Notes
Article history
The research reported in this issue of the journal was commissioned by the HTA programme as project number 09/13/02. The contractual start date was in March 2010. The draft report began editorial review in March 2011 and was accepted for publication in September 2011. As the funder, by devising a commissioning brief, the HTA programme specified the research question and study design. The authors have been wholly responsible for all data collection, analysis and interpretation, and for writing up their work. The HTA editors and publisher have tried to ensure the accuracy of the authors’ report and would like to thank the referees for their constructive comments on the draft document. However, they do not accept liability for damages or losses arising from material published in this report.
Declared competing interests of authors
Amar Rangan has received consultancy fees from DePuy International relating to shoulder replacement prostheses. A division of DePuy is involved in marketing products for surgical treatment of the frozen shoulder. The other authors report no conflicts of interest.
Permissions
Copyright statement
© Queen’s Printer and Controller of HMSO 2012. This work was produced by Maund et al. under the terms of a commissioning contract issued by the Secretary of State for Health. This journal is a member of and subscribes to the principles of the Committee on Publication Ethics (COPE) (http://www.publicationethics.org/). This journal may be freely reproduced for the purposes of private research and study and may be included in professional journals provided that suitable acknowledgement is made and the reproduction is not associated with any form of advertising. Applications for commercial reproduction should be addressed to: NETSCC, Health Technology Assessment, Alpha House, University of Southampton Science Park, Southampton SO16 7NS, UK.
2012 Queen’s Printer and Controller of HMSO
Chapter 1 Background
The decision problem
The commissioning brief requested an evidence synthesis comparing the clinical effectiveness and cost-effectiveness of different treatments for frozen shoulder to determine the most appropriate management strategy. The aims of the project were to:
-
evaluate, by way of a systematic review, the clinical effectiveness (including adverse effects) of strategies currently used in the NHS for the management of frozen shoulder and identify the most appropriate intervention by stage of condition, specifically physical therapies, steroid and other shoulder injections, manipulation under anaesthesia (MUA), arthrographic distension, capsular release, watchful waiting and combinations of these interventions
-
collate, by way of a systematic review, patients’ views and experiences of the interventions being assessed in the review
-
identify, by way of a systematic review, the cost-effectiveness of the different interventions in order to inform the development of a decision model
-
develop a decision-analytic model to estimate the cost-effectiveness of alternative treatment options for frozen shoulder
-
make recommendations for clinical practice
-
identify any gaps in the evidence, undertake value of information analysis to assess the potential value of future research on interventions for frozen shoulder and make specific recommendations for further research.
Frozen shoulder
Frozen shoulder is a painful condition in which movement of the shoulder becomes severely restricted. The condition can vary in severity from mild to severe pain and/or from some to severe restriction in movement. It was first described in 1875 by the French pathologist Duplay, who named it péri-arthrite scapula-humérale. The most well-known definition and name for the condition was provided in 1934 by an American surgeon EA Codman. He defined it as coming on slowly ‘with pain usually felt near the insertion of the deltoid; inability to sleep on the affected side; painful and incomplete elevation and external rotation; restriction of both spasmodic and mildly adherent type; atrophy of the spinati; little local tenderness; [and] X-rays negative except for bone atrophy’ and named it ‘frozen shoulder’. 1 However, there is an acknowledged absence of a specific definition of the condition2,3 and of a diagnostic label,3 with additional names for frozen shoulder including adhesive capsulitis, painful stiff shoulder, retractile capsulitis, Checkrein shoulder, monoarticular arthritis and steroid-sensitive arthritis. 4 Throughout this report the term ‘frozen shoulder’ will be used.
Frozen shoulder is typically characterised as having three overlapping phases:5
-
phase 1, in which there is progressive stiffening and loss of motion in the shoulder with increasing pain on movement, which may be worse at night (months 2–9), usually referred to as the painful phase
-
phase 2, in which there is a gradual decrease in pain but stiffness remains and there is considerable restriction in the range of movement (months 4–12), usually referred to as the stiffening or ‘freezing’ phase
-
phase 3, in which there is an improvement in range of movement (months 12–42), usually referred to as the resolution phase.
There are different views about the underlying fundamental process: inflammation, scarring and scarring produced in reaction to inflammation. 6 Bunker describes a pathology of fibrous contracture of the rotator interval and coracohumeral ligament of the shoulder joint. 7 In addition to the capsular contracture, there is often a reduced joint volume: 3–4 ml compared with the normal volume of 10–15 ml. 8 The formation of new blood vessels in the synovial membrane, which is most marked in the rotator interval area, is a feature of the early stage of the condition. In the stiff phase this declines and thick white scar tissue can be seen and palpated within the capsule at arthroscopic and open surgery. Histology also shows thickening and contracture of the capsule. A significant increase in fibroblasts, which lay down scar tissue and myofibroblasts, which contract scar tissue, has been identified. 8 In addition, pathological studies have identified the presence of inflammatory cells (mast cells, T cells, B cells and macrophages), suggesting a process of inflammation leading to scarring. 9
The cumulative incidence of frozen shoulder is estimated at 2.4 per 1000 population per year based on a Dutch general practice sample. 10 A large UK-based primary care study found that frozen shoulder affected 8.2% of men and 10.1% of women of working age. 11 In contrast, frozen shoulder was estimated to affect only 0.75% of the UK population based on a specialist shoulder surgeon’s hospital care experience. 8 This discrepancy in estimated prevalence may be explained by the fact that only the most resistant cases of frozen shoulder are seen in a hospital setting. 6 Frozen shoulder most commonly occurs in people in their mid-50s and is thought to be slightly more common in women than in men.
Frozen shoulder can be described as either primary (idiopathic), whereby the aetiology is unknown, or secondary, when it can be attributed to another cause. Secondary frozen shoulder has been defined as that associated with diabetes (although some classify this as primary frozen shoulder), trauma, cardiovascular disease and hemiparesis. The incidence of frozen shoulder is reported to be 10–36% amongst people with diabetes, who tend not to respond as well to treatment as those without diabetes. 5 The proportion of frozen shoulder attributed to trauma varies (9–33%) and the trauma is often not particularly severe. There is also a discrepancy between the extent of trauma and severity of subsequent frozen shoulder. 4 These uncertainties mean that in practice it can be difficult to differentiate between primary and secondary frozen shoulder.
Although for most people frozen shoulder is a self-limiting condition of approximately 1–3 years’ duration, it can be extremely painful and debilitating; people with the condition may struggle with basic daily activities and be worn down by sleep disturbance as a result of the pain. 12 As well as interfering with domestic and social activities, it can affect the ability to work. There may not be a complete resolution for all patients and there is variation across case series in the proportion of patients who do not regain full shoulder motion,5 possibly a reflection of variation in how outcome was assessed. Based on the largest series of patients with a mean follow-up of 4.4 years from onset of symptoms, 59% had normal or near normal shoulders, 35% had mild to moderate symptoms with pain being the most common complaint and 6% had severe symptoms at follow-up. 13 Recurrence is unusual although it is estimated that the other shoulder becomes affected in 6–17% of patients within 5 years.
Diagnosis and management
Diagnosis, in both primary and secondary settings, is based on clinical examination and medical history. A key alerting feature is restriction of shoulder movement in all directions – passive and active range of movement. 14 Blood tests, radiography and ultrasound are usually normal and not routinely required unless history or physical examination suggests the need to rule out other pathologies, for example if rheumatoid arthritis or osteoarthritis is suspected. Frozen shoulder is commonly managed in the primary care setting. In a UK study of patterns of referral and diagnosis of shoulder conditions it was estimated that 22% of patients were referred to secondary care, up to 3 years following initial presentation, although most referrals occurred within 3 months. 15 There is little evidence available on referral patterns in relation to frozen shoulder specifically. 12
There are a number of management options, both surgical and non-surgical, but there is no consensus about management. For the purpose of the report we have classified the interventions as conservative and invasive (Table 1).
| Conservative treatment | Steroid injections |
| Physical therapy | |
| Acupuncture | |
| Invasive treatment | Sodium hyaluronate |
| MUA | |
| Distension | |
| Capsular release |
The aims of treatment, depending on stage of condition, are pain relief, increasing arm movement, reducing the duration of symptoms and return to normal activities for the patient. Treatment options include:
-
Watchful waiting or ‘supervised neglect’, which involves explaining the condition to the patient, and education and advice about mobilisation within pain limits and use of pain relief.
-
Oral medications such as non-steroidal anti-inflammatory drugs (NSAIDs) and oral steroids. Although the use of oral steroids is described in the literature they are not a commonly used intervention in the UK.
-
Gentle exercise supervised by a physiotherapist or as part of a home exercise programme.
-
Physical therapies to help regain range of movement and prevent further stiffness. These encompass the wide range of techniques used by physiotherapists as well as acupuncture, and chiropractic and osteopathic techniques. Several different regimes have been described in the literature including supervised exercises, mobilisation, physiotherapy and use of electrotherapeutic interventions such as laser therapy and ultrasound. Mobilisation is therapist-applied passive movement of joints or other structures performed in such a way that they are always within the control of the patient. Electrotherapies include transcutaneous nerve stimulation (TENS), interferential therapy, short-wave diathermy (SWD) and pulsed short-wave diathermy (PSWD), and ultrasound. TENS and interferential therapy consist of electric pulses or currents and have an analgesic effect. SWD and PSWD use radio frequency energy to generate heat in tissues, which has an analgesic effect and reduces muscle spasm and joint stiffness. Ultrasound, mechanical vibration at very high speed, also generates heat and has the same therapeutic effect as SWD and PSWD. 6 Low-level laser therapy is non-thermal and is believed to reduce pain and inflammation, although the exact mechanism of its effect is unknown. 16 Acupuncture is a form of ancient Chinese medicine in which fine needles are inserted into the skin at certain points on the body. Chiropractic uses a range of manual therapies, with an emphasis on manipulation of the spine, whereas osteopathy uses gentle stretching, massage and manipulation of muscles and joints. 17
-
Intra-articular corticosteroid injections to reduce inflammation and provide pain relief. A range of different doses and number of injections is described in the literature. This intervention is usually delivered in the primary care setting but also in the secondary care setting, depending on how services are organised in a particular region.
-
Arthrographic distension (also called hydrodilatation), which involves controlled dilatation of the joint capsule with sterile saline or other solution such as local anaesthetic or steroid, guided by radiological imaging (arthrography). The procedure is performed under local anaesthetic.
-
MUA, in which the shoulder is freed by rotation while the patient is under short general anaesthesia. This can be undertaken as a day procedure.
-
Arthroscopic capsular release, a surgical procedure conducted under general or regional anaesthesia during which the contracted tissue is released. Open capsular release is another surgical option, usually recommended in those resistant to arthroscopic intervention. Both can be undertaken as a day procedure.
These interventions can be used individually or in combination, depending on the disease stage. There is currently not a consensus about the overall management of the condition and the sequence in which treatments should be offered to patients. A recent survey of 303 UK health-care professionals [general practitioner (GPs), including those with a special interest, physiotherapists, advanced scope physiotherapists and orthopaedic surgeons] found that the professional groups had different views on the most appropriate treatment pathway for frozen shoulder. 18 However, there appears to be a fairly consistent view that the treatments used should depend on the phase of the disease and/or that a step-up approach should be adopted in terms of the degree of treatment invasiveness. 5,12,18,19 There is a suggestion that aggressive mobilisation should be avoided in the early, severely painful phase. 12,14,19 Surgical intervention is generally, although not exclusively, used when the condition is resistant to the other interventions,19 although there is no consensus as to what time point or level of pain should indicate surgical intervention. 18
The most commonly used or recommended interventions for the painful phase, in the recent UK survey of health-care professionals, were conservative treatment (watchful waiting, education, oral pain relief) and physical therapy (mainly physiotherapy and mobilisation), each recommended in one-third of responses, and intra-articular steroid injection, recommended in 18% of responses. For patients in the resolution phase, surgery was the most preferred option (mainly MUA and arthroscopic capsular release), recommended in almost half of the responses, followed by conservative treatment (12%) and physical therapy (19%).
