It is also good to hear from a clinician rather than someone with an admin background, as they understand the interventions more.
Six-step model manual
The six-step model manual provided details of the six-step model to good recruitment practices. Eighty-one per cent of RAs found the manual useful or very useful, three RAs said that it was only moderately useful and two RAs said that it was not applicable.
Recruiters’ workshop
The recruiters’ workshop was a half-day training event that was offered at the beginning of the recruitment period. Forty-seven per cent of RAs considered the workshop useful, but 53% of RAs said that it was not applicable.
Feedback on audio-recordings
Individual feedback was offered to RAs who submitted audio-recordings. Thirty-eight per cent of RAs did not receive this type of feedback. Views on how useful this activity was varied from not useful at all (7%) to extremely useful (19%).
Teleconferences
Two teleconferences were conducted at the mid-point of the recruitment period. The teleconferences were considered as not applicable to nine (35%) RAs in this sample. Eleven (44%) RAs found the teleconferences very or extremely useful and six (22%) RAs did not find them useful.
Teleconference summary leaflet
A summary leaflet of the first conference was distributed to sites. RA views of the leaflet mirrored their views of the two teleconferences (see Teleconferences). Twelve (46%) RAs thought that the leaflet was very or extremely useful, seven RAs (25%) thought that the leaflet was not useful and seven RAs (25%) marked it as not applicable.
Note cards
Note cards, containing key trial information, were introduced in the last few months of the trial as a result of extensive analysis of audio-recordings. Consequently, the note cards were not applicable to five (18%) RAs. All RAs who received the note cards found the cards useful, with 70% of RAs saying that they found the cards very or extremely useful and three (11%) RAs saying that they found the cards moderately useful.
Study procedures
In relation to how difficult or easy the study procedures were, the survey covered all aspects, from setting up the trial to follow-up procedures. Procedures catalogued as easy or neutral included communicating with the TMG, working with the local PI, completing CRFs and obtaining clinical data about participants. A small number of RAs reported difficulty in setting up the study, attending clinics, identifying eligible patients, approaching eligible patients and obtaining informed consent.
Two aspects of the study procedures that were rated as difficult by RAs were (1) having a separate recruitment consultation after the diagnostic consultation (n = 7, 28%) and (2) recording consultations (n = 7, 28%). Notably, the RAs said that these difficulties differed from other trials in which they had been involved. However, overall, RAs were satisfied with the study procedures.
Research associates were very satisfied with the trial management support, specifically in relation to dealing with treatment-specific queries, general trial queries and telephone and e-mail contact with the trial team. A RA made the following comment:
The trial management team were excellent in this study. Easily accessible and very helpful and professional.
Study documentation
Research associates’ ratings of study documentation (including information about FAI and treatments, the trial site file, the patient information sheet, screening logs, CRFs, the trial manual and the newsletter) ranged from good to excellent in all questions. A comment from one RA reads:
Having worked on many studies now, I appreciate how hard is to design and produce such high quality of documentation.
When we asked RAs to compare FASHIoN trial procedures and documentation with that of other trials they had been working on, ratings ranged from good to excellent. We noticed a few not applicable responses from RAs who had not worked in other trials.
Overall, RAs reported satisfaction with study procedures and documentation, as well as with the range of recruitment training activities. The trial was well resourced in terms of staff and budget, and this allowed consistent communication between the trial and site teams and resulted in highly professional documentation.
Emphasis on RAs talking to patients in a separate recruitment appointment and the use of a recruiter training programme made the FASHIoN trial somewhat different from other orthopaedic trials. It is not surprising that RAs found these extra activities more challenging than other common trial procedures. For example, there were suggestions to remove recordings from future trials, as RAs found recordings awkward and feedback was inconsistent and may have felt critical at times. However, RAs appreciated the note cards, which could not have been created without data from these recordings. Another strength of our approach was having a variety of activities for recruiting staff who usually have a high turnover. This meant that recruiters were well supported, as evidenced in their feedback.
Discussion and conclusions
We reported various processes aimed at optimising recruitment to the FASHIoN trial. The first phase of the recruitment intervention provided an understanding of the reasons for recruitment rate differences between sites that recruited well and sites that struggled. Equipped with this knowledge, the TMG and QRI teams modified or created processes to support sites in improving or maintaining their recruitment rates (e.g. monthly monitoring of screening logs and extensive RA training). Recruitment rates were maintained and the recruitment target sample was achieved, albeit a few months later than estimated. In the remainder of this discussion, we will discuss some limitations of these findings, then we will consider the relationship between this work and prior QRIs.
There are two limiting conditions to the conclusions that we can draw from our findings. First, the recruitment intervention was applied to only one specific RCT dealing with one specific patient group. Therefore, action plans may be applicable to this trial only, for example whether or not the six-step model can be adapted to other RCTs remains an open question. Second, we obtained a large number of audio-recordings of consultations; however, most sites perceived these recordings as a barrier to conducting the trial, which was more prominent on sites that struggled to recruit. It is possible that our data collection had an embedded bias towards positive results because of hard-to-reach struggling sites. Efforts to overcome this shortcoming were made by including audio-recordings as an integral part of the recruitment process at the site initiation.
Findings from the FASHIoN trial QRI did not differ from other QRIs reported in the literature. 40,76 For example, our classification of the main recruitment issues reported in Phase I reflected those found in the qualitative research synthesis of Donovan et al. 40 Recruiters readily identified organisational difficulties and patients’ preferences as key barriers to recruitment, but hidden issues emerged in relation to how comfortable recruiters were recruiting to this particular trial and their different levels of equipoise. Another expected finding echoed previous research that focused on the importance of research teams working together. 76 Our view is that the TMG imparted strong messages about how to recruit, but good communication and relationships within the research teams made these messages bear fruit in terms of achieving recruitment targets.
Perhaps the most innovative aspects of this QRI were the recruitment training plan and the final survey evaluation. The six-step model provided a guide to create a comprehensive training programme and additional resources to address lack of recruitment experience and skills. We found evidence that the majority of recruiters used a structured model of recruitment in their conversation with patients and through the survey it was possible to discern which resources RAs valued the most.
More generally, this report shows that recruitment interventions tend to be multilayered and complex, and, as in the FASHIoN trial, interventions required the full support of, and commitment from, the TMG and participating site teams to be successful. What stands out in this work is how feasible it is to not only construct a well-crafted message promoting patient equipoise, but also to get a large number of researchers to use this message in their recruitment consultations. The effort of understanding and intervening on recruitment is worthwhile when answering important clinical questions.
Chapter 4 Results
Screening
A total of 6028 patients attending the participating surgeons’ hip clinics were screened between 20 July 2012 and 15 July 2016 (Figure 6).
FIGURE 6.
The FASHIoN study CONSORT flow diagram.
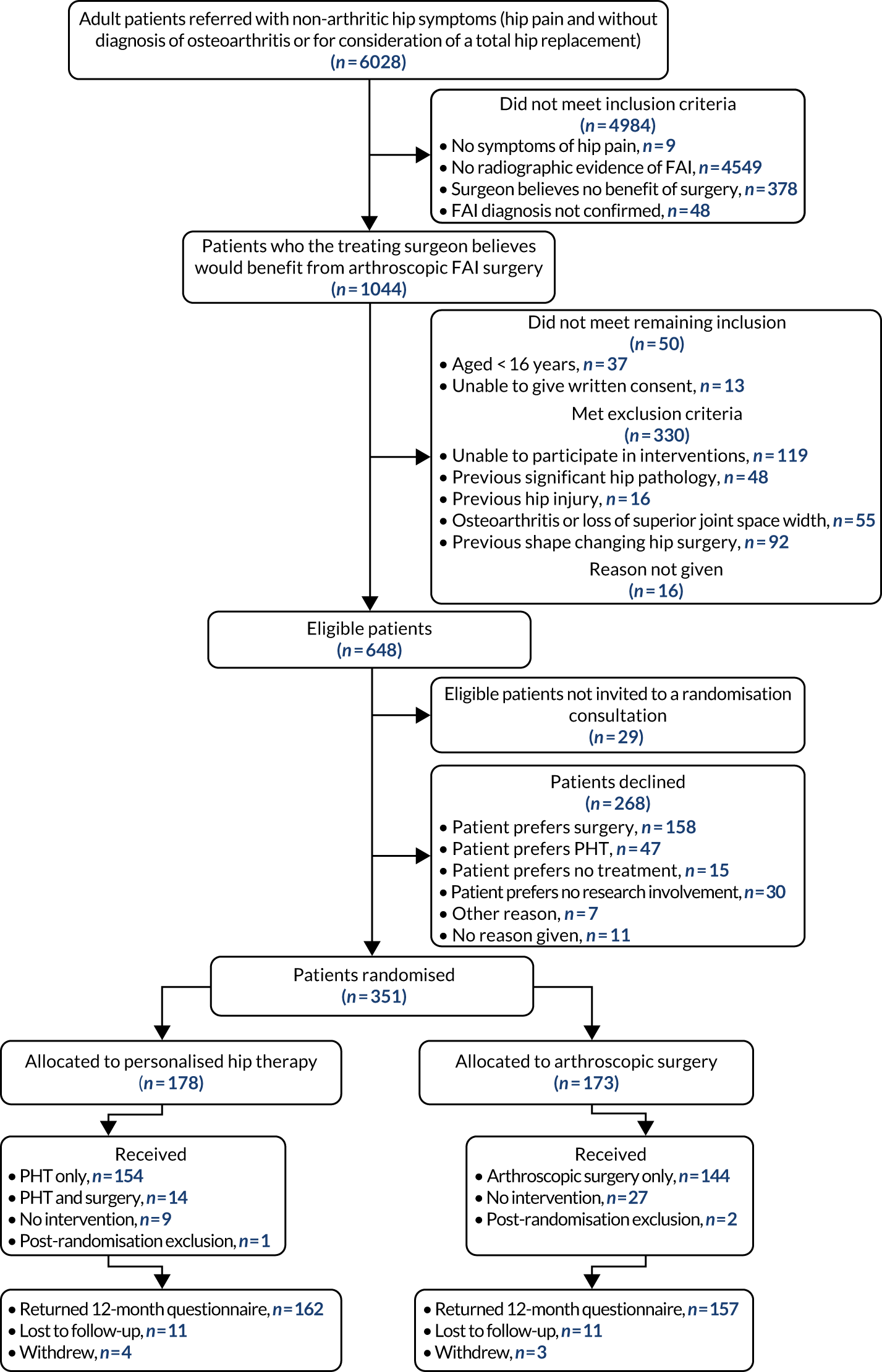
Recruitment
The number of patients approached and recruited by site is shown in Table 6. Overall, 94% of eligible participants were approached, 61% of whom consented to participate in the trial. In total, 351 participants were recruited. There were three participants who were randomised; however, post randomisation, these participants were found to not meet the inclusion criteria and were excluded and withdrawn from the study (referred to as post-randomisation exclusions). For the remainder of the report, data are presented for the 348 participants who were not post-randomisation exclusions. In total, 171 patients were allocated to hip arthroscopy and 177 patients were allocated to PHT.
| Site | Number of months open | Number of participants eligible | Number of participants approached | Number of participants recruited | Approach percentage (approached/eligible, %) | Success percentage (recruited/approached, %) |
|---|---|---|---|---|---|---|
| University Hospitals Coventry and Warwickshire | 44 | 115 | 112 | 78 | 97 | 69 |
| Yeovil District Hospital | 42 | 38 | 37 | 22 | 97 | 59 |
| Royal Devon and Exeter Hospital | 41 | 22 | 22 | 18 | 100 | 82 |
| Royal Orthopaedic Hospital | 41 | 93 | 93 | 39 | 100 | 42 |
| Wrightington Hospital | 37 | 27 | 18 | 12 | 67 | 67 |
| The Royal Cornwall | 15 | 11 | 9 | 7 | 82 | 78 |
| Elective Orthopaedic Centre | 6 | 7 | 7 | 2 | 100 | 29 |
| Northumbria Healthcare NHS Foundation Trust | 36 | 57 | 55 | 14 | 96 | 25 |
| The Royal London | 15 | 8 | 8 | 5 | 100 | 63 |
| Doncaster and Bassetlaw Teaching Hospital NHS Foundation Trust | 36 | 32 | 28 | 16 | 88 | 57 |
| Royal National Orthopaedic Hospital | 35 | 15 | 13 | 10 | 87 | 77 |
| Frimley Park Hospital | 33 | 35 | 35 | 23 | 100 | 66 |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital | 29 | 44 | 44 | 32 | 100 | 73 |
| South Tees Hospitals NHS Foundation Trust | 15 | 36 | 31 | 8 | 86 | 26 |
| University College Hospital | 14 | 15 | 15 | 8 | 100 | 53 |
| Guys’ and St Thomas’ Hospital | 13 | 31 | 31 | 11 | 100 | 35 |
| Cardiff and Vale Hospitals | 13 | 12 | 12 | 11 | 100 | 92 |
| Glasgow Royal Infirmary | 11 | 5 | 5 | 5 | 100 | 100 |
| Wrexham Maelor Hospital | 13 | 4 | 4 | 4 | 100 | 100 |
| King’s College Hospital | 13 | 22 | 22 | 13 | 100 | 59 |
| North Bristol NHS Trust | 10 | 13 | 13 | 7 | 100 | 54 |
| Spire Manchester Hospital | 4 | 4 | 4 | 3 | 100 | 75 |
| Total | 646 | 618 | 348 | 96 | 56 |
Participant characteristics
The two treatment groups of consented participants were well matched in terms of both demographics and pre-randomisation hip-related quality of life (Table 7), with both groups of participants having had symptoms for approximately 3 years.
| Characteristic | Treatment group | Total | |
|---|---|---|---|
| Surgery (N = 171)a | PHT (N = 177)a | ||
| Age (years) | |||
| Mean | 35.4 | 35.2 | 35.3 |
| SD | 9.7 | 9.4 | 9.6 |
| Median | 35.8 | 34.7 | 35.5 |
| Minimum | 16.7 | 16.4 | 16 |
| Maximum | 66.4 | 68.4 | 68.4 |
| Hip side considered for treatment, n (%) | |||
| Left | 75 (44) | 74 (42) | 149 (43) |
| Right | 95 (56) | 103 (58) | 198 (57) |
| Sex, n (%) | |||
| Female | 71 (42) | 64 (36) | 136 (39) |
| Male | 100 (58) | 113 (64) | 215 (61) |
| Current smoking status, n (%) | |||
| Yes | 31 (18) | 25 (14) | 56 (16) |
| No | 136 (80) | 151 (85) | 287 (82) |
| If yes, how many cigarettes smoked, on average, per day | |||
| Mean | 10 | 10 | 10 |
| SD | 5.4 | 4.6 | 5.1 |
| Median | 10 | 10 | 10 |
| Minimum | 2 | 2 | 2 |
| Maximum | 20 | 20 | 20 |
| If yes, total number of years as a smoker | |||
| Mean | 17 | 14 | 16 |
| SD | 9.7 | 8.1 | 9.1 |
| Median | 15 | 15 | 15 |
| Minimum | 1 | 1 | 1 |
| Maximum | 36 | 26 | 36 |
| Duration (months) of hip symptoms | |||
| Mean | 37 | 40 | 39 |
| SD | 36.6 | 40.8 | 38.6 |
| Median | 24 | 24 | 24 |
| Minimum | 0 | 4 | 0 |
| Maximum | 228 | 240 | 240 |
| Impingement type, n (%) | |||
| Cam | 129 (75) | 133 (75) | 262 (75) |
| Mixed | 29 (17) | 30 (17) | 59 (17) |
| Pincer | 13 (8) | 14 (8) | 27 (8) |
| Units of alcohol consumed in an average week | |||
| Mean | 6.2 | 6.0 | 6.1 |
| SD | 8.6 | 7.7 | 8.1 |
| Median | 2 | 3 | 3 |
| Minimum | 0 | 0 | 0 |
| Maximum | 40 | 40 | 40 |
| Diabetes, n (%) | |||
| Yes | 2 (1) | 4 (2) | 6 (2) |
| No | 165 (96) | 171 (97) | 336 (97) |
| Chronic renal failure, n (%) | |||
| Yes | 1 (1) | 0 (0) | 1 (< 1) |
| No | 166 (96) | 176 (99) | 342 (97) |
| Physical activity (UCLA Activity Scale) | |||
| Mean | 4.3 | 4.4 | 4.3 |
| SD | 2.5 | 2.5 | 2.5 |
| Median | 5 | 5 | 5 |
| Minimum | 1 | 1 | 1 |
| Maximum | 9 | 9 | 9 |
| Hip-related quality of life (iHOT-33 score) | |||
| Mean | 39 | 36 | 37 |
| SD | 20.9 | 18.2 | 20.0 |
| Median | 38 | 33 | 37 |
| Minimum | 0 | 2 | 0 |
| Maximum | 82 | 80 | 82 |
| SF-12: physical component score | |||
| Mean | 44 | 44 | 44 |
| SD | 7.6 | 5.9 | 6.8 |
| Median | 44 | 44 | 44 |
| Minimum | 24 | 29 | 24 |
| Maximum | 61 | 63 | 63 |
| SF-12: mental component score | |||
| Mean | 42 | 42 | 42 |
| SD | 7.1 | 7.3 | 7.2 |
| Median | 43 | 43 | 43 |
| Minimum | 24 | 18 | 18 |
| Maximum | 61 | 57 | 61 |
| EQ-5D-3L/EQ-5D-5L index score | |||
| Mean | 0.576 | 0.557 | 0.566 |
| SD | 0.26 | 0.25 | 0.25 |
| Median | 0.689 | 0.642 | 0.654 |
| Minimum | –0.239 | –0.181 | –0.239 |
| Maximum | 1 | 1 | 1 |
| EQ-5D-5L VAS | |||
| Mean | 67 | 67 | 67 |
| SD | 20.2 | 18.7 | 19.4 |
| Median | 70 | 70 | 70 |
| Minimum | 5 | 10 | 5 |
| Maximum | 100 | 100 | 100 |
Treatment allocation and adherence
The adherence to treatment allocation is displayed in Table 8 with reasons for not adhering to allocation given in Appendix 2. With respect to treatment adherence, adherence to PHT was considered to be attending at least one session with the PHT physiotherapist.
| Treatment received | Treatment allocated, n (%) | Total | |
|---|---|---|---|
| Surgery | PHT | ||
| Surgery only | 144 (84) | 0 (0) | 144 |
| PHT only | 0 (0) | 154 (87) | 154 |
| PHT and surgery | 0 (0) | 14 (8) | 14 |
| Neither trial treatment | 27 (16) | 9 (5) | 36 |
| Total | 171 | 177 | 348 |
Fourteen participants who were allocated to PHT had all or part of this intervention, but then, at their request, went on to have hip arthroscopy within 12 months after randomisation. No patients allocated to hip arthroscopy had PHT. Twenty-seven participants allocated to surgery did not receive surgery within the 12-month follow-up period.
Patients allocated to PHT were able to commence their treatment a mean of 48 days after randomisation, whereas patients allocated surgery did not commence their treatment until a mean of 132 days after randomisation (Table 9 and Figure 7).
| Days between date of randomisation and date of intervention | ||
|---|---|---|
| Surgery group | PHT group | |
| Mean | 132 | 48 |
| n | 144 | 154 |
| SD | 71 | 43 |
| Median | 122 | 37 |
| Minimum | 11 | 0 |
| Maximum | 359 | 245 |
FIGURE 7.
Delay from day of randomisation to date at which intervention started, for those who received allocated treatment. (a) Surgery; and (b) PHT.
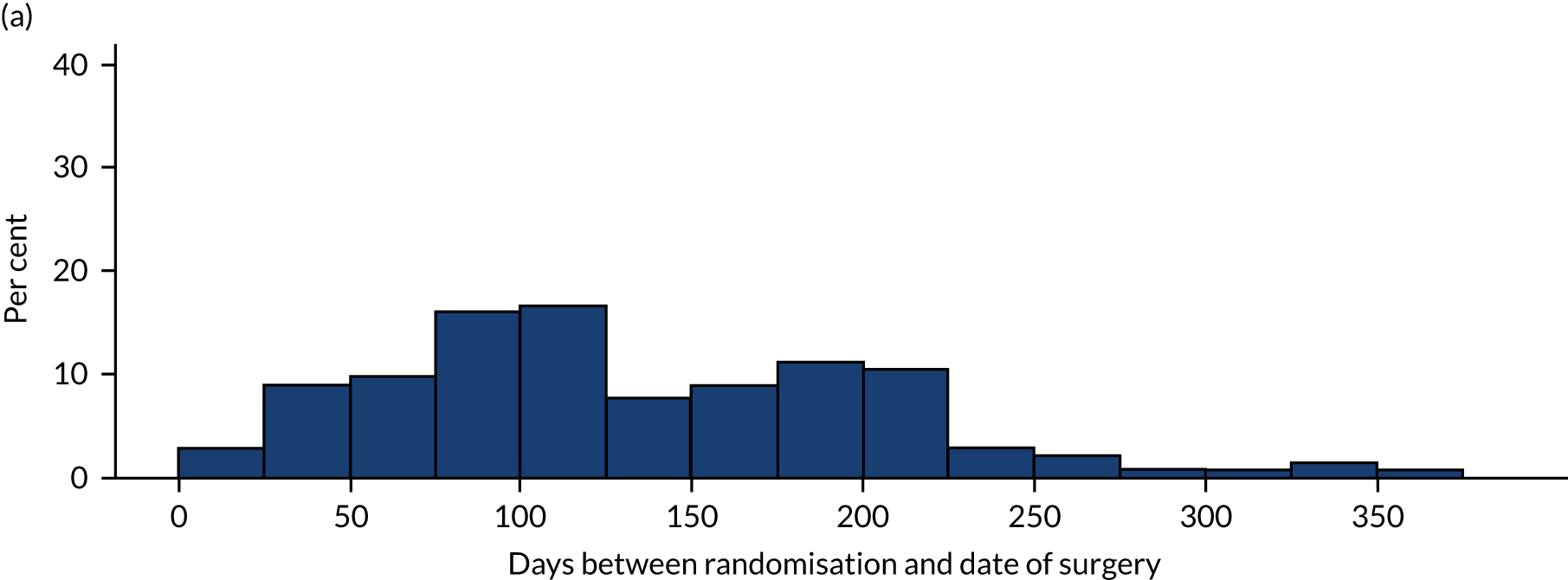
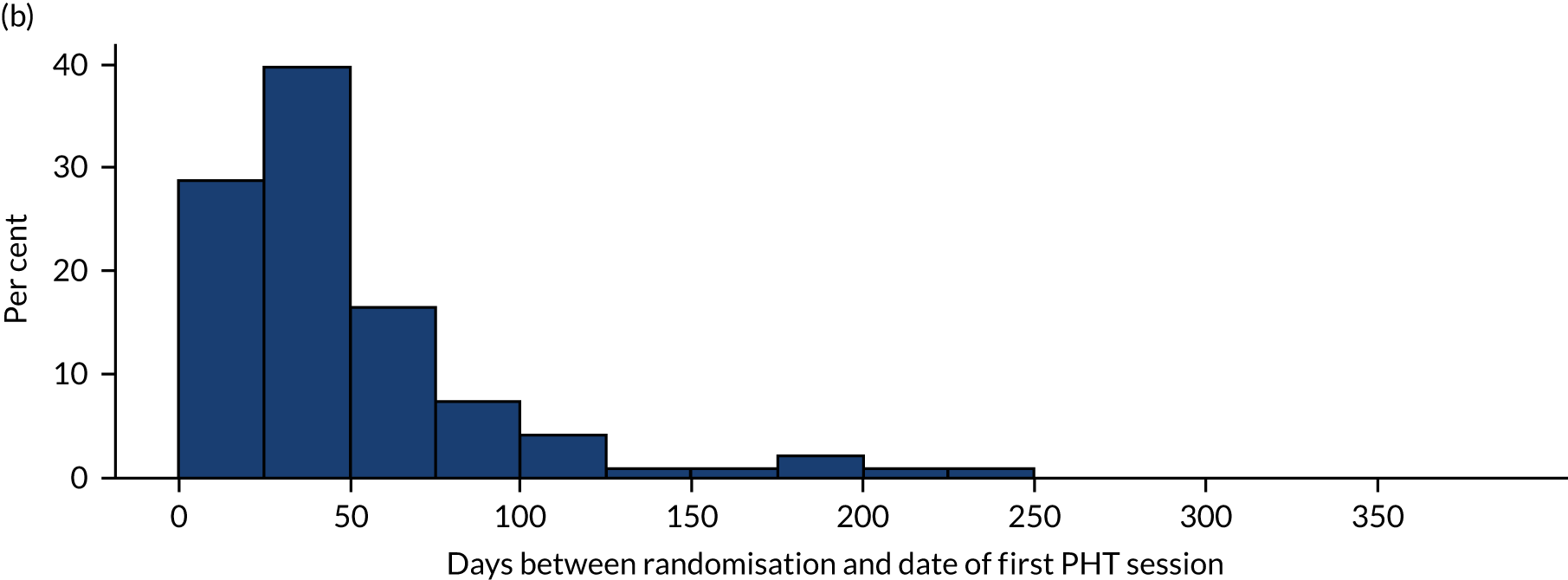
Interventions
Arthroscopic surgery
Surgeons
Surgery was delivered by 1 out of 27 consultants registered with the General Medical Council and on the specialty trauma and orthopaedic register. Surgeons had been on the specialty register for a mean of 11 (SD 7) years. Trial surgeons had been performing hip arthroscopy as a consultant for a mean of 9 (SD 3.6) years, having received the following dedicated hip preservation training: specialist registrar (n = 13), courses (n = 24), fellowship (n = 14, mean duration 9 months) and travelling fellowship (n = 8, mean duration 1.6 months). Six surgeons were directors of hip preservation fellowships and 17 surgeons were faculty on dedicated hip arthroscopy courses. Trial surgeons performed a mean of 112 (SD 55) hip arthroscopies per year, of which 81 (SD 45) were for FAI syndrome. Each surgeon treated a mean of 5.3 (SD 5.3) patients with surgery within the FASHIoN trial (Table 10).
| Number of participants treated | |
|---|---|
| Mean | 5 |
| SD | 5 |
| Median | 3 |
| Minimum | 1 |
| Maximum | 21 |
Surgery performed
A total of 144 patients received arthroscopic hip surgery. Twenty-seven patients allocated to hip arthroscopy did not have surgery in the 12-month follow-up period. A total of 121 patients received postoperative MRI. In 141 operations the surgeon examined the central compartment of the hip (note that three patients had no details available) and in 140 operations the peripheral compartment of the hip was examined (note that four patients had no details available). A cam reshaping was undertaken in isolation in 103 patients, a pincer reshaping in eight patients, cam and pincer reshaping in 27 patients, and in four patients no reshaping was undertaken, as the hip was found to be degenerate. In two patients the reshaping was not detailed.
Postoperative rehabilitation
A total of 141 patients received the surgeons’ routine postoperative rehabilitation. There was a high degree of heterogeneity in the postoperative rehabilitation protocols between sites. Details on the postoperative rehabilitation are available in Appendix 2. Postoperative rehabilitation was most typically structured in stages over several months and included:
-
an immediate postoperative phase that restored hip movement as pain improved
-
a phase to restore static stability and movement
-
a phase to restore dynamic stability and movement
-
sports-specific training.
Surgical fidelity
Of those who received surgery, 84% (121/144) had a postoperative MRI and their case was assessed by the surgical review panel. Of these cases, 87% (105/121) were deemed satisfactory and 13% (16/121) were deemed unsatisfactory. The most common reason for unsatisfactory surgery was an inadequate bony resection (n = 7) and a sharp transition from the femoral head to neck (n = 5) as a result of reshaping surgery. Table 11 reports the grading for postoperative hip shape (see also Appendix 1).
| Grade | ||||
|---|---|---|---|---|
| 1 (satisfactory) | 2 (borderline) | 3 (inadequate) | 4 (no change) | |
| Head sphericity | 80 | 29 | 9 | 2 |
| Head–neck junction | 73 | 38 | 7 | 2 |
| Rim morphology | 109 | 7 | 0 | 2 |
Personalised hip therapy
Physiotherapists
Personalised hip therapy was delivered at 23 hospitals by a total of 47 chartered physiotherapists who were registered with the Health and Care Professions Council (London, UK). In terms of clinical experience, experience ranged from NHS Agenda for Change band 5 to band 8a (band 5, n = 1; band 6, n = 11; band 7, n = 19; band 8a, n = 5; band unknown, n = 11). Agenda for Change is the current NHS grading and pay system in which a band 5 physiotherapist is usually a recently qualified physiotherapist or one with < 3 years of experience and a band 8a physiotherapist represents a specialist physiotherapist or an extended scope practitioner.
All PHT physiotherapists treated patients with musculoskeletal conditions within their normal clinical practice. Forty-one (90%) physiotherapists had previously treated patients with FAI syndrome before involvement in the trial. All physiotherapists attended at least one of eight workshops held between 2012 and March 2016. Physiotherapists delivered a median of seven PHT sessions and each physiotherapist treated a median of two patients (Table 12). Typically, each site had two trained PHT physiotherapists and they often changed jobs and, therefore, the median number of participants treated by each PHT physiotherapist was smaller than that of participants operated on by each surgeon.
| Number of sessions delivered | Number of participants treated | |
|---|---|---|
| Mean | 16 | 3 |
| SD | 27 | 4.1 |
| Median | 7 | 2 |
| Minimum | 1 | 1 |
| Maximum | 187 | 28 |
Personalised hip therapy delivered
The physiotherapists delivered a total of 947 PHT treatment contacts for trial participants, of which 878 (93%) were face-to-face contacts, 31 (3%) were telephone contacts and 4 (0.4%) were contacts via e-mail. For 34 (3%) contacts, physiotherapists did not record the mode of contact on the CRF. Analysis of the CRF data highlighted that 72 (65%) patients randomised to the PHT intervention received six or more treatment contacts in accordance with the PHT protocol. Fifty-six (35%) patients received fewer than six treatment sessions. Of these patients, nine (16%) were formally discharged by the physiotherapist and 47 (84%) were not. Only 20 (11%) participants failed to attend any session of their PHT treatment, with a further 23 (13%) participants receiving fewer than three treatment contacts (Table 13 and Appendix 2).
| Number of sessions attended | Count, n (%) | Cumulative, n (%) |
|---|---|---|
| 1 | 17 (11) | 17 (11) |
| 2 | 6 (4) | 23 (15) |
| 3 | 10 (6) | 33 (21) |
| 4 | 13 (8) | 46 (29) |
| 5 | 10 (6) | 56 (35) |
| 6 | 32 (20) | 88 (56) |
| 7 | 18 (11) | 106 (67) |
| 8 | 20 (13) | 126 (80) |
| 9 | 14 (9) | 140 (89) |
| 10 | 16 (10) | 156 (99) |
| 11 | 2 (1) | 158 (100) |
| Total | 158 | 158 |
Treatment sessions lasted a mean of 30 minutes (SD 11 minutes), with the first assessment and treatment session usually lasting longer. The content of treatment sessions included assessment or reassessment (in 88% of treatment sessions), education and advice (in 77% of sessions) and help with pain relief (in 74% of treatment sessions). There was evidence that the participant’s exercise programme was supervised in clinic in 78% of treatment sessions and that exercise diaries were used with 46% of participants to support monitoring of exercise behaviour at home, although in 4% of cases the exercise diary was forgotten by participants at further treatment sessions.
In terms of the specific content of the PHT exercise prescription for participants, for 158 PHT participants there was a total of 3657 exercises recorded on the CRFs from 947 treatment sessions (highlighting that most participants were prescribed three or four different exercises at each treatment session). A detailed summary of the frequency of each exercise prescribed is provided in Appendix 2. The five most frequent exercises prescribed were (1) bridge (n = 337), (2) clam (n = 326), (3) bent knee fall out (n = 226), (4) straight leg raise through hip abduction in side lying (n = 186) and (5) hip extension in four-point kneeling (n = 172). These five most commonly prescribed exercises (i.e. exercises 4, 7, 3, 8 and 5, respectively) aim to establish pelvic control while strengthening the gluteus maximus and gluteus medius. Other frequently prescribed exercises were stretches targeting the hip flexors and external rotators (see Appendix 2).
Personalised hip therapy fidelity
A source verification audit was undertaken, with 19 sites visited. Seventy-three (41%) study patients’ physiotherapy treatment notes were reviewed and compared with the data recorded on CRFs. All CRFs were judged to accurately reflect the treatment documented in the treatment record. These data were then shared with the central fidelity review panel on a per case basis.
Of the patients who received PHT (n = 154), 107 (69%) were judged to have received the intervention to a high fidelity. The most common reason for the low fidelity of PHT was participants not receiving the minimum of six therapy sessions (34/47, 72%). Other reasons for the low fidelity of PHT were no progression of exercises by the physiotherapist (11/47, 23%) and the patient not complying with the exercise programme (2/47, 4%).
Outcome data completeness
A total of 319 (92%) participants completed questionnaires at 12 months after randomisation, seven participants withdrew from follow-up questionnaire completion and 22 participants were lost to follow-up (Table 14). Table 15 highlights the timings of follow-up assessments by treatment group. Appendix 2, Table 46, reports the completeness of the questionnaire data at different time points.
| Follow-up status | 6-month follow-up, n (%) | 12-month follow-up, n (%) | ||||
|---|---|---|---|---|---|---|
| Surgery group | PHT group | Total | Surgery group | PHT group | Total | |
| Completed questionnaire | 160 (92) | 154 (87) | 314 (89) | 158 (92) | 163 (92) | 321 (92) |
| Loss to follow-up | 8 (5) | 19 (11) | 27 (8) | 10 (6) | 10 (6) | 20 (6) |
| Consent withdrawn | 3 (2) | 4 (2) | 7 (2) | 3 (2) | 4 (2) | 7 (2) |
| Timing of follow-up assessment | Treatment group | |
|---|---|---|
| Surgery | PHT | |
| Days between date of randomisation and date of 6-month follow-up form | ||
| Mean | 190 | 190 |
| n | 161 | 154 |
| SD | 36 | 35 |
| Median | 186 | 185 |
| Minimum | 71 | 83 |
| Maximum | 307 | 296 |
| Days between date of randomisation and date of 12-month follow-up form | ||
| Mean | 370 | 379 |
| n | 158 | 163 |
| SD | 30.7 | 54.5 |
| Median | 362 | 364 |
| Minimum | 292 | 264 |
| Maximum | 470 | 812 |
Outcomes
Primary outcome
The iHOT-33 score increased between baseline and 12 months in both groups (Table 16 and Figures 8 and 9). iHOT-33 scores improved from a mean of 36 points at baseline to 50 points at 12 months in the PHT group and from a mean of 39 points at baseline to 59 points at 12 months in the hip arthroscopy group. In the primary intention-to-treat analysis at 12 months, the adjusted estimate of treatment effect measured with iHOT-33 was 6.8 (95% CI 1.7 to 12.0) in favour of the hip arthroscopy group compared with the PHT group. The p-value of 0.009 indicates that there is evidence for a statistically significant difference (the 95% CI includes the prespecified MCID and does not include zero).
| Time point | Treatment group | Difference (95% CI) | p-value | ||||
|---|---|---|---|---|---|---|---|
| Surgery | PHT | ||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | ||
| 6 months | 46.6 (25) | 161 | 45.6 (23) | 154 | 1.0 | –0.7 (–5.2 to 3.7) | 0.743 |
| 12 months | 58.8 (27) | 158 | 49.7 (25) | 163 | 9.1 | 6.8 (1.7 to 12.0) | 0.009 |
FIGURE 8.
Box plots of iHOT-33 baseline and follow-up scores.
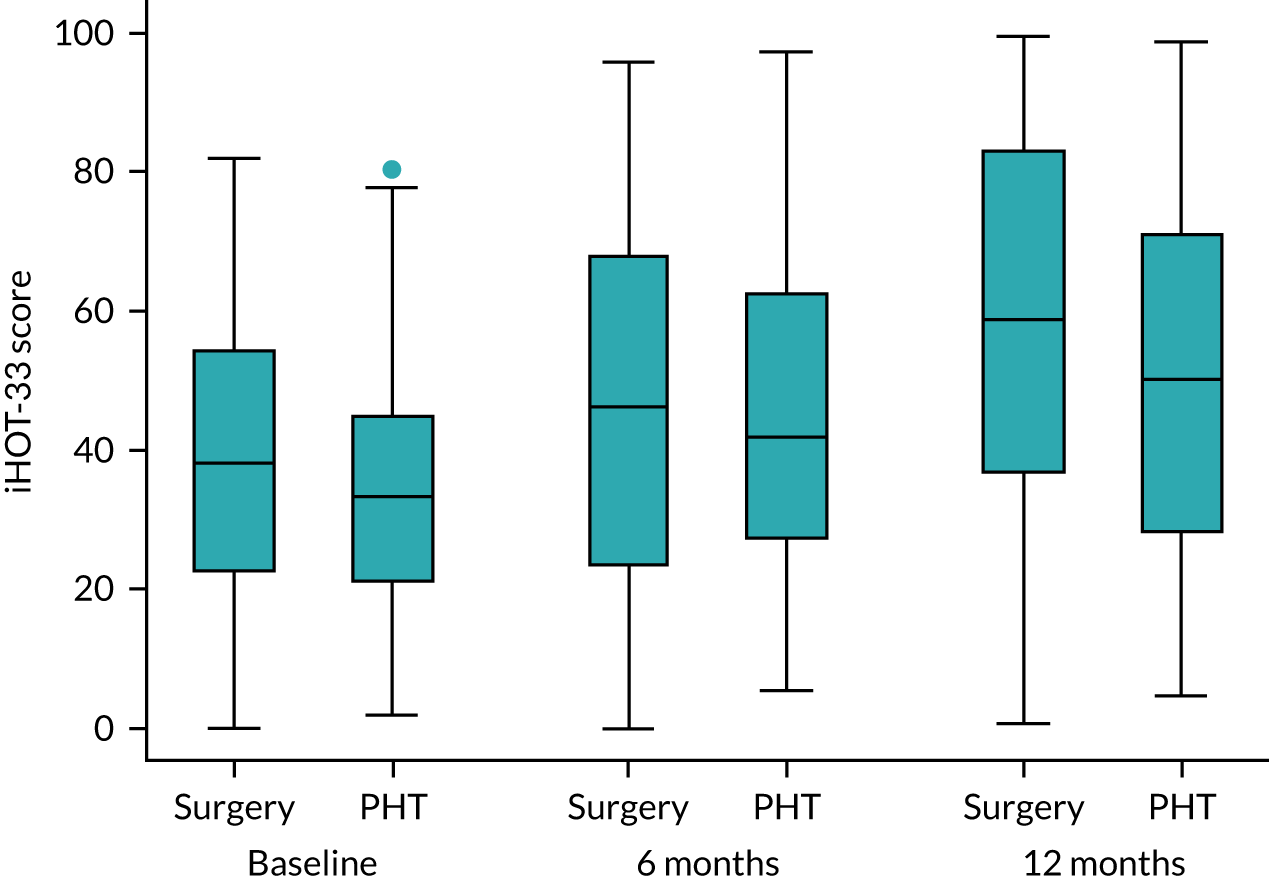
FIGURE 9.
Overall trend in iHOT-33 unadjusted mean scores and 95% CIs.