Previous systematic reviews
Several systematic reviews had previously been undertaken on interventions for frozen shoulder, some of which focused on shoulder pain in general and included a range of conditions. Those interventions evaluated were oral steroids,20 corticosteroid injections,21–23 physiotherapy,24–26 acupuncture,27 arthrographic distension28 and multiple interventions29 (Table 2). None of the literature searches was recent and the reviews required updating (Table 2). In addition, some of the reviews did not report results by type of shoulder complaint. A recent review of systematic reviews on frozen shoulder noted the tendency to focus on a single intervention and that updated reviews were required. 30
| Study | Intervention | End date for literature search |
|---|---|---|
| Alvado 200126 | Physical therapy | 1999 |
| Arroll 200523 | Corticosteroids | 2004 |
| Buchbinder 200620 | Oral steroids | 2005 |
| Buchbinder 200828 | Arthrographic distension | 2006 |
| Buchbinder 200321 | Corticosteroids | 2002 |
| Cleland 200225 | Physical therapy | 2000 |
| Green 199829 | Multiple intervention | 1995 |
| Green 200324 | Physiotherapy | 2002 |
| Green 200527 | Acupuncture | 2003 |
| Shah 200722 | Corticosteroid injections | 2006 |
It is apparent from previous reviews that there is variation in how frozen shoulder is defined across studies. A review of 21 randomised controlled trials (RCTs) of interventions for frozen shoulder could not derive a consistent description of the condition from the trials investigating this patient group. 2 Although the RCTs required that participants had restricted shoulder movement, there was inconsistency across trials in the number of degrees of restriction, the type of restriction (active or passive) and the direction of the restriction (abduction or external rotation). 2 This highlights the difficulty of applying a strict definition for frozen shoulder within the context of a systematic review.
Focus of the synthesis
We undertook a systematic review of a range of NHS-relevant interventions for the treatment of primary frozen shoulder with the aim of informing a decision-analytic model. Previous reviews have tended to assess single treatments. The main focus was on comparing the main treatment options for frozen shoulder and identifying the most appropriate treatment by stage of frozen shoulder. Although variations within treatments were included in the synthesis, the main interest was in questions such as whether there is additional benefit from providing physiotherapy following steroid injection. Given the range of interventions being considered and the possibility that all treatment options would not have been compared in head-to-head in trials we planned to undertake a mixed-treatment comparison (MTC) in addition to pair-wise meta-analysis. MTC is an extension of traditional meta-analysis in which trials comparing the same intervention and the same comparator are pooled to estimate an overall treatment effect. A MTC overcomes the limitations of standard meta-analysis when there are no or few head-to-head comparisons or when the decision problem requires the comparison of several interventions. 31,32 A ranking of interventions, based on the probability that each treatment is best,33 can be produced. This is of particular value when several treatment options are under consideration.
Chapter 2 Methods
Overview
There were four planned components for the assessment: a systematic review of the clinical effectiveness of the interventions, a systematic review of patients’ views and experiences of the treatments, a systematic review of studies assessing the cost-effectiveness of the treatments and an assessment of the cost-effectiveness of the alternative treatment options for primary frozen shoulder. At various stages of the assessment the review team consulted with an advisory group including clinicians, an economist, researchers undertaking other research on frozen shoulder, service users and a specialist in dissemination.
Review of clinical effectiveness and cost-effectiveness
The systematic review of studies of clinical effectiveness and cost-effectiveness was undertaken following the general principles outlined in Centre for Reviews and Dissemination (CRD) guidance. 34
Literature searches
The literature searches sought to identify papers on the use of physical therapies, steroid and other shoulder injections, MUA, arthrographic distension, capsular release, watchful waiting and combinations of these interventions for people with a primary diagnosis of frozen shoulder. A range of study types [systematic reviews, RCTs, economic evaluations, quasi-experimental studies (i.e. with a control group), case series of at least 50 participants] were eligible for inclusion and so the use of a study type filter in the search strategy was not considered appropriate.
The following databases were searched: MEDLINE, MEDLINE In-Process & Other Non-Indexed Citations, Cumulative Index to Nursing and Allied Health (CINAHL), EMBASE, Science Citation Index, BIOSIS Previews, Physiotherapy Evidence Database (PEDro), Cochrane Database of Systematic Reviews (CDSR), Database of Abstracts of Reviews of Effects (DARE), Health Technology Assessment (HTA) database, Cochrane Central Register of Controlled Trials (CENTRAL), PASCAL, Manual, Alternative and Natural Therapy (MANTIS), Latin American and Caribbean Health Sciences Literature (LILACS) and NHS Economic Evaluation Database (NHS EED). In the study protocol the Office of Health Economics Health Economic Evaluations Database (OHE HEED) was listed as one of the databases to be searched but access to this database was not available at the time that the searches were conducted. The searches were not restricted by language. The databases were searched from inception up to March 2010 (22 March for MEDLINE). Update searches were undertaken of MEDLINE and EMBASE on 5 January 2011.
In addition, information on studies in progress, unpublished research or research reported in the grey literature was sought by searching a range of relevant databases including Conference Proceedings Citation Index: Science, Health Management Information Consortium (HMIC), ClinicalTrials.gov and National Technical Information Service (NTIS). Furthermore, the reference lists of relevant systematic reviews were checked to identify further studies.
The websites of some relevant organisations were also scanned: the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) (www.niams.nih.gov/default.asp), the British Elbow & Shoulder Society (BESS) (www.bess.org.uk/), the National Physiotherapy Research Network (www.csp.org.uk/director/members/research/networkingandsupport/nationalphysiotherapyresearchnetwork.cfm) and the Primary Care Rheumatology Society (www.pcrsociety.org/).
The search strategy was developed using the MEDLINE database as the primary database (Table 3). In terms of the PICOS formula (patient, intervention, comparator, outcome, study design), the search strategy focused on population and intervention. The strategy was constructed so that lines 1–18 contain the various terms for frozen shoulder with these being combined at line 19. In the remainder of the strategy potential interventions for this condition are grouped together and then subsequently combined with line 19. For example, lines 28–30 seek to retrieve records for arthroscopy, arthroscopic release or capsular release, all combined in line 31 and subsequently further combined with line 19 so that line 32 potentially identifies records containing terms for both frozen shoulder and arthroscopy. A similar approach has been used throughout the strategy with each of the potential interventions.
| OvidSP interface; Ovid MEDLINE(R), 1950 to March Week 2 2010 |
| 3158 records identified, update search (5 January 2011) identified 98 records |
|
One of the particular challenges in constructing this search strategy was that there is no MeSH directly corresponding to ‘frozen shoulder’ or ‘adhesive capsulitis’. The nearest MeSH available, ‘bursitis’, refers to knee joints and elbow joints as well as the shoulder joint so it is inevitable that redundant records will be identified by the search. The inclusion of free-text terms in the strategy is therefore essential.
During the development of the search strategy a small number of records were identified in which neither the title of the paper nor the abstract contained reference to a potential intervention. As the article titles appeared to indicate that the papers could be potentially useful, lines 1–6 at the beginning of the MEDLINE search strategy focus simply on identifying and retrieving records that have ‘frozen shoulder’, etc. in the title of the record. This assumes that when the term appears in the title there is a high likelihood that it could be relevant even if no intervention is specifically mentioned.
The strategies from all of the databases are given in full in Appendix 1.
Inclusion and exclusion criteria
Studies were included in the review if they met the criteria below, which are also summarised in Table 4.
| Population | Participants with idiopathic (primary) frozen shoulder (adhesive capsulitis) as defined by the authors, with or without diabetes |
| Intervention | Physical therapies, arthrographic distension, steroid injection, sodium hyaluronate injection, MUA, capsular release, watchful waiting |
| Comparator | Any of the above, no treatment or placebo |
| Outcomes | Pain; ROM; function and disability; quality of life; time to recovery, return to work and recreation; adverse events |
| Study design | RCTs; in the absence of randomised trials, quasi-experimental studies (i.e. with a control group). If controlled trials were not available for MUA or capsular release, case series of at least 50 participants |
| Studies of economic evaluations | Full economic evaluations that also met the population and intervention inclusion criteria |
Population
Participants with idiopathic (primary) frozen shoulder were included. We took a pragmatic approach and included studies based on the authors’ definition of frozen shoulder to ensure that we identified all of the relevant evidence. Ideally, only patients with loss of active and passive external rotation of the involved shoulder with normal radiographic findings would be included. This would allow for exclusion of patients with arthritis of the shoulder, which can present as a similar clinical picture. However, such information is not always available in reports of trials.
Studies of general shoulder conditions were included only if outcome data were reported separately for participants with primary frozen shoulder, and studies of mixed populations of primary and secondary frozen shoulder were included only when at least 90% of the participants had primary frozen shoulder or data were reported separately for the two groups. Frozen shoulder in people with diabetes is defined as primary in some classifications and as secondary in others. Patients with diabetes were included as primary frozen shoulder.
Intervention
The following interventions, or combinations of these, were eligible for inclusion:
-
physical therapies (including physiotherapy, acupuncture, chiropractic and osteopathy interventions); physiotherapy encompasses a wide range of techniques including mobilisation, biofeedback, ultrasound and laser therapy and all therapies falling under the physiotherapy umbrella were included
-
distension
-
steroid and other shoulder injections such as sodium hyaluronate
-
MUA
-
capsular release (arthroscopic and open)
-
the approach of ‘watchful waiting’ (as defined by the authors but including education and advice about mobilisation within pain limits, home exercise and use of pain relief).
There are a number of other treatments that have been researched for frozen shoulder that are not commonly used on the NHS, such as radiotherapy, collagenase injection, salmon calcitonin and antibodies to tumour necrosis factor-α. These interventions were excluded. Sodium hyaluronate is not licensed for use in frozen shoulder but a recent survey reported its use by a few respondents and therefore it was included in the review to establish the evidence base.
Comparator
Studies using any of the above treatments as a comparator (including studies comparing different regimens of the same intervention), no treatment or placebo were included. The two exceptions to this were acupuncture and sodium hyaluronate. Dose-ranging studies of sodium hyaluronate were excluded. Studies of acupuncture were included only when the comparator was one of the other interventions of interest in the review. Therefore, studies comparing more than one type of acupuncture or comparing acupuncture to an alternative therapy such as moxibustion were excluded.
Outcomes
The outcomes of interest were pain (e.g. at rest, on movement, at night); range of movement (e.g. internal and external rotation, elevation); function and disability; quality of life; time to recovery, return to work and recreation; and adverse events.
Study design
Only RCTs were eligible for inclusion where this level of evidence was available on an intervention/management strategy. In the absence of randomised trials, quasi-experimental studies (i.e. with a control group) were included. If controlled trials were not available for MUA or capsular release, case series of at least 50 participants were included. It is unclear what size a good-quality case series should be and therefore this was chosen as an arbitrary cut-off; it was considered an achievable size of case series for this field while maximising the possibility of a representative sample of patients.
The intention was to include and update systematic reviews if (1) they fulfilled all the relevant inclusion criteria, (2) they had no significant sources of error and bias and (3) they were reported in detail and the raw data are available from the report or authors to allow an update of the synthesis (if searches are more than 12 months out of date). Otherwise they would be screened as an additional source of studies. Previous systematic reviews focused on individual interventions and had variable inclusion criteria and therefore updating of individual reviews was not appropriate. The reference lists of reviews were screened for additional studies.
Full economic evaluations
Full economic evaluations that met the population and intervention inclusion criteria were eligible for inclusion. A full economic evaluation was defined as any study in which a comparison of two or more relevant alternatives was undertaken with costs and outcomes examined separately for each alternative. This included cost-effectiveness analyses (including cost–consequence analysis), in which health outcomes are expressed in natural units; cost–utility analysis, in which benefits are measured in utility units or utility-weighted life-years; and cost–benefit analyses, in which benefits are measured in monetary form using approaches such as ‘willingness to pay’ or ‘human capital approach’.
Screening and study selection
Two researchers independently screened all titles and abstracts identified from the searches to identify potentially relevant studies. Full manuscripts of potentially relevant studies were ordered and two researchers independently assessed the relevance of each study using the criteria above. Disagreements were resolved by consensus and, if necessary, a third researcher was consulted.
Data extraction
Descriptive data extracted included study design, number randomised, loss to follow-up, country, setting, inclusion criteria, population characteristics, description of the intervention including duration and intensity, concomitant treatments and outcome measures used. These data were extracted by one researcher using a pre-piloted standardised data extraction form in Evidence for Policy and Practice Information (EPPI)-Reviewer 3 (EPPI-Centre, Institute of Education, University of London, UK). Number of participants randomised, number included in each analysis, mean and standard deviation (SD) were extracted into a pre-piloted Microsoft Excel 2007 spreadsheet (Microsoft Corporation, Redmond, WA, USA). Descriptive and outcome data were checked by a second reviewer and discrepancies were resolved through discussion and, if necessary, a third opinion was sought.
For continuous outcomes the post-intervention (final value) mean and SD for each group were extracted, where available, as first preference. Otherwise, change scores (the difference between baseline and follow-up) and SD for each group were extracted, and the between-group difference in change and SD where data for the individual groups were not available. Data available only in graph format were not extracted; authors were contacted for the actual data. Where only median and ranges were reported, these were extracted. Unadjusted data were extracted to allow for covariate analyses in the MTC. Where unadjusted data were not available, adjusted data were extracted and the type of adjustment recorded (two studies35,36). Standard data imputation methods were used, where necessary, to calculate SDs. 37 If the SD or standard error (SE) was not reported, the variance was calculated from the 95% confidence interval (CI) or p-value (three studies38–40). Where this information was not available, the SD was imputed based on the average SD across all interventions for that outcome (two studies35,41). As a final means of imputing SD, a simulation model written in the R programme was used. The model repeatedly sampled from the possible range of values of the outcome measure being used (e.g. 0–9 in the case of a Likert 0–9 scale used to measure pain) to identify sequences whose mean equalled the mean reported in the trial of interest. Where the two means matched, the model calculated a SD; finally, the mean of these SDs was calculated and used in our analysis (two studies42,43). The R code is presented in Appendix 3.
Where the number of participants in an analysis was unclear, and the information was not available from the authors, the number randomised minus the number of dropouts was used.
Assessment of risk of bias
Quality assessment was also undertaken by one researcher and checked by a second with discrepancies resolved by consensus or recourse to a third researcher if necessary. Studies were assessed using the checklist in Appendix 4. The criteria for assessing randomised and non-randomised trials were based on recent CRD guidance;34 the criteria for case series are based on those used in a recent systematic review including case series. 44 The quality of economic evaluations was assessed using a modified version of the Drummond checklist. 45
Synthesis
The synthesis had two main components: (1) a narrative synthesis, including pair-wise comparisons and (2) a MTC. The analysis was based on the protocol and an a priori analysis plan. The primary outcomes of interest were patient-assessed pain intensity, function and disability, quality of life and range of movement. Given that the symptoms of frozen shoulder change over time (with pain being the strongest characteristic of the early stages but not later), it was not appropriate to use a single primary outcome.
Narrative synthesis and pair-wise comparisons
A narrative and tabular summary of key study characteristics, quality assessment and results was undertaken. Studies were grouped by the main intervention of interest in the study and then by comparator. Where appropriate, based on clinical and statistical heterogeneity and the necessary data being available, individual study results were combined in a pair-wise meta-analysis based on type of intervention and comparator using RevMan 5 (The Cochrane Collaboration, The Nordic Cochrane Centre, Copenhagen, Denmark). 46 A random-effects model was used except when there were fewer than three studies when a fixed-effect model was used. Heterogeneity was assessed using the chi-squared test and the I2 statistic. A chi-squared test with p-values < 0.1, and I2 values > 50% were taken to indicate that there was substantial statistical heterogeneity. 34,47 When a quantitative synthesis was not appropriate the results of studies were discussed in a narrative. Studies reporting median rather than mean values were discussed in the narrative synthesis only.
Outcomes
Pain was measured using several different outcome scales; therefore, the standardised mean difference (SMD) was calculated. Final value and change scores were not combined together as SMDs, as the difference in SD reflects differences in measurement. Scales were amended, where necessary, so that an increase in score equated to an increase in pain according to the Cochrane Handbook. 48 The SMD measure used was Hedges’ g as this is considered appropriate for studies with small sample sizes. 49
The interpretation of the SMD is not as intuitive as the interpretation of the original measures of treatment effect. To address this issue, the pooled SMD was back transformed to an original measure by multiplying it with a ‘typical’ SD of one of the pain measures. A visual analogue scale (VAS) 0–100 mm was chosen, as this measure of pain, compared with others used by studies included in the review, had the largest volume of literature regarding minimum clinically important differences (MCIDs) in pain. This included one study performed in a shoulder-specific population. 50 Transformation from SMD to VAS 0–100 mm was made using a representative pooled SD at baseline from one of the included trials. 51 It should be noted that this was only appropriate for pooled rather than individual study SMDs. 52 The reduction in pain recorded as a SMD can be interpreted on a VAS 0–100 mm using Table 5. No data could be located on the MCID in pain for individuals with frozen shoulder; however, it is suggested that a difference of 14 mm on a 100-mm VAS scale is the MCID for individuals with rotator cuff disease, a shoulder disorder also characterised by pain and restricted range of movement. 50 Given that the between-group MCID is thought to approximate 40% of that within individuals,6,53 the MCID between groups for rotator cuff disorder can be estimated to be approximately 5.6 mm. This value is therefore used as a proxy for the between-group MCID for frozen shoulder.
| Reduction in pain score | |||||
|---|---|---|---|---|---|
| SMD | –0.3 | –0.5 | –1.0 | –1.5 | –2.0 |
| VAS 0–100 mm | –5.6 | –9.15 | –18.3 | –27.45 | –36.6 |
A wide range of function and disability scales were reported in the studies. These outcomes were not converted to a SMD as they were not considered similar enough. This was because there were differences between some scales in the aspects of function and disability assessed and in the weighting given to similar components. In addition, the correlation between some of the included scales was at best moderate. 54–56
Because of the large number of different range of movement measures used, following discussion with the advisory group these were prioritised for the analyses. The passive range of movement outcomes of interest were passive external rotation, passive internal rotation and passive abduction; and the active range of movement outcomes were active external rotation, active internal rotation and active abduction. When passive and active range of movement was not reported separately, unspecified external rotation, internal rotation and abduction were used in the synthesis. When active internal rotation was not reported but hand behind back was, this was used as a proxy measure, although there is some evidence that the correlation between the two is low to moderate. 57
Interventions
As stated above, studies were grouped by type of intervention in the narrative synthesis and pair-wise comparisons. In addition, subgroups were created within interventions. These subgroups were specified a priori to identify clinically similar studies for pooling in the pair-wise comparisons and MTC to guide the exploration of similarities and differences within classes of interventions in the narrative synthesis. These subgroups were as follows:
-
Active physical therapy and physical therapy without mobilisation. Active physical therapy (or physical therapy with mobilisation) was defined as an intervention in which at least part of the intervention involved the patient’s body being wholly or partly in motion, such as exercise, mobilisation and stretching (with or without passive techniques such as heat treatment). Interventions in which there was no such active mobilisation or stretching and the participant was the passive recipient of a therapy such as laser therapy, TENS, ultrasound, heat treatment or ice packs alone were classified as physical therapy without mobilisation.
-
Steroid injections were grouped by the number of injections – three or fewer injections and more than three injections. This was based on advice from the advisory group that patients in the UK are unlikely to receive more than three injections for frozen shoulder; therefore, this was the cut-off used.
Length of follow-up
Follow-up of ≤ 4 weeks was not included in the analysis as it was not considered to be informative. Where studies did not report the same length of follow-up, outcomes were pooled grouped by short-, medium- and long-term follow-up. For short-term follow-up the data point from each study at 3 months’ follow-up or the closest data point before 3 months’ follow-up was used. For the outcome of pain, where pair-wise comparisons were possible, the same short-term follow-up data used for the MTC at 3 months were pooled. This was to enable comparison of results from the pair-wise comparisons and the MTC. For medium-term follow-up the data point at 6 months or the closest data point before 6 months was used. For long-term follow-up the data point at 12 months or the closest data point before 12 months was used. Data were also presented (when reported) at multiple times within a follow-up period (e.g. 6 weeks and 3 months). When only a narrative synthesis was possible, data were discussed using the same categories.
Unit of analysis error
Some of the included studies had a unit of analysis error, that is, patients were randomised to the intervention but outcome was reported by shoulder when a participant had two frozen shoulders. In these instances the number of patients was used as the denominator in the analysis as only a few patients had more than one shoulder involved; therefore, the difference between number of shoulders and number of participants was small. 58
Multi-arm trials
Where pooling of multi-arm trials required the use of a control group more than once in the same meta-analysis, the number of participants in the control group was divided approximately evenly among the comparisons, with the means and SDs left unchanged. Where different arms were assessing minor variations of a class of intervention these were combined before pooling. 37
Subgroup analyses
Subgroup analyses were planned to explore the influence of the following study characteristics in the MTC and pair-wise comparisons:
-
inclusion of patients with diabetes
-
stage of frozen shoulder
-
study quality (with studies stratified by concealment of allocation and by blinding of outcome assessors).
Because of the small number of studies suitable for pooling it was only possible to explore these factors in the narrative.
Mixed-treatment comparison
A MTC was undertaken. A MTC, also referred to as network meta-analysis, goes beyond the scope of the standard pair-wise meta-analysis in which direct within-trial results are pooled in a weighted manner, and provides a mechanism for bringing together both direct and indirect evidence. The approach allows data networks that include A versus B trials, B versus C trials and A versus C trials to be used to make inferences about the relative efficacy of all treatments. 33 This allows estimation of comparisons not previously considered in a clinical trial to be made. As the summary treatment effect from each trial is used to inform the MTC, the randomisation of the trial is maintained within the analysis.
As with standard meta-analysis, to facilitate this type of analysis a number of conditions need to hold:
-
Homogeneity assumption – that trials are sufficiently homogeneous to be quantitatively combined.
-
Consistency assumption – that had treatment C been included in a trial comparing interventions A and B, then the treatment effect dAC would be equivalent to that obtained from a trial of interventions A and C. 33 Assuming consistency, the treatment effect dAC is the sum of the treatment effects dAB and dBC.