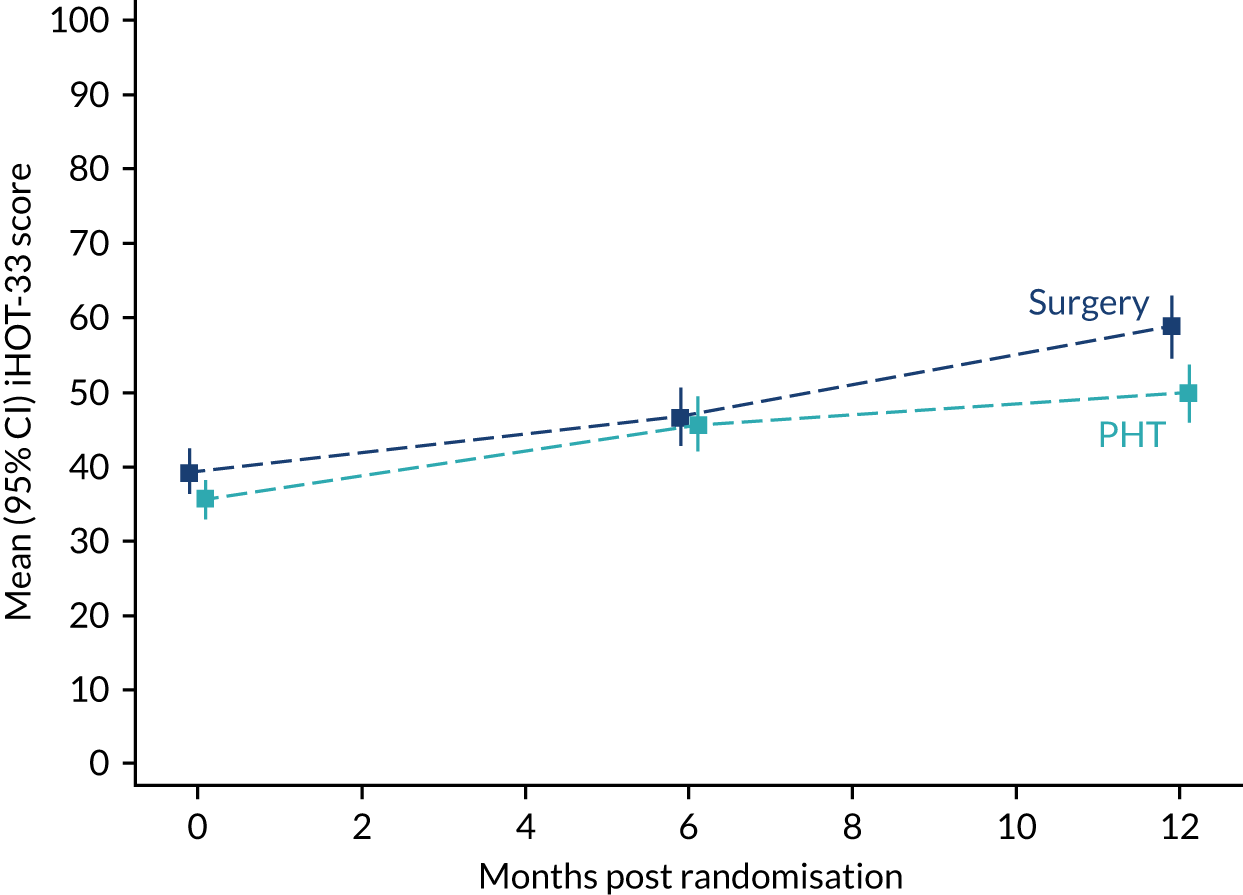
Secondary outcomes
There were no were significant differences between treatment groups at 6 or 12 months in the EuroQol-5 Dimensions (EQ-5D) or SF-12 (Figures 10–13 and Table 17).
FIGURE 10.
Box plots of EQ-5D-5L baseline and follow-up scores.
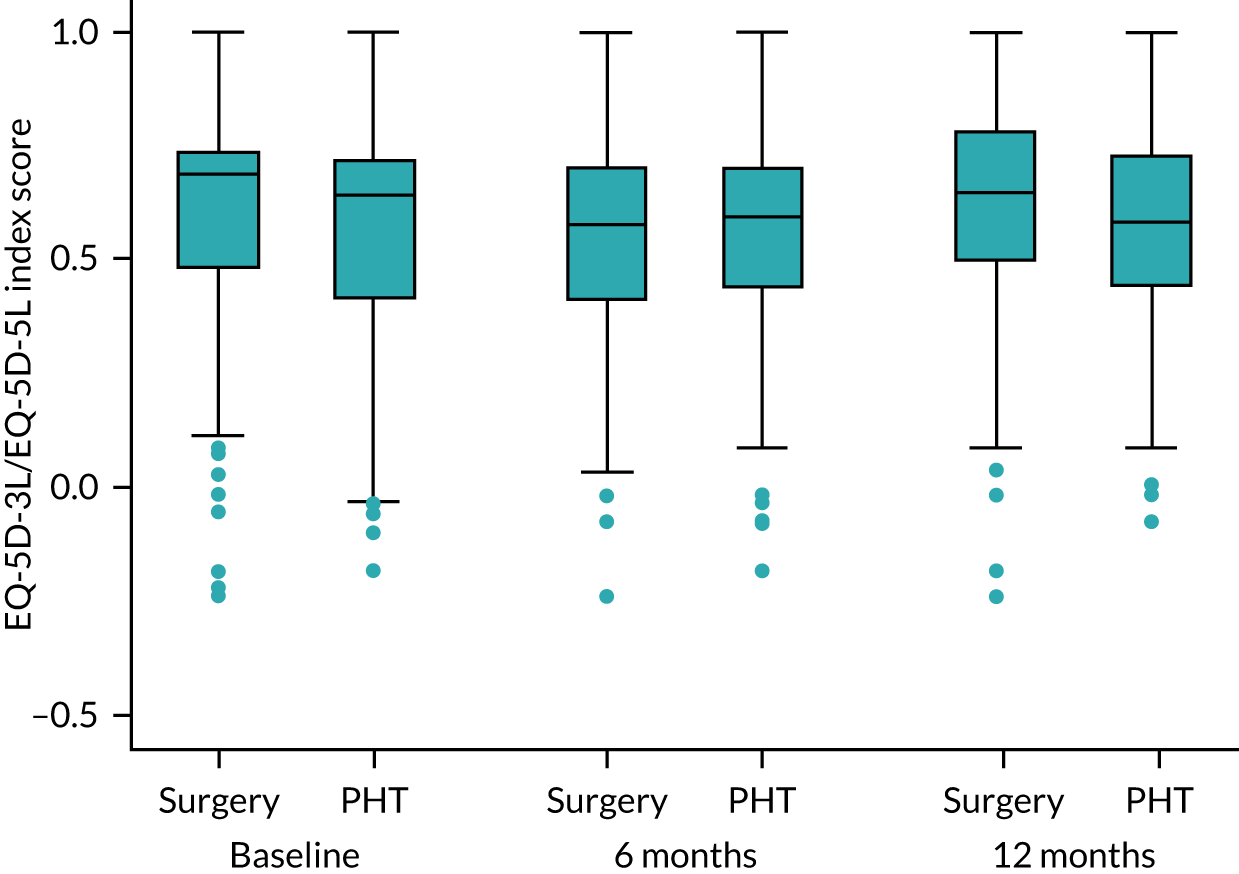
FIGURE 11.
Box plots of EQ-5D VAS baseline and follow-up scores.
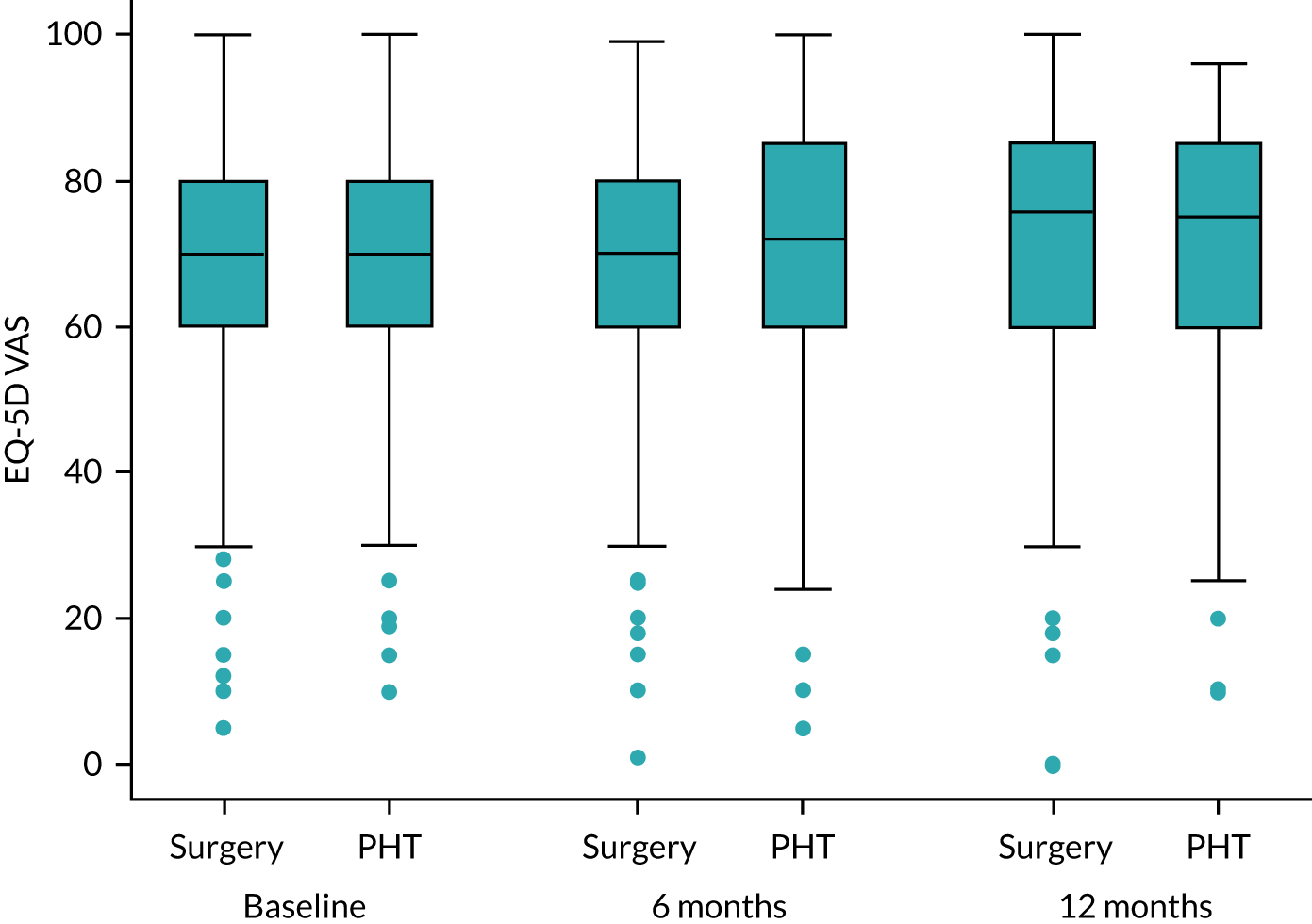
FIGURE 12.
Box plots of SF-12 (physical component score) baseline and follow-up scores.
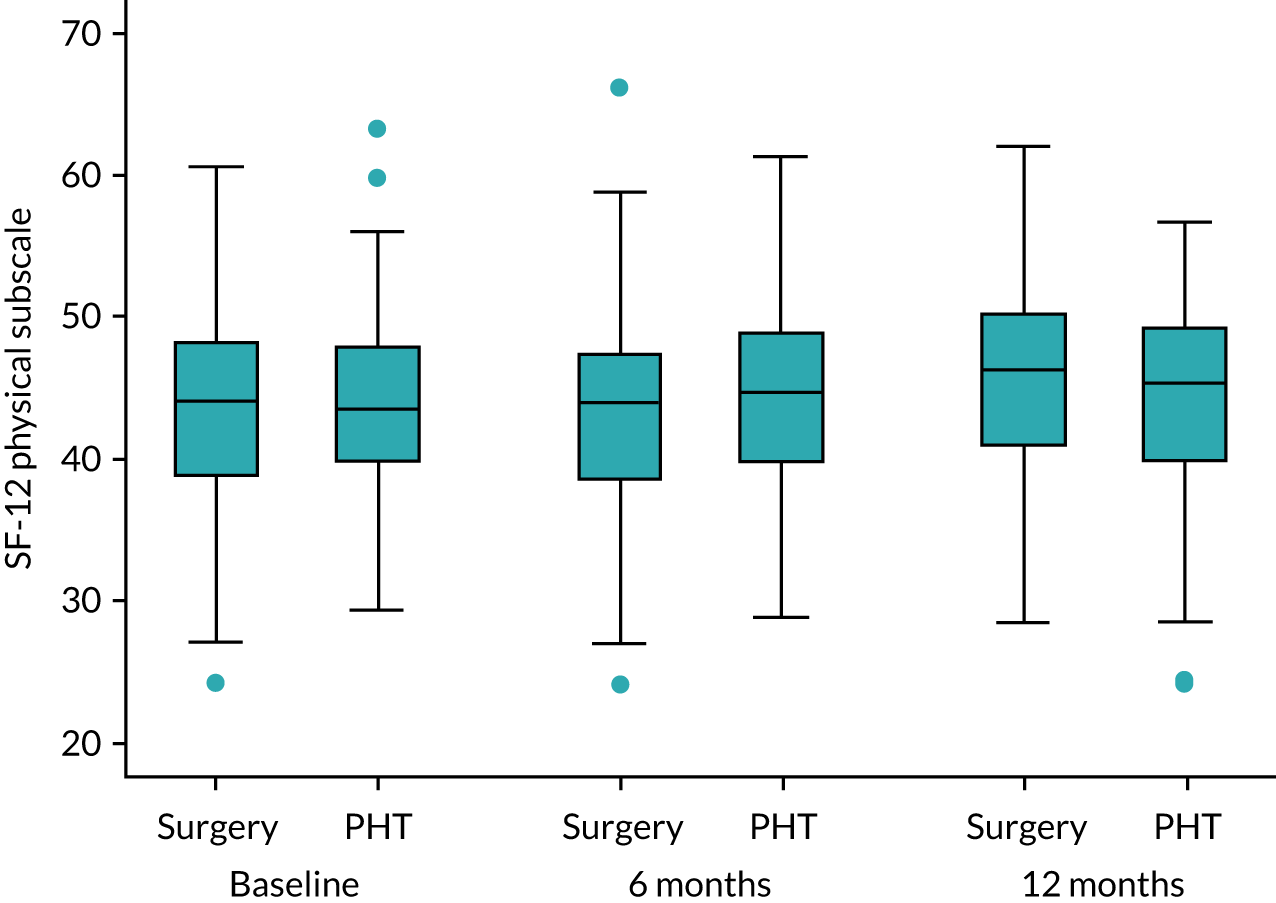
FIGURE 13.
Box plots of SF-12 (mental component score) baseline and follow-up scores.
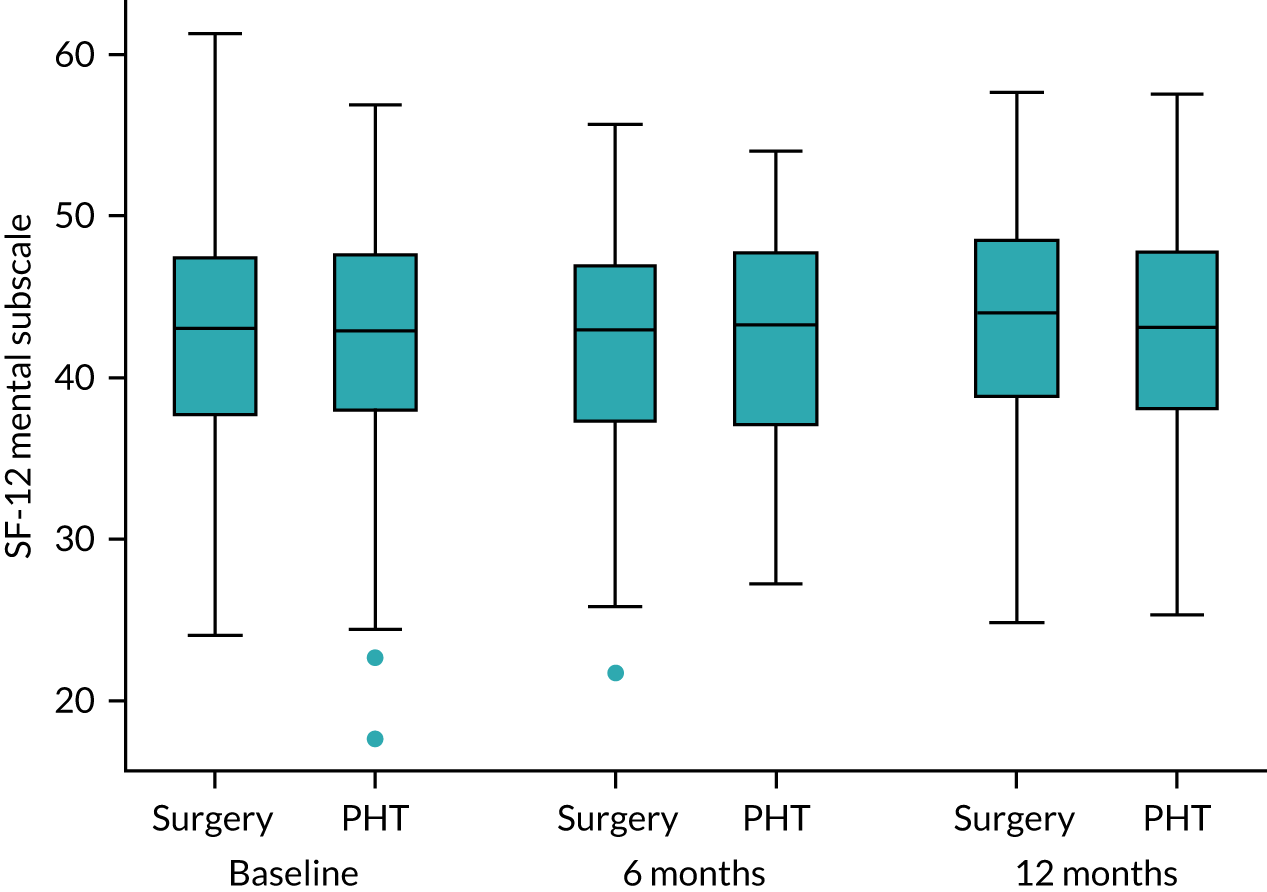
| Outcome | Time point (months) | Treatment group | Difference (95% CI) | p-value | ||||
|---|---|---|---|---|---|---|---|---|
| Surgery | PHT | |||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | |||
| EQ-5D-5L score | 6 | 0.544 (0.26) | 144 | 0.573 (0.23) | 147 | –0.029 | –0.042 (–0.088 to 0.005) | 0.081 |
| 12 | 0.615 (0.25) | 152 | 0.578 (0.24) | 147 | 0.037 | 0.020 (–0.027 to 0.067) | 0.397 | |
| EQ-5D VAS score | 6 | 67.8 | 145 | 70.3 | 145 | –2.5 | –2.1 (–5.7 to 1.4) | 0.241 |
| 12 | 71.9 | 150 | 69.2 | 145 | 2.7 | 2.6 (–1.2 to 6.4) | 0.180 | |
| SF-12 physical component score | 6 | 43.4 | 146 | 44.2 | 142 | –0.8 | –0.7 (–2.1 to 0.7) | 0.304 |
| 12 | 45.1 | 145 | 44.1 | 132 | 1.0 | 1.1 (–0.2 to 2.5) | 0.099 | |
| SF-12 mental component score | 6 | 42.1 | 146 | 42.1 | 142 | –0.1 | –0.1 (–1.5 to 1.3) | 0.929 |
| 12 | 43.2 | 145 | 42.6 | 132 | 0.6 | 0.4 (–1.2 to 2.0) | 0.589 | |
Per-protocol analyses
Two per-protocol analyses were conducted. The first per-protocol analysis was for patients who received surgery only (n = 144) or PHT only (n = 154), irrespective of the quality of the treatment they received (Table 18). The adjusted estimate of treatment effect measured with iHOT-33 was 8.2 (95% CI 2.8 to 13.6) in favour of hip arthroscopy.
| Time point | Treatment group | Difference (95% CI) | p-value | ||||
|---|---|---|---|---|---|---|---|
| Surgery | PHT | ||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | ||
| 12 months | 58.5 (27.9) | 140 | 49.3 (25.4) | 147 | 9.2 | 8.2 (2.8 to 13.6) | 0.003 |
A second exploratory per-protocol analysis was conducted that included only those participants who received their allocated intervention and whose treatment was deemed to be of a satisfactory quality by the respective review panels. In this analysis, 95 surgical cases and 106 PHT participants were included (Table 19). In this exploratory per-protocol analysis, the adjusted estimate of treatment effect measured with the iHOT-33 was favour of surgery (Table 20).
| Treatment received | Treatment allocated, n (%) | ||
|---|---|---|---|
| Surgery | PHT | Total | |
| Adequate | 95 (56) | 106 (60) | 201 (58) |
| Not adequate | 45 (26) | 39 (22) | 84 (24) |
| Received other treatment | 27 (16) | 23 (13) | 50 (14) |
| Missing review | 4 (2) | 9 (5) | 13 (4) |
| Total | 171 | 177 | 348 |
| Time point | Treatment group | Difference (95% CI) | p-value | ||||
|---|---|---|---|---|---|---|---|
| Surgery | PHT | ||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | ||
| 12 months | 56.9 (28.2) | 92 | 49.3 (25.9) | 104 | 7.6 | 6.9 (0.26 to 13.4) | 0.041 |
Missing outcome analyses
There was a low level of missing item-level data in all patient-reported outcome measures at all time points. After imputation for missing data, the adjusted estimate of treatment effect was similar at 6.6 (95% CI 1.7 to 11.4) points in favour of hip arthroscopy (Table 21).
| Time point | Treatment group | Difference (95% CI) | p-value | ||||
|---|---|---|---|---|---|---|---|
| Surgery | PHT | ||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | ||
| 12 months | 58.7 (26.2) | 171 | 49.8 (24.6) | 177 | 8.9 | 6.6 (1.7 to 11.4) | 0.008 |
Subgroup analyses
Planned subgroup analyses were conducted for those participants with cam, pincer and mixed-type FAI syndrome and those aged < 40 years and ≥ 40 years.
There was no evidence of a subgroup effect, with the adjusted estimate of treatment effect measured by the iHOT-33 being as follows: 5.0 (95% CI –1.2 to 11.3) in participants aged < 40 years, 10.9 (95% CI 1.7 to 20.1) in participants aged ≥ 40 years, 8.3 (95% CI 2.5 to 14.2) in participants with cam morphology, 1.1 (95% CI –11.5 to 13.7) in participants with mixed cam and pincer morphology and 4.0 (95% CI –14.6 to 22.7) in participants with pincer morphology, in favour of surgery (Table 22).
| Subgroup | Treatment group | Difference (95% CI) | p-valueb | ||||
|---|---|---|---|---|---|---|---|
| Surgery | PHT | ||||||
| Mean (SD) | n | Mean (SD) | n | Raw | Adjusteda | ||
| Age group | |||||||
| < 40 years | 59.1 (26.6) | 103 | 50.0 (24.5) | 117 | 9.1 | 5.0 (–1.2 to 11.3) | 0.302 |
| ≥ 40 years | 58.1 (28.4) | 55 | 48.8 (27.9) | 46 | 9.3 | 10.9 (1.7 to 20.1) | |
| Impingement type | |||||||
| Cam | 59.4 (27.7) | 120 | 49.1 (24.3) | 124 | 10.4 | 8.3 (2.5 to 14.2) | 0.567 |
| Mixed | 56.3 (22.4) | 26 | 51.5 (31.1) | 27 | 4.8 | 1.1 (–11.5 to 13.7) | |
| Pincer | 57.3 (33.4) | 12 | 51.8 (25.4) | 12 | 5.5 | 4.0 (–14.6 to 22.7) | |
Sensitivity analyses
In a post hoc analysis, there was no significant difference in iHOT-33 score at 12 months for patients in the hip arthroscopy group who were treated within 6 months of randomisation compared with those treated ≥ 6 months after randomisation (0.9, 95% CI −10.7 to 8.8).
Complications
Complications at the 6-week assessment
A total of 138 and 146 participants following surgery and PHT, respectively, returned a 6-week complication log. The most commonly reported complication in both groups was muscle soreness (Table 23). Eight (6%) patients reported a superficial wound infection following surgery, but antibiotics were prescribed in only four (3%) cases. Thirty-five (24%) participants reported groin, leg or foot numbness after surgery.
| Complication | Treatment group, n (%) | |
|---|---|---|
| Surgery (N = 138) | PHT (N = 146) | |
| Patient-reported superficial wound infection | 8 (6) | n/a |
| Requiring antibiotic prescription | 4 (3) | n/a |
| Deep-wound infection | 0 | n/a |
| Requiring further surgery | 0 | n/a |
| Patient-reported numbness in groin, leg or foot | 35 (24) | n/a |
| Hip fracture | 0 | n/a |
| Further surgery following hip arthroscopy | 0 | n/a |
| Problems with pain medication | 10 (7) | 7 (5) |
| Problems with hip joint injections | 0 | 5 (3) |
| Muscle soreness from exercises | 57 (41) | 68 (47) |
| Deep-vein thrombosis | 0 | 0 |
| Unscheduled hospital appointments | 5 (4) | 2 (1) |
| Persistent hip-related symptoms | 11 (8) | 9 (6) |
| Other complications related to intervention | 6 (4) | 3 (12) |
| Other complications not related to intervention | 5 (4) | 10 (7) |
| Of which patient reported lower back pain | 2 (2) | 5 (3) |
Complications reported at the 6- and 12-month follow-up time points
Complications at 12 months post randomisation are reported in Table 24.
| AE | Treatment group | p-valuec | |
|---|---|---|---|
| Surgery (n = 138a) | PHT (n = 146b) | ||
| Numbness in groin, leg or foot | 35 | n/a | n/a |
| Superficial wound problems | 9 (4 required antibiotics) | n/a | n/a |
| Deep infection | 1 | n/a | n/a |
| Fracture | 0 | n/a | n/a |
| Deep-vein thrombosis | 0 | n/a | n/a |
| Muscle soreness at 6 weeks post intervention | 58 | 69 | 0.404 |
| Hip pain or stiffness at 6 weeks post intervention | 13 | 8 | 0.258 |
| Unscheduled hospital appointments | 13 | 6 | 0.096 |
| Other complications related to intervention | 8 (2 numbness proximal thigh, 1 scrotal infection, 1 scrotal bruising, 1 labial swelling, 1 ankle pain, 1 erratic INR, 1 nausea secondary to analgesia and 1 numbness to tip of tongue for 2 weeks post operation)d | 1 (muscle spasms) | 0.0168 |
| Other complications not related to intervention | 10 (3 knee pain, 2 lower back pain, 1 shingles, 1 UTI, 1 essential thrombocythaemia, 1 hernia surgery and 1 contralateral foot pain) | 18 (7 lower back pain, 2 knee pain, 2 road traffic collisions, 2 abdominal pain under investigation, 1 viral illness, 1 endometriosis, 1 chronic pain referred to rheumatologist, 1 skin discolouration and 1 multiple sclerosis) | 0.168 |
At 12 months, seven SAEs had been reported, six of which were in patients who received surgery (Table 25). Two patients allocated to surgery required hospitalisation and four required further medical intervention. One patient who received PHT required hospitalisation.
| Treatment group | SAE | p-valuea |
|---|---|---|
| Surgery (n = 138) | 6 (1 failed discharge from day surgery unit required overnight admission, 1 scrotal haematoma required readmission, 2 superficial wound infection required oral antibiotics, 1 deep infection required further surgery and ultimately a total hip replacement and 1 fall unrelated to surgery) | 0.060 |
| PHT (n = 146) | 1 (biliary sepsis unrelated to PHT) |
Three-year follow-up: further procedures or physiotherapy
In the 24-month period from 12 to 36 months post randomisation, about one-third of all patients had a further procedure and about half had further physiotherapy sessions (Tables 26 and 27). A (further) one-quarter of PHT patients chose to have a hip arthroscopy and 12% of hip arthroscopy patients had a further hip arthroscopy. Two per cent and 7% of the patients in surgery and PHT groups, respectively, had a hip replacement.
| Surgical procedure | Surgery (N = 154), n (%) | PHT (N = 157), n (%) |
|---|---|---|
| Any procedure | 46 (30) | 51 (32) |
| Hip arthroscopy | 19 (12) | 38 (24) |
| Hip replacement | 3 (2) | 11 (7) |
| Hip injection | 24 (16) | 6 (4) |
| Other procedure | 1 (1) | 1 (1) |
| Physiotherapy session | Surgery (N = 154), n | PHT (N = 157), n |
|---|---|---|
| Any treatment | 91 | 78 |
| If yes, setting | ||
| NHS treatment | 74 | 60 |
| Private treatment | 17 | 18 |
| If yes, type of treatment | ||
| Exercises | 66 | 52 |
| Other | 25 | 26 |
| If yes, number (SD) of sessions | 8.8 (10) | 8.9 (7) |
Chapter 5 Economic evaluation results
Study population
The study sample comprised a total of 351 patients, of whom 173 patients were randomised to receive surgery and 178 patients were randomised to receive PHT. Three patients (two from the surgery group and one from the PHT group) were excluded post randomisation. The final sample, therefore, consisted of 348 patients (surgery group, n = 171; PHT group, n = 177). At baseline, completion rates for resource use data were between 96.5% and 99.8% in the surgery group and between 98.9% and 99.4% in the PHT group. At the 6-month assessment point, completion rates for resource use data were between 82% and 84% in the surgery group and between 78% and 81% in the PHT group. At the 12-month assessment point, completion rates for resource use data were between 83% and 87% in the surgery group and between 73% and 78% in the PHT group (Table 28). Health utility values were derived from the EQ-5D-3L/EQ-5D-5L and the SF-6D (via SF-12) and were available from > 98%, 79% and > 74% of patients from both groups at baseline, the 6-month assessment point and the 12-month assessment point, respectively.
| Assessment point and resource use category | Completion rate (%) | |
|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |
| Baseline | ||
| Hospital inpatient care | 98.8 | 99.4 |
| Hospital outpatient attendances | 98.8 | 98.9 |
| Community health-care services | 98.8 | 98.9 |
| Social care services | 98.2 | 99.4 |
| Medications | 96.5 | 99.4 |
| Aids and adaptations | 97.7 | 99.4 |
| Additional (indirect) costs | 98.8 | 99.4 |
| Benefit payments | 98.8 | 99.4 |
| EQ-5D-3L/EQ-5D-5L utility values | 98.2 | 99.4 |
| EQ-5D-3L/EQ-5D-5L VAS values | 100 | 99.4 |
| SF-6D (via SF-12) values | 98.2 | 99.4 |
| 6 months post randomisation | ||
| Hospital inpatient care | 83.0 | 78 |
| Hospital outpatient attendances | 82.5 | 78 |
| Community health-care services | 82.5 | 79.1 |
| Social care services | 83.0 | 79.1 |
| Medications | 82.5 | 78.5 |
| Aids and adaptations | 81.9 | 77.6 |
| Additional (indirect) costs | 84.2 | 81.4 |
| Benefit payments | 84.2 | 81.4 |
| EQ-5D-3L/EQ-5D-5L utility values | 82.5 | 82.5 |
| EQ-5D-3L/EQ-5D-5L VAS values | 90.6 | 94.9 |
| SF-6D (via SF-12) values | 85.4 | 79.7 |
| 12 months post randomisation | ||
| Hospital inpatient care | 85.5 | 76.3 |
| Hospital outpatients attendances | 84.2 | 76.3 |
| Community health-care services | 84.8 | 75.1 |
| Social care services | 84.2 | 74.6 |
| Medications | 84.2 | 75.1 |
| Aids and adaptations | 82.5 | 73.4 |
| Additional (indirect) costs | 86.5 | 78.0 |
| Benefit payments | 86.5 | 78.0 |
| EQ-5D-3L/EQ-5D-5L utility values | 88.3 | 82.5 |
| EQ-5D-3L/EQ-5D-5L VAS values | 95.3 | 89.3 |
| SF-6D (via SF-12) values | 84.8 | 74.6 |
Assessment of resource use and costs results
Cost of personalised hip therapy
Table 29 summarises PHT attendance by type of consultation, impingement classification, missed appointments and recruitment site. A total of 1219 physiotherapy appointments were offered to 166 (94%) of the 177 patients in the PHT group. Of these patients, 909 (75%) were face-to-face consultations, 38 (3%) were telephone consultations, seven (0.6%) were e-mail contacts and 256 (21.2%) were recorded as unknown or missed appointments. A total of 166 (94%) of the 177 PHT patients had at least one physiotherapy contact, 105 (59%) had six or more contacts (as recommended in the treatment protocol for PHT) and 11 (6.2%) did not receive the intervention at all. Excluding missed appointments, the mean number of physiotherapy contacts per patient was 5.6 (range 1–11) and the mean duration of contact for all sessions attended was 178.2 (range 30–375) minutes per patient. Among those who had the recommended six or more physiotherapy contacts, the mean number of contacts per patient was 7.7 (range 6–11) and the mean total duration across all sessions attended was 222.6 (range 140–375) minutes per patient.
| Attendance/consultation type | n (%)a | Mean (SE) | ||
|---|---|---|---|---|
| Number of consultations | Duration (minutes) | Total cost (£)b | ||
| Overall attendance | ||||
| Did not receive intervention | 11 (6.2) | 0.00 (0.00) | 0.00 (0.00) | 0.00 (0.00) |
| Offered one or more appointments (excluding DNAs) | 166 (93.8) | 7.31 (0.22) | 220.87 (6.45) | 192.16 (5.62) |
| Attended one or more sessions (excluding DNAs) | 166 (93.8) | 5.99 (0.21) | 178.17 (6.04) | 155.01 (5.25) |
| Offered six or more appointments (excluding DNAs) | 110 (62.1) | 7.74 (0.17) | 225.93 (5.24) | 196.56 (4.56) |
| Attended six or more sessions (excluding DNAs) | 105 (59.3) | 7.68 (0.15) | 222.59 (4.81) | 193.65 (4.18) |
| Attendance by type of consultation | ||||
| Face to face | 160 (90.4) | 5.66 (0.21) | 170.94 (6.02) | 148.71 (5.23) |
| Telephone | 27 (15.3) | 1.15 (0.07) | 13.65 (1.56) | 11.88 (1.35) |
| 4 (2.3) | 1.00 (0.00) | 27.70 (9.66) | 24.10 (8.41) | |
| Unknown | 22 (12.4) | 2.00 (0.35) | 65.37 (11.76) | 56.87 (10.23) |
| Attendance by type of impingement | ||||
| Cam | 129 (72.9) | 3.68 (0.18) | 110.51 (5.47) | 96.15 (4.76) |
| Pincer | 13 (7.3) | 4.96 (0.64) | 151.64 (19.06) | 131.93 (16.58) |
| Mixed | 28 (15.8) | 3.48 (0.38) | 106.95 (12.04) | 93.05 (10.48) |
| Missed appointments (i.e. DNA), total | ||||
| Attended at least one consultation | 94 (53.1) | 2.34 (0.19) | 75.41 (6.28) | 65.60 (5.46) |
| Attended at least six consultations | 6 (3.4) | 7.50 (0.85) | 246.75 (27.85) | 214.67 (24.23) |
| Missed appointments by type of consultation | ||||
| Face to face | 3 (1.7) | 1.00 (0.00) | 26.67 (3.33) | 23.20 (2.90) |
| Telephone | 5 (2.8) | 1.20 (0.20) | 13.24 (1.83) | 11.52 (1.59) |
| 1 (0.6) | 1.00 (n/a) | 32.90 (–) | 28.62 (–) | |
| Unknown | 96 (54.2) | 2.24 (0.19) | 73.68 (6.16) | 64.10 (5.36) |
| Attendance by recruitment site | ||||
| University Hospitals Coventry and Warwickshire | 33 (18.6) | 7.09 (0.48) | 25.81 (0.77) | 152.90 (8.97) |
| Yeovil District Hospital | 8 (4.5) | 3.50 (0.96) | 36.88 (3.55) | 104.34 (27.03) |
| Royal Devon and Exeter Hospital | 8 (4.5) | 6.62 (0.65) | 30.72 (1.48) | 180.53 (22.96) |
| Royal Orthopaedic Hospital | 14 (7.9) | 6.93 (0.71) | 25.64 (1.23) | 148.20 (14.66) |
| Frimley Park Hospital | 4 (2.3) | 6.25 (1.75) | 31.72 (1.18) | 175.09 (50.18) |
| Royal Cornwall Hospital | 3 (1.7) | 4.33 (0.88) | 33.57 (1.99) | 126.50 (25.19) |
| Elective Orthopaedic Centre (Epsom General Hospital) | 1 (0.6) | 6.00 (–) | 23.33 (–) | 121.80 (–) |
| Guy’s and St Thomas’ Hospital | 7 (4.0) | 7.00 (1.02) | 33.37 (1.40) | 206.18 (31.91) |
| Barts Health NHS Trust | 3 (1.7) | 5.33 (1.67) | 33.95 (2.64) | 150.28 (41.57) |
| University College Hospital | 7 (4.0) | 7.43 (0.95) | 34.83 (0.82) | 223.71 (27.93) |
| Wrightington Hospital | 5 (2.8) | 6.40 (1.12) | 34.70 (1.37) | 190.20 (30.82) |
| Northumbria Healthcare NHS Foundation Trust | 9 (5.1) | 5.22 (0.68) | 32.49 (1.86) | 140.45 (16.53) |
| Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust | 11 (6.2) | 6.27 (0.70) | 29.85 (0.29) | 162.84 (18.34) |
| Royal National Orthopaedic Hospital | 3 (1.7) | 7.67 (0.33) | 32.68 (0.80) | 217.50 (4.35) |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital | 3 (1.7) | 8.00 (0.00) | 31.67 (1.10) | 220.40 (7.67) |
| South Tees Hospitals NHS Foundation Trust | 4 (2.3) | 7.00 (1.08) | 30.17 (0.81) | 181.96 (25.87) |
| University Hospital Llandough | 2 (1.1) | 3.50 (1.50) | 47.25 (5.25) | 137.02 (45.67) |
| Glasgow Royal Infirmary | 2 (1.1) | 9.50 (0.50) | 34.11 (1.89) | 282.75 (30.45) |
| Wrexham Maelor Hospital | 2 (1.1) | 7.00 (1.00) | 31.72 (0.88) | 193.93 (32.98) |
| King’s College Hospital | 4 (2.3) | 5.25 (0.48) | 36.15 (1.46) | 163.93 (12.54) |
| North Bristol NHS Trust | 4 (2.3) | 6.75 (0.48) | 27.75 (1.89) | 162.04 (11.42) |
| Spire Healthcare (London, UK) | 1 (0.6) | 6.00 (–) | 40.00 (–) | 208.80 (–) |
| Recruitment site (missed appointments) | ||||
| University Hospitals Coventry and Warwickshire | 7 (4.0) | 3.00 (1.36) | 32.90 (0.00) | 85.87 (39.01) |
| Yeovil District Hospital | 1 (0.6) | 5.00 (–) | 32.90 (–) | 143.11 (–) |
| Royal Devon and Exeter Hospital | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Royal Orthopaedic Hospital | 4 (2.3) | 4.00 (1.78) | 31.29 (1.61) | 111.69 (52.05) |
| Frimley Park Hospital | 2 (1.1) | 2.50 (0.50) | 18.95 (13.95) | 47.29 (38.59) |
| Royal Cornwall Hospital | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Elective Orthopaedic Centre (Epsom General Hospital) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Guy’s and St Thomas’ Hospital | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Barts Health NHS Trust | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| University College Hospital | 1 (0.6) | 5.00 (–) | 32.90 (–) | 143.11 (–) |
| Wrightington Hospital | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Northumbria Healthcare NHS Foundation Trust | 2 (1.1) | 4.50 (0.50) | 32.90 (0.00) | 128.80 (14.31) |
| Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust | 3 (1.7) | 3.00 (0.00) | 32.90 (0.00) | 85.87 (0.00) |
| Royal National Orthopaedic Hospital | 1 (0.6) | 1.00 (–) | 32.90 (–) | 28.62 (–) |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital (AJ and RH) | 1 (0.6) | 7.00 (–) | 32.90 (–) | 200.36 (–) |
| South Tees Hospitals NHS Foundation Trust | 1 (0.6) | 2.00 (–) | 32.90 (–) | 57.25 (–) |
| University Hospital Llandough | 4 (2.3) | 2.00 (0.71) | 32.90 (0.00) | 57.24 (20.24) |
| Glasgow Royal Infirmary | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Wrexham Maelor Hospital | 1 (0.6) | 1.00 (–) | 32.90 (–) | 28.62 (–) |
| King’s College Hospital | 2 (1.1) | 2.00 (0.00) | 32.17 (0.72) | 55.98 (1.27) |
| North Bristol NHS Trust | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Spire Healthcare | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
A total of 225 out of the 1219 PHT appointments offered were missed by 94 patients (53% of the PHT sample), giving an overall non-attendance rate of 18.5%. Among the 94 patients who missed an appointment, the mean number of appointments missed was 2.3 per patient, the mean total duration of all appointments missed was 75.4 minutes and the mean total cost of missed physiotherapy time was £65.60 per patient. The mean cost of PHT was £192.16 per patient, including missed appointments, and £155.01 per patient, excluding missed appointments. Among those attending six or more consultations, the mean cost of PHT was £196.56 per patient, including missed appointments, and £193.65 per patient, excluding missed appointments.