-
Methodological similarity – that trial methods are similar as relative effects from trials may be associated with the quality of the study, particularly when subjective outcome measures are used. 59
The MTC analysis was conducted in WinBUGS version 1.4.3 (MRC Biostatistics Unit, Cambridge, UK),60 which is a Bayesian analysis software that utilises Markov chain Monte Carlo (MCMC) methods. A Bayesian framework involves a formal combination of a prior probability distribution (which reflects our belief about the possible values of the pooled effect) and a likelihood function (which informs the distribution of the pooled effect based on the observed data) to obtain a posterior probability distribution of the pooled effect. As the outcome for this analysis was the treatment effect difference, a normal likelihood distribution was used for the treatment effect data. A random-effects model was specified as it is likely that, although the treatment effects may be drawn from a common distribution, because of clinical and methodological variations, the true effect size is unlikely to be exactly the same.
Mixed-treatment comparison methods can be applied only to a connected network of RCTs. Because of the diversity of outcomes reported in the clinical trials the number of network options was limited. We were unable to standardise function and disability measures and given the variety reported no network was available. Range of movement outcomes were also not reported in a systematic manner in trials, leaving no network available. As previously stated it was possible to standardise the different pain outcome measures using the SMD; therefore, pain was the only outcome available to undertake a MTC.
Some studies reported more than one type of pain outcome; therefore, for the MTC one pain measure was selected from each study using a hierarchy based on the clinical significance of the type of pain. This was established following discussions with the advisory group, including individuals with frozen shoulder, and after exploring the literature. When more than one pain outcome was reported in a study a single measure was chosen for the MTC analysis in the following order: overall pain, pain at night, pain on activity, any other type of pain (excluding pain at rest) and pain at rest.
Only RCTs reporting final values and SDs (or when data were available to calculate final values or to impute SDs) were eligible for inclusion in the MTC. The model fit was assessed using the residual deviance. A value close to the number of data points in the data set reflects a ‘good fit’. The MCMC simulation requires initial values to be specified for all parameters that are modelled as distributions. It is anticipated that following a sufficient burn-in period the chain will approach a stationary distribution. Convergence to a stable estimate should take place regardless of initial values. This was assessed using diagnostic tools provided within the WinBUGS package, including graphical time series trace of starting values for the selected parameters, the Brooks–Gelman–Rubin statistic and posterior distributions. The first 10,000 iterations were used as ‘burn-in’ and discarded; subsequently, a further 100,000 iterations were performed. Uncertainty was presented using the upper and lower limits of 95% credible intervals (CrIs), which describe the bounds within which it is believed there is a 95% chance that the true value lies.
The selection of the prior distributions is extremely important, particularly when there is limited effectiveness data. In a situation in which we have no information and we wish to include non-informative priors, it is important to check that the selections are truly non-informative. Prior distributions are given to those parameters being estimated by the model. The combining of the prior and the data gives us the posterior distributions from which we sample. The prior for the between-study SD was set to be a uniform distribution with a range 0 to 2. This covered the range of treatment effects within a particular comparison. However, when data are sparse, non-informative priors have an unintentionally large influence on the precision of the treatment effect, which can lead to different statistical inferences. 61 For this reason we undertook a number of sensitivity analyses to ensure that our prior was non-informative and allowed the model to reach convergence. For each network, a sensitivity analysis was performed by changing the range of the uniform prior distribution on the SD from 0–2 to 0–0.8, 0–5, 0–10 and, as a final check for stability for network 1, 0–15. An additional sensitivity analysis was also performed by repeating each analysis using a burn-in of 30,000 iterations.
Systematic review of patients’ views of interventions for frozen shoulder
Given the range of possible treatment options for frozen shoulder, patient preference is an important factor in the treatments received by people with frozen shoulder as well as the sequence in which they try treatments when more than one type of treatment is necessary. We therefore undertook a systematic review of studies of patients’ views of treatments for frozen shoulder. Three databases (CINAHL, MEDLINE and PsycINFO) were searched on 3 June 2010 to identify qualitative studies, with searches being restricted to English-language papers published from 1980 onwards. The search strategies used combined a set of terms for frozen shoulder/adhesive capsulitis with a qualitative search filter. 62 The search strategies used are given in full in Appendix 2.
Studies investigating patients’ views or experiences about the treatments included in the main review were eligible for inclusion. Only English-language qualitative studies were included; expert opinion, letters containing no data on patient views, editorials and discussion papers were excluded.
The processes for study selection, data extraction and quality assessment followed those of the main review. Studies were selected independently by two researchers and disagreements were resolved through discussion.
The intention was to extract data on study aim, participant characteristics, methods of collecting data on patient views and experiences, method of analysis, results in the form of a summary of key themes arising from the analysis and authors’ conclusions. Study quality was to be assessed using a tool developed by Hawker et al. 63 and a narrative synthesis undertaken.
Assessment of cost-effectiveness
The intention was to develop a decision-analytic model, following National Institute for Health and Clinical Excellence (NICE) guidance on methods for technology appraisal,64 to estimate the cost-effectiveness of the different treatments for frozen shoulder. The specific objectives outlined in the proposal for the cost-effectiveness analysis were to (1) assess the cost-effectiveness of the named interventions for frozen shoulder to inform clinical practice and (2) identify the key uncertainties relating to the cost-effectiveness analysis and to use these to inform future research priorities. We reviewed the literature to:
-
identify any existing modelling in a similar population
-
identify treatments for this condition
-
identify sequences or order of treatments
-
explore whether treatments (or sequences of treatment) were influenced by phase of condition
-
identify any potential sources of data to populate the model
-
identify studies most relevant to the UK decision-maker.
The economic literature for this clinical area was found to be scant. The development of a full economic model was not possible. We present a full discussion around the issues of modelling treatments for frozen shoulder in Chapter 3 (see Decision model), but, in brief, a lack of clinical evidence and consensus regarding treatment options made the structuring and populating of a model unachievable.
In an attempt to present some information that will be useful to the decision-maker we undertook a number of supplementary investigative exercises. First, we used the advisory group to elicit details of resource use associated with the interventions for which evidence has been identified in the systematic review. In conjunction with the advisory group, cost estimates for these procedures were produced. Second, as our initial aim was to express health benefits in terms of quality-adjusted life-years (QALYs), we investigated the available evidence to identify utility outcomes. A search of the published literature was undertaken to find any published studies that had measured and reported health utility as an outcome. Following the NICE reference case64 the European Quality of Life-5 Dimensions (EQ-5D) should be used to elicit individuals’ preference for health states. The EQ-5D measures health on five attributes: mobility, self-care, usual activity, pain/discomfort and anxiety/depression. These data are then combined with the UK social tariff, which is based on 10-year time trade-off data,65 and used to weight the time spent in a given health state. Third, we explored the possibility of mapping outcomes presented in the clinical trials from the systematic review to the EQ-5D using established methods. The aim of the mapping was to generate changes in EQ-5D and QALYs for the interventions assessed in the clinical review. We then present some basic exploratory calculations to identify interventions that might be cost-effective and worthy of further investigation, although we acknowledge that these calculations are extremely uncertain.
Chapter 3 Results
Assessment of clinical effectiveness
Study selection
The searches identified 8883 records, 8341 from the original searches undertaken in March 2010, 521 from the update searches undertaken in MEDLINE and EMBASE in January 2011 and 21 from subsequent reference checking (Figure 1). On the basis of reviewing title and abstracts 8161 papers were excluded; 722 papers were ordered for a more detailed evaluation. Additionally, an author sent in one paper. Of the 723 papers, 67 were published in languages other than English. These included Chinese (22 papers), Russian (19 papers), Italian (6 papers), Japanese (6 papers), Dutch (5 papers), French (2 papers), Croatian (1 paper), German (1 paper), Hebrew (1 paper), Korean (1 paper), Norwegian (1 paper), Slovak (1 paper) and Turkish (1 paper).
FIGURE 1.
Flow chart of study selection.
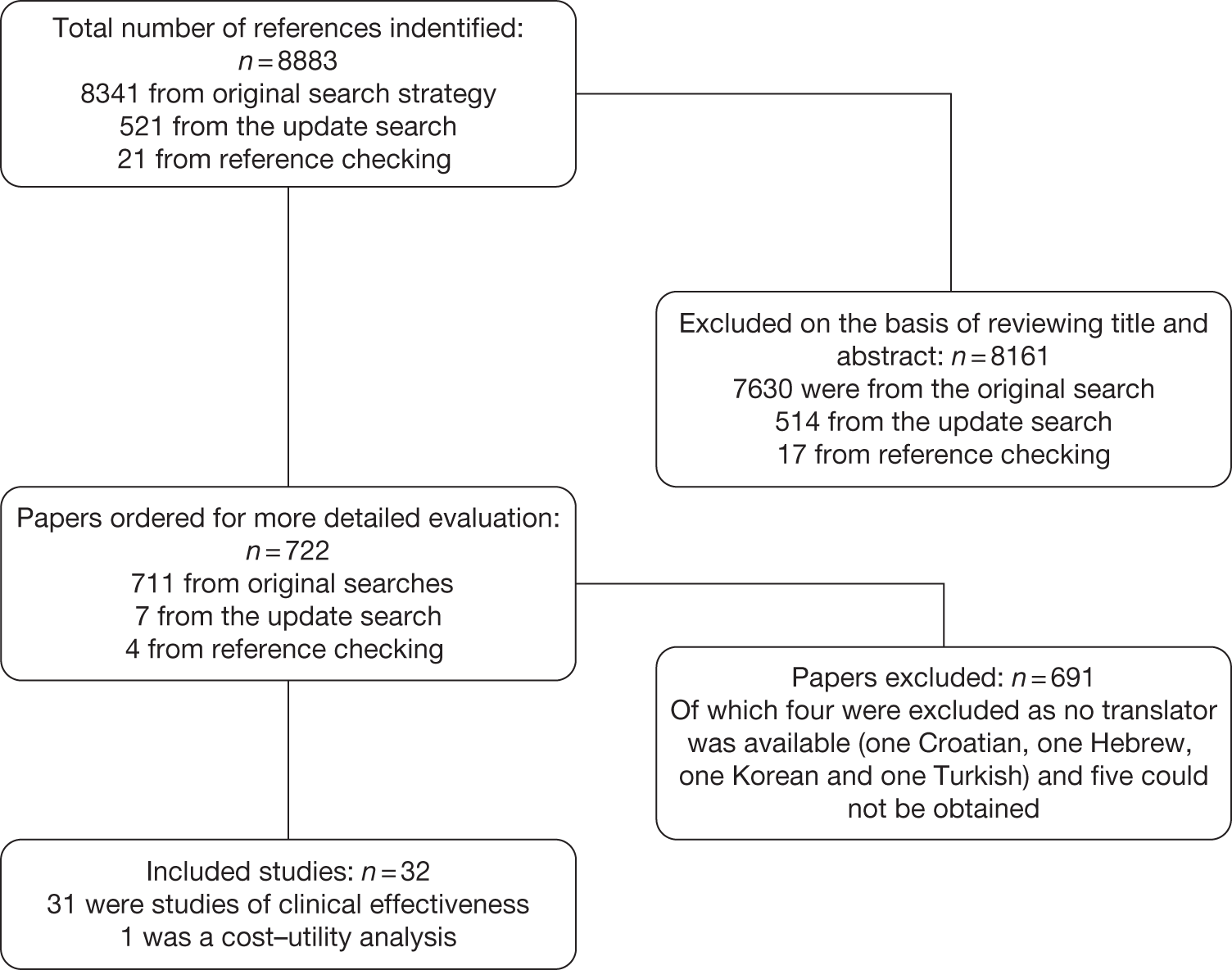
After a detailed evaluation, 691 papers were excluded from the review, including four for which there was no translator available, and five that could not be obtained through the British Library (see Appendix 5 for a list of excluded studies). The main reasons for excluding studies from the review were that they were not a clinical study (146 studies) or they did not meet the study design criteria (193 studies) or the population criteria (244 studies). Of the studies excluded on the basis of population, in 42 studies it was unclear if the patients had primary frozen shoulder, whereas in 17 studies < 90% of patients in each study had primary frozen shoulder.