Resource use and cost of arthroscopic surgery for FAI
Resource use data associated with the delivery of arthroscopic surgery for FAI were collected for 47 (27%) of the 173 patients in the surgery group and one patient in the PHT group across 18 (82%) of the 22 orthopaedic centres participating in the study. (Note that the one patient in the PHT group was randomised to PHT, but had surgery immediately after the 12-month follow-up period had ended.) Six (13%) of 46 patients were excluded because data on duration of surgery and/or post-surgical inpatient length of stay were missing. The mean age among the remaining 40 patients included in the micro-costing of the surgery sample was 34 (range 18–54) years, 60% were male and 68% had the cam impingement type. Compared with participants in the surgery arm of the trial and who were not included in the micro-costing sample (Table 30), those patients included were, on average, 2 years younger (mean age 34 vs. 35 years; p = 0.405), as likely to be male (60% vs. 58% male; p = 0.997) and had similar presentation with respect to type of impingement (68% vs. 78% cam; p = 0.118).
| Characteristic | Included in surgery costing study | ||
|---|---|---|---|
| Yes (N = 40) | No (N = 132) | p-value | |
| Age (years) | |||
| Mean (SD) | 34 (10) | 36 (10) | 0.405 |
| Median (IQR) | 32 (27–43) | 36 (29–42) | |
| Sex, n (%) | |||
| Female | 16 (40) | 55 (42) | 0.997 |
| Male | 24 (60) | 77 (58) | |
| Impingement type, n (%) | |||
| Cam | 27 (68) | 103 (78) | 0.118 |
| Mixed | 7 (18) | 22 (17) | |
| Pincer | 6 (15) | 7 (5) | |
Estimates of resource use associated with the surgery and sources of unit cost data for resource inputs are presented in Appendix 3, Table 34 and Table 48, respectively. Resource categories included operating theatre use/time, clinical staff, anaesthetic drugs, disposable surgical equipment and implants, and inpatient length of stay for post-surgical recuperation. The mean duration of surgery was 2.12 (range 1–3) hours and the mean length of inpatient stay was 1.6 (range 1–3) days (see Appendix 3, Table 42). The composition of the surgical team/staff remained broadly similar across centres and consisted of two surgeons (a consultant and an assistant or registrar), one anaesthetist, a radiographer, one or two nurses, two operating department practitioners and a health-care assistant.
Unit costs of clinical staff time were obtained from the PSSRU Unit Costs of Health and Social Care 201652 services and ranged from £28 per hour for a health-care assistant to £137 per hour for consultant surgeon (including qualifications and overheads). The running cost of an operating theatre was estimated based on data published by the Information Services Scotland. 54 The Information Services Scotland data showed that a total of £157,150,194.90 was allocated to operating theatres across Scotland for the 2015–16 financial year. The total number of theatre hour use recorded for the same period was 526,145.14 hours, generating a running cost of £298.68 per hour (see Appendix 3, Table 42). Allocated costs included non-clinical staff costs, property and equipment maintenance, domestics and cleaning, heating, lighting and power, and capital charges (e.g. the purchase of new equipment). 55 Equivalent data on the running costs of operating theatres in England and Wales were not publicly available and so only the Scottish data were used in our cost calculations. An inpatient stay was assumed to cost £332.77 per day, which is the excess bed-day cost for elective orthopaedics procedures in the 2016 reference costs schedules. 77 The unit costs of anaesthetic drugs were obtained from prescription cost analysis database,53 electronic searches of the BNF58 and searches of the literature, when necessary. Unit cost of syringes and needles and other medical consumables were obtained from online sources when more direct NHS sources were unavailable (see Appendix 3, Table 42).
The quantity and unit costs of disposal equipment and implants such as blades, shavers, burrs and sutures used during surgery are presented in Appendix 3, Table 44. The cost of implements ranged from £35 per item for a Smith & Nephew-supplied banana blade (Smith & Nephew, London, UK) to £349.32 per item for a TAC-S radio-frequency probe (Smith & Nephew), and were primarily extracted from the NHS supply chain catalogue held within hospital finance and procurement departments. 56
Cost estimates for the surgery resource use are summarised in Table 31 by resource category and recruitment centre. The mean cost for the surgery ranged from £2286 per patient at North Bristol NHS Trust to £4076 per patient at the Royal National Orthopaedic Hospital. Across all the centres, the overall mean cost was £3042, 35.3% of which was staffing costs, 23.5% disposal surgical equipment and implants, 19.4% theatre running costs, 17.8% inpatient costs and 4% represent the cost of anaesthesia, including drugs, syringes and needles. These figures represent estimates associated with delivery of hip arthroscopy and do not account for pre- and post-surgery consultations, diagnostic scans and post-surgical rehabilitation costs. These additional costs were, however, taken into account in the cost-effectiveness analysis as follow-up costs if they were reported by trial participants at the 6- and 12-month assessment points.
| Centre (number of patients) | Mean cost (£) (SE) | |||||
|---|---|---|---|---|---|---|
| Equipment | Staff | Theatre running costs | Anaesthetic drugs and disposables | Inpatient stay | Total | |
| University Hospitals Coventry and Warwickshire (n = 7) | 1083 (155) | 1154 (96) | 626 (45) | 122 (0) | 711 (0) | 3695 (202) |
| Yeovil District Hospital (n = 2) | 458 (32) | 1119 (172) | 571 (86) | 122 (0) | 711 (0) | 2980 (290) |
| Royal Devon and Exeter Hospital (n = 1) | 495 (–) | 845 (–) | 606 (–) | 122 (–) | 711 (–) | 2779 (–) |
| Royal Orthopaedic Hospital (n = 0)a | – | – | – | – | – | – |
| Frimley Park Hospital (n = 1) | 502 (–) | 558 (–) | 358 (–) | 122 (–) | 355 (–) | 1895 (–) |
| Guy’s and St Thomas’ Hospital (n = 1) | 657 (–) | 1244 (–) | 728 (–) | 122 (–) | 355 (–) | 3105 (–) |
| University College Hospital (n = 3) | 902 (305) | 1197 (180) | 674 (96) | 122 (0) | 592 (118) | 3487 (506) |
| Wrightington Hospital (n = 3) | 856 (221) | 1260 (74) | 749 (40) | 122 (0) | 355 (0) | 3343 (200) |
| Northumbria Healthcare NHS Foundation Trust (n = 2) | 685 (7) | 958 (404) | 515 (157) | 122 (0) | 355 (0) | 2634 (567) |
| Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust (n = 6) | 522 (122) | 1058 (126) | 564 (61) | 122 (0) | 533 (79) | 2798 (256) |
| Royal National Orthopaedic Hospital (n = 1) | 734 (–) | 1556 (–) | 598 (–) | 122 (–) | 1066 (–) | 4076 (–) |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital (AJ and RH) (n = 1) | 1014 (–) | 1097 (–) | 753 (–) | 122 (–) | 711 (–) | 3697 (–) |
| South Tees Hospitals NHS Foundation Trust (n = 2) | 925 (380) | 1172 (78) | 587 (0) | 122 (0) | 355 (0) | 3161 (458) |
| University Hospital Llandough (n = 1) | 563 (98) | 970 (77) | 564 (32) | 122 (0) | 355 (0) | 2574 (200) |
| Wrexham Maelor Hospital (n = 1) | 446 (–) | 1446 (–) | 846 (–) | 122 (–) | 711 (–) | 3570 (–) |
| King’s College Hospital (n = 2)a | – | – | – | – | – | – |
| North Bristol NHS Trust (n = 4) | 429 (145) | 827 (154) | 465 (76) | 122 (0) | 444 (89) | 2286 (448) |
| Spire (n = 1) | 843 (–) | 455 (–) | 282 (–) | 122 (–) | 711 (–) | 2412 (–) |
| Spire Healthcare (n = 40) | 719 (58) | 1067 (47) | 591 (23) | 122 (0) | 542 (31) | 3042 (116) |
Follow-up resource use and costs
Descriptive summaries of self-reported health and social care service use are presented in Appendix 3, Table 45, by assessment point, resource category and treatment group. The proportion of missing data across all categories of resource inputs ranged from 1.2% at baseline to 17.5% at 12 months post randomisation in the surgery group and from 0.6% at baseline to 26.6% at 12 months post randomisation in the PHT group. Across the majority of resource categories, the baseline values displayed in Appendix 3, Table 45, suggest no more than 8.2% and 10.7% of the patients in surgery and PHT groups, respectively, reported use of health and social care services in the 3-month period prior to randomisation. The only exceptions were GP attendance, medication use and orthopaedic outpatient attendance in the same period. Slightly over one-third of patients (36% in the surgery group and 41% in the PHT group) reported attending at least one GP consultation, with a similar proportion of patients reporting being prescribed medication and 57% attending an orthopaedic outpatient appointment. Overall, the treatment groups were generally balanced with respect to reported resource use across all categories of health and social care services at baseline.
Rates of service use over 12 months of follow-up were also broadly similar between the two treatment groups for most resource categories. However, at the 6-month assessment point, the surgery group reported, on average, 0.035 (95% CI 0.011 to 0.118; p = 0.038) more inpatient hospitalisation days for the category ‘other inpatient admissions’, 0.362 (95% CI 0.116 to 1.032; p = 0.016) more orthopaedic outpatient consultations, 0.499 (95% CI 0.224 to 1.588; p = 0.002) more outpatient physiotherapy attendance for the category ‘your hip/leg’, and significant use of prescribed medication, walking aids and adaptations, than the PHT group. At the 12-month assessment point, the surgery group also reported, on average, 0.596 (95% CI 0.089 to 2.544; p = 0.002) more inpatient days, 0.041 (95% CI 0.007 to 0.129; p = 0.030) more day case admissions and 1.789 (95% CI 1.379 to 4.554; p < 0.001) more outpatient physiotherapy attendances than the PHT group. Compared with the PHT group, attendance rates at outpatient orthopaedic and physiotherapy clinics were also higher among the surgery group at 12 months post randomisation, as were the use of clutches, dressing aids and number of prescriptions received.
Unit cost of resource inputs together with corresponding sources of unit costs are presented in Appendix 3, Table 46. Appendix 3, Table 47, presents health and social service costs generated by assigning the unit cost data to resource inputs summarised by resource category, treatment group and assessment point. The mean total cost across all categories of resource use at baseline, covering the 3-month period prior to randomisation, was £502.12 in the surgery group and £508.53 in the PHT group, generating an unadjusted mean total cost difference of –£6.41 (95% CI –£235.01 to £246.59; p = 0.880). Total cost across all resource categories based on the 6- and 12-month data was generally higher in the surgery group than in the PHT group, but the unadjusted between-group difference in costs was only statistically significant at the 6-month assessment point. Over the 12 months of follow-up, the mean total cost was £1640.91 in the surgery group and £941.02 in the PHT group, generating a statistically significant unadjusted cost difference of £699.88 (95% CI £274.36 to £1121.23; p < 0.001).
Private health expenditure, lost income and other costs
Private health-care service use and related out-of-pocket expenses by patients, friends and family members in the 3-month period prior to randomisation and the 12-month post-randomisation period are summarised in Appendix 3, Tables 48 and 49, respectively. The proportion of missing data across all categories of resource inputs ranged from 1.2% at baseline to 18.1% and 17.5% at 6 and 12 months post randomisation, respectively, in the surgery group and from 0.6% at baseline to 26.6% at 12 months post randomisation in the PHT group. Private health-care services included private physiotherapy consultations, purchase of over-the-counter medication and purchase of walking aids, such as crutches, sticks and specialised shoes. There was no statistically significant difference between the two treatment groups across all categories of private health-care utilisation at baseline and over the 12 months of follow-up (see Appendix 3, Table 48). The mean cost of private health utilisation in the 3-month period prior to randomisation (baseline figures) was £10.89 in the surgery group and £14.95 in the PHT group, generating an unadjusted cost difference of –£4.06 (95% CI –£25.07 to £11.16; p = 0.760) (see Appendix 3, Table 49). Over the 12 months of follow-up, private health-care and out-of-pocket expenditure increased from the baseline figures to about £32.67, on average, in the surgery group and £27.44 in the PHT group, generating a mean cost difference of £5.23 (95% CI –£23.55 to £32.41; p = 0.704).
Appendix 3, Table 46, presents a summary of additional (indirect or non-health service) costs, for example the value of lost income incurred by patients and their families for the 3-month period prior to randomisation (baseline figures) and over the 12-month period of follow-up. Only costs incurred as a result of inability to work or perform tasks because of their hip pain were included in these additional (indirect) cost calculations. These costs included lost earnings, help with house work, child care and expenditure on specialised equipment. The mean additional cost reported by patients for the 3-month period prior to randomisation was £164.15 in the surgery group and £111.49 in the PHT group, generating a mean additional cost difference of £52.66 (95% CI –£189.36 to £470.01; p = 0.748). Over the 12 months of follow-up, the corresponding costs reported increased to £1143.20 in the surgery group and to £184.81 in the PHT group, generating a cost difference of £958.39 (95% CI £219.74 to £2001.32; p = 0.004). Lost income accounted for approximately 92% of the additional costs in the surgery group, but only 60% of additional costs in the PHT group at baseline. Over the 12 months of follow-up, lost income accounted for approximately 83% and 76% of the additional costs in the surgery and PHT groups, respectively.
Total economic costs
Table 32 summarises the total costs during the 12-month follow-up period by treatment group, cost category and cost perspective. Among the complete cases, the mean total cost from a NHS and Personal Social Service Perspective was £3712.77 in the surgery group and £1282.93 in the PHT group, generating an unadjusted cost difference of £2429.84 (95% CI £1865.92 to £2911.70; p < 0.001). Surgery costs accounted for approximately 72% of total costs in the surgery group, whereas the treatment costs (including surgery costs for PHT patients who had surgery) accounted for only 28% of the overall costs in the PHT group. The corresponding mean total cost estimated from a societal perspective was £4993.84 in the surgery group, of which 53.5% was surgery costs, and £1481.96 in the PHT group, of which 24% was accounted for by treatment costs, generating an unadjusted cost difference of £3511.88 (95% CI £2490.57 to £4784.53; p < 0.001).
| Costing perspective | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| % missing | % zero costs | Mean (£) (SE) | % missing | % zero costs | Mean (£) (SE) | Mean difference (£) (bootstrap 95% CI)a | p-valueb | |
| NHS/Personal Social Services perspective | ||||||||
| Treatment costs | 1.2 | 15.8 | 2673.57 (83.52) | 0.0 | 0.0 | 352.88 (81.61) | 2320.69 (2101.32 to 2550.03) | < 0.001 |
| Follow-up costs | 33.3 | 5.3 | 884.53 (149.28) | 40.7 | 7.3 | 950.64 (155.54) | –66.11 (–555.66 to 334.16) | 0.818 |
| Total NHS/Personal Social Services costs | 33.3 | 1.2 | 3712.77 (177.02) | 40.7 | 0.0 | 1282.93 (184.45) | 2429.84 (1865.92 to 2911.70) | < 0.001 |
| Societal perspective | ||||||||
| Treatment costs | 1.2 | 15.8 | 2673.57 (83.52) | 0.0 | 0.0 | 352.88 (81.61) | 2320.69 (2096.77 to 2546.05) | < 0.001 |
| Follow-up costs (NHS/Personal Social Services) | 33.3 | 5.3 | 884.53 (149.28) | 40.7 | 7.3 | 950.64 (155.54) | –66.11 (–547.32 to 338.69) | 0.844 |
| Follow-up costs (non-NHS/Personal Social Services) | 33.3 | 19.9 | 1281.08 (376.1) | 40.7 | 22.6 | 199.03 (391.89) | 1082.04 (233.70 to 2236.23) | 0.002 |
| Total societal costs | 33.3 | 1.2 | 4993.84 (418.83) | 40.7 | 0.0 | 1481.96 (436.41) | 3511.88 (2490.57 to 4784.53) | < 0.001 |
Benefit payments
Patient self-reports of benefit payments, pensions and statutory sick pay are presented in Appendix 3, Table 51. The proportion of patients with missing data on benefit payments ranged from 2.3% at baseline to 17% and 14% at the 6- and 12-month assessment points, respectively, in the surgery group and from 0.6% at baseline to 20.3% and 22% at 6 and 12 months post randomisation in the PHT group. Examples of benefit payments that were reported during follow-up include Attendance Allowance, Income Support, Housing Benefit, Carer’s Allowance, Child Tax Credit, Council Tax Reduction, Employment and Support Allowance, and Disability Living Allowance. The proportion of patients in receipt of at least one of these payments increased from 10.6% at baseline to 17.6% over the 12 months of follow-up in the surgery group, but decreased in the PHT group from 6.2% at baseline to 5.7% over the 12 months of follow-up. The average reported payment per patient at baseline was £17.62 in the surgery group and £11.90 in the PHT group, generating a between-group difference of £5.72 (95% CI –£20.89 to £36.78; p = 0.512) in the 3-month period prior to randomisation. Over the 12 months of follow-up, receipts increased from baseline values to £57.93 in the surgery group and £12.94 in the PHT group, generating an unadjusted mean difference of £44.99 (95% CI £10.88 to £97.42; p < 0.001).
Health-related quality of life
Of the 351 patients randomised, the first 97 patients who were recruited into the feasibility study sample completed the EQ-5D-3L questionnaire, whereas the remaining 251 patients were recruited into the main study sample and completed the EQ-5D-5L questionnaire. In addition, health-related quality-of-life data were collected for both feasibility and main trial samples using version 2 of the SF-12 questionnaire. Appendix 3, Tables 52–54, present descriptive summaries of the responses to these questionnaires by treatment group and assessment point. Treatment groups were balanced with respect to function (i.e. optimal vs. suboptimal response to each health dimension) at baseline and there were no statistically significant differences between the two groups in the distribution of responses across all health dimensions and assessment points.
The utility weights generated from the EQ-5D-3L/EQ-5D-5L and the SF-6D (via the SF-12) are summarised in Table 33 by questionnaire instrument, assessment point and treatment group. The proportion of missing data ranged from 0% at baseline to no more than 20% and 15% at the 6- and 12-month assessment points, respectively, in the surgery group and from 0% to no more than 21.4% and 25.5% at the 6- and 12- month assessment points, respectively, in the PHT group. On average, patients in the surgery group had higher utility scores at baseline based on the responses to the EQ-5D-5L and lower scores based on the responses to the EQ-5D-3L and the SF-12, but between-group differences in baseline scores were not statistically significant for all measures. Among patients with complete data, the mean unadjusted utilities generated from the EQ-5D-3L/EQ-5D-5L increased from 0.58 at baseline to 0.59 and 0.67 at 6 and 12 months post randomisation, respectively, in the surgery group and from 0.56 at baseline to 0.62 at both 6 and 12 months post randomisation in the PHT group. The corresponding utility values generated from the SF-6D UK tariff increased from 0.64 at baseline to 0.65 and 0.69 at 6 and 12 months post randomisation, respectively, in the surgery group and from 0.64 at baseline to 0.66 and 0.68 at 6 and 12 months post randomisation, respectively, in the PHT group. 46 There was no statistically significant between-group difference at baseline across all quality-of-life measures, but the surgery group, on average, reported worse EQ-5D-3L/EQ-5D-5L utility scores at 6 months (unadjusted mean difference –0.012, 95% CI –0.04 to 0.015) and improved scores at 12 months after randomisation (unadjusted mean difference 0.049, 95% CI –0.01 to 0.108; p = 0.105) (see Table 33).
| Outcome | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery | PHT | |||||||
| n | % missing | Mean (SE) | n | % missing | Mean (SE) | Mean difference (95% CI) | p-value | |
| EQ-5D-3L/EQ-5D-5L crosswalka | ||||||||
| Baseline | 171 | 1.8 | 0.575 (0.02) | 177 | 0.6 | 0.557 (0.019) | 0.018 (–0.035 to 0.072) | 0.5 |
| 6 months | 171 | 17.5 | 0.586 (0.022) | 177 | 17.5 | 0.617 (0.022) | –0.031 (–0.092 to 0.031) | 0.33 |
| 12 months | 171 | 11.7 | 0.671 (0.021) | 177 | 17.5 | 0.622 (0.021) | 0.049 (–0.01 to 0.108) | 0.105 |
| SF-12 (SF-6D UK tariff) | ||||||||
| Baseline | 171 | 1.8 | 0.639 (0.009) | 177 | 0.6 | 0.642 (0.009) | –0.003 (–0.028 to 0.021) | 0.797 |
| 6 months | 171 | 14.6 | 0.648 (0.01) | 177 | 20.3 | 0.659 (0.01) | –0.011 (–0.04 to 0.017) | 0.421 |
| 12 months | 171 | 15.2 | 0.69 (0.01) | 177 | 25.4 | 0.683 (0.011) | 0.007 (–0.023 to 0.037) | 0.644 |
| EQ-5D-3L | ||||||||
| Baseline | 46 | 4.3 | 0.529 (0.046) | 51 | 2.0 | 0.555 (0.044) | –0.026 (–0.153 to 0.1) | 0.68 |
| 6 months | 46 | 10.9 | 0.602 (0.049) | 51 | 9.8 | 0.611 (0.046) | –0.008 (–0.142 to 0.125) | 0.9 |
| 12 months | 46 | 15.2 | 0.621 (0.052) | 51 | 23.5 | 0.664 (0.052) | –0.043 (–0.189 to 0.103) | 0.558 |
| EQ-5D-5L (crosswalk tariff) | ||||||||
| Baseline | 125 | 0.8 | 0.591 (0.021) | 126 | 0.0 | 0.557 (0.02) | 0.034 (–0.023 to 0.091) | 0.239 |
| 6 months | 125 | 20.0 | 0.579 (0.024) | 126 | 20.6 | 0.619 (0.024) | –0.04 (–0.108 to 0.028) | 0.247 |
| 12 months | 125 | 10.4 | 0.688 (0.022) | 126 | 15.1 | 0.606 (0.022) | 0.082 (0.02 to 0.143) | 0.01 |
| EQ-5D-5L (new UK tariff) | ||||||||
| Baseline | 125 | 0.8 | 0.7 (0.02) | 126 | 0.0 | 0.669 (0.019) | 0.031 (–0.023 to 0.085) | 0.262 |
| 6 months | 125 | 20.0 | 0.691 (0.022) | 126 | 20.6 | 0.724 (0.022) | –0.033 (–0.096 to 0.029) | 0.294 |
| 12 months | 125 | 10.4 | 0.782 (0.021) | 126 | 15.1 | 0.702 (0.022) | 0.08 (0.02 to 0.139) | 0.009 |
| EQ-5D-3L VAS | ||||||||
| Baseline | 46 | 0.0 | 61.587 (3.037) | 51 | 2.0 | 68.9 (2.913) | –7.313 (–15.668 to 1.042) | 0.086 |
| 6 months | 46 | 6.5 | 64.302 (3.339) | 51 | 9.8 | 68.022 (3.228) | –3.719 (–12.95 to 5.511) | 0.425 |
| 12 months | 46 | 13.0 | 67.375 (3.391) | 51 | 15.7 | 70.953 (3.27) | –3.578 (–12.952 to 5.795) | 0.45 |
| EQ-5D-5L VAS | ||||||||
| Baseline | 125 | 0.0 | 69.168 (1.685) | 126 | 0.0 | 66.556 (1.678) | 2.612 (–2.071 to 7.296) | 0.273 |
| 6 months | 125 | 18.4 | 69.324 (1.787) | 126 | 21.4 | 71.323 (1.813) | –2 (–7.02 to 3.02) | 0.433 |
| 12 months | 125 | 12.0 | 73.545 (1.857) | 126 | 19.0 | 68.402 (1.928) | 5.143 (–0.134 to 10.421) | 0.056 |
QALY values were generated from combining health-related quality of life weights (measured at baseline and at 6 and 12 months post randomisation) over the 12 months of follow-up using area-under-the-curve approaches (see Table 35). Mean QALYs based on the combined EQ-5D-3L/EQ-5D-5L utility score were 0.617 in the surgery group and 0.613 in the PHT, generating an unadjusted difference of 0.005 (–0.046 to 0.055, p-value = 0.859) QALYs over the 12 months of follow-up. Unadjusted QALY differences between the surgery and PHT groups generated for the feasibility and the main trial population when variants of the EQ-5D measure were applied, and separately for the whole trial population when SF-6D utility values were applied, were not statistically significant.
Cost-effectiveness results
Base-case analysis results
Table 34 presents estimates of the cost-effectiveness of hip arthroscopy compared with PHT for FAI. In the base-case analysis, surgery was associated with an adjusted mean additional cost of £2483 (95% CI £1533 to £3432) and an adjusted mean additional QALY of –0.018 (95% CI –0.051 to 0.015) per patient compared with PHT over the 12 months of follow-up. On average, surgery was more costly and marginally less effective than PHT in the adjusted analysis during the first year of follow-up. The mean base-case ICER was –£140,361 per QALY gained for surgery compared with PHT. Figure 14 shows the uncertainty around this central estimate of the ICER. Figure 14a displays 1000 simulated replicates of the ICER on a cost-effectiveness plane and Figure 14b displays the probability that surgery is cost-effective compared with PHT for a range of cost-effectiveness thresholds. Almost all simulated replicates of the ICER fell on the left-hand side of the £30,000 and £50,000 per QALY cost-effectiveness threshold lines, with the central estimate (indicated by the black diamond) falling in the north-west quadrant. This suggests that surgery is unlikely to be cost-effective at the £20,000–£30,000 per QALY threshold range (see Figure 14b) that NICE currently uses to determine the cost-effectiveness of health technologies. 67 Figure 14b shows that the probability that surgery is cost-effective compared with PHT is close to zero for threshold values < £100, 0000 per QALY.
| Description | Cost-effectiveness outcome | Probability surgery is cost-effective at cost-effectiveness threshold of | |||||
|---|---|---|---|---|---|---|---|
| Mean incremental costs (£) (95% CI) | Mean incremental QALYs (95% CI) | ICERa | £13,000/QALY | £20,000/QALY | £30,000/QALY | £50,000/QALY | |
| Base-case analysisb | 2483 (1533 to 3432) | –0.018 (–0.051 to 0.015) | –140,361 | 0 | 0 | 0 | 0 |
| Sensitivity analyses | |||||||
| Unadjusted analysis | 2515 (1581 to 3450) | 0.001 (–0.048 to 0.049) | 4,196,009 | 0 | 0 | 0.005 | 0.05 |
| Adjusted complete-case analysis | 2425 (2043 to 2807) | –0.017 (–0.050 to 0.017) | –144,799 | 0 | 0 | 0 | 0 |
| Per-protocol samplec | 2689 (1627 to 3750) | –0.013 (–0.050 to 0.023) | –205,243 | 0 | 0 | 0 | 0 |
| Per-protocol sampled | |||||||
| Assume surgery costs are £2680e | 2498 (1551 to 3444) | –0.018 (–0.052 to 0.016) | –136,562 | 0 | 0 | 0 | 0 |
| Assume surgery costs are £5811f | 5628 (4682 to 6575) | –0.018 (–0.052 to 0.016) | –307,703 | 0 | 0 | 0 | 0 |
| Societal costs | 3689 (2140 to 5238) | –0.023 (–0.056 to 0.010) | –160,577 | 0 | 0 | 0 | 0 |
| SF-12/SF-6D | 2431 (1500 to 3362) | –0.003 (–0.018 to 0.012) | –779,664 | 0 | 0 | 0 | 0 |
| Subgroups | |||||||
| Feasibility sample (EQ-5D-3L) | 2234 (1567 to 2901) | –0.006 (–0.074 to 0.062) | –368,113 | 0 | 0 | 0.02 | 0.077 |
| Main study sample (EQ-5D-5L crosswalk value set) | 2614 (1245 to 3984) | –0.019 (–0.058 to 0.020) | –134,395 | 0 | 0 | 0 | 0.001 |
| Main study sample (EQ-5D-5L new UK value set) | 2719 (1225 to 4214) | –0.016 (–0.049 to 0.016) | –165,227 | 0 | 0 | 0 | 0.001 |
| Impingement type: cam | 2327 (1905 to 2749) | –0.006 (–0.042 to 0.029) | –359,783 | 0 | 0 | 0 | 0.002 |
| Impingement type: pincer/mixed | 2992 (–694 to 6679) | –0.020 (–0.101 to 0.061) | –149,445 | 0.062 | 0.062 | 0.073 | 0.1 |
| Restricted analysis to women only | 2047 (1248 to 2846) | –0.013 (–0.066 to 0.039) | –151,606 | 0 | 0 | 0.003 | 0.027 |
| Restricted analysis to men only | 2826 (1297 to 4354) | –0.012 (–0.053 to 0.029) | –241,044 | 0.002 | 0.003 | 0.005 | 0.013 |
FIGURE 14.
Base-case analysis comparing the cost-effectiveness of arthroscopic surgery with PHT for FAI. The analysis accounted for missing data using multiple imputation and adjusting for age, sex and baseline health-related quality of life (effectiveness regression). (a) Incremental costs; and (b) probability that surgery is cost-effectiveness. WTP, willingness to pay.
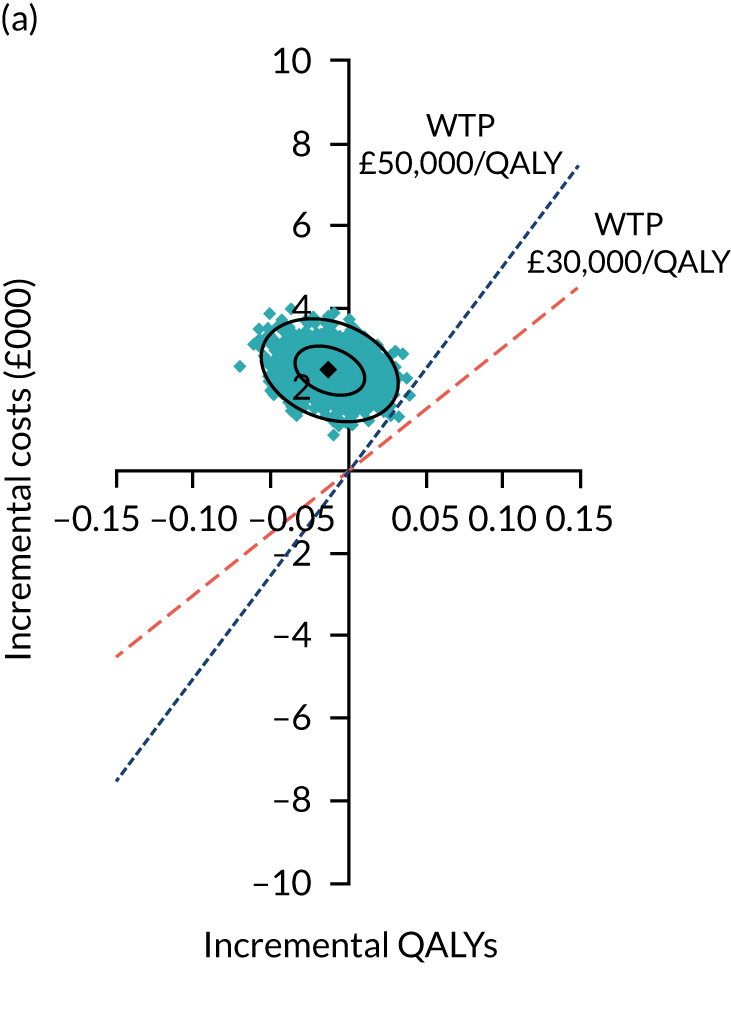
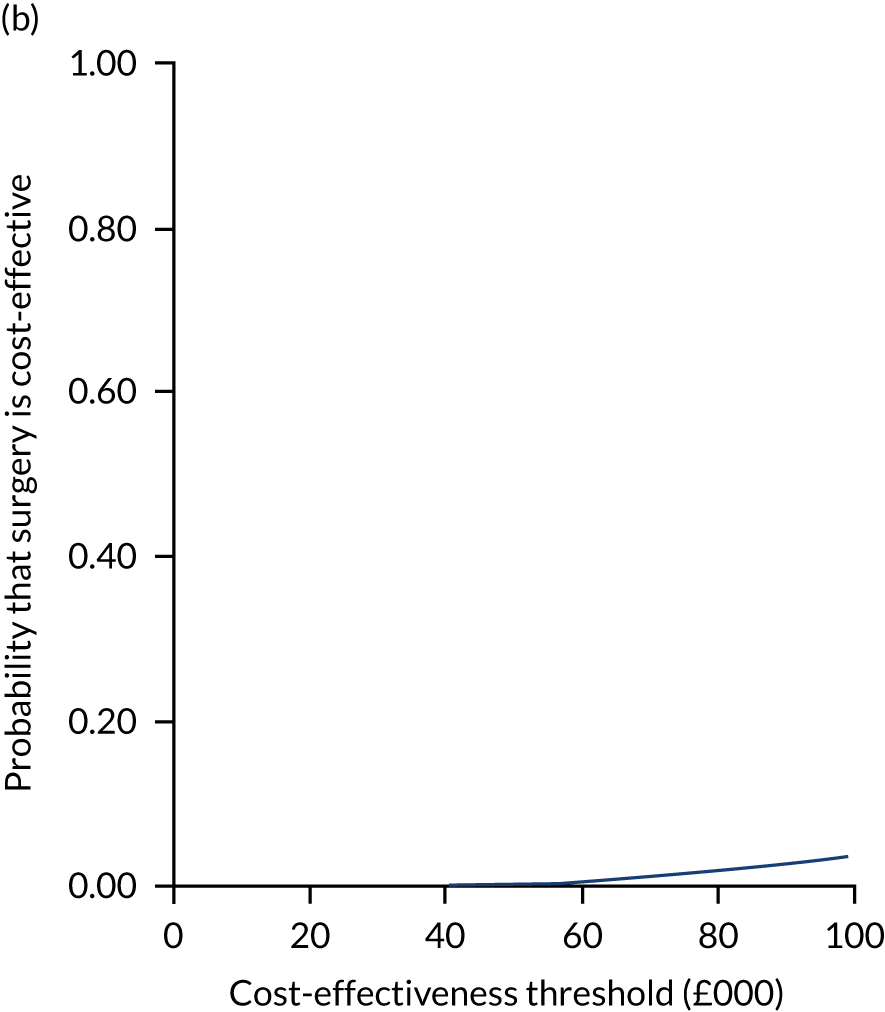
Mean incremental net (monetary) benefit was also generated from the 1-year data for different cost-effectiveness thresholds (Table 35). The mean incremental net benefit was –£2713 if the cost-effectiveness threshold was £13,000 per QALY gained, –£3013 if the cost-effectiveness threshold was £30,000 per QALY gained and –£3367 if the cost-effectiveness threshold was £50,000 per QALY gained. These values (and the associated upper and lower 95% CI confidence limits) are all negative and suggest a net loss to the health and social care service based on the 1-year data if surgery is adopted (or a net gain by the same amount, on average, if PHT is adopted).
| Description | Mean incremental net (monetary) benefit (£ 2016 prices) and 95% CIs at cost-effectiveness threshold of | |||
|---|---|---|---|---|
| £13,000/QALY | £20,000/QALY | £30,000/QALY | £50,000/QALY | |
| Base-case analysisa | –2713 (–3842 to –1583) | –2836 (–4125 to –1548) | –3013 (–4566 to –1460) | –3367 (–5523 to –1211) |
| Sensitivity analyses | ||||
| Unadjusted analysis | –2507 (–3748 to –1267) | –2503 (–4012 to –994) | –2497 (–4441 to –554) | –2485 (–5375 to 405) |
| Adjusted complete-case analysis | –2642 (–3246 to –2038) | –2759 (–3561 to –1957) | –2927 (–4038 to –1816) | –3262 (–5023 to –1500) |
| Per-protocol sampleb | –2859 (–4136 to –1582) | –2951 (–4378 to –1523) | –3082 (–4768 to –1396) | –3344 (–5632 to –1056) |
| Per-protocol samplec | ||||
| Assume surgery costs are £2680d | –2736 (–3869 to –1602) | –2864 (–4165 to –1562) | –3047 (–4624 to –1469) | –3412 (–5608 to –1216) |
| Assume surgery costs are £5811e | –5866 (–7000 to –4732) | –5994 (–7296 to –4692) | –6177 (–7755 to –4599) | –6543 (–8739 to –4347) |
| Societal costs | –3988 (–5678 to –2298) | –4148 (–5931 to –2366) | –4378 (–6332 to –2424) | –4838 (–7232 to –2443) |
| SF-12/SF-6D | –2472 (–3426 to –1517) | –2493 (–3479 to –1508) | –2525 (–3569 to –1480) | –2587 (–3791 to –1382) |
| Subgroups | ||||
| Feasibility sample (EQ-5D-3L) | –2313 (–3501 to –1125) | –2355 (–3972 to –739) | –2416 (–4683 to –150) | –2537 (–6148 to 1073) |
| Main study sample (EQ-5D-5L crosswalk value set) | –2867 (–4391 to –1343) | –3003 (–4668 to –1339) | –3198 (–5114 to –1282) | –3587 (–6115 to –1059) |
| Main study sample (EQ-5D-5L new UK value set) | –2933 (–4567 to –1300) | –3049 (–4794 to –1304) | –3213 (–5155 to –1272) | –3542 (–5966 to –1119) |
| Impingement type: cam | –2411 (–3079 to –1743) | –2456 (–3337 to –1576) | –2521 (–3736 to –1306) | –2651 (–4570 to –731) |
| Impingement type: pincer/mixed | –3253 (–7342 to 837) | –3393 (–7759 to 974) | –3593 (–8436 to 1250) | –3993 (–9989 to 2002) |
| Restricted analysis to women only | –2222 (–3284 to –1160) | –2317 (–3666 to –967) | –2452 (–4275 to –628) | –2722 (–5577 to 134) |
| Restricted analysis to men only | –2978 (–4748 to –1208) | –3060 (–4993 to –1128) | –3177 (–5390 to –965) | –3412 (–6287 to –537) |
Sensitivity analyses results
Only the unadjusted analysis generated a difference in mean QALYs of 0.001 in favour of the surgery. The probability that surgery is cost-effective was 0.005 at £30,000 per QALY and no more than 0.05 at £50,000 per QALY (see Table 34). All other sensitivity analyses adjusted for baseline characteristics, such as age, sex, impingement type, study site, health-care service use prior to randomisation and health-related quality of life. In the adjusted sensitivity analyses, surgery was significantly more expensive (adjusted mean difference in costs ranged from £2047 to £5628) and generated fewer QALYs (adjusted mean difference in QALYs ranged from –0.018 to –0.003), on average, than PHT over 12 months of follow-up (see Table 34). Cost-effectiveness acceptability curves and plots of simulated ICERs, displayed in Appendix 3, Figures 15–21, for the different scenarios evaluated through the sensitivity analyses, show that surgery is unlikely to be cost-effective even at willingness-to-pay threshold values as high as £100,000 per QALY.
Subgroup analyses results
The subgroup analyses results are also summarised in Tables 34 and 35 and displayed graphically in Appendix 3, Figures 22–28. There was substantial uncertainty around the central estimates of incremental costs and incremental QALYs because of the reduced sample size in each subgroup, but the direction of relative cost-effectiveness of the interventions remained mostly the same as in the base-case analysis. Across all subgroups of patients, surgery mostly generated fewer QALYs (adjusted mean difference in QALYs ranged from –0.019 to –0.006) and was significantly more expensive (adjusted mean difference in costs ranged from £2058 to £3260), on average, than PHT.
Long-term modelling
The study protocol had allowed for trial participants to be followed up beyond the initial 12-month period for up to 3 years and outcome data collected at the end of the second and third year post randomisation. Given that the 12-month within-trial economic analysis did not show evidence of cost-effectiveness in favour of the surgery, it is doubtful if long-term economic modelling would be meaningful without this additional data. In addition, 11 patients representing 7.3% of the PHT group had crossed over and received surgery during the 12-month follow-up period. The net effect of patients crossing over to surgery may increase costs in the PHT arm and decrease the incremental costs between surgery and PHT. If more and more PHT patients continue to cross over to surgery in subsequent years, then they will be picked up at the second- and third-year assessments. Therefore, an assessment of the utility of a long-term economic model should be delayed until the second- and third-year data become available. This would provide a more accurate assessment of outcomes over a longer follow-up period and determine whether or not modelling is needed to capture the long-term (i.e. lifetime) costs and consequences of treatment.
Chapter 6 Discussion
To the best of our knowledge, the UK FASHIoN trial is the first RCT evidence that hip arthroscopy with postoperative rehabilitation is effective in patients with FAI syndrome. In this pragmatic trial, after 12 months, there was a mean difference in iHOT-33 scores of 6.8 points in favour of patients allocated to hip arthroscopy, compared with those who were allocated to a best conservative care strategy of PHT. This is a statistically significant difference that exceeds the MCID for the iHOT-33. The results are consistent with the hypothesis that hip arthroscopy is more clinically effective than best conservative care.