There were 32 studies included in the review, one of which was a cost–utility analysis conducted alongside a separately published study of effectiveness. Of the 32 studies, 28 were published in English, 2 in Chinese, 1 in Japanese and 1 in Norwegian.
Studies were grouped by the main treatment intervention of interest: six studies were of steroid injection (with and without physical therapy), three of sodium hyaluronate injection, twelve of physical therapy, three of acupuncture, four of MUA, three of distension and two of capsular release. One study, by Calis et al. ,66 fell into more than one category and was reported in all relevant sections (steroid injection, sodium hyaluronate injection and physical therapy). In one of the physical therapy studies there was a ‘supervised neglect’ comparator. The included studies were from a wide range of countries (Turkey, Canada, UK, USA, Italy, Japan, Hong Kong, China, Taiwan, the Netherlands, Singapore, Thailand, Greece, Pakistan, Finland, Australia, Denmark and Norway).
Although a quantitative synthesis was planned this was largely not possible, as few studies could be pooled. The overall quality of the studies was poor and there were few studies of the same comparators. Within the classes of intervention there was considerable variability between studies in various aspects of the intervention such as dose (e.g. of steroid or sodium hyaluronate injection), and duration and intensity of treatment, especially in terms of physical therapy and home exercise programmes. The physical therapy interventions encompassed a range of combinations of ‘active’ and ‘passive’ components. There was variability in the outcomes reported, the tools used to measure individual outcomes, time of follow-up and type of data reported. As a result there were few situations in which it was appropriate to pool and undertake the planned subgroup analyses. It was therefore necessary to primarily use narrative synthesis to present the findings of this review. The main analysis was the narrative synthesis with pair-wise comparisons where appropriate. This is presented first with studies grouped by intervention. This is followed by the MTC. This was a secondary, exploratory analysis because of the small number of studies that connected in the network.
Steroid injection
Study characteristics
Six RCTs investigated steroid injections, with or without the addition of physiotherapy, in the treatment of primary frozen shoulder. A summary of the study characteristics is reported in Table 6, with further details available in Appendix 6. All were full papers of studies conducted in the UK,41,67 Turkey,66,68 Canada35 and the USA42 between 1981 and 2008. There was one two-armed trial,68 one three-armed trial67 and four four-armed trials35,41,42,66
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Bal 200868 Two-arm RCT |
n = 80 (82 shoulders) Steroid injection: 40 (42) Placebo injection: 40 (40) |
Steroid: methylprednisolone acetate, 40 mg, 1 ml, one injection Placebo: saline, 0.9% sodium chloride, 1 ml, one injection |
Presence of shoulder pain with limitation of both active and passive movements of the glenohumeral joint of ≥ 25% in at least two directions. Symptom duration between 6 weeks and 6 months |
|
Calis 200666 Four-arm RCT |
n = 90 (95 shoulders) Sodium hyaluronate injection: 24 (27) Steroid injection: 25 (26) PT: 21 (22) No intervention (home exercise only): 20 (20) |
Sodium hyaluronate: 30 mg, one injection once weekly for 2 weeks. Steroid: triamcinolone acetonide, 40 mg, one injection PT: 10 daily sessions at least 45 minutes |
History of pain for at least 1 month; limited active and passive shoulder movement; decreased passive ROM of 20% or more, in at least three movements, according to the American Medical Association guide for the evaluation of permanent impairment |
|
Carette 200335 Four-arm RCT |
n = 93 Steroid injection: 23 Placebo injection: 23 Steroid + PT: 26 PT + placebo: 23 |
Steroid: triamcinolone hexacetonide, 40 mg, 2 ml, one injection Placebo: saline, 2 ml, one injection PT: 12 × 1-hour sessions, 3 times per week for 4 weeks |
Adhesive capsulitis defined as the presence of shoulder pain with limitation of both active and passive movements of the glenohumeral joint of ≥ 25% in at least two directions compared with contralateral shoulder or normal values. Symptomatic for < 1 year |
|
Dacre 198967 Three-arm RCT |
n = 66 (data reported for 62) Steroid injection: 22 Steroid + PT: 20 PT: 20 |
Steroid: triamcinolone, 20 mg, 1 ml with 2% lidocaine, one injection PT: 4–6 weeks’ duration |
Painful stiff shoulder for at least 4 weeks; inability to use arm with restriction of movement and loss of full function; pain at night causing sleep disturbance and inability to lie on affected side |
|
Rizk 199142 Four-arm RCT |
n = 48 Steroid, (IA) + PT: 16 Steroid, (IB) + PT: 16 Placebo injection (IA) + PT: 8 Placebo injection (IB) + PT: 8 |
Steroid: methylprednisolone acetate, 40 mg, 1 ml with 2 ml of 1% lidocaine. Once weekly for 3 weeks IA (using anterior approach) or IB (using lateral approach) PT: 11 weeks Placebo: 1% lidocaine once-weekly for 3 weeks IA or IB |
Total ROM < 50% of normal range (i.e. < 320°); shoulder pain for < 6 months, nocturnal accentuation of pain, no effusion in the glenohumeral joint |
|
Ryans 200541 Four-arm RCT |
n = 80 Steroid injection: 20 Steroid + PT: 20 PT + placebo injection: 20 Placebo injection: 20 |
Steroid: triamcinolone, 20 mg, 1 ml and normal saline, 2 ml; 1.5 ml injected by an anterior approach and 1.5 ml by a lateral approach Placebo: saline (3 ml) administered as per steroid PT: eight sessions over 4 weeks |
Painful shoulder in the 5th cervical (C5) dermatome distribution of between 4 weeks’ and 6 months’ duration. Restriction of active and passive ROM in both external rotation and glenohumeral abduction of > 25% compared with other shoulder |
The steroids administered were methylprednisolone acetate and triamcinolone hexacetonide. Two studies used a single injection of 20 mg triamcinolone hexacetonide,41,67 one of which also administered lidocaine,67 and three studies used a single injection of 40 mg methylprednisolone acetate or triamcinolone hexacetonide. 35,66,68 The remaining study administered three 40-mg injections of methylprednisolone acetate plus lidocaine, one each week for 3 weeks. 42 In one of the studies the injection was guided using fluoroscopy. 35 Five of the studies reported administering an intra-articular injection,35,41,42,66,68 one of which compared an anterior and a lateral approach,42 and in one study it was unclear. 67
The comparators were steroid injection plus physiotherapy,35,41,67 placebo injection,35,41,42,68 physiotherapy with35,41 and without66,67 placebo injection and home exercise alone. 66
Five of the six trials evaluated a physical therapy regimen as part of the intervention. 35,41,42,66,67 These varied between studies but all consisted of physiotherapy modalities. They included combinations of exercises, mobilisation techniques, TENS, ultrasound, ice, heat packs and interferential therapy. All of the studies appeared to include some form of mobilisation as part of the physiotherapy, although details were scant in some studies. The duration of physiotherapy ranged from 4 to 11 weeks, although, with the exception of one study, duration was < 6 weeks. One study tailored the physiotherapy depending on whether participants had acute or chronic-like symptoms. 35
Five studies reported that participants in all groups were asked to undertake home exercises, although the level of detail reported varied between studies. Bal et al. 68 had a substantial home exercise programme: participants were asked to undertake shoulder stretching and stabilising exercises five times per day over 12 weeks, using a heat pack before exercise and a cold pack after. Carette et al. 35 asked participants to carry out active and auto-assisted exercises twice daily over 12 weeks. In the study by Calis et al. 66 stretching and Codman’s exercises were performed. Details of home exercise were not reported in the other two studies. 41,42 The study by Dacre et al. 67 did not have a home exercise component.
There was also some variability between studies in concomitant treatments. Four studies reported that paracetamol of varying doses was available to participants: 1500 mg per day when needed in one study,68 a supply (dose unspecified) in one study,35 one to two tablets every 4–6 hours (a maximum of eight per day) in another study41 and paracetamol when needed in the remaining study. 66 Another study stated that all participants were advised to continue NSAIDs. 42 One study did not report whether participants received concomitant treatment. 67
The inclusion criteria varied between the six studies, although four of the studies used similar criteria for the extent of restriction of movement. 35,41,66,68 Three of the studies included participants with frozen shoulder of < 6 months’ duration. 35,41,42 Where reported, frozen shoulder was diagnosed through presence of pain and range of movement, clinical diagnosis, presence of symptoms, laboratory tests and radiography. The method of diagnosis was unclear in two studies. 42,68 Three studies included a small number of participants with diabetes: 6% in the study by Carette et al. ,35 5% in the study by Dacre et al. 67 and 6% in the study by Ryans et al. 41 None of the studies reported results separately for patients with diabetes. The participants’ stage of frozen shoulder at baseline was not reported in any of the studies. Where reported, the mean duration of frozen shoulder amongst the included participants ranged from 1342 to 2135 weeks. None of the studies reported whether participants had received any previous treatment for frozen shoulder. The mean age of participants ranged from 54 to 57 years and the proportion of women ranged from 42% to 63%.
Quality assessment
Quality varied between the included studies. Four studies did not report the method of randomisation;42,66–68 therefore, it was unclear whether these were truly randomised studies as stated by the authors. In addition, in two of these studies it was unclear whether the intervention groups were comparable at baseline. 67,68 These four studies therefore have a potentially high risk of bias even though they met other criteria such as blinding of outcome assessment. Only one of these studies was adequately powered for at least one outcome. 67 Bal et al. 68 also reported substantial loss to follow-up (20%, all in the placebo group). Dacre et al. 67 and Rizk et al. 42 reported considerably less loss to follow-up (6% and 8% respectively), whereas Calis et al. 66 reported that there were no dropouts.
The remaining two studies were considered of satisfactory quality although one had some risk of bias. 41 Both reported an adequate method of randomisation and Carette et al. 35 also reported an adequate method of allocation concealment; it was unclear whether Ryans et al. 41 used an adequate method because, although sealed envelopes were used, it was not stated whether these were opaque or sequentially numbered. Both studies met most of the remaining criteria including reported blinding of outcome assessment. Ryans et al. 41 reported that participants were blinded to type of injection. Carette et al. 35 attempted to blind the injection; however, the method used (covering the syringe with foil) may not have been reliable. Blinding of the physiotherapy treatment was not possible. Carette et al. 35 also reported an intention-to-treat analysis. The main limitation of this study was that it may not have been sufficiently powered; 17% were lost to follow-up and there appears to be an imbalance between treatment groups, with greater loss to follow-up in the steroid group. Ryans et al. 41 did not report an intention-to-treat analysis and there was also a high proportion lost to follow-up (27%), with a higher number of dropouts in the steroid only and placebo groups. As with the Carette study, the Ryans study may not have been sufficiently powered. Full details of study quality are reported in Appendix 8.
Pain
All six studies assessed pain but Calis et al. 66 and Dacre et al. 67 reported data in graphical form only (Table 7). Consequently, a SMD could not be calculated and these two studies could not be included in a meta-analysis. Calis et al. 66 reported that there was significantly greater improvement in pain in the steroid group than in the placebo group (p = 0.02). Dacre et al. 67 evaluated steroid, steroid and physiotherapy, and physiotherapy alone. This study reported that all three groups showed a significant reduction in pain after 6 weeks (p < 0.001) with mean measures improving by 49–66%, and further improvements at 6 months. Improvements were reported to be similar in all three treatment groups.
Of the four remaining studies, three reported final mean values and SDs were reported or could be imputed. Both Carette et al. 35 and Rizk et al. 42 reported pain overall and Ryans et al. 41 reported daytime pain at rest (Table 7). Each of the three studies reported pain using a different scale.
| Study | Pain outcome assesseda | Scale used | Data reported and time of follow-up |
|---|---|---|---|
| Bal 200868 | Pain at night | VAS 0–100 mm | Mean change at 2 and 12 weeks (baseline median and IQR reported only) |
| Calis 200666 | Pain severity | VASb | Data reported in graphs only; p-value for between-group difference reported at 3 months |
| Carette 200335 | Pain overall | SPADI 5-item pain subscale | Change from baseline at 6 weeks and 3, 6 and 12 months |
| Dacre 198967 | Pain at night | VAS 0–10 cm | Data presented in graphs only |
| Rizk 199142 | Pain overall | Likert (0–5) | Final mean values at 4, 11 and 24 weeks. SD not reported |
| Ryans 200541 | Daytime pain at rest | VAS 0–100 mm | Change from baseline at 6 and 16 weeks |
The study by Bal et al. 68 reported mean change and SDs at 12 weeks for pain at night measured on the VAS 0–100 mm. Although a SMD could be calculated for this study, it was inappropriate to pool this SMD with those SMDs calculated from final value means, as the difference in SD reflects not differences in measurement scale, but differences in the reliability of the measurements. 48
Table 8 provides the SMDs and 95% CIs. The outcome data for individual groups in the included trials are available in Appendix 7.
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months |
|---|---|---|---|
| Steroid vs placebo | |||
| Bal 200868 | –0.05 (–0.55 to 0.45)a | ||
| Carette 200335 |
–1.55 (2.22 to –0.89)b |
||
| Ryans 200541 | –1.00 (–1.71 to –0.28)b,d | ||
| Steroid vs physiotherapy + placebo | |||
| Carette 200335 |
–0.94 (–1.53 to –0.35)b –0.39 (–0.96 to 0.18)c |
||
| Ryans 200541 | –0.01 (–0.66 to 0.64)b | ||
| Steroid + physiotherapy vs placebo | |||
| Carette 200335 |
–2.36 (–3.15 to –1.58)b |
||
| Ryans 200541 | –2.24 (–3.11 to –1.36)b,d | ||
| Steroid + physiotherapy vs physiotherapy + placebo | |||
| Carette 200335 |
–1.75 (–2.43 to –1.07)b |
||
| Rizk 199142 | 0.18 (–0.45 to 0.80)e | 0.06 (–0.56 to 0.68)f | |
| Ryans 200541 | –1.25 (–1.97 to –0.54)b,d | ||
| Steroid + physiotherapy vs steroid | |||
| Carette 200335 |
–0.81 (–1.43, -0.19)b –0.39 (–0.99 to 0.21)c |
||
| Ryans 200541 | –1.25 (–1.97 to –0.54)b,d | ||
Steroid versus placebo
Three studies reported pain at short-term follow-up. Two studies reported final value mean pain for short-term follow-up: Carette et al. 35 reported data at 6 weeks and 3 months and Ryans et al. 41 reported data at 6 weeks (Table 8). As stated in the methods section, to allow comparison of the results of the meta-analysis with the MTC for pain, time points closest to 3 months were pooled, that is, 3-month data for Carette et al. 35 were pooled with 6-week data for Ryans et al. 41 There was a significant decrease in pain with steroid injection compared with placebo: pooled SMD –1.15, 95% CI –1.62 to –0.67 (Figure 2). When back transformed to a 0–100 mm VAS scale, this equates to a mean difference (MD) of 21 mm (95% CI –29.7 to –12.3 mm). No data could be located on the MCID in pain for individuals with frozen shoulder. However, it is suggested that a difference of 14 mm on a 100-mm VAS scale is the MCID for individuals with rotator cuff disease, a shoulder disorder also characterised by pain and restricted range of movement. 50 Given that the between-group MCID is thought to approximate 40% of that within individuals,6,53 the MCID between groups for rotator cuff disorder can be estimated to be approximately 5.6 mm. Using this as a proxy for between-group MCID for frozen shoulder, this suggests that there is a clinically significant decrease in pain with steroid compared with placebo. There was no statistical heterogeneity between the studies (I2 = 0%). When 6-week data for Carette et al. 35 and Ryans et al. 41 were pooled the results were similar (pooled SMD –1.30, 95% CI –1.78 to –0.81; MD 23.8 mm, 95% CI 32.6 to 14.8 mm).
FIGURE 2.
Standardised mean difference at up to 3 months: steroid vs placebo.
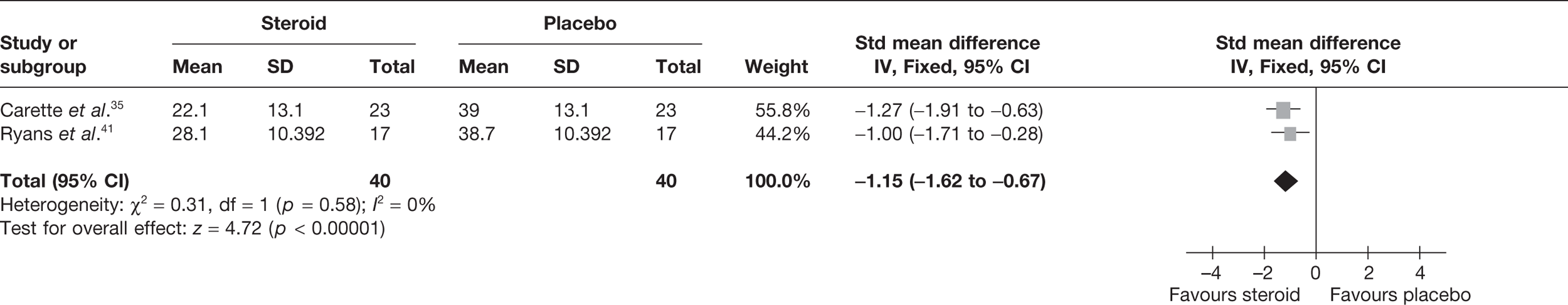
Bal et al. 68 reported mean change in pain at 12 weeks. There was no significant difference in pain with steroid compared with placebo (Table 8).
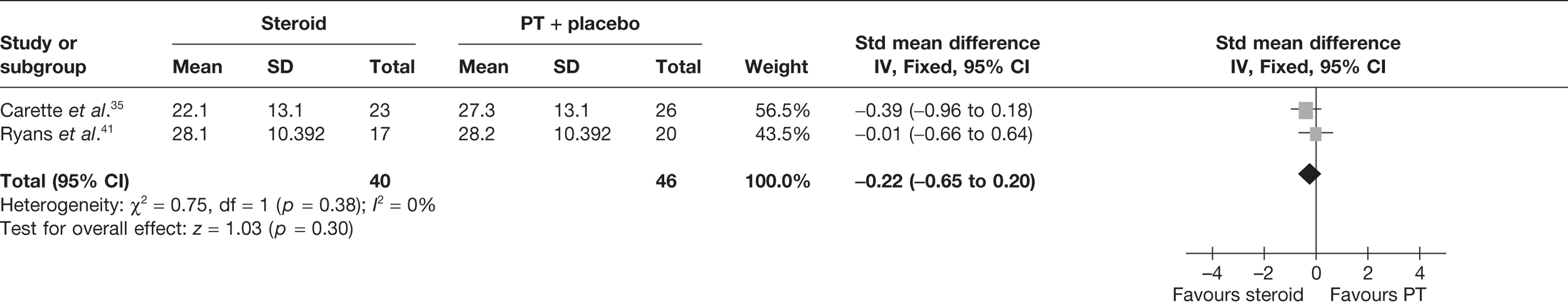
Steroid versus physiotherapy combined with placebo
Two studies reported data at short-term follow-up: Carette et al. 35 reported data at 6 weeks and 3 months and Ryans et al. 41 reported data at 6 weeks (Table 8). In both studies physiotherapy was combined with placebo. When 3-month data from Carette et al. 35 were pooled with 6-week data from Ryans et al. 41 there was no significant difference in pain between steroid and physiotherapy (Figure 3). When back transformed to a 0–100 mm VAS scale, this equates to a MD of –4 mm (95% CI –11.9 to 3.7 mm), which suggests that there may be no clinically significant difference between treatments. There was no statistical heterogeneity between studies (I2 = 0%). When 6-week data for Carette et al. 35 and Ryans et al. 41 were pooled there was a marginally significant decrease in pain with steroid compared with physiotherapy combined with placebo (pooled SMD –0.51, 95% CI –0.94 to –0.07). However, when back transformed to a 0–100 mm VAS scale, this equates to a MD of –9.3 mm (95%–17.2 to –1.2 mm), which, as with the pooled 3-month and 6-week data, suggests that there may be no clinically significant difference between treatments.
FIGURE 3.
Standardised mean difference at up to 3 months: steroid vs physiotherapy combined with placebo.
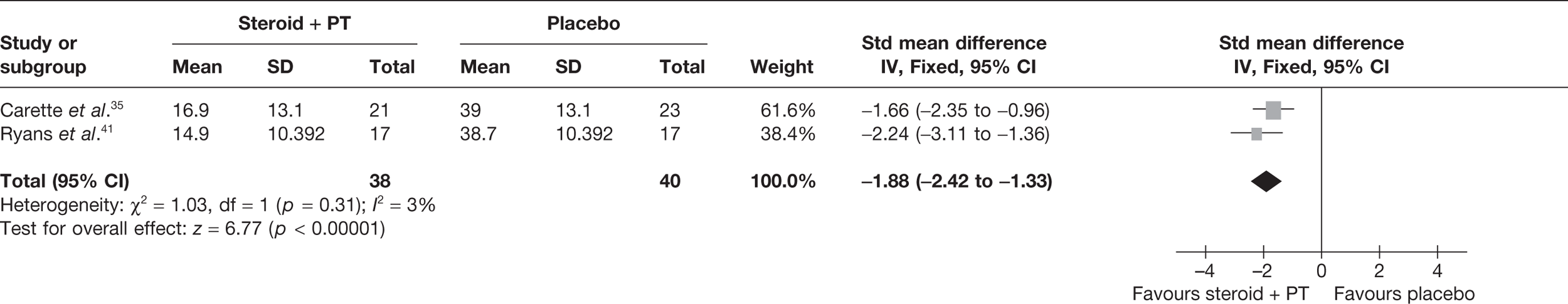
Steroid combined with physiotherapy versus placebo
Two studies reported pain at short-term follow-up: Carette et al. 35 reported data at 6 weeks and 3 months and Ryans et al. 41 reported data at 6 weeks (Table 8). When 3-month data from Carette et al. 35 were pooled with 6-week data from Ryans et al. 41 there was a statistically significant decrease in pain with steroid combined with physiotherapy compared with placebo: pooled SMD –1.88, 95% CI –2.43 to –1.33 (Figure 4). When back transformed to a 0–100 mm VAS scale, this equates to a MD of –34.40 mm (95% CI –44.47 to –24.34 mm). This suggests that there is a clinically significant decrease in pain with steroid combined with physiotherapy compared with placebo. There was no statistical heterogeneity between studies (I2 = 3%). When 6-week data for Carette et al. 35 and Ryans et al. 41 were pooled the results were similar (pooled SMD –2.31, 95% CI –2.89 to –1.72; MD –42.3 mm, 95% CI –52.9 to –31.5 mm).
FIGURE 4.
Standardised mean difference at up to 3 months: steroid combined with physiotherapy vs placebo.