There have been many observational studies showing benefit from hip arthroscopy; however, these studies generally did not have control groups for comparison and were at high risk of bias. 3 Our previous systematic review16 showed that there had been no previous relevant RCTs. One RCT has recently reported no difference in outcome between hip arthroscopy and conservative care. 78 This study was small, conducted in a military setting, with a single surgeon in a single centre and with a very high rate of crossover (70%) from conservative care to hip arthroscopy. 78 When the authors performed a per-protocol comparison of those who had hip arthroscopy (n = 66) with those who had conservative care (n = 14), they reported that ‘power was lost making type II errors possible’. 78 The authors concluded that ‘large cohorts across multiple sites are needed to make definitive conclusions’. 78 This trial is much larger and, therefore, has greater power to detect between-group differences as statistically significant, and was conducted in 23 centres and in a more generalisable population. 78
There was no difference in secondary outcome measures of general health-related quality of life (i.e. EQ-5D-5L and SF-12) between PHT and arthroscopic surgery. This could either be because treatment for FAI syndrome does not have an effect on health-related quality of life or because the measures are not sufficiently sensitive to detect the changes that occur. A further possibility is that the trial was not sufficiently powered to detect changes in health-related quality of life.
Complications in the hip arthroscopy group were more frequent than in the PHT group. However, there was only one serious surgical complication in which a patient developed a deep hip infection. Hip arthroscopy is considered relatively safe, with a reported minor complication rate of 7.9% and major complication rate of 0.45% in a large systematic review. 79 The UK FASHIoN trial results are consistent with this. 80 The delay in surgical treatment may have affected the collection of complication data; but most complications from surgery occur within the first 6 weeks and complications data were available for 138 of the 144 participants who received a hip arthroscopy compared with 146 of the 154 participants who received PHT. This suggests that the delay in treatment did not cause an under-reporting of complications.
The within-trial health economic evaluation suggests that hip arthroscopy is not cost-effective in comparison with PHT. However, our economic models were able to assess cost-effectiveness at only 12 months from randomisation. This must also be set in the context of the high initial treatment costs of hip arthroscopy, the treatment timing (i.e. a long delay in hip arthroscopy, reducing the period of potential benefit during follow-up), the period of economic inactivity during the period of postoperative recovery and subsequent hip arthroscopy in one-quarter of PHT patients in the first 3 years after randomisation. There may be longer-term differences between groups that were not assessed in this economic analysis, and further follow-up points at 5 and 10 years will inform the life time cost-effectiveness of both surgery and PHT. For example, hip replacement rates appear to have diverged at 3 years and the longer-term comparison of this rate between groups will help establish whether or not surgery affects the risk of osteoarthritis and consequent societal and health-care costs. 7,81
The differences in the primary outcome reached statistical significance at 12 months and the mean value was greater than the MCID of the iHOT-33. However, the CIs overlap the MCID, raising the possibility that hip arthroscopy is superior to PHT, but not to a clinically significant amount. These differences must be set in the context of other factors, such as the greater delay to treatment and crossovers, which would be likely to reduce the reported effect of hip arthroscopy in the trial. Any benefit of hip arthroscopy over conservative care must also be weighed against the complication profile of surgery.
Strengths of this trial include the consent to participate rate among eligible patients (54%) and the follow-up rate (92%). Both of these are high compared with similar trials in orthopaedics, and especially to trials of surgery compared with no surgery, contributing to external and internal validity. 82 The integrated qualitative research optimised recruitment, as it has in other trials. 71 This trial was thoroughly pragmatic, exploring the effectiveness of a strategy of offering hip arthroscopy and conservative care in the everyday reality of an NHS where patients may not always receive or comply with the treatment they are offered, where surgeons and physiotherapists have varying levels of training, skill and expertise, where postoperative care is variable and where there are waiting lists for treatment. The large number of centres (n = 23), surgeons (n = 27) and physiotherapists (n = 43) involved in this trial is a strength, which contributes to the generalisability of our findings. The comparator for this trial was PHT, a fair representation of the best conservative care that can realistically be provided in the UK NHS for these patients. PHT was designed through international consensus and developed, supported and tested in similar ways to other physiotherapist-led conservative care protocols. 83 PHT meets the standards expected of a complex intervention in a RCT and was delivered by musculoskeletal physiotherapists who attended additional training and support events. 3,5,84
Limitations of this trial include the non-blinding of participants and treating clinicians to allocation. A blinded allocation trial, with a placebo control, would have been better suited to measuring the underlying effect of surgery. In this trial, the pragmatic research question was whether hip arthroscopy or best conservative care was the most effective treatment strategy, leading to an inevitable lack of blinding. Data collection and analysis were performed without revealing treatment allocation. An unexpected difficulty in the performance of the trial was the frequent delay in the delivery of surgery for those patients allocated to hip arthroscopy. It was anticipated that this would be < 3 months in most patients because when the trial was designed there was a strongly enforced NHS target to treat patients within 18 weeks from referral to surgery. However, during the study, this target was a challenge in many hospitals. Therefore, patients allocated to hip arthroscopy often experienced longer times to treatment and, because outcome was measured 12 months after randomisation, were often still within a few months (and in some cases a few weeks) of their operation when the primary outcome was measured. Patients in the hip arthroscopy group had, on average, less time to recover before the primary outcome measurement. A comparison of the outcome of hip arthroscopy participants who had surgery in the first 6 months after randomisation with those who had surgery in the second 6 months showed no significant difference between these groups, suggesting that the systematic difference in time to treatment between groups does not account for the treatment effect. Inferences about the effectiveness of hip arthroscopy compared with PHT are limited to 12-month post-randomisation data. Longer-term follow-up is required to establish if this effect is maintained and if further treatments are required. There will be further follow-up points at 5 and 10 years. The fact that not all surgery or PHT was deemed to be of a high fidelity is also a reflection of the real-world setting in which this trial was conducted. Some surgery was not satisfactory and some participants allocated PHT did not engage with it or complete it. The fidelity assessment showed ‘good’ treatment in 87% and 70% of hip arthroscopy and PHT groups, respectively. However, these proportions are comparable to other studies and reflect the pragmatism of the trial. 85 Crossover was minimised using techniques developed in the feasibility study. No participants allocated hip arthroscopy received PHT and 14 participants allocated to PHT subsequently changed their mind and decided to have surgery within 12 months. These crossovers cannot account for the results of the trial. Indeed, such crossovers should dilute and so reduce the estimate of the real underlying effect of hip arthroscopy. A further limitation is the timings of the collection of outcome. In both treatment groups, the mean and median data were collected at 6 and 12 months; however, the SD around this was over 30 days in both groups.
Personalised hip therapy is believed to work by improving muscle control, strength around the hip and movement patterns, leading to the avoidance of hip impingement. Surgery is thought to work by reshaping the bone to prevent impingement and by treating painful injuries to articular cartilage and labrum. In this trial, the observed effect of hip arthroscopy over conservative care might be attributable to the surgical procedure, the placebo effect of surgery (given the unblinded nature of this trial), post-surgical rehabilitation or a combination of these factors. The subgroup analysis of those with only cam morphology is suggestive of an increased treatment effect of hip arthroscopy compared with other shapes and this would support the idea that the removal of a cam shape has a specific therapeutic effect. The small number of patients with pincer or mixed cam and pincer morphology in this study leads to less confidence about the influence of reshaping the acetabular rim. Future research should focus on investigating the mechanism of benefit from both arthroscopic surgery and PHT, and on which patients, including impingement types, benefit most from hip arthroscopy or PHT. A priority for further research is an assessment to establish the longer-term clinical effectiveness and cost-effectiveness of the treatments, given the short horizon in the analysis reported.
Chapter 7 Conclusion
Hip arthroscopy in patients with FAI syndrome led to better patient-reported hip-specific outcomes at 12 months after randomisation than best conservative care, supporting the use of this technology in clinical practice. This improvement comes at some cost, and this study did not demonstrate cost-effectiveness of hip arthroscopy compared with conservative care within the first 12 months. Costs continue to be incurred in both groups up to at least 3 years, including operations in more than one-quarter of those allocated to best conservative care. The lifetime cost-effectiveness will be established only when long-term follow-up data are available. Further follow-up at 5 and 10 years, including patient-reported outcome and hip replacement rates, will provide guidance on the best long-term strategy for patients with FAI syndrome. Integrated qualitative research into recruitment was able to maximise study enrolment and may be of benefit in future surgical trials.
Acknowledgements
Contributions of authors
Damian R Griffin (https://orcid.org/0000-0003-0952-7848) (Chief Investigator, Professor, Trauma and Orthopaedic Surgery) conceived, designed and led the study, and prepared this report.
Edward J Dickenson (https://orcid.org/0000-0001-8369-1262) (Research Fellow, Trauma and Orthopaedic Surgery) designed and performed the feasibility and full studies, respectively, and prepared this report.
Felix Achana (https://orcid.org/0000-0002-8727-9125) (Research Fellow, Health Economics) performed the economic analysis.
James Griffin (https://orcid.org/0000-0002-6364-0414) (Research Fellow, Statistics) performed the statistical analyses.
Joanna Smith (Research Fellow, Physiotherapy) designed and implemented PHT.
Peter DH Wall (https://orcid.org/0000-0003-3149-3373) (Research Fellow, Trauma and Orthopaedic Surgery) designed and performed the feasibility and full studies, respectively, and prepared this report.
Alba Realpe (https://orcid.org/0000-0001-9502-3907) (Research Fellow, Qualitative Health Services) ran the qualitative study.
Nick Parsons (https://orcid.org/0000-0001-9975-888X) (Senior Lecturer, Statistics) performed the statistical analyses.
Rachel Hobson (https://orcid.org/0000-0002-1535-6339) (Trial Manager) set up the administrative processes for the trial.
Jeremy Fry (lay member) was the patient representative on the TSC.
Marcus Jepson (https://orcid.org/0000-0003-3261-1626) (Senior Research Fellow, Qualitative Health Science) ran the qualitative study.
Stavros Petrou (https://orcid.org/0000-0003-3121-6050) (Professor, Health Economics) performed the economic analysis.
Charles Hutchinson (https://orcid.org/0000-0003-3387-9229) (Professor, Clinical Imaging) provided the radiological support to the trial.
Nadine Foster (https://orcid.org/0000-0003-4429-9756) (Professor, Physiotherapy) designed and implemented PHT.
Jenny Donovan (https://orcid.org/0000-0002-6488-5472) (Professor, Social Medicine) ran the qualitative study.
Publications
Griffin DR, Dickenson EJ, O'Donnell J, Agricola R, Awan T, Beck M, et al. The Warwick Agreement on femoroacetabular impingement syndrome (FAI syndrome): an international consensus statement. Br J Sports Med 2016;50:1169–76.
Griffin DR, Dickenson EJ, Wall PD, Donovan JL, Foster NE, Hutchinson CE, et al. Protocol for a multi-centre, parallel-arm, 12-month, randomised controlled trial of arthroscopic surgery versus conservative care for femoroacetabular impingement syndrome (FASHIoN). BMJ Open 2016;6:e012453.
Griffin DR, Dickenson EJ, Wall PDH, Realpe A, Adams A, Parsons N, et al. The feasibility of conducting a randomised controlled trial comparing arthroscopic hip surgery to conservative care for patients with femoroacetabular impingement syndrome: the FASHIoN feasibility study. J Hip Preserv Surg 2016;3:304–11.
Wall PD, Dickenson EJ, Robinson D, Hughes I, Realpe A, Hobson R, et al. Personalised Hip Therapy: development of a non-operative protocol to treat femoroacetabular impingement syndrome in the FASHIoN randomised controlled trial. Br J Sports Med 2016;50:1217–23.
Murphy NJ, Eyles J, Bennell KL, Bohensky M, Burns A, Callaghan FM, et al. Protocol for a multi-centre randomised controlled trial comparing arthroscopic hip surgery to physiotherapy-led care for femoroacetabular impingement (FAI): the Australian FASHIoN trial. BMC Musculoskelet Disord 2017;18:406.
Griffin DR, Dickenson EJ, Wall PDH, Achana F, Donovan JL, Griffin J, et al. Hip arthroscopy versus best conservative care for the treatment of femoroacetabular impingement syndrome (UK FASHIoN): a multicentre randomised controlled trial. Lancet 2018;391:2225–35.
Dickenson EJ, Wall PDH, Hutchinson CE, Griffin DR. The prevalence of cam hip morphology in the general population. Osteoarthritis Cartilage 2019;27:444–8.
Hunter DJ, Eyles J, Murphy NJ, Spiers L, Burns A, Davidson E, et al. Multi-centre randomised controlled trial comparing arthroscopic hip surgery to physiotherapist-led care for femoroacetabular impingement (FAI) syndrome on hip cartilage metabolism: the Australian FASHIoN trial. BMC Musculoskelet Disord 2021;22:697.
Realpe AX, Foster NE, Dickenson EJ, Jepson M, Griffin DR, Donovan JL. Patient experiences of receiving arthroscopic surgery or personalised hip therapy for femoroacetabular impingement in the context of the UK fashion study: a qualitative study. Trials 2021;22:211.
Data-sharing statement
All available data can be obtained from the corresponding author.
Patient data
This work uses data provided by patients and collected by the NHS as part of their care and support. Using patient data is vital to improve health and care for everyone. There is huge potential to make better use of information from people’s patient records, to understand more about disease, develop new treatments, monitor safety, and plan NHS services. Patient data should be kept safe and secure, to protect everyone’s privacy, and it’s important that there are safeguards to make sure that it is stored and used responsibly. Everyone should be able to find out about how patient data are used. #datasaveslives You can find out more about the background to this citation here: https://understandingpatientdata.org.uk/data-citation.
Disclaimers
This report presents independent research funded by the National Institute for Health Research (NIHR). The views and opinions expressed by authors in this publication are those of the authors and do not necessarily reflect those of the NHS, the NIHR, NETSCC, the HTA programme or the Department of Health and Social Care. If there are verbatim quotations included in this publication the views and opinions expressed by the interviewees are those of the interviewees and do not necessarily reflect those of the authors, those of the NHS, the NIHR, NETSCC, the HTA programme or the Department of Health and Social Care.
References
- Griffin DR, Dickenson EJ, Wall PD, Donovan JL, Foster NE, Hutchinson CE, et al. Protocol for a multi-centre, parallel-arm, 12-month, randomised controlled trial of arthroscopic surgery versus conservative care for femoroacetabular impingement syndrome (FASHIoN). BMJ Open 2016;6. https://doi.org/10.1136/bmjopen-2016-012453.
- Griffin DR, Dickenson EJ, Wall PDH, Realpe A, Adams A, Parsons N, et al. The feasibility of conducting a randomised controlled trial comparing arthroscopic hip surgery to conservative care for patients with femoroacetabular impingement syndrome: the FASHIoN feasibility study. J Hip Preserv Surg 2016;3:304-11. https://doi.org/10.1093/jhps/hnw026.
- Griffin D, Wall P, Realpe A, Adams A, Parsons N, Hobson R, et al. UK FASHIoN: feasibility study of a randomised controlled trial of arthroscopic surgery for hip impingement compared with best conservative care. Health Technol Assess 2016;20. https://doi.org/10.3310/hta20320.
- Wall PDH. Treatments for Femoroacetabular Impingement 2013.
- Wall PD, Dickenson EJ, Robinson D, Hughes I, Realpe A, Hobson R, et al. Personalised hip therapy: development of a non-operative protocol to treat femoroacetabular impingement syndrome in the FASHIoN randomised controlled trial. Br J Sports Med 2016;50:1217-23. https://doi.org/10.1136/bjsports-2016-096368.
- Griffin DR, Dickenson EJ, Wall PDH, Achana F, Donovan JL, Griffin J, et al. Hip arthroscopy versus best conservative care for the treatment of femoroacetabular impingement syndrome (UK FASHIoN): a multicentre randomised controlled trial. Lancet 2018;391:2225-35. https://doi.org/10.1016/S0140-6736(18)31202-9.
- Ganz R, Parvizi J, Beck M, Leunig M, Nötzli H, Siebenrock KA. Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res 2003;417:112-20. https://doi.org/10.1097/01.blo.0000096804.78689.c2.
- Lavigne M, Parvizi J, Beck M, Siebenrock KA, Ganz R, Leunig M. Anterior femoroacetabular impingement: part I. Techniques of joint preserving surgery. Clin Orthop Relat Res 2004;418:61-6. https://doi.org/10.1097/00003086-200401000-00011.
- Ganz R, Leunig M, Leunig-Ganz K, Harris WH. The etiology of osteoarthritis of the hip: an integrated mechanical concept. Clin Orthop Relat Res 2008;466:264-72. https://doi.org/10.1007/s11999-007-0060-z.
- Hack K, Di Primio G, Rakhra K, Beaulé PE. Prevalence of cam-type femoroacetabular impingement morphology in asymptomatic volunteers. J Bone Joint Surg Am 2010;92:2436-44. https://doi.org/10.2106/JBJS.J.01280.
- Gosvig KK, Jacobsen S, Sonne-Holm S, Gebuhr P. The prevalence of cam-type deformity of the hip joint: a survey of 4151 subjects of the Copenhagen osteoarthritis study. Acta Radiol 2008;49:436-41. https://doi.org/10.1080/02841850801935567.
- Montgomery SR, Ngo SS, Hobson T, Nguyen S, Alluri R, Wang JC, et al. Trends and demographics in hip arthroscopy in the United States. Arthroscopy 2013;29:661-5. https://doi.org/10.1016/j.arthro.2012.11.005.
- Botser IB, Smith TW, Nasser R, Domb BG. Open surgical dislocation versus arthroscopy for femoroacetabular impingement: a comparison of clinical outcomes. Arthroscopy 2011;27:270-8. https://doi.org/10.1016/j.arthro.2010.11.008.
- Matsuda DK, Carlisle JC, Arthurs SC, Wierks CH, Philippon MJ. Comparative systematic review of the open dislocation, mini-open, and arthroscopic surgeries for femoroacetabular impingement. Arthroscopy 2011;27:252-69. https://doi.org/10.1016/j.arthro.2010.09.011.
- National Institute for Health and Care Excellence (NICE) . Arthroscopic Femoro-Acetabular Surgery for Hip Impingement Syndrome. Interventional Procedures Guidance [IPG408] 2011.
- Wall PD, Brown JS, Parsons N, Buchbinder R, Costa ML, Griffin D. Surgery for treating hip impingement (femoroacetabular impingement). Cochrane Database Syst Rev 2014;9. https://doi.org/10.1002/14651858.CD010796.pub2.
- Britton A, McKee M, Black N, McPherson K, Sanderson C, Bain C. Choosing between randomised and non-randomised studies: a systematic review. Health Technol Assess 1997;2. https://doi.org/10.3310/hta2130.
- Ross S, Grant A, Counsell C, Gillespie W, Russell I, Prescott R. Barriers to participation in randomised controlled trials: a systematic review. J Clin Epidemiol 1999;52:1143-56. https://doi.org/10.1016/S0895-4356(99)00141-9.
- Ergina PL, Cook JA, Blazeby JM, Boutron I, Clavien PA, Reeves BC, et al. Challenges in evaluating surgical innovation. Lancet 2009;374:1097-104. https://doi.org/10.1016/S0140-6736(09)61086-2.
- Wall PD, Fernandez M, Griffin DR, Foster NE. Nonoperative treatment for femoroacetabular impingement: a systematic review of the literature. PM R 2013;5:418-26. https://doi.org/10.1016/j.pmrj.2013.02.005.
- NHS Berkshire West Clinical Commissioning Group . Thames Valley Priorities Committee Commissioning Policy Statement: Policy No. TVPC33 – Surgical Treatment Of Femoro Acetabular Hip Impingement (FAI) (Open or Arthroscopic) 2018.
- Fransen M, McConnell S. Exercise for osteoarthritis of the knee. Cochrane Database Syst Rev 2008;4. https://doi.org/10.1002/14651858.CD004376.pub2.
- Hayden JA, van Tulder MW, Malmivaara AV, Koes BW. Meta-analysis: exercise therapy for nonspecific low back pain. Ann Intern Med 2005;142:765-75. https://doi.org/10.7326/0003-4819-142-9-200505030-00013.
- Chartered Society of Physiotherapy . Frontline Magazine n.d. www.csp.org.uk/news/frontline-magazine (accessed 23 September 2021).
- Palmer AJ, Thomas GE, Pollard TC, Rombach I, Taylor A, Arden N, et al. The feasibility of performing a randomised controlled trial for femoroacetabular impingement surgery. Bone Joint Res 2013;2:33-40. https://doi.org/10.1302/2046-3758.22.2000137.
- Donovan JL, Lane JA, Peters TJ, Brindle L, Salter E, Gillatt D, et al. Development of a complex intervention improved randomization and informed consent in a randomized controlled trial. J Clin Epidemiol 2009;62:29-36. https://doi.org/10.1016/j.jclinepi.2008.02.010.
- Wade J, Donovan JL, Lane JA, Neal DE, Hamdy FC. It’s not just what you say, it’s also how you say it: opening the ‘black box’ of informed consent appointments in randomised controlled trials. Soc Sci Med 2009;68:2018-28. https://doi.org/10.1016/j.socscimed.2009.02.023.
- Realpe A, Adams A, Wall P, Griffin D, Donovan JL. A new simple six-step model to promote recruitment to RCTs was developed and successfully implemented. J Clin Epidemiol 2016;76:166-74. https://doi.org/10.1016/j.jclinepi.2016.02.002.
- Quintana JM, Escobar A, Bilbao A, Arostegui I, Lafuente I, Vidaurreta I. Responsiveness and clinically important differences for the WOMAC and SF-36 after hip joint replacement. Osteoarthritis Cartilage 2005;13:1076-83. https://doi.org/10.1016/j.joca.2005.06.012.
- Wamper KE, Sierevelt IN, Poolman RW, Bhandari M, Haverkamp D. The Harris hip score: do ceiling effects limit its usefulness in orthopedics? A systematic review. Acta Orthop 2010;81:703-7. https://doi.org/10.3109/17453674.2010.537808.
- Mohtadi NG, Griffin DR, Pedersen ME, Chan D, Safran MR, Parsons N, et al. The development and validation of a self-administered quality-of-life outcome measure for young, active patients with symptomatic hip disease: the International Hip Outcome Tool (iHOT-33). Arthroscopy 2012;28:595-60. https://doi.org/10.1016/j.arthro.2012.03.013.
- Christensen CP, Althausen PL, Mittleman MA, Lee J, McCarthy JC. The nonarthritic hip score: reliable and validated. Clin Orthop Relat Res 2003;406:75-83. https://doi.org/10.1097/00003086-200301000-00013.
- Phillips L, Mohtadi N, Chan D. The responsiveness and minimal clinically important difference (MCID) of the MAHORN Quality of Life Tool. Arthroscopy 2011;27. https://doi.org/10.1016/j.arthro.2010.11.053.
- Harris-Hayes M, McDonough CM, Leunig M, Lee CB, Callaghan JJ, Roos EM. Clinical outcomes assessment in clinical trials to assess treatment of femoroacetabular impingement: use of patient-reported outcome measures. J Am Acad Orthop Surg 2013;21:39-46. https://doi.org/10.5435/JAAOS-21-07-S39.
- World Medical Association . Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 2013;310:2191-4. https://doi.org/10.1001/jama.2013.281053.
- Nötzli HP, Wyss TF, Stoecklin CH, Schmid MR, Treiber K, Hodler J. The contour of the femoral head-neck junction as a predictor for the risk of anterior impingement. J Bone Joint Surg Br 2002;84:556-60. https://doi.org/10.1302/0301-620x.84b4.12014.
- Nepple JJ, Prather H, Trousdale RT, Clohisy JC, Beaulé PE, Glyn-Jones S, et al. Diagnostic imaging of femoroacetabular impingement. J Am Acad Orthop Surg 2013;21:S20-6. https://doi.org/10.5435/JAAOS-21-07-S20.
- Tönnis D, Heinecke A. Current concepts review – acetabular and femoral anteversion: relationship with osteoarthritis of the hip. J Bone Joint Surg Am 1999;81:1747-70. https://doi.org/10.2106/00004623-199912000-00014.
- Philippon MJ, Briggs KK, Yen YM, Kuppersmith DA. Outcomes following hip arthroscopy for femoroacetabular impingement with associated chondrolabral dysfunction: minimum two-year follow-up. J Bone Joint Surg Br 2009;91:16-23. https://doi.org/10.1302/0301-620X.91B1.21329.
- Donovan JL, Paramasivan S, de Salis I, Toerien M. Clear obstacles and hidden challenges: understanding recruiter perspectives in six pragmatic randomised controlled trials. Trials 2014;15. https://doi.org/10.1186/1745-6215-15-5.
- Emara K, Samir W, Motasem el H, Ghafar KA. Conservative treatment for mild femoroacetabular impingement. J Orthop Surg 2011;19:41-5. https://doi.org/10.1177/230949901101900109.
- Walters SJ, Brazier JE. Comparison of the minimally important difference for two health state utility measures: EQ-5D and SF-6D. Qual Life Res 2005;14:1523-32. https://doi.org/10.1007/s11136-004-7713-0.
- Oppe M, Devlin NJ, van Hout B, Krabbe PF, de Charro F. A program of methodological research to arrive at the new international EQ-5D-5L valuation protocol. Value Health 2014;17:445-53. https://doi.org/10.1016/j.jval.2014.04.002.
- Ostendorf M, van Stel HF, Buskens E, Schrijvers AJ, Marting LN, Verbout AJ, et al. Patient-reported outcome in total hip replacement. A comparison of five instruments of health status. J Bone Joint Surg Br 2004;86:801-8. https://doi.org/10.1302/0301-620x.86b6.14950.
- Jenkinson C, Layte R. Development and testing of the UK SF-12. J Health Serv Res Policy 1997;2:14-8. https://doi.org/10.1177/135581969700200105.
- Brazier JE, Roberts J. The estimation of a preference-based measure of health from the SF-12. Med Care 2004;42:851-9. https://doi.org/10.1097/01.mlr.0000135827.18610.0d.
- Foster NE, Thomas E, Barlas P, Hill JC, Young J, Mason E, et al. Acupuncture as an adjunct to exercise based physiotherapy for osteoarthritis of the knee: randomised controlled trial. BMJ 2007;335. https://doi.org/10.1136/bmj.39280.509803.BE.
- van den Brink M, van den Hout W, Stiggelbout A, Kievit J, van de Velde C. Self-Reports of Health Care Utilization: Can a Questionnaire Replace a Diary n.d.
- Amstutz H, Thomas B, Jinnah R, Kim W, Grogan T, Yale C. Treatment of primary osteoarthritis of the hip. A comparison of total joint and surface replacement arthroplasty. J Bone Joint Surg Am 1984;66:228-41. https://doi.org/10.2106/00004623-198466020-00010.
- Ng VY, Arora N, Best TM, Pan X, Ellis TJ. Efficacy of surgery for femoroacetabular impingement: a systematic review. Am J Sports Med 2010;38:2337-45. https://doi.org/10.1177/0363546510365530.
- Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) – explanation and elaboration: a report of the ISPOR Health Economic Evaluation Publication Guidelines Good Reporting Practices Task Force. Value Health 2013;16:231-50. https://doi.org/10.1016/j.jval.2013.02.002.
- Curtis L. Unit Costs of Health and Social Care 2016. Canterbury: PSSRU, University of Kent; 2016.
- NHS Digital . Prescription Cost Analysis, England – 2016 n.d. www.content.digital.nhs.uk/catalogue/PUB23631 (accessed April 2017).
- ISD Scotland . Theatres: Costs – Detailed Tables n.d. www.isdscotland.org/Health-Topics/Finance/Costs/Detailed-Tables/Theatres.asp (accessed April 2017).
- NHS Scotland . Scottish Health Service Costs Book User Manual June 2016 Version: 3.0 2016.
- NHS Supply Chain . NHS Supply Chain Catalogue 2016 n.d. www.supplychain.nhs.uk/clinical-and-consumables/ (accessed April 2017).
- Department of Health and Social Care . National Reference Costs 2014 to 2015 n.d. www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015 (accessed 4 October 2021).
- Joint Formulary Committee . British National Formulary 2016.
- EuroQol Group . EuroQol – a new facility for the measurement of health-related quality of life. Health Policy 1990;16:199-208. https://doi.org/10.1016/0168-8510(90)90421-9.
- Herdman M, Gudex C, Lloyd A, Janssen M, Kind P, Parkin D, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 2011;20:1727-36. https://doi.org/10.1007/s11136-011-9903-x.
- Tarlov AR, Ware JE, Greenfield S, Nelson EC, Perrin E, Zubkoff M. The Medical Outcomes Study. An application of methods for monitoring the results of medical care. JAMA 1989;262:925-30. https://doi.org/10.1001/jama.262.7.925.
- Kind P, Dolan P, Gudex C, Williams A. Variations in population health status: results from a United Kingdom national questionnaire survey. BMJ 1998;316:736-41. https://doi.org/10.1136/bmj.316.7133.736.
- van Hout B, Janssen MF, Feng YS, Kohlmann T, Busschbach J, Golicki D, et al. Interim scoring for the EQ-5D-5L: mapping the EQ-5D-5L to EQ-5D-3L value sets. Value Health 2012;15:708-15. https://doi.org/10.1016/j.jval.2012.02.008.
- Devlin NJ, Shah KK, Feng Y, Mulhern B, van Hout B. Valuing health-related quality of life: an EQ-5D-5L value set for England. Health Econ 2018;27:7-22. https://doi.org/10.1002/hec.3564.
- Ramsey SD, Willke RJ, Glick H, Reed SD, Augustovski F, Jonsson B, et al. Cost-effectiveness analysis alongside clinical trials II – an ISPOR Good Research Practices Task Force report. Value Health 2015;18:161-72. https://doi.org/10.1016/j.jval.2015.02.001.
- Faria R, Gomes M, Epstein D, White IR. A guide to handling missing data in cost-effectiveness analysis conducted within randomised controlled trials. PharmacoEconomics 2014;32:1157-70. https://doi.org/10.1007/s40273-014-0193-3.
- National Institute of Health and Care Excellence (NICE) . Guide to the Methods of Technology Appraisal 2013 2013.
- Claxton K, Martin S, Soares M, Rice N, Spackman E, Hinde S, et al. Methods for the estimation of the National Institute for Health and Care Excellence cost-effectiveness threshold. Health Technol Assess 2015;19. https://doi.org/10.3310/hta19140.
- Glick HA, Doshi JA, Sonnad SS, Polsky D. Economic Evaluation in Clinical Trials. Oxford: Oxford University Press; 2014.
- Sculpher MJ, Claxton K, Drummond M, McCabe C. Whither trial based economic evaluation for health care decision making?. Health Econ 2006;15:677-87.
- Donovan JL, Rooshenas L, Jepson M, Elliott D, Wade J, Avery K, et al. Optimising recruitment and informed consent in randomised controlled trials: the development and implementation of the Quintet Recruitment Intervention (QRI). Trials 2016;17. https://doi.org/10.1186/s13063-016-1391-4.
- de Salis I, Tomlin Z, Toerien M, Donovan J. Qualitative research to improve RCT recruitment: issues arising in establishing research collaborations. Contemp Clin Trials 2008;29:663-70. https://doi.org/10.1016/j.cct.2008.03.003.
- Mills N, Donovan JL, Wade J, Hamdy FC, Neal DE, Lane JA. Exploring treatment preferences facilitated recruitment to randomised controlled trials. J Clin Epidemiol 2011;64:1127-36. https://doi.org/10.1016/j.jclinepi.2010.12.017.
- Paramasivan S, Huddart R, Hall E, Lewis R, Birtle A, Donovan JL. Key issues in recruitment to randomised controlled trials with very different interventions: a qualitative investigation of recruitment to the SPARE trial (CRUK/07/011). Trials 2011;12. https://doi.org/10.1186/1745-6215-12-78.
- Paramasivan S, Strong S, Wilson C, Campbell B, Blazeby JM, Donovan JL. A simple technique to identify key recruitment issues in randomised controlled trials: Q-QAT – Quanti-Qualitative Appointment Timing. Trials 2015;16. https://doi.org/10.1186/s13063-015-0617-1.
- Strong S, Paramasivan S, Mills N, Wilson C, Donovan JL, Blazeby JM. ‘The trial is owned by the team, not by an individual’: a qualitative study exploring the role of teamwork in recruitment to randomised controlled trials in surgical oncology. Trials 2016;17. https://doi.org/10.1186/s13063-016-1341-1.
- Department of Health and Social Care . National Reference Costs 2015 to 2016 n.d. www.gov.uk/government/publications/nhs-reference-costs-2015-to-2016 (accessed April 2017).
- Mansell NS, Rhon DI, Meyer J, Slevin JM, Marchant BG. Arthroscopic surgery or physical therapy for patients with femoroacetabular impingement syndrome: a randomized controlled trial with 2-year follow-up. Am J Sports Med 2018;46:1306-14. https://doi.org/10.1177/0363546517751912.
- Weber AE, Harris JD, Nho SJ. Complications in hip arthroscopy: a systematic review and strategies for prevention. Sports Med Arthrosc Rev 2015;23:187-93. https://doi.org/10.1097/JSA.0000000000000084.
- Nakano N, Lisenda L, Jones TL, Loveday DT, Khanduja V. Complications following arthroscopic surgery of the hip: a systematic review of 36 761 cases. Bone Joint J 2017;99–B:1577-83. https://doi.org/10.1302/0301-620X.99B12.BJJ-2017-0043.R2.
- Agricola R, Heijboer M, Bierma-Zeinstra S, Verhaar J, Weinans H, Waarsing E. Cam impingement causes end-stage osteoarthritis of the hip: a nationwide prospective study (CHECK). HIP Int 2012;22. https://doi.org/10.1136/annrheumdis-2012-201643.
- Walters SJ, Bonacho Dos Anjos Henriques-Cadby I, Bortolami O, Flight L, Hind D, Jacques RM, et al. Recruitment and retention of participants in randomised controlled trials: a review of trials funded and published by the United Kingdom Health Technology Assessment Programme. BMJ Open 2017;7. https://doi.org/10.1136/bmjopen-2016-015276.
- Foster NE, Healey EL, Holden MA, Nicholls E, Whitehurst DG, Jowett S, et al. A multicentre, pragmatic, parallel group, randomised controlled trial to compare the clinical and cost-effectiveness of three physiotherapy-led exercise interventions for knee osteoarthritis in older adults: the BEEP trial protocol (ISRCTN: 93634563). BMC Musculoskelet Disord 2014;15. https://doi.org/10.1186/1471-2474-15-254.
- Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I, Petticrew M. Medical Research Council Guidance. Developing and evaluating complex interventions: the new Medical Research Council guidance. BMJ 2008;337. https://doi.org/10.1136/bmj.a1655.
- Hay EM, Foster NE, Thomas E, Peat G, Phelan M, Yates HE, et al. Effectiveness of community physiotherapy and enhanced pharmacy review for knee pain in people aged over 55 presenting to primary care: pragmatic randomised trial. BMJ 2006;333. https://doi.org/10.1136/bmj.38977.590752.0B.
- Villar Bajwa Protocol n.d. www.villarbajwa.com/pdfs/Rehabilitation%20protocol%20following%20Hip%20Arthroscopy%20(May%202010).pdf (accessed 20 February 2021).
- Shepherd J, Jones J, Frampton G, Bryant J, Baxter L, Cooper K. Clinical effectiveness and cost-effectiveness of depth of anaesthesia monitoring (E-Entropy, Bispectral Index and Narcotrend): a systematic review and economic evaluation. Health Technol Assess 2013;17. https://doi.org/10.3310/hta17340.
- Joint Formulary Committee . British National Formulary 2017.
- Curtis L. Unit Costs of Health and Social Care 2014. Canterbury: PSSRU, University of Kent; 2014.
- Curtis L. Unit Costs of Health and Social Care 2015. Canterbury: PSSRU, University of Kent; 2015.
- NHS Foundation Trust . Sensory Equipment Supplier Information n.d. www.swft.nhs.uk/download_file/223/185 (accessed 8 March 2017).
- South & West Devon Formulary and Referral . 18.2 Catheters n.d. https://southwest.devonformularyguidance.nhs.uk/formulary/chapters/continence-formulary/catheters (accessed 20 February 2021).
- The Physiotherapy Centre n.d. www.thephysiocentre.co.uk/how_much/ (accessed 30 November 2016).
- amazon.co.uk . Faithfull FAILITPICK Litter Picker, Pick Up Grabber Tool 87 cm (34 in) n.d. www.amazon.co.uk/product-reviews/B004EK3NOC (accessed 4 October 2021).
- Sports Direct . Adidas Galaxy Elite Trainers n.d. www.sportsdirect.com/adidas-galaxy-elite-trainers-ladies-271462?colcode=27146203 (accessed 20 February 2021).
- Sports Direct . Slazenger Gel Insoles n.d. www.sportsdirect.com/dunlop-gel-insole-mens-750268?colcode=75026890 (accessed 20 February 2021).
- Sports Direct . Character Umbrella n.d. www.sportsdirect.com/character-umbrella-infants-750143?colcode=75014393unknown (accessed 20 February 2021).
- amazon.co.uk . Oypla Kneeling Orthopaedic Ergonomic Posture Office Stool Chair Seat n.d. www.amazon.co.uk/Oypla-Kneeling-Orthopaedic-Ergonomic-Posture/dp/B00YS8NLJC/ref=sr_1_1?dchild=1%26keywords=Oypla+Kneeling+Orthopaedic+Ergonomic+Posture+Office+Stool+Chair+Seat%26qid=1633348842%26sr=8-1 (accessed 20 February 2021).
- amazon.co.uk . Lexington Office Chair – Black Computer Chair – Desk Chair – Gas Lift – Adjustable Seat – Mesh Design n.d. amazon.co.uk (accessed 20 February 2021).
- amazon.co.uk . Foam Bed Wedge With Quilted Cover Multi-Purpose Cushion for Back, Neck &Amp; Leg Support n.d. amazon.co.uk (accessed 20 February 2021).
- amazon.co.uk . YAHILL® 1pc or 2 Pcs Folding Trekking Pole Collapsible Climbing Stick Ultralight Adjustable, Alpenstocks With EVA Foam Handle, for Traveling Hiking Camping Climbing Backpacking Walking n.d. www.amazon.co.uk/Collapsible-Ultralight-Adjustable-Alpenstocks-Backpacking/dp/B0140KEYUM/ref=sr_1_7?ie=UTF8%26qid=1485185655%26sr=8-7%26keywords=Trekking+pole (accessed 20 February 2021).