Steroid combined with physiotherapy versus physical therapy combined with placebo
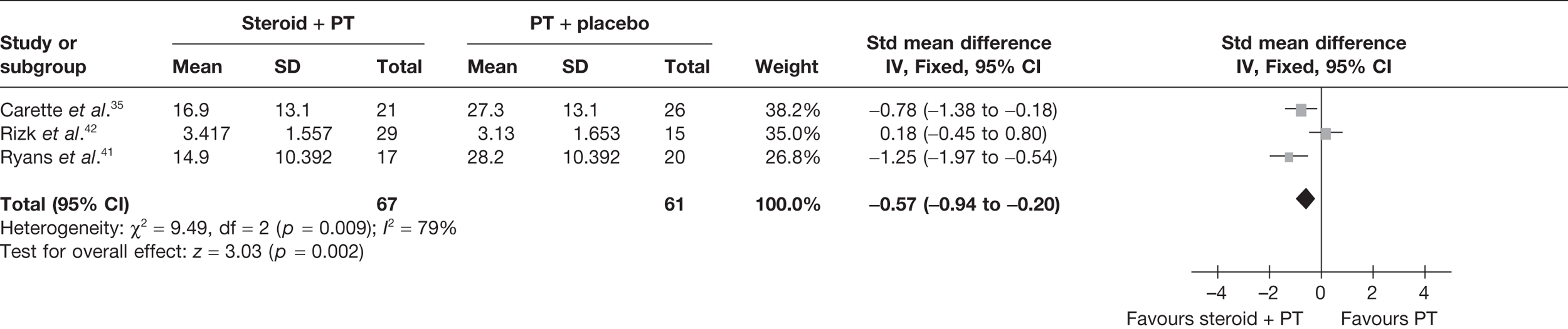
Three studies reported pain for short-term follow-up. In all studies physiotherapy was combined with a placebo injection. Carette et al. 35 reported pain at 6 weeks and 3 months, Rizk et al. 42 reported pain at 11 weeks and Ryans et al. 41 reported pain at 6 weeks (Table 8).
When 3-month data from Carette et al. 35 were pooled with 11-week data from Rizk et al. 42 and 6-week data from Ryans et al. 41 there was a significant decrease in pain with steroid combined with physiotherapy compared with physiotherapy: pooled SMD –0.57, 95% CI –0.94 to –0.20 (Figure 5). However, there was evidence of substantial statistical heterogeneity (I2 = 79%). When 6-week data for Carette et al. 35 were pooled with 11-week data from Rizk et al. 42 and 6-week data from Ryans et al. ,41 the results were similar (pooled SMD –0.86, 95% CI –1.25 to –0.47, I2 = 89%). Potential sources of heterogeneity may be the type of steroid used, different lengths of follow-up and proportion of patients with diabetes. Furthermore, the studies by Carette et al. 35 and Ryans et al. 41 were assessed to have a potentially lower risk of bias than the study by Rizk et al. 42
FIGURE 5.
Standardised mean difference at up to 3 months: steroid combined with physiotherapy vs physiotherapy combined with placebo.
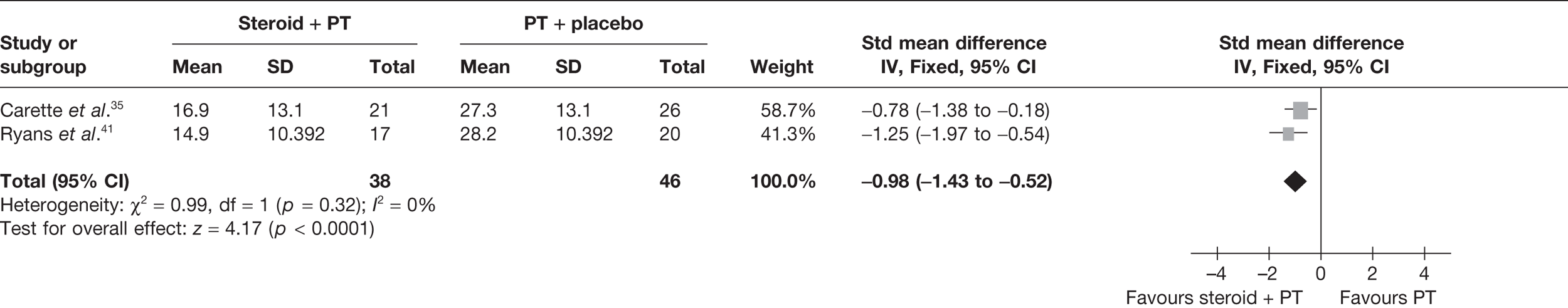
Given the differences in study quality, a sensitivity analysis was performed by excluding Rizk et al. 42 from the meta-analyses (Figure 6). Pooling 3-month data from Carette et al. 35 and 6-week data from Ryans et al. 41 resulted in a significant decrease in pain with steroid combined with physiotherapy compared with physiotherapy: pooled SMD –0.98, 95% CI –1.43 to –0.52. Furthermore, there was no longer evidence of statistical heterogeneity (I2 = 0%). When back transformed to a 0–100 mm VAS, the pooled SMD equates to a MD of –17.93 mm (95% CI –26.2 to –9.5 mm), which suggests that there is also a clinically significant decrease in pain with steroid combined with physiotherapy compared with physiotherapy. When 6-week data for Carette et al. 35 and Ryans et al. 41 were pooled the results were similar (pooled SMD –1.51, 95% CI –2.00 to –1.02; MD 27.6 mm, 95% CI 36.6 to 18.7 mm).
FIGURE 6.
Standardised mean difference at up to 3 months (sensitivity analysis): steroid combined with physiotherapy vs physiotherapy combined with placebo.
The study by Rizk et al. 42 also reported pain for medium-term follow-up. At 24 weeks there was no significant difference between steroid combined with physiotherapy and physiotherapy (see Table 8).
Steroid combined with physiotherapy versus steroid
Carette et al. 35 reported data at 6 weeks and 3 months and Ryans et al. 41 reported data at 6 weeks (Table 8). When 3-month data from Carette et al. 35 were pooled with 6-week data from Ryans et al. 41 there was a significant difference in pain with steroid combined with physiotherapy compared with steroid alone: pooled SMD –0.75, 95% CI –1.20 to –0.29 (Figure 7). When back transformed to a 0–100 mm VAS, this equates to a MD of –13.7 mm (95% CI –21.96 to –5.3 mm), which suggests that there may be no clinically significant difference between treatments. However, there was evidence of substantial statistical heterogeneity (I2 = 70%); therefore, this result should be interpreted with caution. When 6-week data for Carette et al. 35 and Ryans et al. 41 were pooled, there was a significant decrease in pain with steroid combined with physiotherapy compared with steroid alone (pooled SMD –1.00, 95% CI –1.47 to –0.53). When back transformed to a 0–100 mm VAS, this equates to a MD of –18 mm (95% CI 26.9 to 9.7 mm). This suggests that there may be a clinically significant reduction in pain with steroid combined with physiotherapy compared with steroid alone. Furthermore, there was no evidence of statistical heterogeneity (I2 = 0%).
FIGURE 7.
Standardised mean difference at up to 3 months: steroid combined with physiotherapy vs steroid.
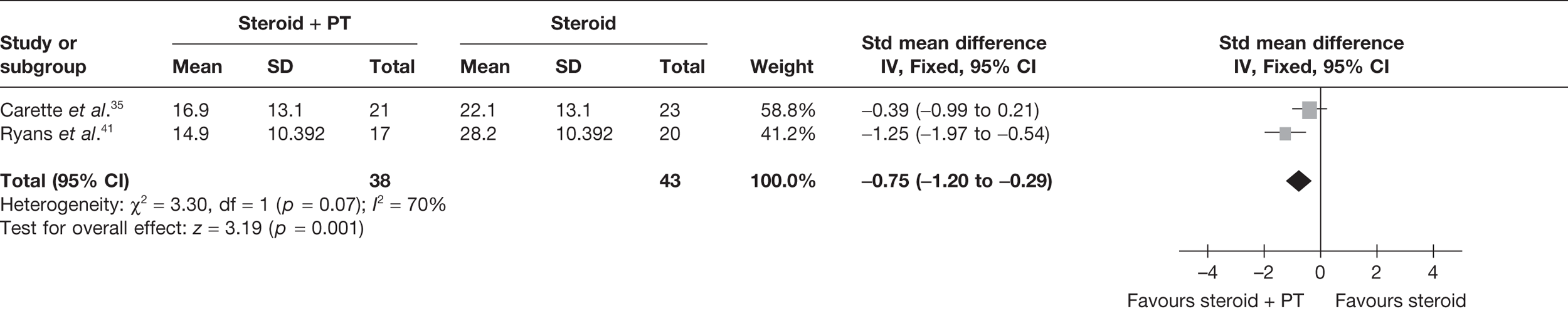
Function and disability
Four of the six studies assessed function and disability; however, only the Shoulder Pain and Disability Index (SPADI) total score and SPADI 8-item disability subscales were common to more than one study and follow-up times varied (Table 9). Results were presented as change from baseline data for all but one of the studies. 66 Table 10 provides the between-group difference in means and 95% CIs. The outcome data for the individual groups in the included trials are available in Appendix 7.