Appendix 1 Case report forms
Baseline data
Follow-up case report forms
Follow-up case report form: 6 and 12 months
Follow-up case report form: 2 and 3 years – further procedures
Surgical case report forms
Postoperative magnetic resonance imaging Instructions
Personalised hip therapy case report form
Personalised hip therapy manual
Diary
Appendix 2 Results
Recruitment: randomisation by site
| Site | Treatment group, n | ||
|---|---|---|---|
| Surgery (N = 171) | PHT (N = 177) | Total (N = 348) | |
| University Hospitals Coventry and Warwickshire | 37 | 41 | 78 |
| Yeovil District Hospital | 11 | 11 | 22 |
| Royal Devon and Exeter Hospital | 9 | 9 | 18 |
| Royal Orthopaedic Hospital | 20 | 19 | 39 |
| Wrightington Hospital | 6 | 6 | 12 |
| The Royal Cornwall | 3 | 4 | 7 |
| Elective Orthopaedic Centre | 1 | 1 | 2 |
| Northumbria Healthcare NHS Foundation Trust | 7 | 7 | 14 |
| The Royal London | 2 | 3 | 5 |
| Doncaster and Bassetlaw Teaching Hospital NHS Foundation Trust | 8 | 8 | 16 |
| Royal National Orthopaedic Hospital | 5 | 5 | 10 |
| Frimley Park Hospital | 11 | 12 | 23 |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital | 16 | 16 | 32 |
| South Tees Hospitals NHS Foundation Trust | 5 | 3 | 8 |
| University College Hospital | 4 | 4 | 8 |
| Guys’ and St Thomas’ Hospital | 6 | 5 | 11 |
| Cardiff and Vale Hospitals | 5 | 6 | 11 |
| Glasgow Royal Infirmary | 3 | 2 | 5 |
| Wrexham Maelor Hospital | 1 | 3 | 4 |
| King’s College Hospital | 7 | 6 | 13 |
| North Bristol NHS Trust | 3 | 4 | 7 |
| Spire Manchester Hospital | 1 | 2 | 3 |
| Total | 171 | 177 | 348 |
| Site | FAI type, n | |||||
|---|---|---|---|---|---|---|
| Cam | Mixed | Pincer | ||||
| Surgery (N = 171) | PHT (N = 177) | Surgery (N = 171) | PHT (N = 177) | Surgery (N = 171) | PHT (N = 177) | |
| University Hospitals Coventry and Warwickshire | 34 | 36 | 1 | 2 | 2 | 3 |
| Yeovil District Hospital | 8 | 8 | 2 | 2 | 1 | 1 |
| Royal Devon and Exeter Hospital | 6 | 6 | 3 | 3 | 0 | 0 |
| Royal Orthopaedic Hospital | 16 | 14 | 4 | 3 | 0 | 2 |
| Wrightington Hospital | 6 | 6 | 0 | 0 | 0 | 0 |
| The Royal Cornwall | 3 | 3 | 0 | 1 | 0 | 0 |
| Elective Orthopaedic Centre | 1 | 1 | 0 | 0 | 0 | 0 |
| Northumbria Healthcare NHS Foundation Trust | 6 | 7 | 1 | 0 | 0 | 0 |
| The Royal London | 2 | 3 | 0 | 0 | 0 | 0 |
| Doncaster and Bassetlaw Teaching Hospital NHS Foundation Trust | 3 | 2 | 4 | 5 | 1 | 1 |
| Royal National Orthopaedic Hospital | 3 | 0 | 2 | 5 | 0 | 0 |
| Frimley Park Hospital | 8 | 8 | 1 | 3 | 2 | 1 |
| The Robert Jones and Agnes Hunt Orthopaedic Hospital | 10 | 12 | 3 | 2 | 3 | 2 |
| South Tees Hospitals NHS Foundation Trust | 2 | 3 | 2 | 0 | 1 | 0 |
| University College Hospital | 2 | 2 | 3 | 1 | 0 | 0 |
| Guys’ and St Thomas’ Hospital | 4 | 5 | 1 | 0 | 1 | 0 |
| Cardiff and Vale Hospitals | 3 | 4 | 1 | 1 | 1 | 1 |
| Glasgow Royal Infirmary | 3 | 1 | 0 | 0 | 0 | 1 |
| Wrexham Maelor Hospital | 1 | 1 | 0 | 1 | 0 | 1 |
| King’s College Hospital | 5 | 5 | 1 | 0 | 1 | 1 |
| North Bristol NHS Trust | 3 | 2 | 0 | 2 | 0 | 0 |
| Spire Manchester Hospital | 1 | 2 | 0 | 0 | 0 | 0 |
| Total | 130 | 131 | 29 | 31 | 13 | 14 |
| Type of withdrawal | Treatment group, n (%) | Total (N = 348), n (%) | |
|---|---|---|---|
| Surgery (N = 171) | PHT (N = 177) | ||
| Participant requested to withdraw from trial | |||
| Yes | 13 | 18 | 31 |
| No | 158 | 159 | 317 |
| Patient level of withdrawal | |||
| From treatment only | 9 (69) | 12 (67) | 21 (68) |
| From treatment and follow-up | 3 (23) | 4 (22) | 7 (23) |
| Missing | 1 (8) | 2 (11) | 3 (10) |
| Participant reason for withdrawal | |||
| Participant’s decision | 2 (15) | 3 (17) | 5 (16) |
| Participant does not want to continue with treatment | 8 (62) | 9 (50) | 17 (55) |
| No reason given | 0 (0) | 1 (6) | 1 (3) |
| Patient does not want to complete questionnaire | 0 (0) | 3 (17) | 5 (16) |
| Other reason | 2 (15) | 3 (17) | 3 (10) |
| Missing | 1 (8) | 2 (11) | 3 (10) |
Surgery
| Key stage of procedure undertaken | Hip arthroscopy (N = 144), n (%) |
|---|---|
| General anaesthetic with muscle relaxation | |
| Yes | 140 (97) |
| No with reason | 3 (2) |
| Missing | 1 (1) |
| Supine or lateral patient positioning | |
| Yes | 143 (99) |
| No with reason | 0 (0) |
| Missing | 1 (1) |
| Operating table used with facility for traction and range of movement testing | |
| Yes | 143 (99) |
| No with reason | 0 (0) |
| Missing | 1 (1) |
| Arthroscopy of central compartment | |
| Yes | 141 (98) |
| No with reason | 0 (0) |
| Missing | 3 (2) |
| Arthroscopy of peripheral compartment | |
| Yes | 140 (97) |
| No with reason | 0 (0) |
| Missing | 4 (3) |
| Entire acetabular labrum examined | |
| Yes | 142 (99) |
| No with reason | 1 (1) |
| Missing | 1 (1) |
| Entire articular surface examined | |
| Yes | 142 (99) |
| No with reason | 1 (1) |
| Missing | 1 (1) |
| Confirmed impingement has been relieved using either range of movement testing or an image intensifier | |
| Yes | 138 (96) |
| No with reason | 4 (3) |
| Missing | 2 (1) |
| Did the patient have any intraoperative complications (e.g. fracture, iatrogenic)? | |
| Yes | 4 (3) |
| No with reason | 135 (94) |
| Missing | 5 (3) |
| Was standard postoperative rehabilitation/physiotherapy prescribed for this patient? | |
| Yes | 141 (98) |
| No with reason | 2 (1) |
| Missing | 1 (1) |
Postoperative rehabilitation protocols
Twenty of the 21 sites responded to the request for information about postoperative rehabilitation protocols. Eighteen (90%) sites had a protocol in place and two (10%) sites had no specific protocol but delivered ‘usual-care’ physiotherapy.
Four themes emerged from the protocols:
-
Seven (25%) sites delivered a prescriptive format adherent to a timeline according to postoperative weeks. Three (15%) sites adopted the Villar Bajwa protocol. 86
-
Four (20%) sites used a phased rehabilitation programme for which there was an entry criteria stipulated for each level. These phases were described as early (immediate postoperative), intermediate and advanced rehabilitation.
-
Seven (35%) sites offered an additional phase of sport-specific training.
-
Two (10%) sites delivered a prescriptive early immediate postoperative phase. This phase was a continuation to usual care and was left to the discretion of the treating physiotherapist.
Personalised hip therapy
| PHT session detail | PHT sessionsa (N = 947) |
|---|---|
| Number of sessions | 891 |
| Length of consultation (minutes) | |
| Mean | 30 |
| SD | 11 |
| Median | 30 |
| Minimum | 0 |
| Maximum | 60 |
| Type of session, n (%) | |
| Face to face | 878 (93) |
| Telephone | 31 (3) |
| 4 (1) | |
| Missing | 34 (3) |
| Assessment/reassessment, n (%) | |
| Ticked | 834 (88) |
| Not ticked | 102 (11) |
| Education and advice, n (%) | |
| Ticked | 729 (77) |
| Not ticked | 203 (21) |
| Help with pain relief, n (%) | |
| Ticked | 696 (74) |
| Not ticked | 235 (25) |
| Supervised exercises in clinic, n (%) | |
| Ticked | 741 (78) |
| Not ticked | 191 (20) |
| Exercise prescription given, n (%) | |
| Ticked | 566 (60) |
| Not ticked | 366 (39) |
| Exercise diary given/reviewed, n (%) | |
| Ticked | 399 (42) |
| Not ticked | 491 (52) |
| Diary forgotten | 39 (4) |
| Diary lost | 0 (0) |
| Number and type of exercise | Frequency, n |
|---|---|
| Other | 671 |
| Stability exercise r | 337 |
| Stability exercise p/clam | 326 |
| Stability exercise q | 226 |
| Stability exercise s | 186 |
| Stability exercise t | 172 |
| Abdominal exercise | 170 |
| Kneeling/hip flexor stretching | 157 |
| External rotation stretches | 142 |
| Pelvic tilt exercises | 129 |
| Stability exercise h | 127 |
| Stability exercise l/lunge | 121 |
| Stability exercise o | 117 |
| Gluteal dip strength | 81 |
| Stability exercise z | 78 |
| Hip joint glides | 78 |
| Crab walk strength | 76 |
| Side plank | 75 |
| Inverted hamstring strength | 72 |
| Gym ball exercise | 69 |
| Three-way lunge strength | 48 |
| VMO exercise | 46 |
| VMO exercise | 45 |
| Stability exercise g | 38 |
| Anterior capsule hip joint mobilisation | 16 |
Follow-up completeness of data
| iHOT-33 item | Number missing | ||
|---|---|---|---|
| Baseline (N = 348) | 6 months (N = 315) | 12 months (N = 321) | |
| Compulsory item, n | |||
| Hip ache | 1 | 0 | 1 |
| Hip stiffness from sitting all day | 2 | 2 | 1 |
| Long-distance walk difficulty | 1 | 1 | 1 |
| Hip pain while sitting | 3 | 0 | 1 |
| Standing on feet trouble | 1 | 3 | 2 |
| Getting off the floor difficulty | 2 | 0 | 1 |
| Walking on uneven floor | 1 | 0 | 1 |
| Lying on affected hip difficulty | 2 | 1 | 2 |
| Stepping over objects trouble | 2 | 3 | 1 |
| Climbing up stairs | 5 | 0 | 1 |
| Rising from sitting position | 4 | 1 | 4 |
| Long-stride discomfort | 4 | 1 | 2 |
| Getting out of a car trouble | 1 | 0 | 1 |
| Grinding, catching or clicking | 6 | 1 | 5 |
| Taking off socks, shoes and stockings | 2 | 0 | 1 |
| Overall pain in hip | 6 | 1 | 1 |
| Concern about fitness level | 3 | 2 | 5 |
| Post-activity hip pain | 3 | 4 | 8 |
| Concern about pain from sport | 1 | 5 | 6 |
| Quality-of-life deterioration | 1 | 5 | 6 |
| Performance level during sport | 3 | 9 | 10 |
| Frustration because of your hip | 4 | 6 | 0 |
| Distraction because of hip | 4 | 5 | 0 |
| Tension and stress relief difficulty | 4 | 6 | 0 |
| Discouragement as a result of your hip | 6 | 7 | 3 |
| Awareness of hip disability | 5 | 5 | 0 |
| Non-compulsory item, n/Na | |||
| Changing direction during sport | 3/245 | 0/223 | 0/231 |
| Heavy objects at work | 0/207 | 0/178 | 0/198 |
| Crouching or squatting trouble | 5/320 | 4/266 | 2/280 |
| Concern about job worsening hip pain | 7/320 | 5/266 | 0/280 |
| Reduced hip mobility difficulty at work | 8/320 | 4/266 | 0/280 |
| Sexual activity as a result of hip | 0/311 | 0/279 | 0/284 |
| Carrying children concerns | 0/234 | 0/193 | 1/209 |
| SF-12 item, n | N = 348 | N = 291 | N = 278 |
| General health | 1 | 1 | 0 |
| Moderate activities | 1 | 0 | 0 |
| Climbing stairs | 1 | 0 | 0 |
| Accomplished less physical health | 2 | 0 | 1 |
| Limited activities | 4 | 0 | 1 |
| Accomplished less emotional problems | 2 | 0 | 1 |
| Did not do work or activities carefully | 4 | 2 | 4 |
| Pain interference with normal work | 1 | 3 | 1 |
| Calm and peaceful | 0 | 3 | 1 |
| Lot of energy | 0 | 3 | 1 |
| Downhearted and blue | 0 | 4 | 1 |
| Social activities | 0 | 3 | 1 |
| EQ-5D-5L item, n | N = 348 | N = 293 | N = 302 |
| Mobility | 0 | 0 | 0 |
| Self-care | 0 | 0 | 0 |
| Usual activities | 0 | 0 | 0 |
| Pain | 0 | 1 | 0 |
| Anxiety | 0 | 0 | 0 |
| VAS | 1 | 3 | 7 |
Appendix 3 Health economics
| Resource category | Sample (n = 40) | |||
|---|---|---|---|---|
| Mean (SE) | Unit cost (£) | Unit | Source of unit cost | |
| Theatre time (hours) | 2.09 (0.08) | 298.68 | Hour | ISD Scotland (2016)54 |
| Inpatient length of stay (days) | 1.57 (0.09) | 332.77 | Day | Reference costs (2016)77 |
| Clinical staff | ||||
| Consultant surgeon | 1.00 (0.00) | 137 | Hour | PSSRU 2016, section 1552 |
| Consultant anaesthetist | 1.00 (0.00) | 135 | Hour | PSSRU 2016, section 1552 |
| Assistant surgeon | 1.00 (0.07) | 59 | Hour | PSSRU 2016, section 1552 |
| Radiographer (band 6) | 0.95 (0.05) | 46.00 | Hour | PSSRU 2016, p. 22152 |
| Nurse (band 6) | 0.86 (0.10) | 44.00 | Hour | PSSRU 2016, section 1452 |
| Nurse (band 5) | 0.81 (0.09) | 35 | Hour | PSSRU 2016, section 1452 |
| ODP (band 4) | 1.07 (0.07) | 30.00 | Hour | PSSRU 2016, p. 22152 |
| Health-care assistant (band 4) | 1.20 (0.09) | 28.00 | Hour | PSSRU 2016, section 1452 |
| Anaesthesia | ||||
| Propofol (Aspen Pharma Trading Ltd, Dublin, Ireland) 10-mg/ml amp (1% emulsion) | 1 | 2.94 | Per item | BNF 201658 |
| Rocuronium (Bowmed Ibisqus Ltd, Brynkinallt, UK) 10 mg/ml, 10-ml vial | 2 | 5.79 | Per item | BNF 201658 |
| Cyclizine 50 mg/ml, 1-ml amp | 1 | 2.57 | Per item | Prescription costs analysis database 201653 |
| Dexamethasone 3.8 mg/ml, 1-ml vial | 2 | 1.49 | Per item | Prescription costs analysis database 201653 |
| Cefuroxime 1.5-g vial | 1 | 5.05 | Per item | Prescription costs analysis database 201653 |
| Sugammadex [Merck Sharp & Dohme (UK) Ltd, Hoddesdon, UK] 100-mg/ml vial | 1 | 59.64 | Per item | BNF 201658 |
| Desflurane o2/w20 | 2 | 11.96 | Per item | Shepherd et al.87 |
| Hartmann’s solution 1000 mls | 1 | 6.80 | Per item | Prescription costs analysis database 201653 |
| Fentanyl | 1 | 4.99 | Per item | Prescription costs analysis database 201653 |
| Paracetamol infusion (B. Braun Medical Ltd, Sheffield, UK) 10 mg/ml | 1 | 1.20 | Per item | BNF 201788 |
| Equipment/implant | Quantity (N = 40), mean (SE) | Unit cost (£) | Supplier number | Source |
|---|---|---|---|---|
| Smith & Nephew | ||||
| Ligament chisel (radio-frequency probe) | 0.100 (0.048) | 340.79 | 72200682 | NHS Supply Chain Catalogue 2016 56 |
| Ablator (radio-frequency probe) | 0.650 (0.105) | 340.79 | 72200683 | |
| Tac-s (radio-frequency probe) | 0.125 (0.053) | 349.31 | 72200681 | |
| Hook (radio-frequency probe) | 0.125 (0.053) | 149.32 | 7209646 | |
| Incisor plus elite shaver | 0.075 (0.042) | 104.92 | 72200081 | |
| 4.5-mm-long curved shaver | 0.200 (0.082) | 137.25 | 7205332 | |
| 4.0/5.5-mm abrader burr | 0.175 (0.071) | 161.47 | 72200082 | |
| 4.0/5.5-mm flat top burr | 0.025 (0.025) | 161.47 | 72203130 | |
| 4.0/5.5-mm barrel burr | 0.200 (0.073) | 161.47 | 72203132 | |
| ACCU-PASS suture | 0.225 (0.076) | 104.29 | 7210425 | |
| All suture cefix | 0.125 (0.089) | 255.93 | 72201993 | |
| Banana blade | 0.150 (0.057) | 35.00 | 72203307 | |
| Ambient super multivac 50 | 0.075 (0.042) | 167.07 | ASHA4830-01 | |
| Hip pac | 0.025 (0.025) | 38.56 | 7209874 | |
| Dyonics water pump | 0.075 (0.042) | 66.95 | 7211006 | |
| STARVAC 90 | 0.025 (0.025) | 142.27 | ASC4251-01 | |
| SUPER TURBOVAC 90 | 0.050 (0.035) | 154.64 | ASH4250-01 | |
| Hip disposable needle | 0.025 (0.025) | 96.52 | 72201811 | |
| 110-mm hip cannula | 0.050 (0.035) | 40.60 | 72200436 | |
| Cross 50 | 0.025 (0.025) | 139.05 | 72202140 | |
| Arthrex (Naples, FL, USA) | ||||
| CoolCut™ ablator 90 | 0.100 (0.048) | 123.04 | AR-9705A-90 | NHS Supply Chain Catalogue 2016 56 |
| CoolCut™ ablator 30/50 | 0.025 (0.025) | 125.45 | AR-9703A-90 | |
| 4.2-mm bone cutter (Excalibur) | 0.025 (0.025) | 77.20 | AR-6420EX | |
| 4.2-mm bone cutter | 0.050 (0.035) | 90.46 | AR-6420XBC | |
| 4.2-mm sabre tooth shaver | 0.025 (0.025) | 77.20 | AR-6420CST | |
| 4.2-mm dissector | 0.025 (0.025) | 90.46 | AR-6420XDS | |
| 4-mm burr | 0.250 (0.069) | 77.20 | AR-8550RBE | |
| ConMed Linvatec (Utica, NY, USA) | ||||
| 4.2-mm great white shaver | 0.025 (0.025) | 81.35 | HPS-C001 | NHS Supply Chain Catalogue 2016 56 |
| 4.2-mm full radius resector | 0.050 (0.035) | 193.86 | C9144 | |
| 4.5/5.5-mm spherical burr | 0.050 (0.035) | 193.86 | C9014 | |
| 4.5/5-mm oval burr | 0.050 (0.035) | 59.12 | 702139600 | |
| Stryker Corporation (Kalamazoo, MI, USA) | ||||
| 5-mm resector | 0.100 (0.048) | 86.60 | 385552000 | NHS Supply Chain Catalogue 2016 56 |
| 5.5-mm pear burr | 0.075 (0.055) | 94.39 | 5820016050 | |
| 5.5-mm barrel burr | 0.100 (0.048) | 94.39 | 5820017050 | |
| 4.5-mm burr | 0.025 (0.025) | 86.60 | 375941000 | |
| Pivot SlingShot | 0.025 (0.025) | 120.00 | CAT02589 | |
| Pivot NanoPass® | 0.050 (0.035) | 120.00 | CAT02298 | |
| Pivot InJector® | 0.025 (0.025) | 95.00 | CAT01857 | |
| Pivot CinchLock® SS | 0.225 (0.121) | 230.00 | CAT02462 | |
| Pivot NanoTack | 0.100 (0.070) | 200.00 | CAT01858 | |
| Samurai blade | 0.050 (0.035) | 124.21 | CAT00227 | |
| MicroFX blade | 0.025 (0.025) | 151.60 | 234-200-200 | |
| Other manufacturer | ||||
| JuggerKnot® (Zimmer Biomet, Warsaw, IN, USA) | 0.075 (0.075) | 173.00 | 912068 | NHS Supply Chain Catalogue 2016 56 |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| Missing, % | Zero counts, % | Mean (SE) | Missing, % | Zero counts, % | Mean (SE) | Mean difference (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Inpatient stay (days) | ||||||||
| Orthopaedics (hip/leg) | 1.2 | 91.8 | 0.071 (0.022) | 0.6 | 89.3 | 0.105 (0.022) | –0.034 (–0.158 to 0.040) | 0.248 |
| Orthopaedics (other) | 1.2 | 98.8 | 0.000 (0.000) | 0.6 | 98.9 | 0.006 (0.006) | –0.006 (–0.028 to 0.000) | |
| Rehabilitation unit | 1.2 | 98.8 | 0.000 (0.000) | 0.6 | 98.9 | 0.006 (0.006) | –0.006 (–0.028 to 0.000) | |
| Day case | 1.2 | 94.7 | 0.041 (0.013) | 0.6 | 97.7 | 0.017 (0.013) | 0.024 (–0.014 to 0.097) | 0.162 |
| Pain injection | 1.2 | 98.2 | 0.006 (0.006) | 0.6 | 98.9 | 0.006 (0.006) | 0.000 (–0.027 to 0.029) | 0.876 |
| Other inpatient staya | 1.2 | 96.5 | 0.024 (0.016) | 0.6 | 96.6 | 0.04 (0.016) | –0.016 (–0.100 to 0.041) | 0.452 |
| Outpatient attendance | ||||||||
| Orthopaedics (hip/leg) | 1.2 | 43.3 | 1.047 (0.09) | 1.1 | 41.2 | 1.074 (0.088) | –0.027 (–0.481 to 0.427) | 0.84 |
| Physiotherapy (hip/leg) | 1.2 | 90.6 | 0.219 (0.078) | 1.1 | 88.7 | 0.291 (0.076) | –0.072 (–0.492 to 0.219) | 0.532 |
| Physiotherapy (other) | 1.2 | 97.7 | 0.018 (0.018) | 1.1 | 96.6 | 0.04 (0.018) | –0.022 (–0.123 to 0.040) | 0.368 |
| Accident and emergency | 1.2 | 94.7 | 0.047 (0.02) | 1.1 | 94.9 | 0.051 (0.019) | –0.004 (–0.095 to 0.076) | 0.86 |
| Radiology (number of scans) | 1.2 | 95.3 | 0.047 (0.02) | 1.1 | 93.8 | 0.057 (0.02) | –0.010 (–0.097 to 0.070) | 0.698 |
| Other outpatient attendanceb | 1.2 | 88.9 | 0.207 (0.074) | 1.1 | 89.3 | 0.229 (0.073) | –0.021 (–0.350 to 0.290) | 0.842 |
| Primary health-care contact (minutes) | ||||||||
| GP surgery | 1.2 | 64.3 | 31.585 (14.972) | 1.1 | 58.8 | 9.205 (14.713) | 22.380 (–3.255 to 112.987) | 0.268 |
| GP home visit | 1.2 | 98.8 | 0.000 (0.000) | 1.1 | 98.9 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| GP telephone | 1.2 | 90.6 | 0.995 (0.671) | 1.1 | 94.4 | 1.286 (0.66) | –0.290 (–3.640 to 1.744) | 0.842 |
| Practice nurse | 1.2 | 95.9 | 2.426 (1.51) | 1.1 | 94.9 | 0.671 (1.484) | 1.755 (–1.412 to 10.429) | 0.676 |
| District nurse | 1.2 | 98.8 | 0.000 (0.000) | 1.1 | 98.3 | 0.114 (0.114) | –0.114 (–0.572 to 0.000) | |
| Community physiotherapy | 1.2 | 96.5 | 1.746 (3.087) | 1.1 | 92.1 | 10.343 (3.033) | –8.597 (–27.504 to –2.263) | 0.01 |
| Other primary carec | 1.2 | 95.9 | 3.728 (2.062) | 1.1 | 96.6 | 3.434 (2.026) | 0.294 (–8.669 to 9.443) | 0.962 |
| Social care contacts (minutes) | ||||||||
| Community physiotherapy | 1.8 | 98.2 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Home care worker | 1.8 | 97.7 | 1.786 (1.770) | 0.6 | 99.4 | 0.000 (0.000) | 1.786 (0.000 to 9.186) | 0.724 |
| Other social cared | 1.8 | 98.2 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Medications (item) | ||||||||
| Medications | 3.5 | 63.7 | 13.77 (2.511) | 0.6 | 71.8 | 11.528 (2.431) | 2.241 (–7.168 to 15.641) | 0.536 |
| Aids and adaptations (per item/pair where appropriate) | ||||||||
| Crutches | 2.3 | 95.3 | 0.036 (0.018) | 0.6 | 97.7 | 0.028 (0.018) | 0.008 (–0.065 to 0.085) | 0.738 |
| Stick | 2.3 | 97.1 | 0.006 (0.006) | 0.6 | 99.4 | 0.000 (0.000) | 0.006 (0.000 to 0.031) | 0.722 |
| Grab rail | 2.3 | 97.1 | 0.006 (0.006) | 0.6 | 99.4 | 0.000 (0.000) | 0.006 (0.000 to 0.031) | 0.71 |
| Dressing aids | 2.3 | 97.7 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other aids and adaptationse | 2.3 | 97.7 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| 6 months post randomisation | ||||||||
| Inpatient stay (days) | ||||||||
| Orthopaedics (hip/leg) | 17.0 | 56.7 | 0.669 (0.164) | 22.0 | 71.8 | 0.17 (0.167) | 0.499 (0.224 to 1.588) | 0.002 |
| Orthopaedics (other) | 17.0 | 82.5 | 0.014 (0.011) | 22.0 | 77.4 | 0.007 (0.011) | 0.007 (–0.033 to 0.070) | 0.918 |
| Rehabilitation unit | 17.0 | 81.3 | 0.021 (0.051) | 22.0 | 77.4 | 0.072 (0.052) | –0.051 (–0.332 to 0.063) | 0.674 |
| Day case | 17.0 | 82.5 | 0.007 (0.009) | 22.0 | 76.8 | 0.014 (0.009) | –0.007 (–0.055 to 0.027) | 0.468 |
| Pain injection | 17.0 | 83.0 | 0.000 (0.000) | 22.0 | 77.4 | 0.007 (0.006) | –0.007 (–0.037 to 0.000) | |
| Other inpatient staya | 17.0 | 80.7 | 0.035 (0.017) | 22.0 | 78.0 | 0.000 (0.000) | 0.035 (0.011 to 0.118) | 0.038 |
| Outpatient attendance | ||||||||
| Orthopaedics (hip/leg) | 17.5 | 52.0 | 0.943 (0.237) | 22.0 | 65.5 | 0.623 (0.239) | 0.320 (–0.594 to 1.546) | 0.33 |
| Physiotherapy (hip/leg) | 17.5 | 56.1 | 1.816 (0.284) | 22.0 | 47.5 | 2.217 (0.287) | –0.402 (–2.036 to 0.685) | 0.356 |
| Physiotherapy (other) | 17.5 | 79.5 | 0.113 (0.073) | 22.0 | 73.4 | 0.181 (0.074) | –0.068 (–0.448 to 0.184) | 0.492 |
| Accident and emergency | 17.5 | 80.1 | 0.043 (0.021) | 22.0 | 75.1 | 0.036 (0.021) | 0.006 (–0.067 to 0.105) | 0.91 |
| Radiology (number of scans) | 17.5 | 80.7 | 0.028 (0.016) | 22.0 | 77.4 | 0.014 (0.016) | 0.014 (–0.049 to 0.090) | 0.584 |
| Other outpatient attendanceb | 17.5 | 77.2 | 0.135 (0.046) | 22.0 | 71.8 | 0.123 (0.046) | 0.012 (–0.166 to 0.222) | 0.876 |
| Primary health care contact (minutes) | ||||||||
| GP surgery | 17.5 | 50.9 | 10.839 (2.523) | 20.9 | 58.8 | 9.752 (2.532) | 1.087 (–10.726 to 12.721) | 0.732 |
| GP home visit | 17.5 | 81.9 | 0.142 (0.129) | 20.9 | 79.1 | 0.000 (0.000) | 0.142 (0.000 to 0.752) | 0.758 |
| GP telephone | 17.5 | 76.0 | 1.28 (0.534) | 20.9 | 75.7 | 0.88 (0.535) | 0.400 (–1.907 to 3.001) | 0.56 |
| Practice nurse | 17.5 | 71.9 | 2.447 (0.66) | 20.9 | 75.1 | 1.243 (0.662) | 1.204 (–1.354 to 4.578) | 0.2 |
| District nurse | 17.5 | 77.8 | 0.894 (0.401) | 20.9 | 78.5 | 0.429 (0.403) | 0.465 (–1.175 to 2.481) | 0.39 |
| Community physiotherapy | 17.5 | 76.0 | 10.071 (3.013) | 20.9 | 75.7 | 4.571 (3.024) | 5.499 (–4.179 to 22.121) | 0.192 |
| Other primary carec | 17.5 | 80.1 | 2.943 (2.898) | 21.5 | 75.7 | 5.475 (2.919) | –2.532 (–17.518 to 8.329) | 0.532 |
| Social care contacts (minutes) | ||||||||
| Community physiotherapy | 17.0 | 81.3 | 3.38 (2.054) | 20.9 | 78.5 | 1.286 (2.069) | 2.095 (–4.212 to 13.000) | 0.524 |
| Home care worker | 17.0 | 83.0 | 0.000 (0.000) | 20.9 | 79.1 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other social cared | 17.0 | 83.0 | 0.000 (0.000) | 20.9 | 79.1 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Medications (item) | ||||||||
| Medications | 17.5 | 47.4 | 25.695 (3.945) | 21.5 | 59.3 | 9.108 (3.973) | 16.587 (9.738 to 43.725) | < 0.001 |
| Aids and adaptations (per item/pair where appropriate) | ||||||||
| Crutches | 18.1 | 57.3 | 0.564 (0.055) | 22.6 | 75.7 | 0.036 (0.056) | 0.528 (0.597 to 1.076) | < 0.001 |
| Stick | 18.1 | 80.7 | 0.021 (0.012) | 22.6 | 76.8 | 0.007 (0.013) | 0.014 (–0.024 to 0.084) | 0.496 |
| Grab rail | 18.1 | 81.3 | 0.014 (0.013) | 22.6 | 77.4 | 0.000 (0.000) | 0.014 (0.000 to 0.074) | 0.764 |
| Dressing aids | 18.1 | 77.8 | 0.214 (0.087) | 22.6 | 77.4 | 0.000 (0.000) | 0.214 (0.082 to 0.670) | |
| Other aids and adaptationse | 18.1 | 80.1 | 0.036 (0.019) | 22.6 | 77.4 | 0.000 (0.000) | 0.036 (0.000 to 0.135) | 0.114 |
| 12 months post randomisation | ||||||||
| Inpatient stay (days) | ||||||||
| Orthopaedics (hip/leg) | 14.6 | 69.6 | 0.73 (0.319) | 23.7 | 67.8 | 0.134 (0.331) | 0.596 (0.089 to 2.544) | 0.002 |
| Orthopaedics (other) | 14.6 | 85.4 | 0.000 (0.000) | 23.7 | 74.6 | 0.037 (0.019) | –0.037 (–0.140 to 0.000) | |
| Rehabilitation unit | 14.6 | 84.8 | 0.007 (0.007) | 23.7 | 75.7 | 0.007 (0.007) | –0.001 (–0.035 to 0.031) | 0.712 |
| Day case | 14.6 | 81.3 | 0.048 (0.014) | 23.7 | 75.7 | 0.007 (0.014) | 0.041 (0.007 to 0.129) | 0.03 |
| Pain injection | 14.6 | 84.8 | 0.007 (0.007) | 23.7 | 75.7 | 0.007 (0.007) | –0.001 (–0.034 to 0.024) | 0.676 |
| Other inpatient staya | 14.6 | 83.6 | 0.021 (0.048) | 23.7 | 72.9 | 0.141 (0.05) | –0.120 (–0.448 to 0.007) | 0.062 |
| Outpatient attendance | ||||||||
| Orthopaedics (hip/leg) | 15.8 | 46.2 | 0.66 (0.069) | 23.7 | 65.0 | 0.289 (0.072) | 0.371 (0.264 to 0.898) | < 0.001 |
| Physiotherapy (hip/leg) | 15.8 | 52.0 | 2.611 (0.339) | 23.7 | 61.0 | 0.822 (0.35) | 1.789 (1.379 to 4.554) | < 0.001 |
| Physiotherapy (other) | 15.8 | 80.1 | 0.292 (0.114) | 23.7 | 72.9 | 0.148 (0.118) | 0.144 (–0.231 to 0.789) | 0.39 |
| Accident and emergency | 15.8 | 81.3 | 0.035 (0.023) | 23.7 | 70.1 | 0.096 (0.023) | –0.062 (–0.205 to 0.006) | 0.072 |
| Radiology (number of scans) | 15.8 | 81.9 | 0.028 (0.014) | 23.7 | 74.0 | 0.03 (0.014) | –0.002 (–0.070 to 0.058) | 0.922 |
| Other outpatient attendanceb | 15.8 | 77.8 | 0.139 (0.064) | 23.7 | 63.8 | 0.333 (0.066) | –0.194 (–0.595 to -0.027) | 0.028 |
| Primary health-care contact (minutes) | ||||||||
| GP surgery | 15.2 | 55.0 | 12.464 (2.226) | 24.9 | 49.2 | 11.358 (2.324) | 1.105 (–7.578 to 12.831) | 0.774 |
| GP home visit | 15.2 | 84.8 | 0.000 (0.000) | 24.9 | 75.1 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| GP telephone | 15.2 | 78.9 | 1.041 (0.694) | 24.9 | 67.8 | 1.829 (0.724) | –0.787 (–5.120 to 1.515) | 0.482 |
| Practice nurse | 15.2 | 78.9 | 2.069 (0.762) | 24.9 | 69.5 | 1.695 (0.796) | 0.373 (–2.493 to 4.121) | 0.726 |
| District nurse | 15.2 | 82.5 | 0.586 (1.724) | 24.9 | 72.3 | 4.173 (1.8) | –3.587 (–14.605 to 0.774) | 0.16 |
| Community physiotherapy | 15.2 | 81.3 | 14.69 (7.597) | 24.9 | 71.8 | 5.075 (7.933) | 9.614 (–9.858 to 56.054) | 0.42 |
| Other primary carec | 15.2 | 84.8 | 0.000 (0.000) | 25.4 | 73.4 | 2.576 (1.978) | –2.576 (–12.413 to 0.000) | |
| Social care contacts (minutes) | ||||||||
| Community physiotherapy | 15.8 | 84.2 | 0.000 (0.000) | 25.4 | 74.6 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Home care worker | 15.8 | 84.2 | 0.000 (0.000) | 25.4 | 74.6 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other social cared | 15.8 | 84.2 | 0.000 (0.000) | 25.4 | 74.6 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Medications (item) | ||||||||
| Medications | 15.8 | 56.7 | 19.375 (4.514) | 24.9 | 58.2 | 9.609 (4.697) | 9.766 (–1.582 to 39.771) | 0.094 |
| Aids and adaptations (per item/pair where appropriate) | ||||||||
| Crutches | 17.5 | 69.0 | 0.305 (0.046) | 26.6 | 71.8 | 0.046 (0.048) | 0.259 (0.211 to 0.618) | < 0.001 |
| Stick | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 72.9 | 0.008 (0.007) | –0.008 (–0.039 to 0.000) | |
| Grab rail | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 73.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Dressing aids | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 73.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other aids and adaptationse | 17.5 | 81.3 | 0.035 (0.022) | 26.6 | 71.8 | 0.031 (0.023) | 0.005 (–0.087 to 0.117) | 0.924 |
| Category | Currency code | Unit cost (£) | Source |
|---|---|---|---|
| Inpatients (per day of inpatient stay) | |||
| Orthopaedics (hip/leg) | Weighted average of lower limb orthopaedic HRGs | 1032.31 | Reference costs (2016)77 |
| Rehabilitation unit | VC36Z | 357.42 | |
| Day case | Weighted average of lower limb orthopaedic HRGs | 1872.47 | |
| Pain injection | AB19Z | 691 | |
| Burns and plastics | JB31A, JB31B, JB31C, JB32A, JB32B, JB32C, JB33A, JB33B and JB33C | 966.34 | |
| Cholecystectomy | GA10H, GA10J, GA10K, GA10L, GA10M, GA10N and GA11Z | 1737.81 | |
| General surgery | GA10H, GA10J, GA10K, GA10L, GA10M, GA10N and GA11Z | 1737.81 | |
| Colonoscopy | FZ51Z, FZ52Z and FZ53Z | 793.29 | |
| Hernia | GA10H, GA10J, GA10K, GA10L, GA10M, GA10N and GA11Z | 1703.3 | |
| Surgery general | Weighted average of upper limb orthopaedic HRGs | 1872.47 | |
| Septoplasty | CA11A | 1638.88 | |
| Varicocele embolisation | YR56Z | 1177.53 | |
| Accident and emergency | Accident and emergency, admitted | 203.55 | |
| Outpatients (per contact) | |||
| General surgery | 100 | 130.06 | Reference costs (2016)77 |
| Urology | 101 | 105.19 | |
| Trauma and orthopaedics | 110 | 117.01 | |
| Ear, nose and throat | 120 | 96.87 | |
| Plastics clinic | 160 | 99.95 | |
| Accident and emergency | 180 | 146.86 | |
| Pain clinic | 191 | 139.12 | |
| General medicine | 300 | 309 | |
| Diabetes | 307 | 159.31 | |
| Cardiology | 320 | 127.67 | |
| Dermatology | 330 | 101.63 | |
| Breast clinic | 370 | 170.85 | |
| Neurology | 400 | 175.6 | |
| Rheumatology | 410 | 142.74 | |
| Dentist | 450 | 111.18 | |
| Eye clinic | 460 | 63.46 | |
| Gynaecology | 502 | 133.01 | |
| Midwife | 560 | 75.15 | |
| Osteopath | 650 | 48.33 | |
| Physiotherapy | 650 | 48.33 | |
| Chiropractor | 653 | 42.84 | |
| Podiatrist | 653 | 39.2 | |
| Mental health | 710 | 287.57 | |
| Blood test | DAPS05 | 3 | |
| Fertility clinic | 502 | 133.01 | |
| Gastroscopy | FZ93A | 186.75 | |
| Hydrotherapy | 650 | 48.33 | |
| Occupational health | 300 | 167.05 | |
| Light treatment therapy | Phototherapy | ||
| MRI | RD01A | 145.14 | |
| MRI spinal | RD01A | 145.14 | |
| Computerised tomography | RD20A | 93.93 | |
| DMSA scan | RN30A | 261.28 | |
| Bowl screening | FZ54Z | 198.29 | |
| X-ray | X-rays | 30.23 | |
| Primary and social care (cost/minute) | |||
| Acupuncture | 0.87 | Community health professional (band 7) cost per hour = £52 (PSSRU 2016, section 952) | |
| Chiropractor | 0.87 | ||
| Physiotherapy | 0.87 | ||
| Osteopathy | 0.63 | ||
| Massage | 0.87 | ||
| Pharmacist | 0.87 | ||
| Psychology | 0.87 | ||
| Counsellor | 51.1 | £50 per hour (PSSRU 2014, p. 5189) updated to 2016 prices using Hospital and Community Health Services pay and price inflation index52 | |
| District nurse/health visitor/midwife | 0.87 | Nurse (band 7) cost per hour = £52 (PSSRU 2016, section 10.152) | |
| Practice nurse | 0.72 | Practice nurse cost per hour (with qualifications) = £43 (PSSRU 2016, section 10.252) | |
| GP home visit | 3.9 | GP home visit lasting 11.4 minutes (PSSRU 2015, Table 10.8a90). Added cost of 2.18 minutes of GP time for every home visits | |
| GP surgery | 3.9 | Cost per surgery consultation lasting 9.22 minutes = £36 (PSSRU 2016, Table 10.3b52) | |
| GP telephone | 3.9 | ||
| Health-care assistant | 0.4 | Nurse (band 3) cost per hour = £24 (PSSRU 2016, section 10.152) | |
| Adaptations: NHS supplied (per item) | |||
| Crutches | 5.05 | ||
| Stick | 1.66 | ||
| Walking frame | 1.61 | ||
| Grab rail | 1.66 | ||
| Dressing aids | 3.94 | ||
| Long-handle shoe horns | 63.15 | ||
| Ankle brace | 10.8 | MPC code S385B-1 from supply chain catalogue56 | |
| Bath seat | 11.13 | ||
| Bath sliding | 2.58 | ||
| Bath step | 10.02 | ||
| Bands from physiotherapy | 19.2 | NHS Foundation Trust91 | |
| Catheters | 20.71 | South & West Devon Formulary and Referral92 | |
| Adaptations: private purchase (per item) | |||
| Moon boot | 12.84 | ||
| Perching stool | 46.18 | ||
| Raised toilet seat | 7.42 | ||
| Private costs | |||
| Physiotherapy | 1.17 | The Physiotherapy Centre93 | |
| Toilet frame | 17.8 | ||
| Trigger-operated picker | 12.61 | amazon.co.uk94 | |
| New trainers | 31.99 | Sports Direct95 | |
| Insoles | 9.99 | Sports Direct96 | |
| Umbrella | 34.99 | Sports Direct97 | |
| Kneeling stool for work | 49.99 | amazon.co.uk98 | |
| New workstation chair | 24.99 | amazon.co.uk99 | |
| Wedge cushion | 18.99 | amazon.co.uk100 | |
| Trekking pole | 13.99 | amazon.co.uk101 | |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| Missing, % | Zero costs, % | Mean (£) (SE) | Missing, % | Zero costs, % | Mean (£) (SE) | Mean difference (£) (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Inpatient costs | ||||||||
| Orthopaedics (hip/leg) | 1.2 | 91.8 | 73.3 (22.89) | 0.6 | 89.3 | 108.63 (22.43) | –35.33 (–162.74 to 41.51) | 0.254 |
| Orthopaedics (other) | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 98.9 | 10.72 (10.69) | –10.72 (–53.39 to 0.00) | |
| Rehabilitation unit | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 98.9 | 2.03 (2.03) | –2.03 (–10.12 to 0.00) | |
| Day case | 1.2 | 94.7 | 77.56 (24.17) | 0.6 | 97.7 | 31.92 (23.69) | 45.64 (–25.59 to 181.82) | 0.158 |
| Pain injection | 1.2 | 98.2 | 4.09 (4.05) | 0.6 | 98.9 | 3.93 (3.97) | 0.16 (–18.68 to 19.75) | 0.936 |
| Other inpatient staya | 1.2 | 96.5 | 30.41 (19.18) | 0.6 | 96.6 | 45.55 (18.8) | –15.14 (–112.72 to 59.81) | 0.546 |
| Total inpatient costs | 1.2 | 85.4 | 185.36 (39.95) | 0.6 | 84.2 | 202.77 (39.15) | –17.41 (–210.64 to 141.44) | 0.754 |
| Outpatient costs | ||||||||
| Orthopaedics (hip/leg) | 1.2 | 43.3 | 122.55 (10.48) | 1.1 | 41.2 | 125.7 (10.3) | –3.15 (–56.32 to 49.93) | 0.848 |
| Orthopaedics (other) | 1.2 | 90.6 | 10.58 (3.76) | 1.1 | 88.7 | 14.08 (3.69) | –3.50 (–23.79 to 10.59) | 0.534 |
| Physiotherapy (hip/leg) | 1.2 | 97.7 | 0.86 (0.89) | 1.1 | 96.6 | 1.93 (0.88) | –1.08 (–5.96 to 1.91) | 0.38 |
| Physiotherapy (other) | 1.2 | 94.7 | 6.95 (2.88) | 1.1 | 94.9 | 7.55 (2.83) | –0.60 (–13.96 to 11.10) | 0.864 |
| Radiology | 1.2 | 95.3 | 8.13 (3.48) | 1.1 | 93.8 | 8.13 (3.42) | 0.01 (–14.47 to 16.21) | 0.918 |
| Other outpatientb | 1.2 | 88.9 | 26.1 (8.18) | 1.7 | 89.3 | 23.38 (8.06) | 2.73 (–29.10 to 42.12) | 0.85 |
| Total outpatient costs | 1.2 | 33.3 | 175.17 (13.93) | 1.7 | 29.9 | 181.68 (13.73) | –6.51 (–83.13 to 65.48) | 0.742 |
| Primary care costs | ||||||||
| GP surgery | 1.2 | 64.3 | 123.18 (58.39) | 1.1 | 58.8 | 35.9 (57.38) | 87.28 (–12.69 to 440.65) | 0.268 |
| GP home visit | 1.2 | 98.8 | 0.00 (0.00) | 1.1 | 98.9 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| GP telephone | 1.2 | 90.6 | 3.88 (2.62) | 1.1 | 94.4 | 5.01 (2.57) | –1.13 (–14.20 to 6.80) | 0.842 |
| Practice nurse | 1.2 | 95.9 | 1.75 (1.09) | 1.1 | 94.9 | 0.48 (1.07) | 1.26 (–1.02 to 7.51) | 0.676 |
| District nurse | 1.2 | 98.8 | 0.00 (0.00) | 1.1 | 98.3 | 0.10 (0.10) | –0.10 (–0.50 to 0.00) | |
| Community physiotherapy | 1.2 | 96.5 | 1.52 (2.69) | 1.1 | 92.1 | 9 (2.64) | –7.48 (–23.93 to -1.97) | 0.01 |
| Other primary carec | 1.2 | 95.9 | 3.16 (38.18) | 1.1 | 96.6 | 54.65 (37.52) | –51.49 (–264.13 to 6.74) | 0.482 |
| Total primary care costs | 1.2 | 57.3 | 133.49 (71.28) | 1.1 | 53.1 | 105.15 (70.05) | 28.34 (–253.42 to 396.08) | 0.81 |
| Social care costs | ||||||||
| Community physiotherapy | 1.8 | 98.2 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Home care worker | 1.8 | 97.7 | 1.11 (1.10) | 0.6 | 99.4 | 0.00 (0.00) | 1.11 (0.00 to 5.70) | 0.724 |
| Other social cared | 1.8 | 98.2 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total social care costs | 1.8 | 97.7 | 1.11 (1.10) | 0.6 | 99.4 | 0.00 (0.00) | 1.11 (0.00 to 5.70) | 0.724 |
| Medications | ||||||||
| Medication costs | 3.5 | 63.7 | 0.62 (1.5) | 0.6 | 71.8 | 2.08 (1.45) | –1.47 (–9.81 to 1.68) | 0.71 |
| Aids and adaptations | ||||||||
| Crutches | 2.3 | 95.3 | 0.18 (0.09) | 0.6 | 97.7 | 0.14 (0.09) | 0.04 (–0.33 to 0.43) | 0.754 |
| Stick | 2.3 | 97.1 | 0.01 (0.01) | 0.6 | 99.4 | 0.00 (0.00) | 0.01 (0.00 to 0.05) | 0.722 |
| Grab rail | 2.3 | 97.1 | 0.01 (0.01) | 0.6 | 99.4 | 0.00 (0.00) | 0.01 (0.00 to 0.05) | 0.71 |
| Dressing aids | 2.3 | 97.7 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Other aids and adaptationse | 2.3 | 97.7 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total aids and adaptation costs | 2.3 | 94.2 | 0.2 (0.09) | 0.6 | 97.7 | 0.14 (0.09) | 0.06 (–0.31 to 0.47) | 0.646 |
| Total at baseline costs | 4.1 | 12.3 | 502.12 (90.38) | 1.7 | 13.6 | 508.53 (87.74) | –6.41 (–235.01 to 246.59) | 0.88 |
| 6 months post randomisation | ||||||||
| Inpatient costs | ||||||||
| Orthopaedics (hip/leg) | 17.0 | 56.7 | 690.63 (169.54) | 22.0 | 71.8 | 175.94 (171.98) | 514.69 (231.61 to 1639.05) | 0.002 |
| Orthopaedics (other) | 17.0 | 82.5 | 26.57 (21.16) | 22.0 | 77.4 | 13.67 (21.46) | 12.90 (–62.88 to 132.78) | 0.912 |
| Rehabilitation unit | 17.0 | 81.3 | 7.55 (18.19) | 22.0 | 77.4 | 25.9 (18.45) | –18.35 (–118.58 to 22.59) | 0.674 |
| Day case | 17.0 | 82.5 | 13.19 (16.23) | 22.0 | 76.8 | 27.14 (16.46) | –13.95 (–102.90 to 50.42) | 0.5 |
| Pain injection | 17.0 | 83.0 | 0.00 (0.00) | 22.0 | 77.4 | 5.01 (4.42) | –5.01 (–25.30 to 0.00) | |
| Other inpatientsa | 17.0 | 81.3 | 44.31 (25.66) | 22.0 | 78.0 | 0.00 (0.00) | 44.31 (0.00 to 167.76) | 0.086 |
| Total inpatient costs | 17.0 | 55.0 | 782.25 (178.9) | 22.0 | 69.5 | 247.66 (181.48) | 534.59 (203.05 to 1720.75) | 0.008 |
| Outpatient costs | ||||||||
| Orthopaedics (hip/leg) | 17.5 | 52.0 | 110.37 (27.71) | 22.0 | 65.5 | 72.92 (28.01) | 37.45 (–69.46 to 180.88) | 0.33 |
| Orthopaedics (other) | 17.5 | 56.1 | 87.75 (13.74) | 22.0 | 47.5 | 107.17 (13.89) | –19.42 (–98.42 to 33.10) | 0.356 |
| Physiotherapy (hip/leg) | 17.5 | 79.5 | 5.48 (3.53) | 22.0 | 73.4 | 8.76 (3.57) | –3.27 (–21.63 to 8.90) | 0.492 |
| Physiotherapy (other) | 17.5 | 80.1 | 6.25 (3.02) | 22.0 | 75.1 | 5.32 (3.06) | 0.93 (–9.77 to 15.42) | 0.906 |
| Radiology | 17.5 | 80.7 | 4.12 (2.31) | 22.0 | 77.4 | 2.1 (2.33) | 2.01 (–7.17 to 13.08) | 0.57 |
| Other outpatientsb | 18.1 | 77.2 | 16.2 (5.72) | 22.0 | 71.8 | 14.63 (5.76) | 1.57 (–20.73 to 29.39) | 0.862 |
| Total outpatient costs | 18.1 | 33.9 | 201.41 (28) | 22.0 | 33.9 | 210.9 (28.21) | –9.49 (–161.42 to 118.18) | 0.84 |
| Primary care costs | ||||||||
| GP surgery | 17.5 | 50.9 | 42.27 (9.84) | 20.9 | 58.8 | 38.03 (9.87) | 4.24 (–41.83 to 49.61) | 0.732 |
| GP home visit | 17.5 | 81.9 | 0.54 (0.49) | 20.9 | 79.1 | 0.00 (0.00) | 0.54 (0.00 to 2.86) | 0.758 |
| GP telephone | 17.5 | 76.0 | 4.99 (2.08) | 20.9 | 75.7 | 3.43 (2.09) | 1.56 (–7.44 to 11.70) | 0.56 |
| Practice nurse | 17.5 | 71.9 | 1.76 (0.48) | 20.9 | 75.1 | 0.89 (0.48) | 0.87 (–0.98 to 3.30) | 0.202 |
| District nurse | 17.5 | 77.8 | 0.78 (0.35) | 20.9 | 78.5 | 0.37 (0.35) | 0.40 (–1.02 to 2.16) | 0.39 |
| Community physiotherapy | 17.5 | 76.0 | 8.76 (2.62) | 20.9 | 75.7 | 3.98 (2.63) | 4.78 (–3.63 to 19.24) | 0.194 |
| Other primary carec | 17.5 | 80.1 | 2.47 (2.52) | 21.5 | 75.7 | 4.76 (2.54) | –2.30 (–15.46 to 7.01) | 0.512 |
| Total primary care costs | 17.5 | 41.5 | 61.57 (11.8) | 21.5 | 50.3 | 51.81 (11.88) | 9.76 (–45.49 to 67.60) | 0.556 |
| Social care costs | ||||||||
| Community physiotherapy | 17.0 | 81.3 | 2.13 (1.29) | 20.9 | 78.5 | 0.81 (1.3) | 1.32 (–2.65 to 8.19) | 0.528 |
| Home care worker | 17.0 | 83.0 | 0.00 (0.00) | 20.9 | 79.1 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | – |
| Other social cared | 17.0 | 83.0 | 0.00 (0.00) | 20.9 | 79.1 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | – |
| Total social care costs | 17.0 | 81.3 | 2.13 (1.29) | 20.9 | 78.5 | 0.81 (1.3) | 1.32 (–2.65 to 8.19) | 0.528 |
| Medication costs | ||||||||
| Total medication cost | 17.5 | 47.4 | 0.32 (0.14) | 21.5 | 59.3 | 0.25 (0.14) | 0.07 (–0.51 to 0.72) | 0.788 |
| Aids and adaptations | ||||||||
| Crutches | 18.1 | 57.3 | 2.85 (0.28) | 22.6 | 75.7 | 0.18 (0.28) | 2.67 (3.02 to 5.44) | < 0.001 |
| Stick | 18.1 | 80.7 | 0.04 (0.02) | 22.6 | 76.8 | 0.01 (0.02) | 0.02 (–0.04 to 0.14) | 0.554 |
| Grab rail | 18.1 | 81.3 | 0.02 (0.02) | 22.6 | 77.4 | 0.00 (0.00) | 0.02 (0.00 to 0.12) | 0.764 |
| Dressing aids | 18.1 | 77.8 | 0.84 (0.34) | 22.6 | 77.4 | 0.00 (0.00) | 0.84 (0.32 to 2.64) | 0.004 |
| Other aids and adaptationse | 18.1 | 80.1 | 1.39 (0.76) | 22.6 | 77.4 | 0.00 (0.00) | 1.39 (0.00 to 5.15) | 0.114 |
| Total cost of aids and adaptations | 18.1 | 56.1 | 5.14 (0.79) | 22.6 | 75.1 | 0.2 (0.8) | 4.94 (4.72 to 11.63) | |
| Total at 6-month assessment point | 20.5 | 16.4 | 875.49 (103.01) | 25.4 | 18.1 | 499.33 (104.56) | 376.16 (53.16 to 637.80) | 0.022 |
| 12 months post randomisation | ||||||||
| Inpatient costs | ||||||||
| Orthopaedics (hip/leg) | 14.6 | 69.6 | 753.16 (328.9) | 23.7 | 67.8 | 137.95 (342.03) | 615.21 (91.64 to 2626.04) | 0.002 |
| Orthopaedics (other) | 14.6 | 85.4 | 0.00 (0.00) | 23.7 | 74.6 | 69.87 (36.37) | –69.87 (–263.69 to 0.00) | |
| Rehabilitation unit | 14.6 | 84.8 | 2.45 (2.5) | 23.7 | 75.7 | 2.65 (2.6) | –0.20 (–12.62 to 11.17) | 0.748 |
| Day case | 14.6 | 81.3 | 89.78 (25.67) | 23.7 | 75.7 | 13.87 (26.7) | 75.91 (13.15 to 241.63) | 0.028 |
| Pain injection | 14.6 | 84.8 | 4.73 (4.82) | 23.7 | 75.7 | 5.12 (5.02) | –0.39 (–23.56 to 16.26) | 0.698 |
| Other inpatientsa | 15.2 | 83.6 | 15.15 (47.75) | 24.9 | 72.9 | 117.97 (49.85) | –102.82 (–412.52 to 28.94) | 0.132 |
| Total inpatient costs | 15.2 | 62.6 | 871.13 (338.25) | 24.9 | 63.3 | 339.07 (353.18) | 532.06 (–192.26 to 2520.93) | 0.192 |
| Outpatient costs | ||||||||
| Orthopaedics (hip/leg) | 15.8 | 46.2 | 77.19 (8.12) | 23.7 | 65.0 | 33.8 (8.39) | 43.39 (30.89 to 105.08) | < 0.001 |
| Orthopaedics (other) | 15.8 | 52.0 | 126.19 (16.37) | 23.7 | 61.0 | 39.74 (16.9) | 86.46 (66.66 to 220.12) | < 0.001 |
| Physiotherapy (hip/leg) | 15.8 | 80.1 | 14.1 (5.53) | 23.7 | 72.9 | 7.16 (5.71) | 6.94 (–11.20 to 38.17) | 0.388 |
| Physiotherapy (other) | 15.8 | 81.3 | 5.1 (3.34) | 23.7 | 70.1 | 14.14 (3.44) | –9.04 (–30.13 to 0.83) | 0.072 |
| Radiology | 15.8 | 81.9 | 4.03 (2.23) | 23.7 | 74.0 | 4.78 (2.3) | –0.75 (–12.10 to 8.40) | 0.852 |
| Other outpatientsb | 15.8 | 78.4 | 15.85 (8.15) | 24.9 | 63.8 | 38.86 (8.48) | –23.01 (–73.33 to –0.64) | 0.046 |
| Total outpatient costs | 15.8 | 26.3 | 242.46 (21.84) | 24.9 | 39.5 | 134.43 (22.73) | 108.03 (64.21 to 283.44) | < 0.001 |
| Primary care costs | ||||||||
| GP surgery | 15.2 | 55.0 | 48.61 (8.68) | 24.9 | 49.2 | 44.3 (9.06) | 4.31 (–29.55 to 50.04) | 0.774 |
| GP home visit | 15.2 | 84.8 | 0.00 (0.00) | 24.9 | 75.1 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| GP telephone | 15.2 | 78.9 | 4.06 (2.7) | 24.9 | 67.8 | 7.13 (2.82) | –3.07 (–19.96 to 5.91) | 0.482 |
| Practice nurse | 15.2 | 78.9 | 1.49 (0.55) | 24.9 | 69.5 | 1.22 (0.57) | 0.27 (–1.79 to 2.97) | 0.73 |
| District nurse | 15.2 | 82.5 | 0.51 (1.5) | 24.9 | 72.3 | 3.63 (1.57) | –3.12 (–12.71 to 0.67) | 0.16 |
| Community physiotherapy | 15.2 | 81.3 | 12.78 (6.61) | 24.9 | 71.8 | 4.42 (6.9) | 8.36 (–8.57 to 48.77) | 0.42 |
| Other primary carec | 15.2 | 84.8 | 0.00 (0.00) | 25.4 | 73.4 | 2.24 (1.72) | –2.24 (–10.80 to 0.00) | |
| Total primary care costs | 15.2 | 50.3 | 67.45 (12.11) | 25.4 | 44.1 | 62.88 (12.69) | 4.57 (–43.57 to 67.87) | 0.852 |
| Social care costs | ||||||||
| Community physiotherapy | 15.8 | 84.2 | 0.00 (0.00) | 25.4 | 74.6 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Home care worker | 15.8 | 84.2 | 0.00 (0.00) | 25.4 | 74.6 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Other social cared | 15.8 | 84.2 | 0.00 (0.00) | 25.4 | 74.6 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total social care costs | 15.8 | 84.2 | 0.00 (0.00) | 25.4 | 74.6 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Medications | ||||||||
| Total cost of medications | 15.8 | 56.7 | 0.79 (0.31) | 24.9 | 58.2 | 0.32 (0.33) | 0.47 (–0.40 to 2.31) | 0.248 |
| Aids and adaptations | ||||||||
| Crutches | 17.5 | 69.0 | 1.54 (0.23) | 26.6 | 71.8 | 0.23 (0.24) | 1.31 (1.07 to 3.12) | < 0.001 |
| Stick | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 72.9 | 0.01 (0.01) | –0.01 (–0.06 to 0.00) | |
| Grab rail | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 73.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Dressing aids | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 73.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Other aids and adaptationse | 17.5 | 81.3 | 0.67 (0.53) | 26.6 | 71.8 | 0.83 (0.55) | –0.16 (–2.75 to 2.08) | 0.774 |
| Total cost of aids and adaptations | 17.5 | 68.4 | 2.21 (0.64) | 26.6 | 70.6 | 1.07 (0.67) | 1.14 (–1.21 to 4.69) | 0.216 |
| Total costs at 12-month assessment point | 20.5 | 18.7 | 1236.26 (366.13) | 29.9 | 22.0 | 560.11 (383.44) | 676.15 (–59.37 to 1810.86) | 0.082 |
| Total NHS and Personal Social Services costs over 12 months of follow-up | 32.7 | 5.3 | 1640.91 (148.33) | 40.7 | 7.3 | 941.02 (155.23) | 699.88 (274.36 to 1121.23) | < 0.001 |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| Missing, % | Zero counts, % | Mean (SE) | Missing, % | Zero counts, % | Mean (SE) | Mean difference (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Primary, secondary and social care | ||||||||
| Outpatient physiotherapy (number attended) | 1.2 | 98.8 | 0.000 (0.000) | 1.1 | 98.3 | 0.023 (0.023) | –0.023 (–0.114 to 0.000) | |
| Primary care physiotherapy (minutes) | 1.2 | 97.7 | 1.243 (0.880) | 1.1 | 98.9 | 0.000 (0.000) | 1.243 (0.000 to 5.051) | 0.268 |
| Social care physiotherapy (minutes) | 1.8 | 98.2 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other social care (minutes) | 1.8 | 97.7 | 0.714 (0.708) | 0.6 | 99.4 | 0.000 (0.000) | 0.714 (0.000 to 3.637) | 0.71 |
| Medications (number of items) | 3.5 | 73.7 | 1.024 (0.323) | 0.6 | 74.0 | 0.966 (0.313) | 0.058 (–1.293 to 1.373) | 0.934 |
| Aids and adaptations (item/item pair) | ||||||||
| Crutches | 2.3 | 97.7 | 0.000 (0.000) | 0.6 | 99.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Stick | 2.3 | 95.3 | 0.024 (0.011) | 0.6 | 97.7 | 0.017 (0.011) | 0.007 (–0.036 to 0.061) | 0.65 |
| Grab rail | 2.3 | 97.1 | 0.012 (0.009) | 0.6 | 98.9 | 0.006 (0.009) | 0.006 (–0.020 to 0.060) | 0.832 |
| Dressing aids | 2.3 | 95.9 | 0.018 (0.011) | 0.6 | 98.9 | 0.011 (0.011) | 0.007 (–0.041 to 0.057) | 0.644 |
| Other | 2.3 | 94.7 | 0.084 (0.046) | 0.6 | 94.4 | 0.068 (0.045) | 0.016 (–0.140 to 0.260) | 0.906 |
| 6 months post randomisation | ||||||||
| Primary, secondary and social care | ||||||||
| Outpatient physiotherapy (number attended) | 17.5 | 82.5 | 0.000 (0.000) | 22.0 | 78.0 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Primary care physiotherapy (minutes) | 17.5 | 80.7 | 3.191 (2.083) | 20.9 | 79.1 | 0.000 (0.000) | 3.191 (0.000 to 12.503) | 0.092 |
| Social care physiotherapy (minutes) | 17.0 | 83.0 | 0.000 (0.000) | 20.9 | 79.1 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other social care (minutes) | 17.0 | 83.0 | 0.000 (0.000) | 20.9 | 79.1 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Medications (number of items) | 17.5 | 57.3 | 5.603 (2.732) | 21.5 | 60.5 | 0.619 (2.752) | 4.984 (0.680 to 21.518) | 0.008 |
| Aids and adaptations (item/item pair) | ||||||||
| Crutches | 18.1 | 80.7 | 0.029 (0.018) | 22.6 | 77.4 | 0.000 (0.000) | 0.029 (0.000 to 0.115) | 0.256 |
| Stick | 18.1 | 80.7 | 0.014 (0.013) | 22.6 | 76.3 | 0.022 (0.014) | –0.008 (–0.076 to 0.042) | 0.674 |
| Grab rail | 18.1 | 81.9 | 0.000 (0.000) | 22.6 | 77.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Dressing aids | 18.1 | 80.7 | 0.014 (0.009) | 22.6 | 77.4 | 0.000 (0.000) | 0.014 (0.000 to 0.058) | 0.266 |
| Other | 18.1 | 76.0 | 0.1 (0.044) | 22.6 | 71.2 | 0.139 (0.044) | –0.039 (–0.257 to 0.122) | 0.53 |
| 12 months post randomisation | ||||||||
| Primary, secondary and social care | ||||||||
| Outpatient physiotherapy (number attended) | 15.8 | 84.2 | 0.000 (0.000) | 23.7 | 76.3 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Primary care physiotherapy (minutes) | 15.2 | 83.6 | 3.621 (4.323) | 24.9 | 74.0 | 5.865 (4.514) | –2.244 (–25.070 to 13.874) | 0.736 |
| Social care physiotherapy (minutes) | 15.8 | 83.6 | 0.312 (0.287) | 25.4 | 74.6 | 0.000 (0.000) | 0.312 (0.000 to 1.647) | 0.712 |
| Other social care (minutes) | 15.8 | 84.2 | 0.000 (0.000) | 25.4 | 74.6 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Medications (number of items) | 16.4 | 71.3 | 0.958 (0.463) | 24.9 | 58.8 | 1.256 (0.48) | –0.298 (–2.617 to 1.536) | 0.618 |
| Aids and adaptations (item/item pair) | ||||||||
| Crutches | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 73.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Stick | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 73.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Grab rail | 17.5 | 81.9 | 0.014 (0.013) | 26.6 | 73.4 | 0.000 (0.000) | 0.014 (0.000 to 0.079) | 0.708 |
| Dressing aids | 17.5 | 82.5 | 0.000 (0.000) | 26.6 | 73.4 | 0.000 (0.000) | 0.000 (0.000 to 0.000) | |
| Other | 17.5 | 78.9 | 0.106 (0.063) | 26.6 | 69.5 | 0.115 (0.065) | –0.009 (–0.244 to 0.255) | 0.86 |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| Missing, % | Zero counts, % | Mean (SE) | Missing, % | Zero counts, % | Mean (SE) | Mean difference (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Primary, secondary and social care | ||||||||
| Total outpatient physiotherapy | 1.2 | 98.8 | 0.00 (0.00) | 1.1 | 98.3 | 1.60 (1.59) | –1.60 (–8.00 to 0.00) | |
| Total primary care costs | 1.2 | 97.7 | 1.45 (1.03) | 1.1 | 98.9 | 0.00 (0.00) | 1.45 (0.00 to 5.91) | 0.268 |
| Total social care costs | 1.8 | 98.2 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total cost of medications | 3.5 | 73.7 | 1.08 (0.19) | 0.6 | 74.0 | 1.03 (0.19) | 0.05 (–0.83 to 0.97) | 0.858 |
| Aids and adaptations | ||||||||
| Crutches | 2.3 | 97.7 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Stick | 2.3 | 95.3 | 1.88 (1.08) | 0.6 | 97.7 | 0.44 (1.05) | 1.44 (–0.95 to 7.99) | 0.396 |
| Grab rail | 2.3 | 97.1 | 0.16 (0.11) | 0.6 | 98.9 | 0.02 (0.11) | 0.13 (–0.08 to 0.83) | 0.71 |
| Dressing aids | 2.3 | 95.9 | 0.16 (0.08) | 0.6 | 98.9 | 0.06 (0.08) | 0.10 (–0.18 to 0.53) | 0.392 |
| Other | 2.9 | 94.7 | 1.37 (6.36) | 0.6 | 94.4 | 9.82 (6.18) | –8.45 (–43.72 to 3.68) | 0.386 |
| Total cost of aids and adaptations | 2.9 | 91.2 | 3.57 (6.46) | 0.6 | 92.1 | 10.34 (6.27) | –6.77 (–41.43 to 8.17) | 0.62 |
| Total costs at baseline | 4.7 | 70.2 | 10.89 (7.11) | 1.1 | 69.5 | 14.95 (6.86) | –4.06 (–25.06 to 11.16) | 0.76 |
| 6 months post randomisation | ||||||||
| Primary, secondary and social care | ||||||||
| Total outpatient physiotherapy | 17.5 | 82.5 | 0.00 (0.00) | 22.0 | 78.0 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total primary care costs | 17.5 | 80.7 | 3.73 (2.44) | 20.9 | 79.1 | 0.00 (0.00) | 3.73 (0.00 to 14.63) | 0.092 |
| Total social care costs | 17.0 | 83.0 | 0.00 (0.00) | 20.9 | 79.1 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total cost of medications | 17.5 | 57.3 | 1.12 (0.22) | 21.5 | 60.5 | 1.23 (0.22) | –0.11 (–1.13 to 0.84) | 0.716 |
| Aids and adaptations | ||||||||
| Crutches | 18.1 | 80.7 | 1.46 (1.29) | 22.6 | 77.4 | 0.00 (0.00) | 1.46 (0.00 to 7.14) | 0.256 |
| Stick | 18.1 | 80.7 | 0.21 (0.3) | 22.6 | 76.3 | 0.55 (0.31) | –0.33 (–2.07 to 0.55) | 0.456 |
| Grab rail | 18.1 | 81.9 | 0.00 (0.00) | 22.6 | 77.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Dressing aids | 18.1 | 80.7 | 0.21 (0.14) | 22.6 | 77.4 | 0.00 (0.00) | 0.21 (0.00 to 0.92) | 0.266 |
| Other | 18.1 | 76.0 | 8.05 (4.61) | 22.6 | 71.2 | 5.31 (4.66) | 2.75 (–11.93 to 28.37) | 0.778 |
| Total cost of aids and adaptations | 18.1 | 73.7 | 9.94 (4.73) | 22.6 | 70.6 | 5.85 (4.78) | 4.08 (–10.29 to 31.00) | 0.608 |
| Total private medical costs at 6 months | 19.9 | 50.3 | 19.73 (5.11) | 24.9 | 55.4 | 4.86 (5.19) | 14.87 (3.32 to 31.31) | 0.006 |
| 12 months post randomisation | ||||||||
| Primary, secondary and social care | ||||||||
| Total outpatient physiotherapy | 15.8 | 84.2 | 0.00 (0.00) | 23.7 | 76.3 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Total primary care costs | 15.2 | 83.6 | 4.24 (5.06) | 24.9 | 74.0 | 6.86 (5.28) | –2.63 (–29.33 to 16.23) | 0.736 |
| Total social care costs | 15.8 | 83.6 | 0.37 (0.34) | 25.4 | 74.6 | 0.00 (0.00) | 0.37 (0.00 to 1.93) | 0.712 |
| Total cost of medications | 16.4 | 71.3 | 0.67 (0.2) | 24.9 | 58.8 | 0.92 (0.2) | –0.24 (–1.28 to 0.49) | 0.384 |
| Aids and adaptations | ||||||||
| Crutches | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 73.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Stick | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 73.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Grab rail | 17.5 | 81.9 | 0.57 (0.52) | 26.6 | 73.4 | 0.00 (0.00) | 0.57 (0.00 to 3.16) | 0.708 |
| Dressing aids | 17.5 | 82.5 | 0.00 (0.00) | 26.6 | 73.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Other | 17.5 | 78.9 | 2.01 (4.19) | 26.6 | 69.5 | 9.45 (4.37) | –7.43 (–34.91 to 3.79) | 0.202 |
| Total cost of aids and adaptations | 17.5 | 78.4 | 2.58 (4.21) | 26.6 | 69.5 | 9.45 (4.39) | –6.86 (–34.36 to 4.63) | 0.246 |
| Total costs at 12-month point | 20.5 | 64.3 | 11.43 (7.13) | 27.7 | 53.1 | 19.56 (7.35) | –8.13 (–29.57 to 9.55) | 0.43 |
| Total costs over 12 months of follow-up | 32.2 | 36.3 | 32.67 (10.24) | 37.9 | 38.4 | 27.44 (10.51) | 5.23 (–23.55 to 32.41) | 0.704 |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 172) | PHT (n = 177) | |||||||
| Missing, % | Zero counts, % | Mean (SE) | Missing, % | Zero counts, % | Mean (SE) | Mean difference (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Lost earnings | 1.2 | 91.8 | 151.7 (78.85) | 0.6 | 92.7 | 66.86 (77.27) | 84.83 (–124.67 to 512.25) | 0.502 |
| Help with housework | 1.2 | 98.2 | 1.78 (2.81) | 0.6 | 98.3 | 4.55 (2.75) | –2.77 (–17.57 to 6.34) | 0.468 |
| Child care | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 98.9 | 8.24 (8.22) | –8.24 (–42.24 to 0.00) | |
| Special equipment | 1.2 | 98.2 | 0.3 (2.2) | 0.6 | 97.7 | 4.26 (2.16) | –3.97 (–16.88 to 0.45) | 0.082 |
| Other | 1.2 | 91.2 | 10.38 (9.44) | 0.6 | 88.1 | 27.58 (9.25) | –17.20 (–72.85 to 7.68) | 0.154 |
| Total additional costs at baseline | 1.2 | 84.8 | 164.15 (80.28) | 0.6 | 81.9 | 111.49 (78.66) | 52.66 (–189.36 to 470.01) | 0.748 |
| 6 months post randomisation | ||||||||
| Lost earnings | 15.8 | 73.7 | 309.51 (78.71) | 18.6 | 76.8 | 8.37 (78.71) | 301.15 (162.19 to 842.50) | < 0.001 |
| Help with housework | 15.8 | 83.0 | 3.51 (5.92) | 18.6 | 80.2 | 10.28 (5.92) | –6.77 (–40.58 to 11.43) | 0.432 |
| Child care | 15.8 | 83.6 | 3.47 (2.57) | 18.6 | 80.2 | 1.39 (2.57) | 2.08 (–4.83 to 17.86) | 0.794 |
| Special equipment | 15.8 | 83.0 | 1.25 (0.65) | 18.6 | 80.8 | 0.23 (0.65) | 1.02 (–0.67 to 4.84) | 0.282 |
| Other | 15.8 | 70.8 | 141.78 (87.43) | 18.6 | 70.6 | 12.49 (87.43) | 129.29 (–6.41 to 625.59) | 0.154 |
| Total additional costs at 6 months | 15.8 | 61.4 | 459.53 (116.58) | 18.6 | 66.7 | 32.76 (116.58) | 426.77 (226.04 to 1199.80) | < 0.001 |
| 12 months post randomisation | ||||||||
| Lost earnings | 13.5 | 77.2 | 637.36 (262.83) | 22.0 | 74.6 | 132.1 (272.19) | 505.26 (–40.90 to 2221.03) | 0.074 |
| Help with housework | 13.5 | 86.0 | 0.61 (0.57) | 22.0 | 78.0 | 0.00 (0.00) | 0.61 (0.00 to 3.14) | 0.694 |
| Child care | 13.5 | 86.0 | 0.81 (0.75) | 22.0 | 78.0 | 0.00 (0.00) | 0.81 (0.00 to 4.24) | 0.77 |
| Special equipment | 13.5 | 86.5 | 0.00 (0.00) | 22.0 | 78.0 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Other | 13.5 | 81.3 | 11.22 (3.77) | 22.0 | 73.4 | 4.41 (3.91) | 6.80 (–3.36 to 28.08) | 0.158 |
| Total additional costs at 12 months | 13.5 | 71.9 | 650 (262.76) | 22.0 | 70.6 | 136.51 (272.11) | 513.49 (–31.90 to 2244.99) | 0.064 |
| Total additional costs over 12 months of follow-up | 22.8 | 50.9 | 1143.2 (325.37) | 29.9 | 52.5 | 184.81 (335.7) | 958.39 (219.74 to 2001.32) | 0.004 |
| Assessment point | Treatment group | Surgery vs. PHT | ||||||
|---|---|---|---|---|---|---|---|---|
| Surgery (n = 171) | PHT (n = 177) | |||||||
| Missing, % | Zero counts, % | Mean (SE) | Missing, % | Zero counts, % | Mean (SE) | Mean difference (bootstrap 95% CI) | p-value | |
| Baseline | ||||||||
| Attendance Allowance | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Income Support | 1.2 | 97.7 | 1.05 (0.76) | 0.6 | 99.4 | 0.00 (0.00) | 1.05 (0.00 to 4.36) | 0.266 |
| Housing Benefit | 1.2 | 96.5 | 1.82 (1.63) | 0.6 | 98.3 | 2.36 (1.6) | –0.54 (–8.92 to 4.97) | 0.87 |
| Carer’s Allowance | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 98.9 | 0.34 (0.34) | –0.34 (–1.73 to 0.00) | |
| Child Tax Credit | 1.2 | 94.2 | 4.37 (1.6) | 0.6 | 96.6 | 2.16 (1.57) | 2.21 (–3.41 to 10.76) | 0.348 |
| DLA, mobility | 1.2 | 98.2 | 0.12 (0.15) | 0.6 | 98.9 | 0.17 (0.15) | –0.05 (–0.84 to 0.58) | 0.712 |
| DLA, caring | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Pension Credit | 1.2 | 98.8 | 0.00 (0.00) | 0.6 | 99.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Council Tax Reduction | 1.2 | 98.2 | 0.01 (0.01) | 0.6 | 99.4 | 0.00 (0.00) | 0.01 (0.00 to 0.03) | 0.744 |
| Employment and Support Allowance | 1.2 | 94.2 | 3.11 (0.89) | 0.6 | 98.9 | 0.41 (0.87) | 2.70 (0.79 to 8.70) | 0.014 |
| Personal Independence Payment | 1.2 | 98.2 | 5.77 (5.74) | 0.6 | 99.4 | 0.00 (0.00) | 5.77 (0.00 to 29.26) | 0.77 |
| Statutory Sick Pay | 1.2 | 98.2 | 0.52 (4.17) | 0.6 | 98.3 | 6.18 (4.09) | –5.66 (–28.33 to 1.65) | 0.35 |
| Other paymentsa | 2.3 | 95.9 | 0.65 (0.39) | 0.6 | 98.9 | 0.28 (0.38) | 0.37 (–0.92 to 2.35) | 0.498 |
| Total benefit payments at baseline | 2.3 | 87.1 | 17.62 (6.54) | 0.6 | 93.2 | 11.9 (6.37) | 5.72 (–20.89 to 36.78) | 0.512 |
| 6 months post randomisation | ||||||||
| Attendance Allowance | 15.8 | 84.2 | 0.00 (0.00) | 18.6 | 81.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Income Support | 15.8 | 83.6 | 0.69 (0.64) | 18.6 | 81.4 | 0.00 (0.00) | 0.69 (0.00 to 3.53) | 0.71 |
| Housing Benefit | 15.8 | 81.3 | 2.56 (1.02) | 18.6 | 80.8 | 0.76 (1.02) | 1.80 (–1.33 to 7.60) | 0.234 |
| Carer’s Allowance | 15.8 | 84.2 | 0.00 (0.00) | 18.6 | 81.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Child Tax Credit | 15.8 | 80.1 | 4.03 (1.99) | 18.6 | 78.5 | 3.37 (1.99) | 0.66 (–9.07 to 9.53) | 0.78 |
| DLA, mobility | 15.8 | 84.2 | 0.00 (0.00) | 18.6 | 80.8 | 0.15 (0.13) | –0.15 (–0.77 to 0.00) | |
| DLA, caring | 15.8 | 83.6 | 0.22 (0.31) | 18.6 | 80.8 | 0.38 (0.31) | –0.16 (–1.85 to 0.98) | 0.694 |
| Pension Credit | 15.8 | 84.2 | 0.00 (0.00) | 18.6 | 81.4 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Council Tax Reduction | 15.8 | 82.5 | 0.47 (0.25) | 18.6 | 81.4 | 0.00 (0.00) | 0.47 (0.00 to 1.68) | 0.088 |
| Employment and Support Allowance | 15.8 | 79.5 | 5.00 (1.88) | 18.6 | 81.4 | 0.00 (0.00) | 5.00 (2.60 to 15.14) | 0.002 |
| Personal Independence Payment | 16.4 | 82.5 | 1.86 (1.22) | 19.2 | 80.8 | 0.00 (0.00) | 1.86 (0.00 to 7.38) | 0.282 |
| Statutory Sick Pay | 16.4 | 77.2 | 29.43 (13.69) | 19.2 | 80.2 | 1.57 (13.69) | 27.86 (2.77 to 116.29) | 0.018 |
| Other paymentsa | 15.8 | 83.0 | 1.42 (1.01) | 19.2 | 80.2 | 0.84 (1.01) | 0.58 (–2.99 to 5.35) | 0.744 |
| Total benefit payments at the 6-month assessment point | 17.0 | 68.4 | 46.12 (14.27) | 20.3 | 75.1 | 7.21 (14.32) | 38.91 (14.21 to 135.48) | 0.006 |
| 12 months post randomisation | ||||||||
| Attendance Allowance | 13.5 | 86.0 | 0.07 (0.06) | 22.0 | 78.0 | 0.00 (0.00) | 0.07 (0.00 to 0.34) | 0.818 |
| Income Support | 13.5 | 86.5 | 0.00 (0.00) | 22.0 | 78.0 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Housing Benefit | 13.5 | 84.8 | 0.87 (0.51) | 22.0 | 77.4 | 0.36 (0.53) | 0.51 (–1.22 to 3.35) | 0.532 |
| Carer’s Allowance | 13.5 | 86.5 | 0.00 (0.00) | 22.0 | 78.0 | 0.00 (0.00) | 0.00 (0.00 to 0.00) | |
| Child Tax Credit | 13.5 | 84.8 | 1.1 (1.44) | 22.0 | 76.8 | 2.46 (1.49) | –1.36 (–10.24 to 2.94) | 0.574 |
| DLA, mobility | 13.5 | 85.4 | 0.75 (0.52) | 22.0 | 78.0 | 0.00 (0.00) | 0.75 (0.00 to 3.28) | 0.276 |
| DLA, caring | 13.5 | 84.8 | 1.14 (0.74) | 22.0 | 78.0 | 0.00 (0.00) | 1.14 (0.00 to 5.08) | 0.112 |
| Pension Credit | 14.0 | 85.4 | 2.04 (1.89) | 22.0 | 78.0 | 0.00 (0.00) | 2.04 (0.00 to 10.23) | 0.788 |
| Council Tax Reduction | 13.5 | 85.4 | 0.14 (0.35) | 22.0 | 77.4 | 0.51 (0.36) | –0.37 (–2.41 to 0.51) | 0.622 |
| Employment and Support Allowance | 13.5 | 84.2 | 2.72 (1.1) | 22.0 | 76.8 | 1.06 (1.14) | 1.67 (–1.84 to 8.20) | 0.282 |
| Personal Independence Payment | 14.0 | 84.2 | 2.20 (1.47) | 22.0 | 78.0 | 0.00 (0.00) | 2.20 (0.00 to 9.57) | 0.102 |
| Statutory Sick Pay | 13.5 | 82.5 | 5.1 (2.07) | 22.6 | 76.3 | 0.65 (2.16) | 4.45 (0.10 to 18.65) | 0.048 |
| Other paymentsa | 14.0 | 84.8 | 0.98 (0.6) | 22.0 | 77.4 | 0.51 (0.62) | 0.47 (–1.73 to 3.66) | 0.698 |
| Total benefit payments at the 12-month assessment point | 14.6 | 74.9 | 17.31 (4.3) | 22.6 | 74.6 | 5.58 (4.44) | 11.72 (1.49 to 38.39) | 0.038 |
| Total benefit payments over 12 months of follow-up | 25.1 | 57.3 | 57.93 (16.34) | 31.6 | 62.7 | 12.94 (16.8) | 44.99 (10.88 to 97.42) | < 0.001 |
| Assessment point | Treatment group, n (%)a | p-valueb | |
|---|---|---|---|
| Surgery (N = 46) | PHT (N = 51) | ||
| Baseline | |||
| Mobility | |||
| No problems | 19 (40.4) | 21 (41.2) | 1 |
| Some problems | 25 (53.2) | 30 (58.8) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 3 (6.4) | 0 (0.0) | |
| Self-care | |||
| No problems | 35 (74.5) | 47 (92.2) | 0.138 |
| Some problems | 9 (19.1) | 4 (7.8) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 3 (6.4) | 0 (0.0) | |
| Usual activities | |||
| No problems | 13 (27.7) | 14 (27.5) | 1 |
| Some problems | 27 (57.4) | 33 (64.7) | |
| Severe problems | 4 (8.5) | 4 (7.8) | |
| Missing | 3 (6.4) | 0 (0.0) | |
| Pain and discomfort | |||
| No problems | 2 (4.3) | 3 (5.9) | 1 |
| Some problems | 31 (66.0) | 39 (76.5) | |
| Severe problems | 11 (23.4) | 9 (17.6) | |
| Missing | 3 (6.4) | 0 (0.0) | |
| Anxiety and depression | |||
| No problems | 22 (46.8) | 30 (58.8) | 0.444 |
| Some problems | 18 (38.3) | 16 (31.4) | |
| Severe problems | 4 (8.5) | 4 (7.8) | |
| Missing | 3 (6.4) | 1 (2.0) | |
| 6 months post randomisation | |||
| Mobility | |||
| No problems | 23 (48.9) | 22 (43.1) | 0.662 |
| Some problems | 19 (40.4) | 24 (47.1) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 5 (10.6) | 5 (9.8) | |
| Self-care | |||
| No problems | 37 (78.7) | 42 (82.4) | 0.885 |
| Some problems | 5 (10.6) | 4 (7.8) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 5 (10.6) | 5 (9.8) | |
| Usual activities | |||
| No problems | 13 (27.7) | 18 (35.3) | 0.563 |
| Some problems | 25 (53.2) | 24 (47.1) | |
| Severe problems | 4 (8.5) | 4 (7.8) | |
| Missing | 5 (10.6) | 5 (9.8) | |
| Pain and discomfort | |||
| No problems | 6 (12.8) | 7 (13.7) | 1 |
| Some problems | 28 (59.6) | 33 (64.7) | |
| Severe problems | 7 (14.9) | 6 (11.8) | |
| Missing | 6 (12.8) | 5 (9.8) | |
| Anxiety and depression | |||
| No problems | 21 (44.7) | 28 (54.9) | 0.418 |
| Some problems | 18 (38.3) | 14 (27.5) | |
| Severe problems | 3 (6.4) | 4 (7.8) | |
| Missing | 5 (10.6) | 5 (9.8) | |
| 12 months post randomisation | |||
| Mobility | |||
| No problems | 21 (44.7) | 18 (35.3) | 0.651 |
| Some problems | 18 (38.3) | 21 (41.2) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 8 (17.0) | 12 (23.5) | |
| Self-care | |||
| No problems | 31 (66.0) | 34 (66.7) | 0.543 |
| Some problems | 8 (17.0) | 5 (9.8) | |
| Severe problems | 0 (0.0) | 0 (0.0) | |
| Missing | 8 (17.0) | 12 (23.5) | |
| Usual activities | |||
| No problems | 16 (34.0) | 16 (31.4) | 1 |
| Some problems | 21 (44.7) | 22 (43.1) | |
| Severe problems | 2 (4.3) | 1 (2.0) | |
| Missing | 8 (17.0) | 12 (23.5) | |
| Pain and discomfort | |||
| No problems | 9 (19.1) | 8 (15.7) | 1 |
| Some problems | 24 (51.1) | 26 (51.0) | |
| Severe problems | 6 (12.8) | 5 (9.8) | |
| Missing | 8 (17.0) | 12 (23.5) | |
| Anxiety and depression | |||
| No problems | 25 (53.2) | 26 (51.0) | 1 |
| Some problems | 9 (19.1) | 13 (25.5) | |
| Severe problems | 5 (10.6) | 0 (0.0) | |
| Missing | 8 (17.0) | 12 (23.5) | |
FIGURE 15.
Sensitivity analysis 1: unadjusted analysis based on multiple imputed data sets under the missing-at-random assumption. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
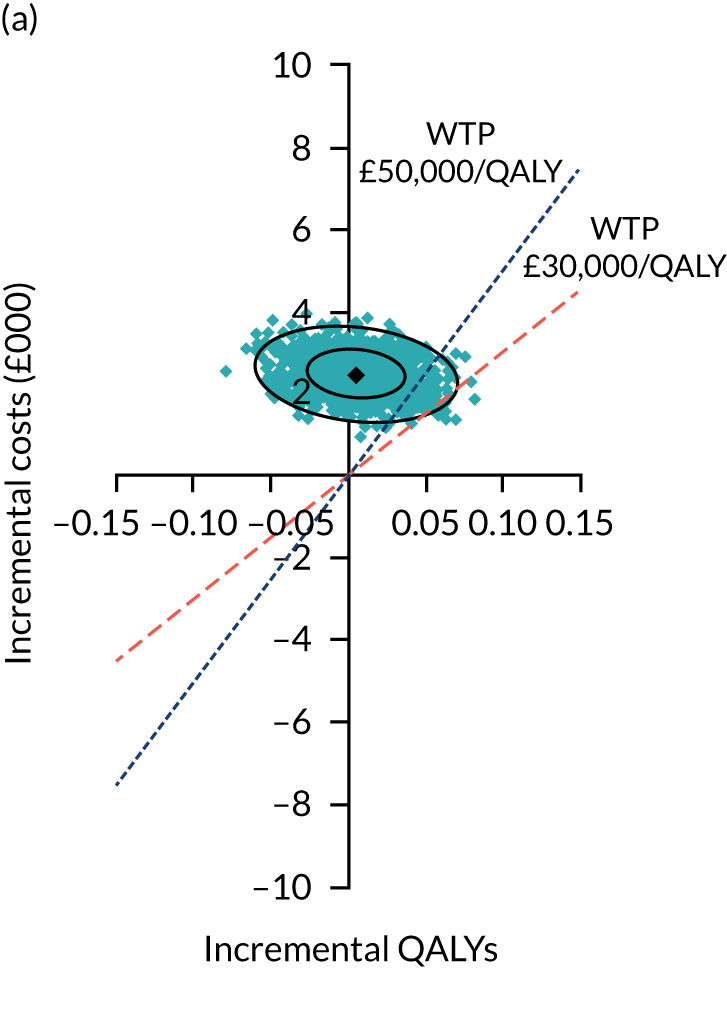
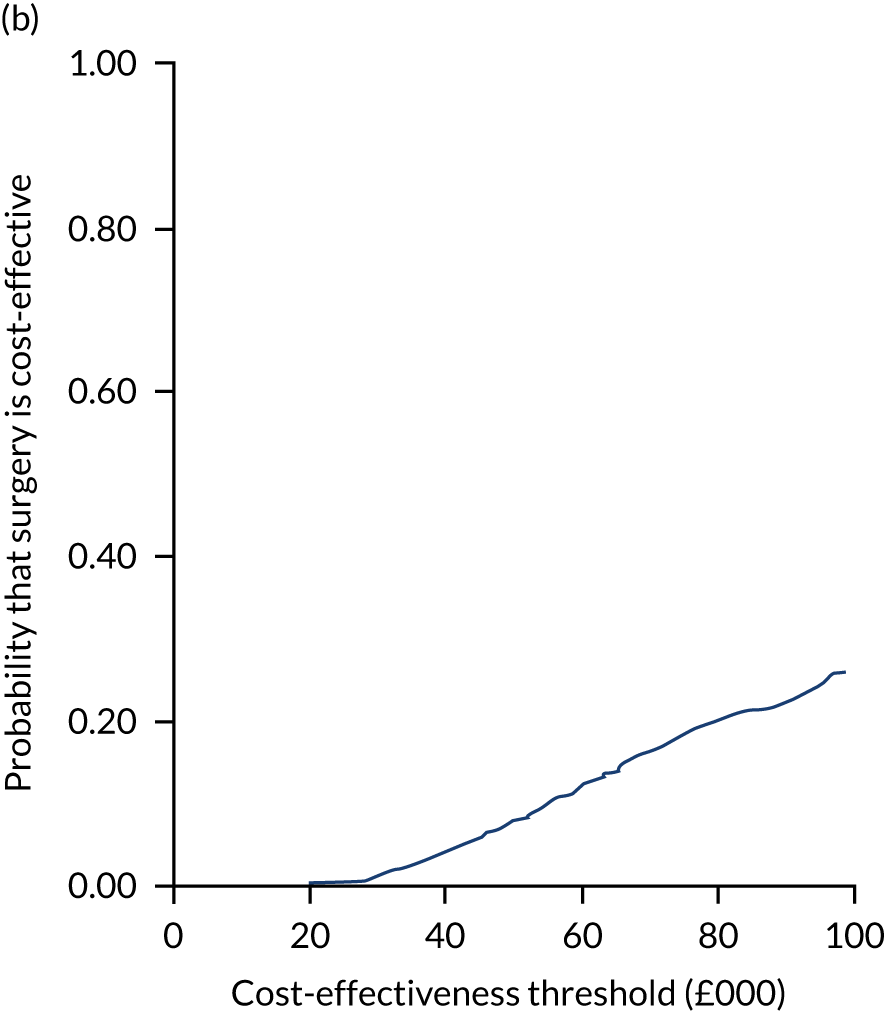
FIGURE 16.
Sensitivity analysis 2: complete-case (adjusted) analysis. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
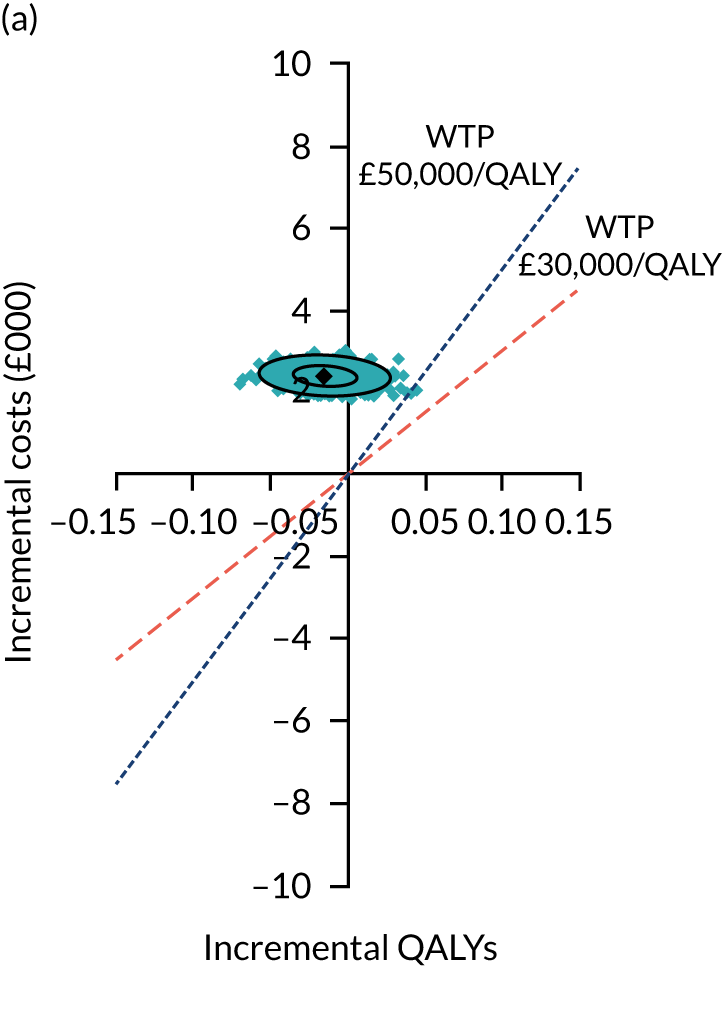
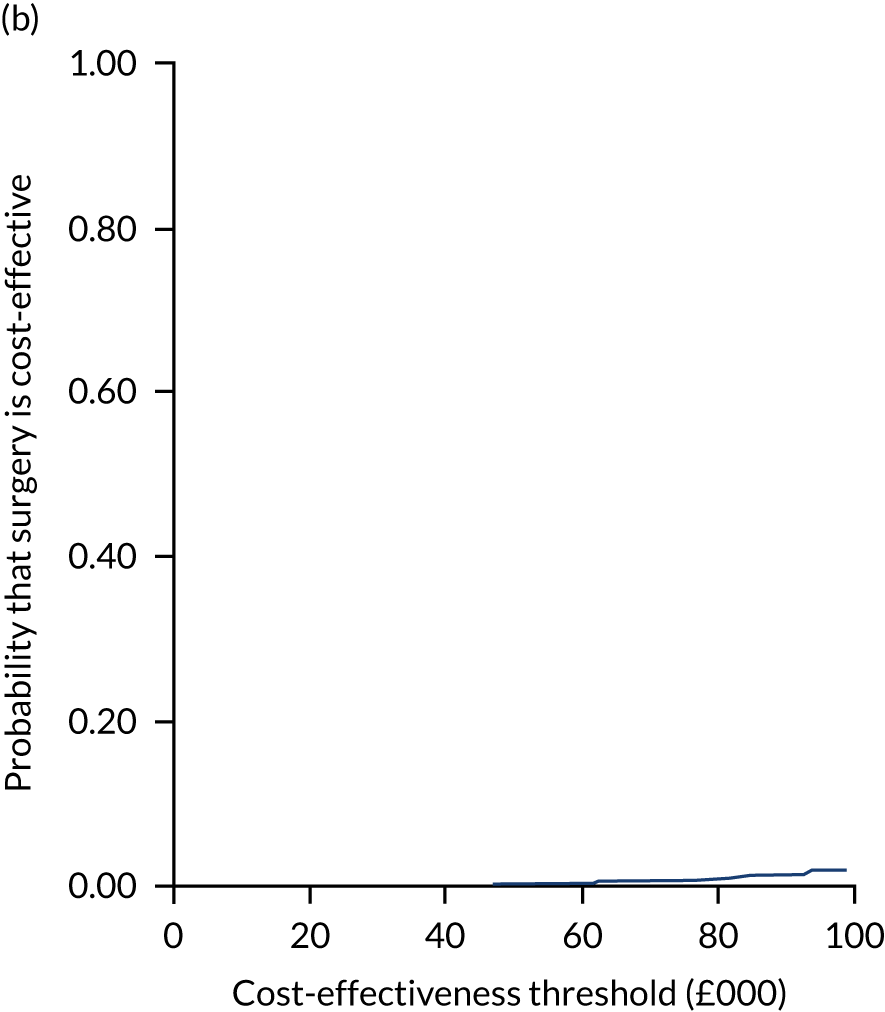
FIGURE 17.
Sensitivity analysis 3: per-protocol (adjusted) analysis based on imputed data. Patients who crossed over from PHT to surgery were included in the surgery group; those who did not receive the surgery or PHT were excluded. (a) Incremental costs; and (b) probability that surgery is cost-effectiveness’. WTP, willingness to pay.
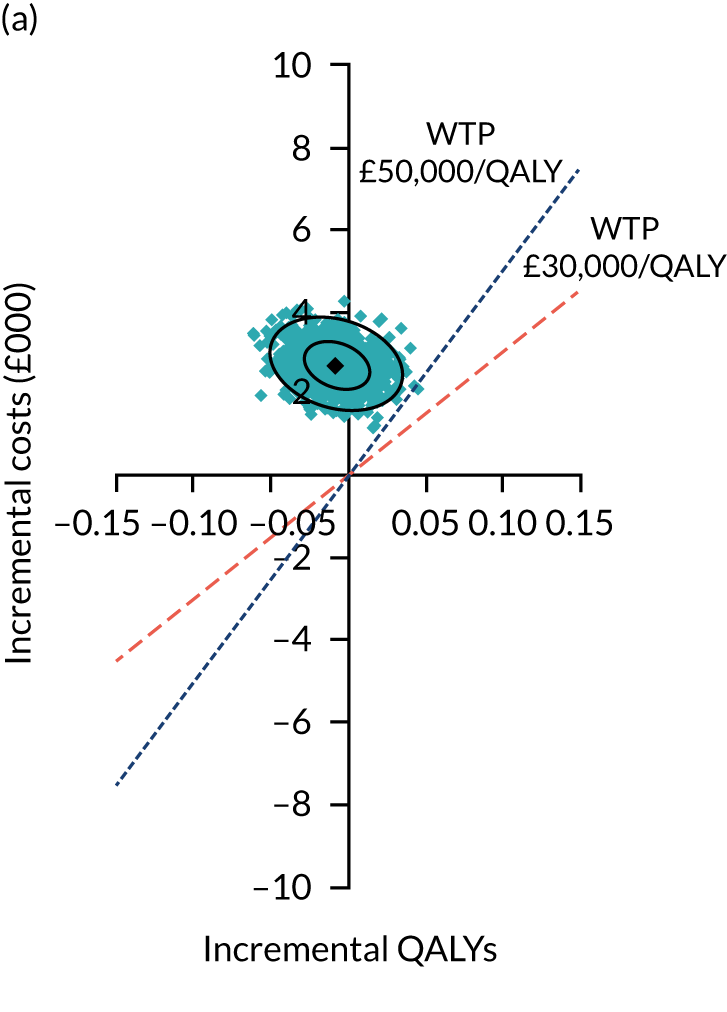
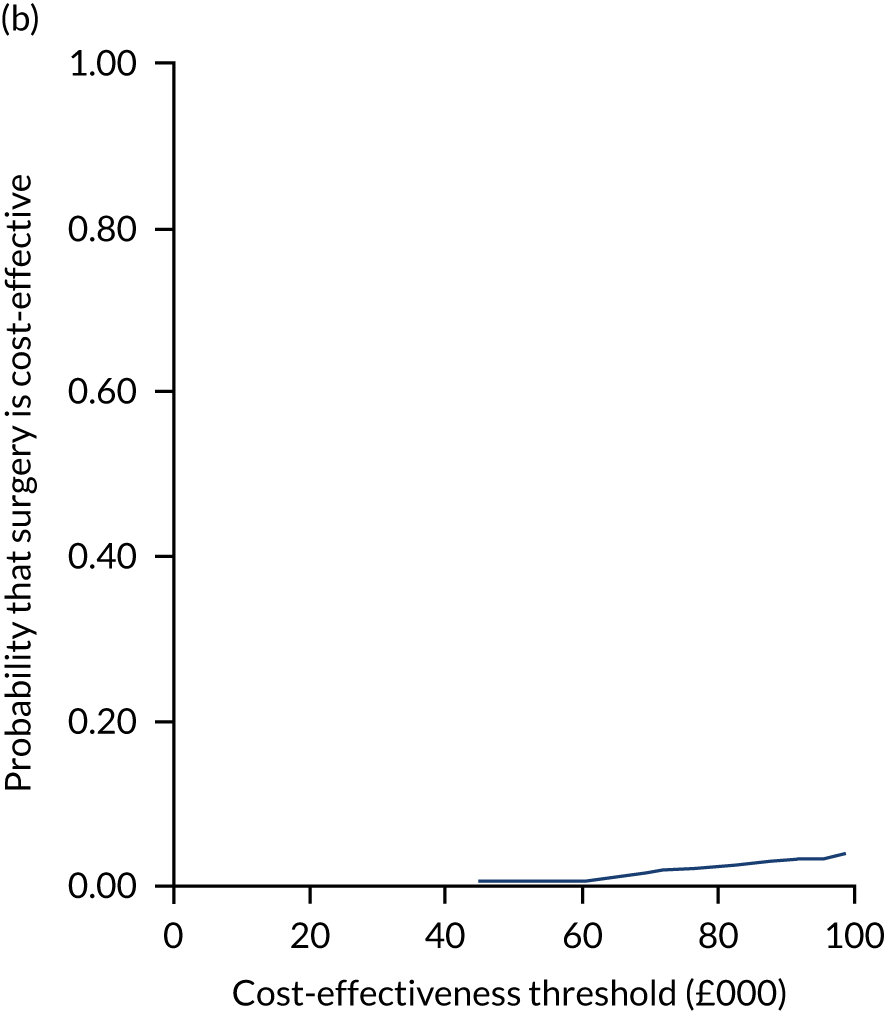
FIGURE 18.
Sensitivity analysis 4: adjusted analysis based on imputed data with the cost of surgery changed from £3045 to £2680 based on HRG code HT15Z (Minor Hip Procedures for Trauma, elective long stay). (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
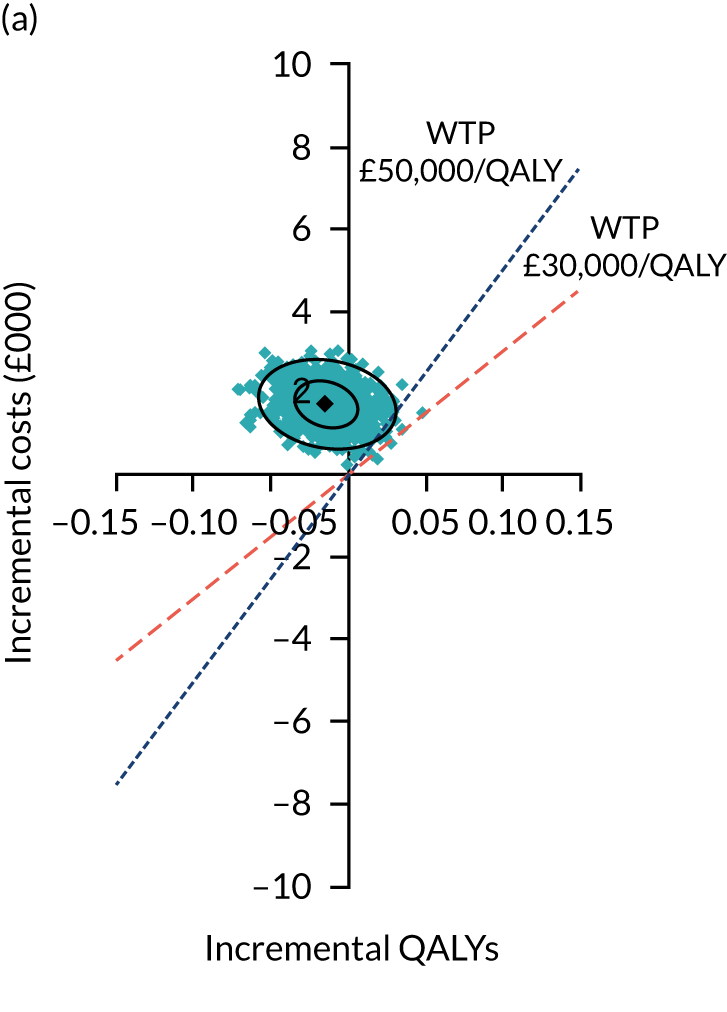
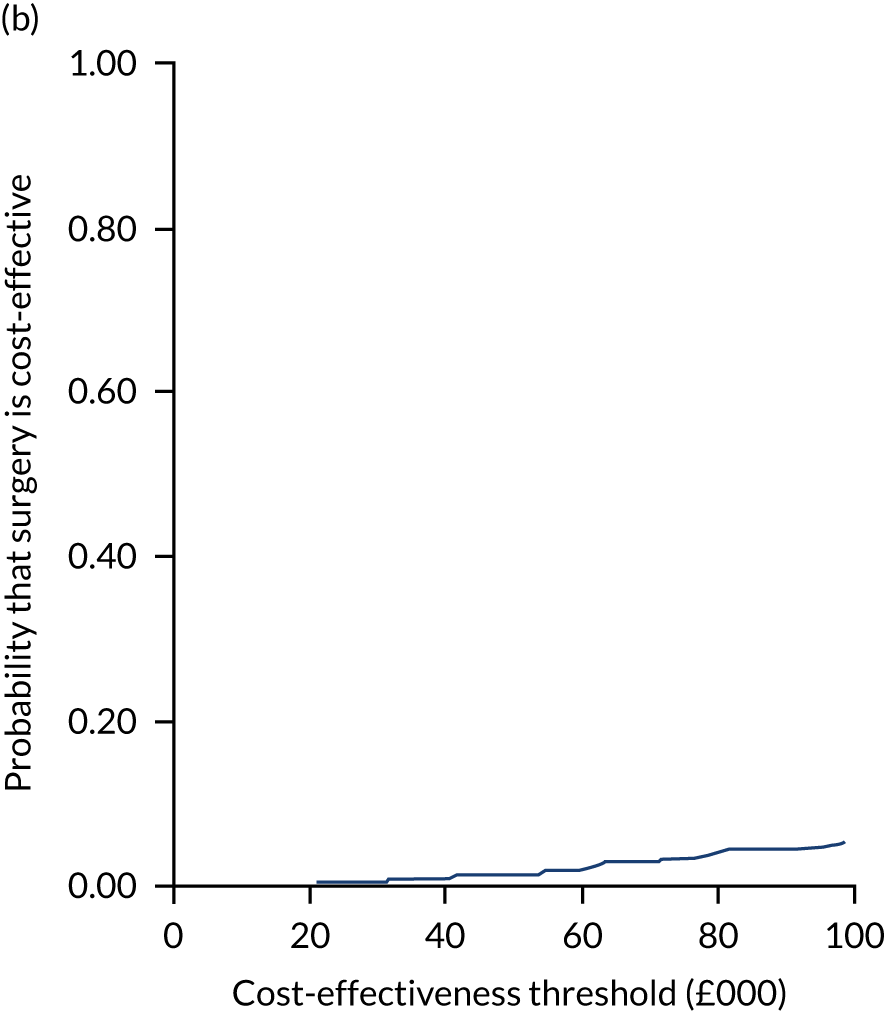
FIGURE 19.
Sensitivity analysis 5: adjusted analysis based on imputed data with the cost of surgery changed from £3045 to £5811 based on HRG code HT12A (Very Major Hip Procedures for Trauma with CC Score 12 +, elective long stay). (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
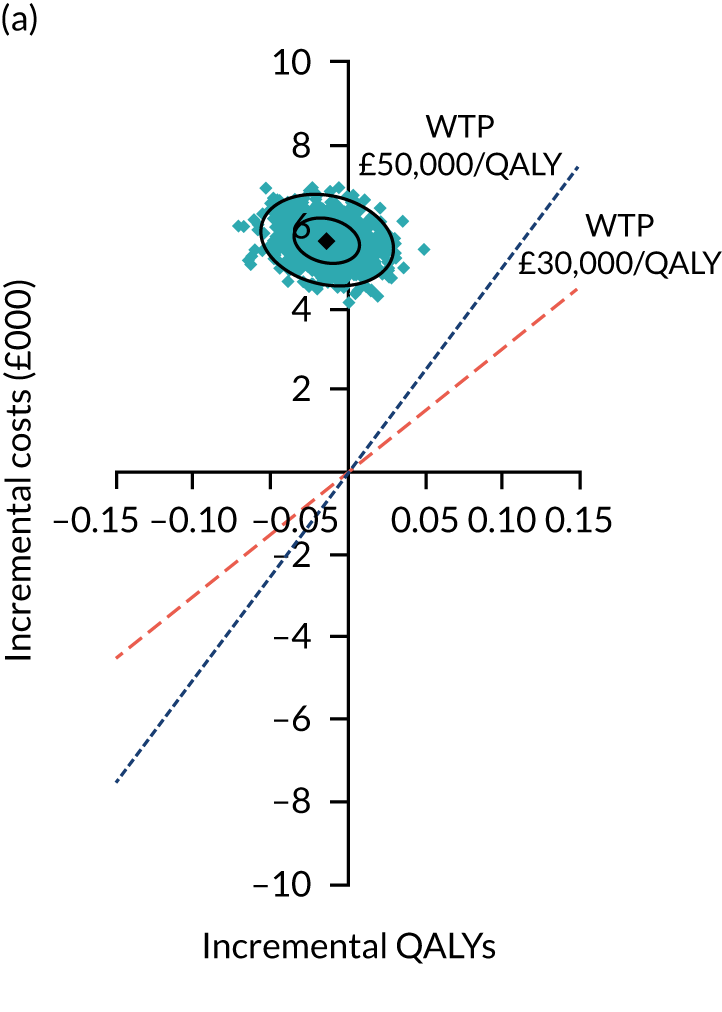
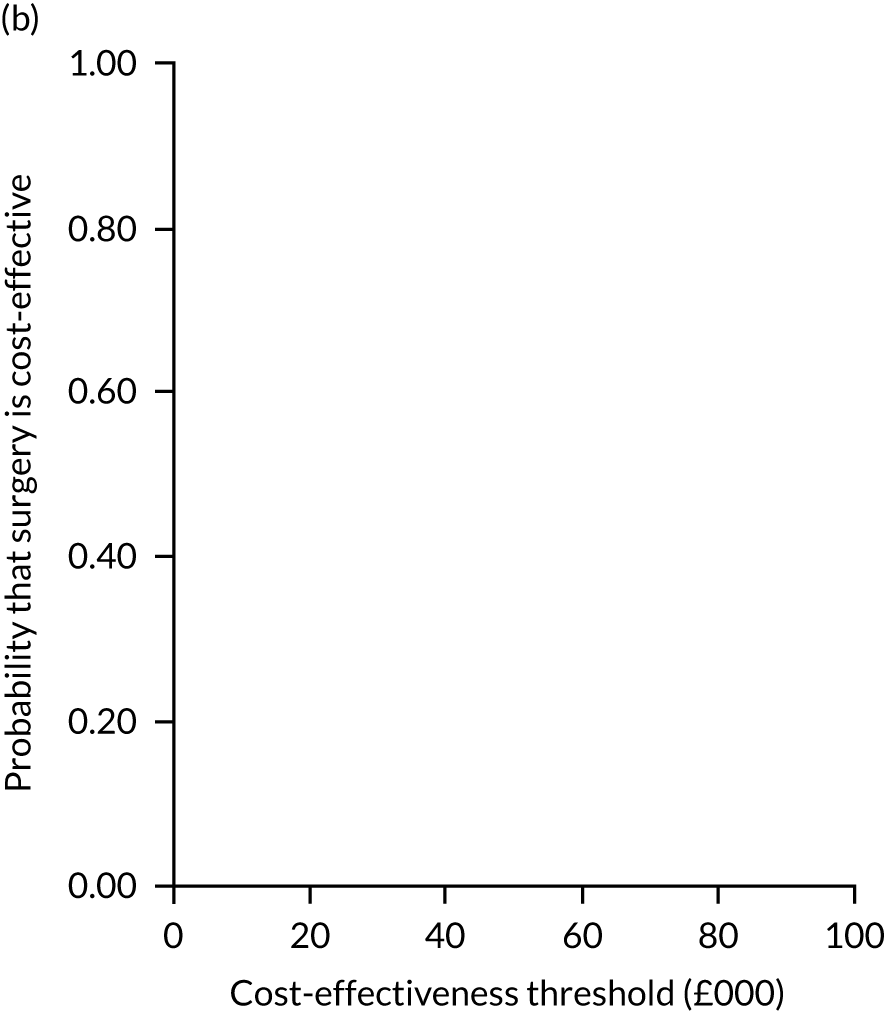
FIGURE 20.
Sensitivity analysis 6: adjusted analysis based on imputed data. Economic costs calculated from a societal perspective (including NHS/Personal Social Services costs, private health costs and non-health-related costs, such as lost income due to illness). (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
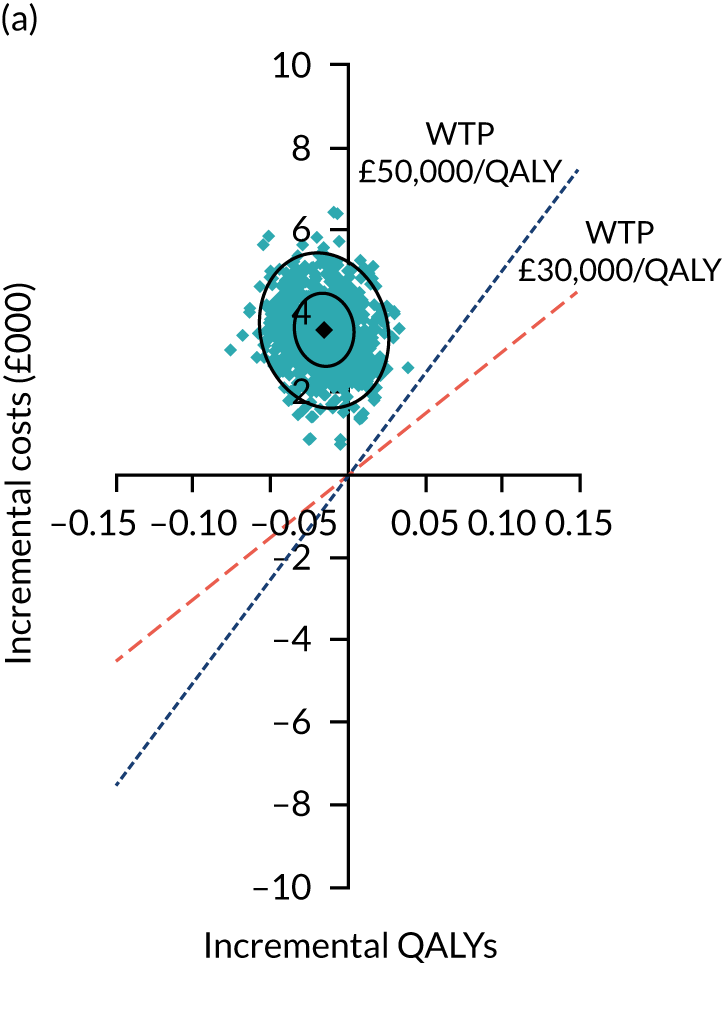
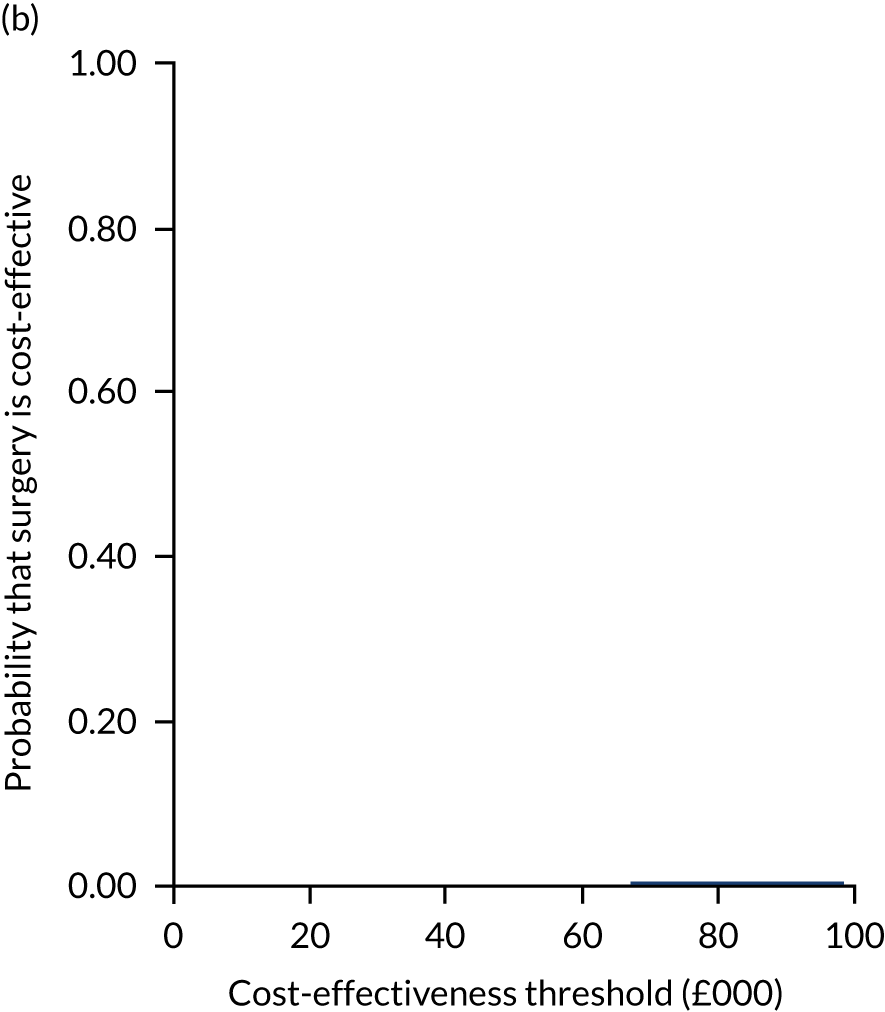
FIGURE 21.
Sensitivity analysis 8: adjusted analysis based on imputed data. Adjusted analysis based on imputed data sets using QALYs generated from SF-6D (via SF-12). (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
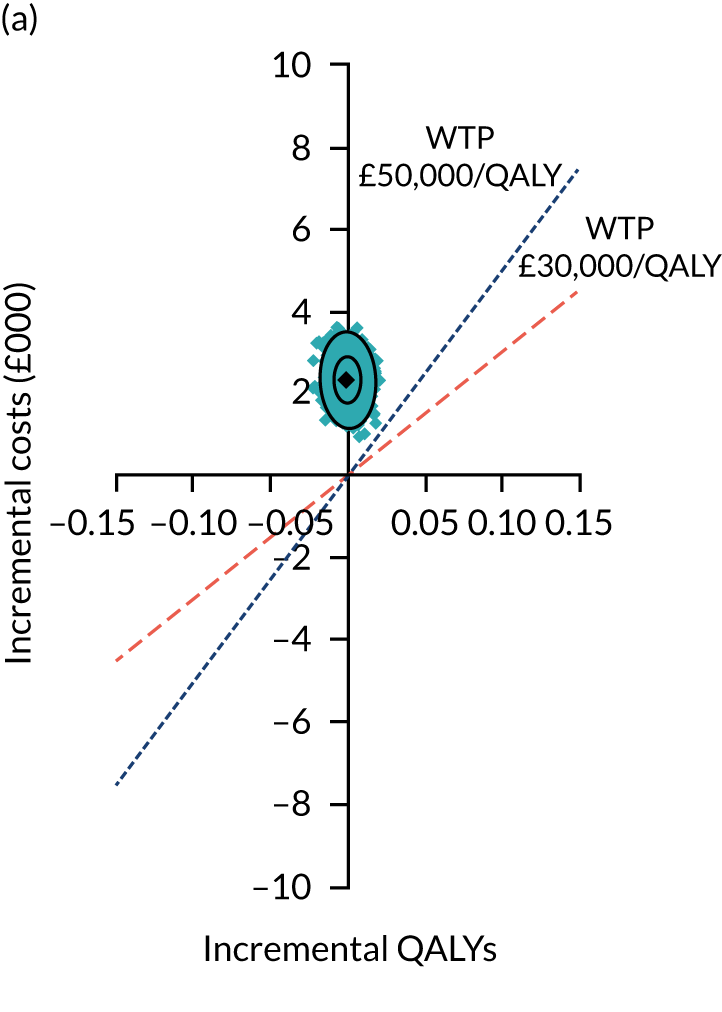
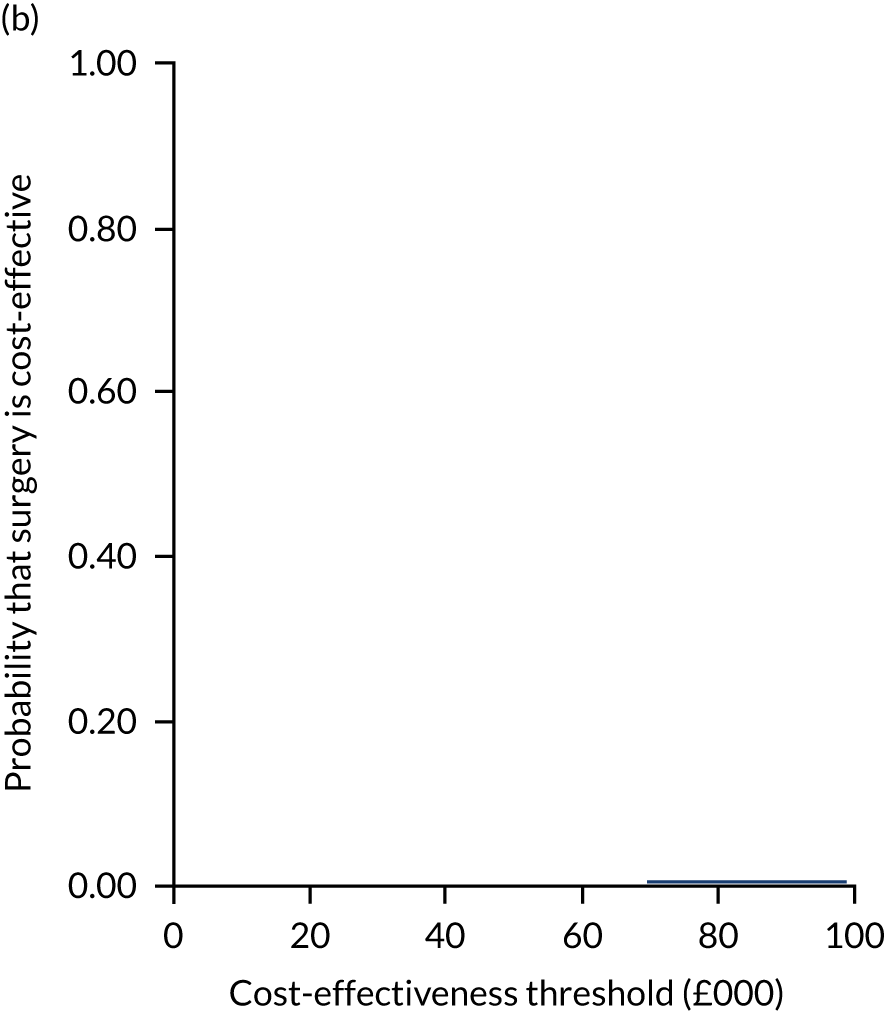
FIGURE 22.
Subgroup of patients recruited into the feasibility study (unadjusted analysis based on multiple imputed data sets). (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
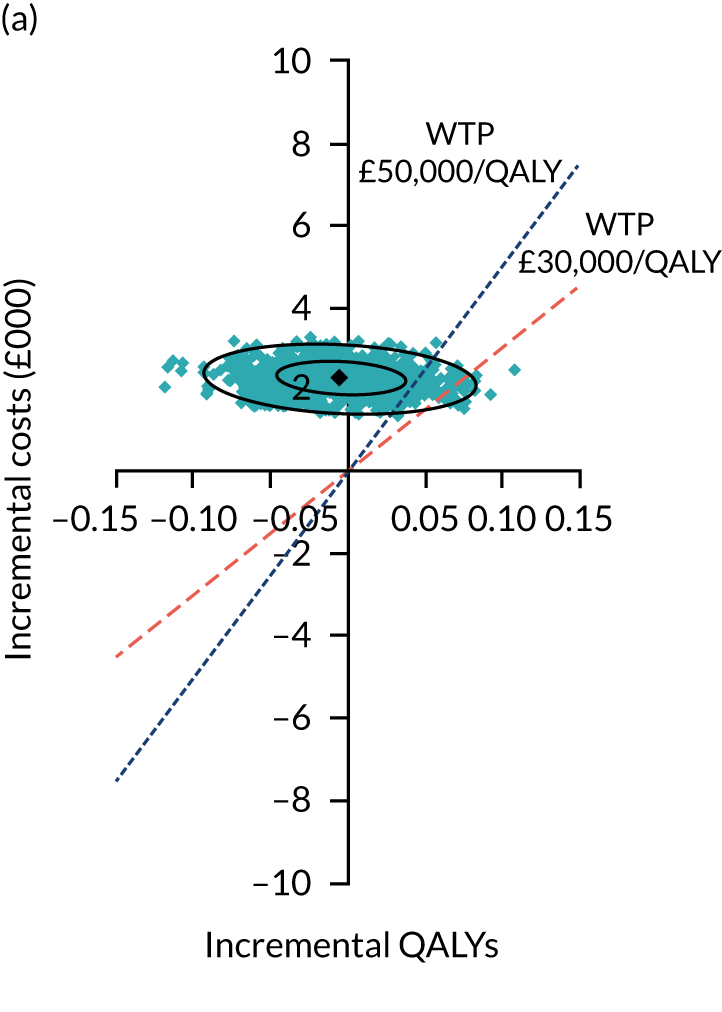
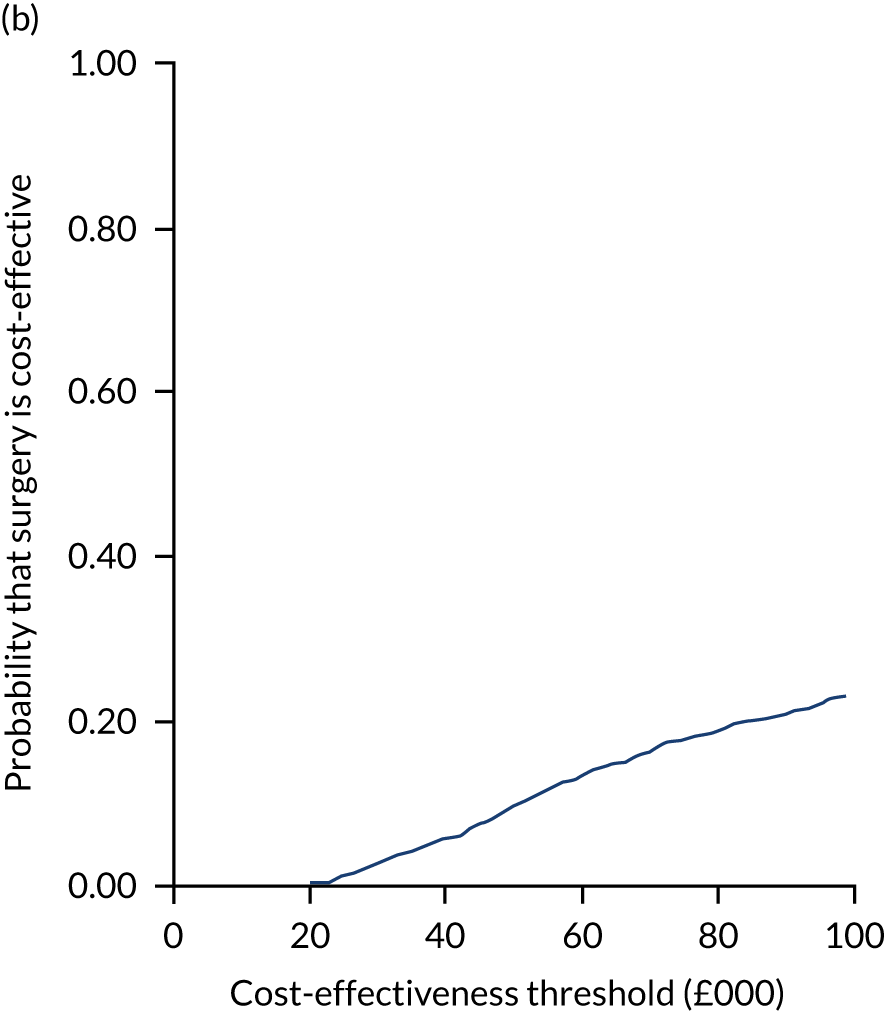
FIGURE 23.
Subgroup of patients recruited into the main study sample (unadjusted analysis based on multiple imputed data sets) with QALYs generated using the interim EQ-5D-5L to EQ-5D-3L UK crosswalk tariffs. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
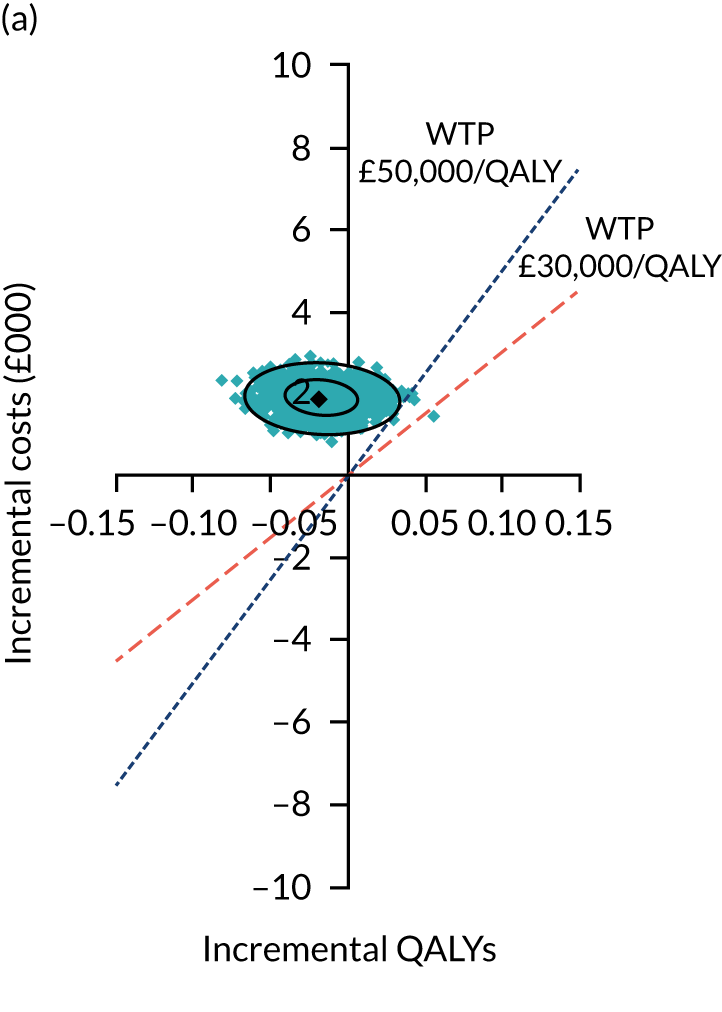
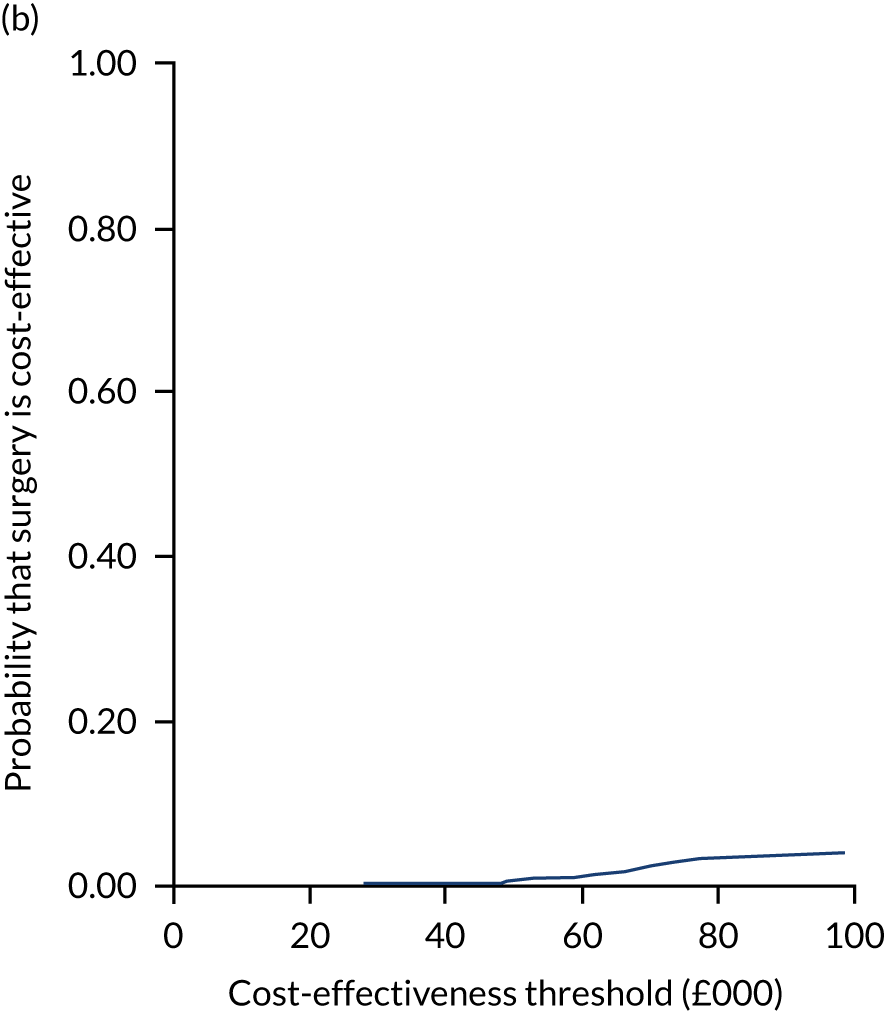
FIGURE 24.
Subgroup of patients recruited into the main study sample (unadjusted analysis based on multiple imputed data sets) with QALYs generated using the new UK EQ-5D-5L tariffs derived by Devlin et al. 64 (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
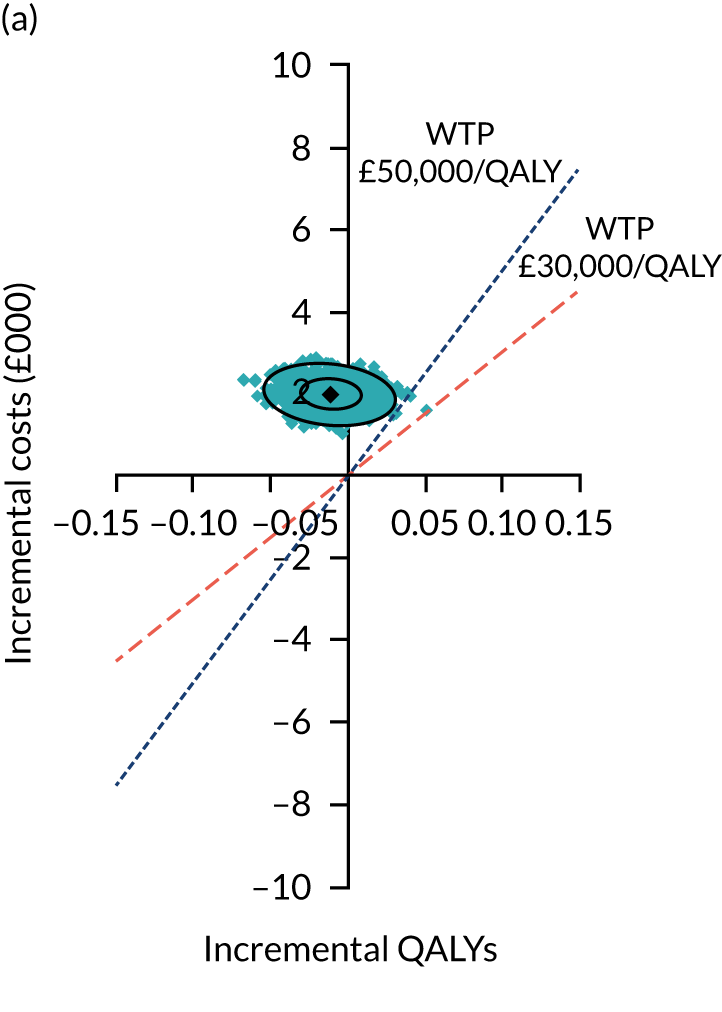
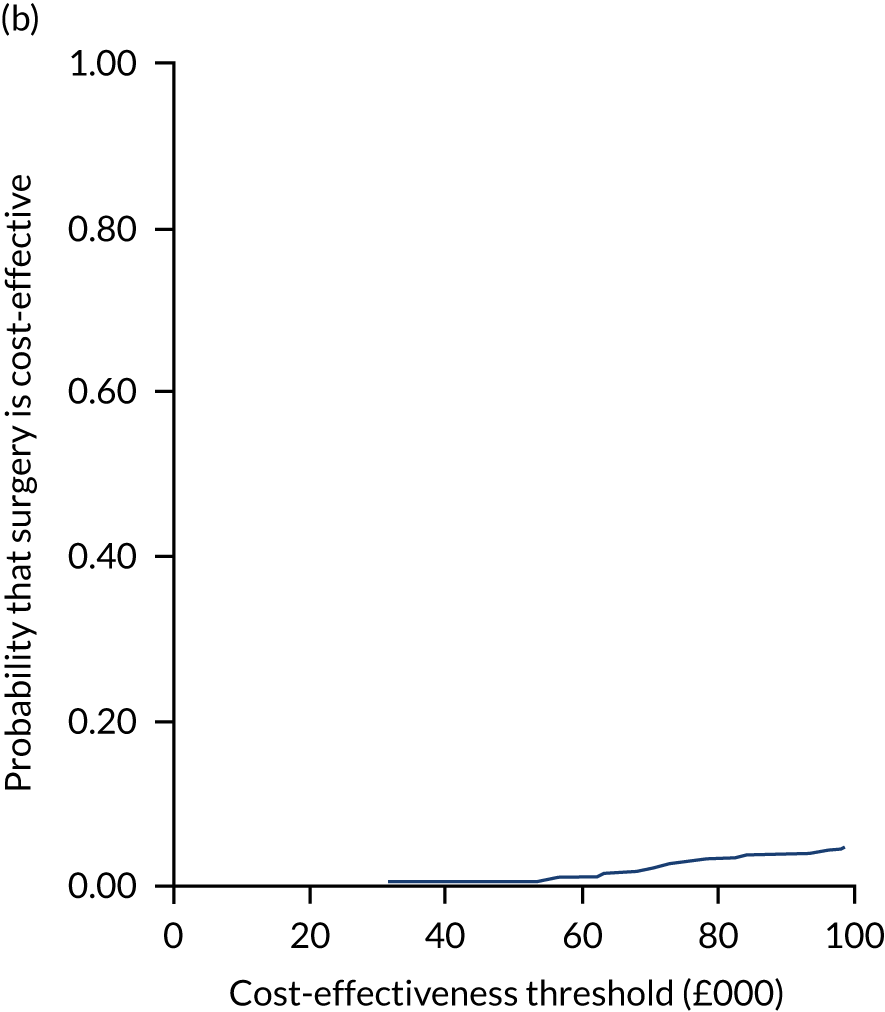
FIGURE 25.
Subgroup of patients with cam FAI syndrome. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
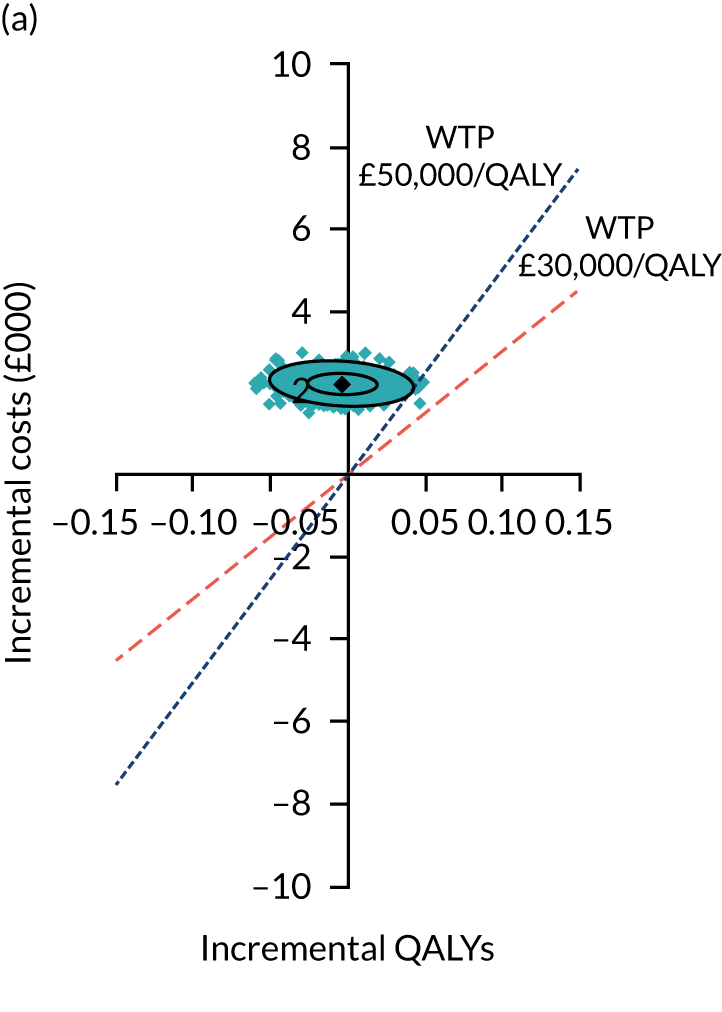
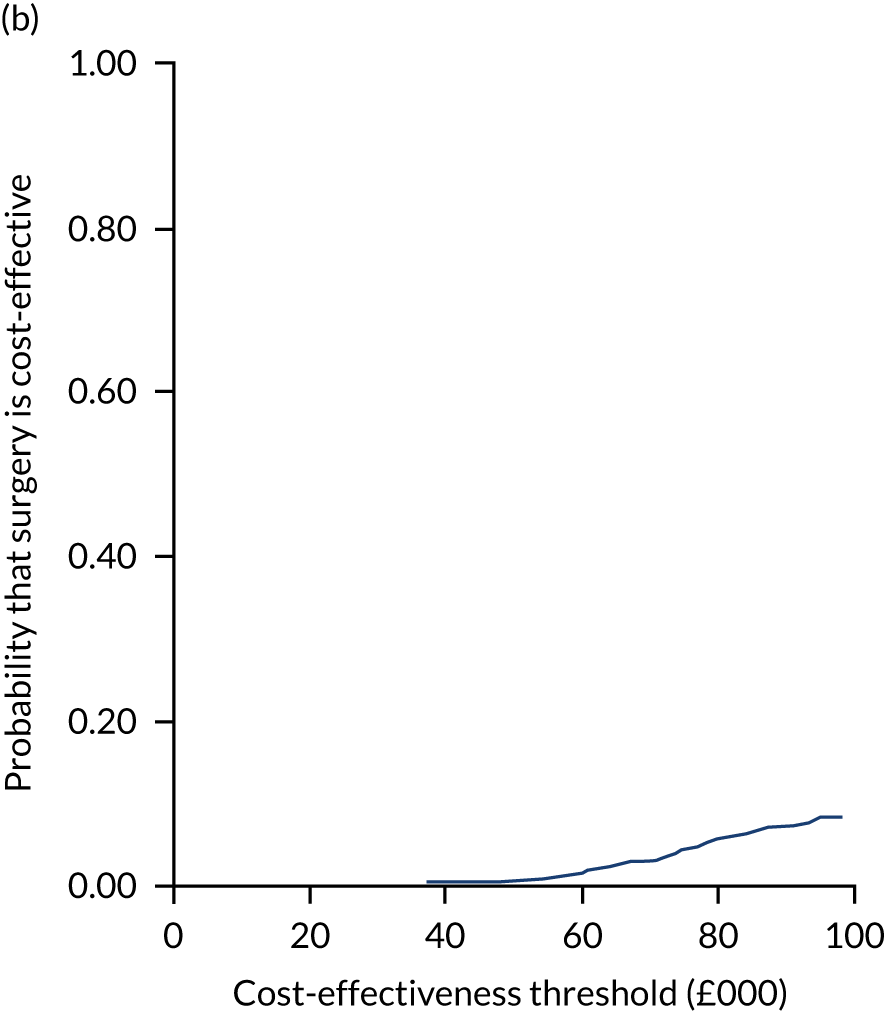
FIGURE 26.
Subgroup of patients with mixed or pincer (non-cam) FAI syndrome. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
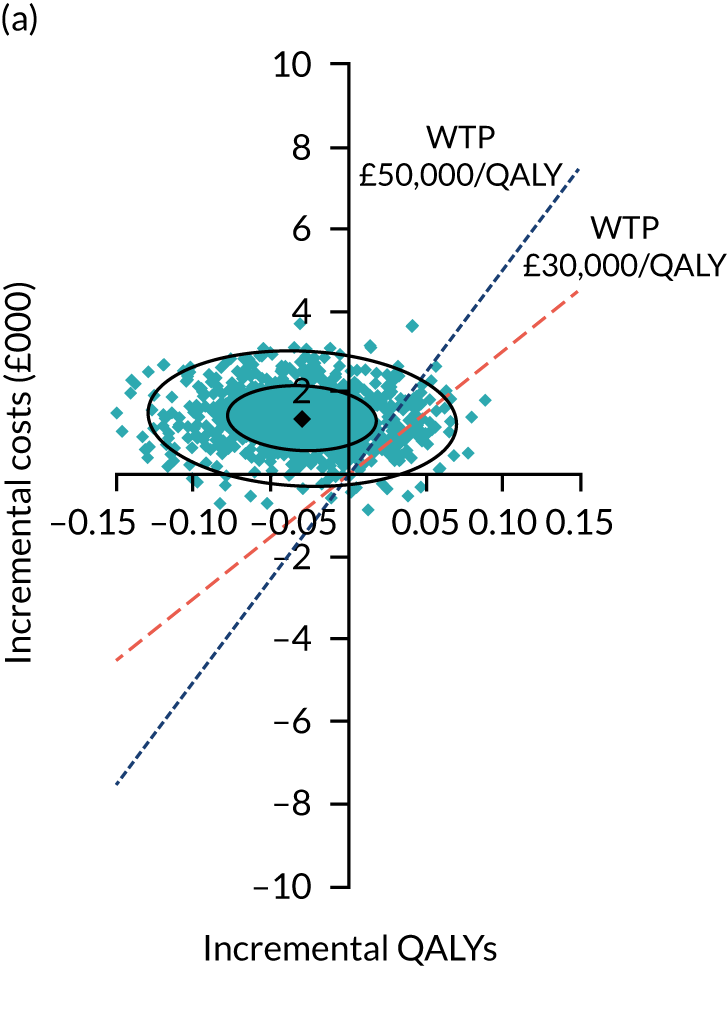
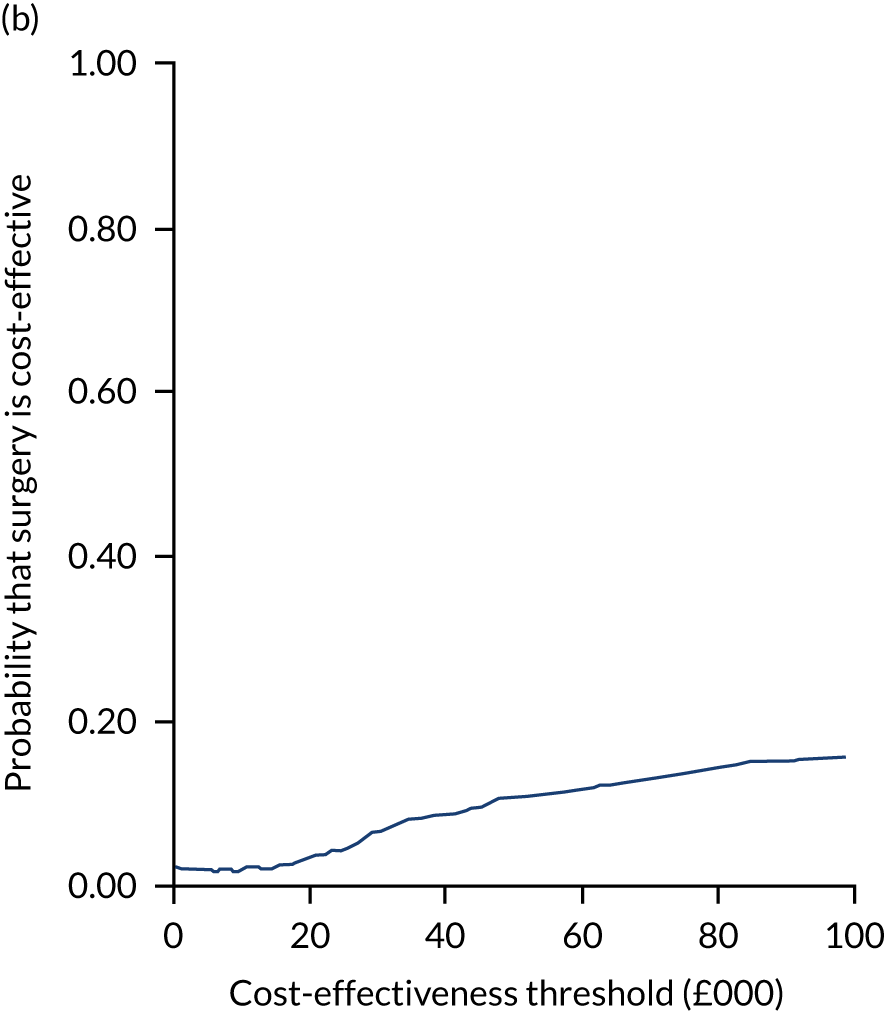
FIGURE 27.
Restricted analysis to women. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
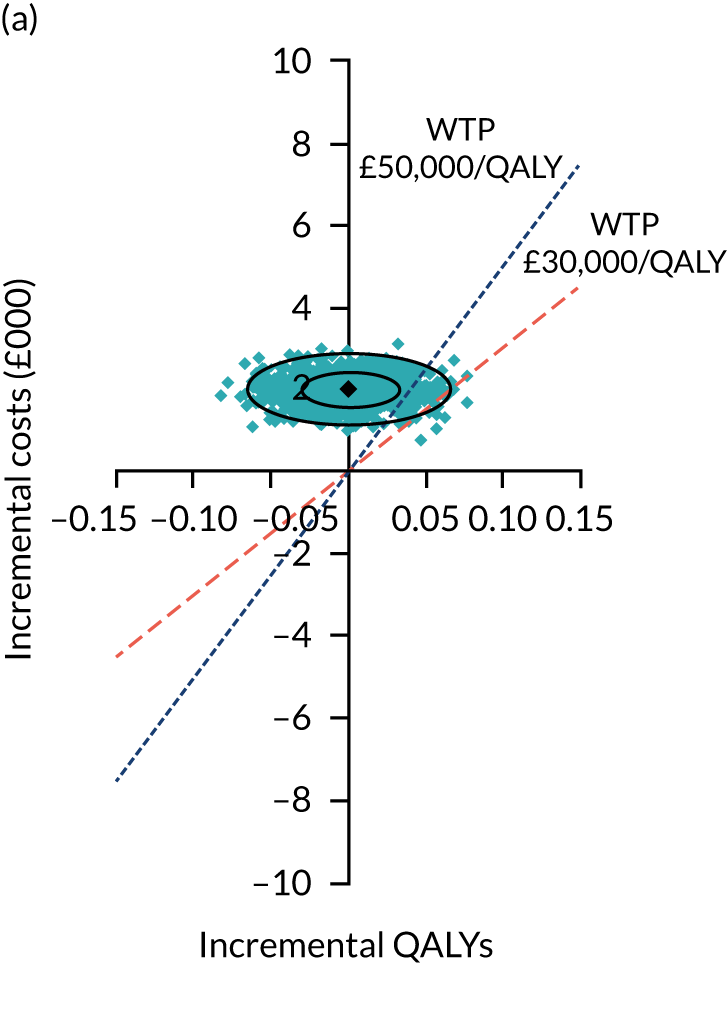
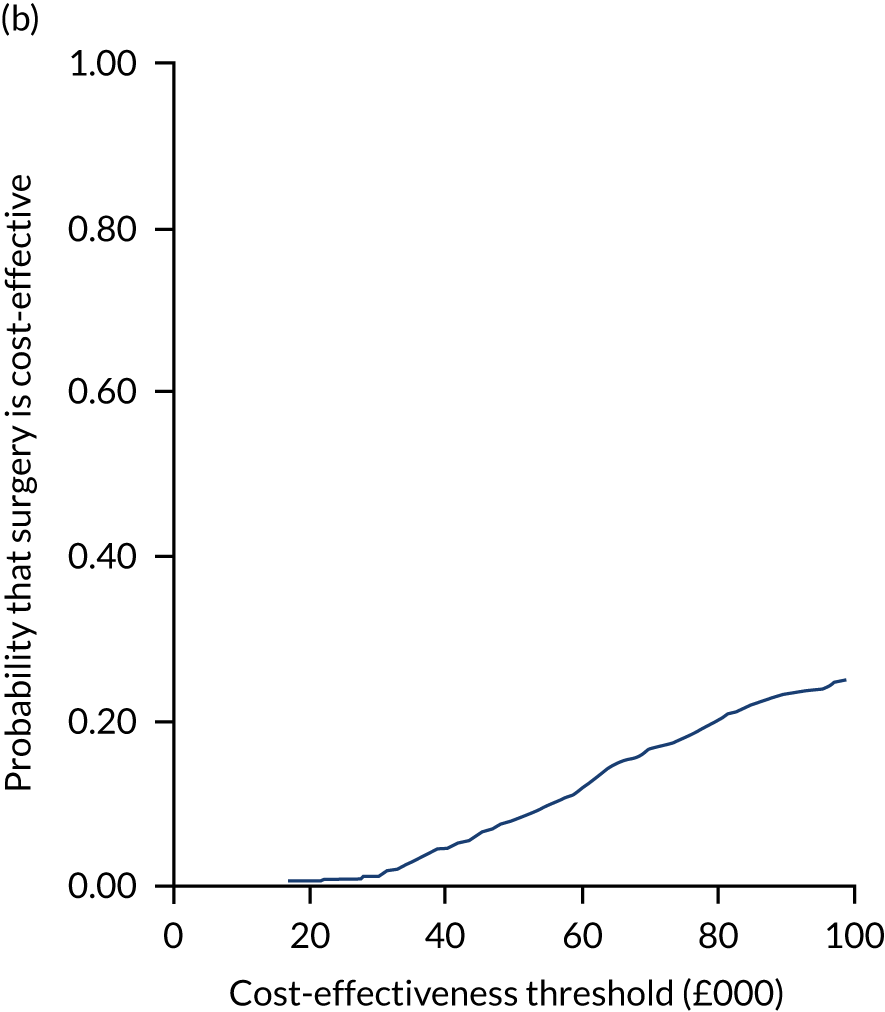
FIGURE 28.
Restricted analysis to men. (a) Incremental costs; and (b) probability that surgery is cost-effective. WTP, willingness to pay.
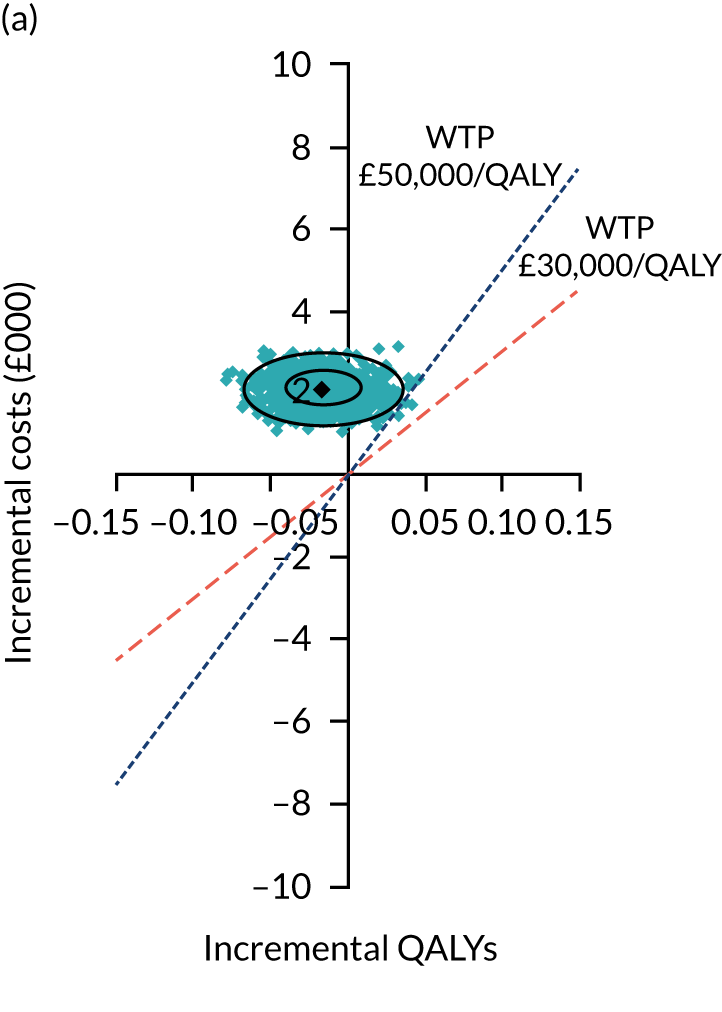
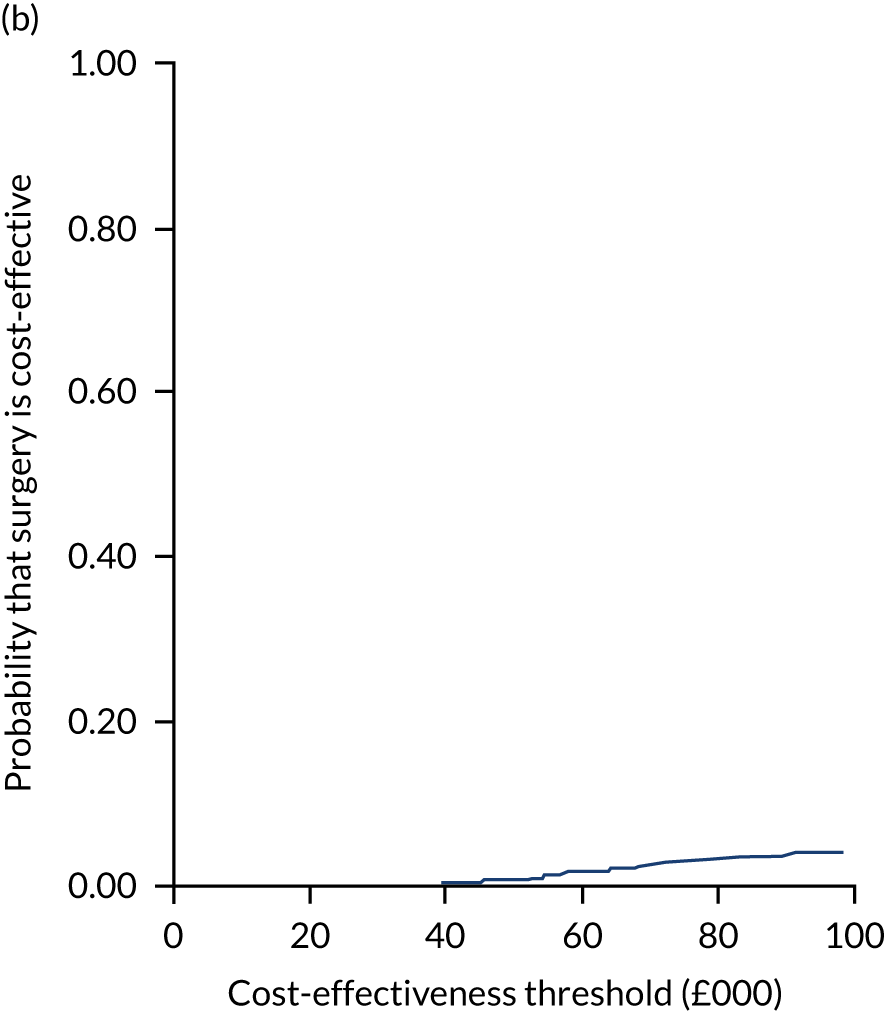
List of abbreviations
- AE
- adverse event
- BNF
- British National Formulary
- CI
- confidence interval
- CONSORT
- Consolidated Standards of Reporting Trials
- CRF
- case report form
- CSP
- Chartered Society of Physiotherapy
- DMC
- Data Monitoring Committee
- EQ-5D
- EuroQol-5 Dimensions
- EQ-5D-3L
- EuroQol-5 Dimensions, three-level version
- EQ-5D-5L
- EuroQol-5 Dimensions, five-level version
- FAI
- femoroacetabular impingement
- GP
- general practitioner
- HRG
- Healthcare Resource Group
- ICER
- incremental cost-effectiveness ratio
- iHOT-33
- International Hip Outcome Tool-33
- IQR
- interquartile range
- MAHORN
- Multicenter Arthroscopy of the Hip Outcomes Research Network
- MCID
- minimum clinically important difference
- MRI
- magnetic resonance imaging
- NICE
- National Institute for Health and Care Excellence
- PHT
- personalised hip therapy
- PI
- principal investigator
- PSSRU
- Personal Social Services Research Unit
- QALY
- quality-adjusted life-year
- QRI
- qualitative recruitment intervention
- R&D
- research and development
- RA
- research associate
- RCT
- randomised controlled trial
- SAE
- serious adverse event
- SD
- standard deviation
- SE
- standard error
- SF-6D
- Short Form questionnaire-6 Dimensions
- SF-12
- Short Form questionnaire-12 items
- SOP
- standard operating procedure
- TMG
- Trial Management Group
- TSC
- Trial Steering Committee
- VAS
- visual analogue scale
- WCTU
- Warwick Clinical Trials Unit