| Study | Function and disability outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Bal 200868 |
UCLA Shoulder score (35-point scale: < 28 poor, 29–33 good, 34–35 excellent as defined by the authors) SPADI total score (0–100)a SPADI 8-item disability subscale (0–100)a |
Change from baseline at 12 weeks |
| Calis 200666 | Constant score | Final value data at 15 days and 3 months (3-month data used in analysis) |
| Carette 200335 |
SPADI total score (0–100)a SPADI 8-item disability subscale (0–100)a |
Change from baseline at 6 weeks and 3, 6 and 12 monthsb |
| Ryans 200541 |
Shoulder Disability Questionnaire (SDQ) (0–22)a,c (score ≥ 5 was defined by authors as significant disability) Self-assessed global function (100-mm VAS, 0–100)a |
Change from baseline at 6 and 16 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Steroid vs placebo | |||
| SPADI total score | |||
| Bal 200868 | 3.80 (–6.09 to 13.69)a | ||
| Carette 200335 | –12.90 (–27.06 to 1.26)d | –2.90 (–17.06 to 11.26)e | |
| SPADI 8-item subscale | |||
| Bal 200868 | 7.60 (–3.49 to 18.69)a | ||
| Carette 200335 | –7.10 (–20.83 to 6.63)d | 0.80 (–12.93 to 14.53)e | |
| SDQ | |||
| Ryans 200541 | –3.00 (–6.44 to 0.44)c | –1.20 (–5.63 to 3.23)f | |
| Global function | |||
| Ryans 200541 | –11.60 (–27.11 to 3.91)c | 5.10 (–15.92 to 26.12)f | |
| UCLA Shoulder score | |||
| Bal 200868 |
Steroid: median 32.5 (IQR 6.2)a Placebo: median 31.5 (IQR 7.7)a No p-value |
||
| Steroid vs home exercise | |||
| Constant score | |||
| Calis 200666 | 9.10 (2.98 to 15.22)a,b | ||
| Steroid vs physiotherapy | |||
| Constant score | |||
| Calis 200666 | –5.80 (11.80 to 0.20)a | ||
| Steroid vs physiotherapy + placebo | |||
| SPADI total score | |||
| Carette 200335 |
–14.50 (–28.25 to –0.75)c –7.60 (–21.35 to 6.15)a |
–8.20 (–21.95 to 5.55)d | –4.60 (–18.35 to 9.13)e |
| SPADI 8-item subscale | |||
| Carette 200335 |
–11.50 (–24.97 to 1.97)c –5.30 (–18.77 to 8.17)a |
–5.30 (–18.77 to 8.17)d | –2.60 (–16.07 to 10.87)e |
| SDQ | |||
| Ryans 200541 | –2.60 (–6.32 to 1.12)c | –2.20 (–6.49 to 2.09)f | |
| Global function | |||
| Ryans 200541 | –2.30 (–18.68 to 14.08)c | 7.10 (–13.43 to 27.63)f | |
| Steroid + physiotherapy vs placebo | |||
| SPADI total score | |||
| Carette 200335 | –14.10 (–28.53 to 0.33)d | –1.10 (–15.53 to 13.33)e | |
| SPADI 8-item subscale | |||
| Carette 200335 | –11.60 (–25.59 to 2.39)d | 0.30 (–13.69 to 14.29)e | |
| SDQ | |||
| Ryans 200541 | –4.70 (–7.85 to –1.55)b,c | –1.00 (–5.12 to 3.12)f | |
| Global function | |||
| Ryans 200541 | –21.00 (–34.54 to –7.46)b,c | 1.50 (–17.96 to 20.96)f | |
| Steroid + physiotherapy vs physiotherapy + placebo | |||
| SPADI total score | |||
| Carette 200335 |
–12.50 (–26.53 to 1.53)a |
–9.40 (–23.43 to 4.63)d | –2.80 (–16.83 to 11.23)e |
| SPADI 8-item subscale | |||
| Carette 200335 |
–11.00 (–24.74 to 2.74)a |
–9.80 (–23.54 to 3.94)d | –3.10 (–16.84 to 10.64)e |
| SDQ | |||
| Ryans 200541 | –4.30 (–7.76 to –0.84)b,c | –2.00 (–5.96 to 1.96)f | |
| Global function | |||
| Ryans 200541 | –11.70 (–26.22 to 2.82)c | 3.50 (–15.42 to 22.42)f | |
| Steroid + physiotherapy vs steroid | |||
| SPADI total score | |||
| Carette 200335 |
9.80 (–4.63 to 24.23)c 4.9 (–9.53 to 19.33)a |
1.20 (–13.23 to 15.63)d | –1.8 (–16.23 to 12.63)e |
| SPADI 8-item subscale | |||
| Carette 200335 |
10.10 (–4.04 to 24.24)c 5.70 (–8.44 to 19.84)a |
4.50 (–9.64 to 18.64)d | 0.50 (–13.64 to 14.64)e |
| SDQ | |||
| Ryans 200541 | 1.70 (–2.37 to 5.77)b | –0.20 (–4.43 to 4.03)f | |
| Global function | |||
| Ryans 200541 | 9.40 (–4.93 to 23.73)c | 3.60 (–15.49 to 22.69)f | |
Steroid versus placebo
Two studies reported SPADI total score and SPADI 8-item subscale score for steroid versus placebo injection (Table 10). 35,68
Carette et al. 35 reported SPADI total score at 6 weeks and found a significant effect in favour of the steroid injection group (MD –17.8, 95% CI –31.96 to –3.64).
Both studies reported 3-month follow-up and were pooled at this time point, although it should be noted that the study by Bal et al. 68 was at high risk of bias. When the two studies were pooled (Figure 8) statistical heterogeneity was high (I2 = 80%); therefore, the treatment effect should be treated with considerable caution. The study by Carette et al. 35 reported a significantly greater decrease in SPADI total score in the steroid group than in the placebo and home exercise group (MD –16.10, 95% CI –30.26 to –1.94), whereas in the Bal et al. study68 the treatment effect was in favour of placebo and home exercise, although the CIs crossed the line of no effect. Only Carette et al. 35 reported SPADI total score for medium- and long-term follow-up; there was no significant difference between groups for these later follow-up times (Table 10).
FIGURE 8.
Shoulder Pain and Disability Index total score at 3 months: steroid vs placebo.
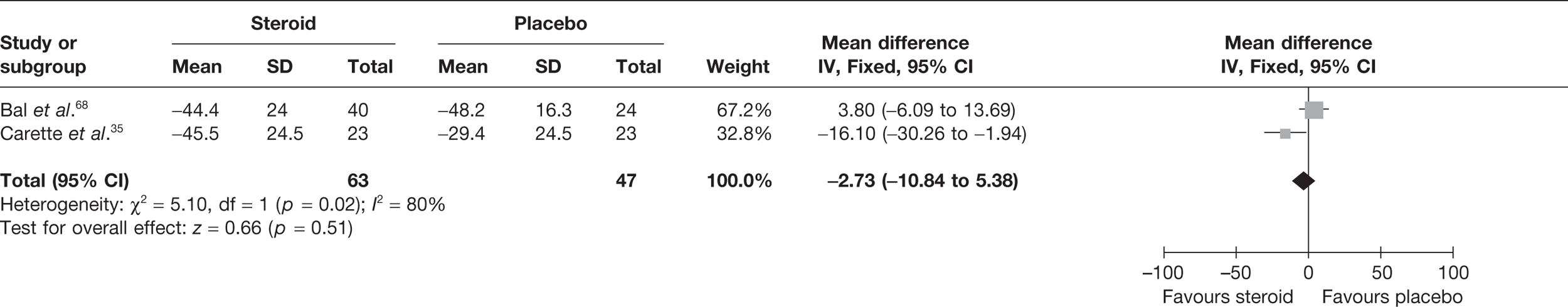
Carette et al. 35 reported SPADI 8-item disability subscale score at 6 weeks and found a significant effect in favour of the steroid injection group (MD –13.80, 95% CI –27.53 to –0.07).
As with SPADI total score, the same two studies reported SPADI 8-item disability subscale score and were available for meta-analysis at 3 months. 35,68 Again, the pooled treatment effect should be treated with considerable caution because of high heterogeneity (Figure 9). Carette et al. 35 reported a significantly greater decrease in SPADI 8-item subscale score in the steroid group than with placebo and home exercise (MD –14.30, 95% CI –28.03 to –0.57), whereas in the Bal et al. study68 the treatment effect was in favour of placebo and home exercise, although the CIs crossed the line of no effect.
FIGURE 9.
Shoulder Pain and Disability Index 8-item subscale score at 3 months: steroid vs placebo.
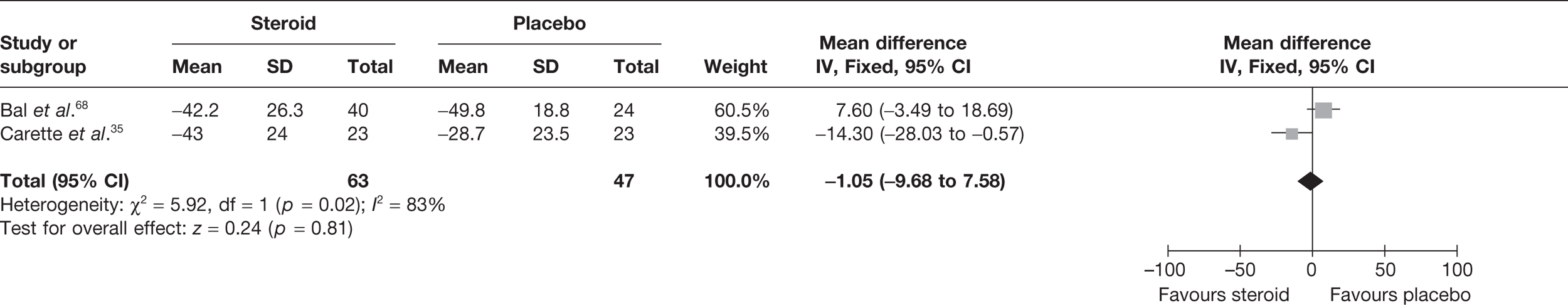
Only Carette et al. 35 reported SPADI 8-item subscale score for medium- and long-term follow-up; there was no significant difference between groups for this outcome (Table 10).
Ryans et al. 41 reported results of the Shoulder Disability Questionnaire (SDQ) and global function at 6 and 16 weeks. There was no significant difference between the steroid group and the placebo group at either time point for either outcome (Table 10). Bal et al. 68 reported University of California Los Angeles (UCLA) Shoulder score with steroid injection versus placebo and both groups were similar at the 3-month follow-up [steroid group median 32.5, interquartile range (IQR) 6.2; placebo group median 31.5, IQR 7.7; no p-value reported].
Steroid versus home exercise
Calis et al. 66 reported Constant score for short-term follow-up; steroid injection was associated with a significantly better outcome than home exercise alone (MD 9.10, 95% CI 2.98 to 15.22).
Steroid versus physiotherapy
Calis et al. 66 compared steroid with physiotherapy (without placebo injection) using the Constant score. At 3 months there was no significant difference between the two treatment groups (MD –5.80, 95% CI –11.80 to 0.20).
Steroid versus physiotherapy combined with placebo
Two studies compared steroid with physiotherapy plus placebo injection. 35,41 One study reported SPADI total score35 and the other the SDQ and a global function score. 41
There was a significantly greater improvement in SPADI total score in the steroid group than in the physiotherapy group at 6 weeks in the study by Carette et al. 35 (MD –14.50, 95% CI –28.25 to –0.75), but not at 3 months or for medium- and long-term follow-up or in the SPADI 8-item disability subscale score between treatment groups at any follow-up time in the same study (Table 10). 35 There was also no significant difference between treatment groups in either SDQ or global function score at 6 or 16 weeks in the study by Ryans et al. 41
Steroid combined with physiotherapy versus placebo
Two studies investigated steroid combined with physiotherapy versus placebo. 35,41 One study reported SPADI total score35 and the other the SDQ and a global function score. 41
There was a significant improvement in total SPADI score with steroid in combination with physiotherapy compared with placebo in the study by Carette et al. 35 at 6 weeks (MD –27.60, 95% CI –42.03 to –13.17) and 3 months (MD –21.00, 95% CI –35.43 to –6.57), although not at medium- (MD –14.10, 95% CI –28.53 to 0.33) or long-term follow-up (MD –1.10, 95% CI –15.53 to 13.33) (Table 10). 35 There was also a significant improvement in SPADI 8-item disability subscale score with steroid in combination with physiotherapy compared with placebo in the same study at 6 weeks (MD –23.90, 95% CI –37.89 to –9.91) and 3 months (MD –20.00, 95% CI –33.99 to –6.01) but not at medium- and long-term follow-up.
There was a significant improvement in both SDQ (MD –4.70, 95% CI –7.85 to –1.55) and global function score (MD –21.00, 95% CI –34.54 to –7.46) with steroid in combination with physiotherapy compared with placebo at 6 weeks in the study by Ryans et al. 41 At 16 weeks there was no significant difference between groups in this study (Table 10).
Steroid combined with physiotherapy versus physiotherapy combined with placebo
Two studies investigated the effect of steroid combined with physiotherapy versus physiotherapy (plus placebo injection) on function and disability using SPADI total score35 and the SDQ and a global function score. 41
There was a significant improvement in total SPADI score with steroid in combination with physiotherapy compared with physiotherapy in the study by Carette et al. 35 at 6 weeks (MD –24.30, 95% CI –38.33 to –10.27), although not at 3 months (MD –12.50, 95% CI –26.53 to 1.53) or for medium- and long-term follow-up (Table 10). 35 Similarly, there was a significant improvement in SPADI 8-item disability subscale score with steroid in combination with physiotherapy compared with physiotherapy in the same study at 6 weeks (MD –21.60, 95% CI –35.34 to –7.86) but not at any of the other follow-up points. 35
Ryans et al. 41 also showed a significant improvement in SDQ score with steroid combined with physiotherapy compared with physiotherapy at 6 weeks (MD –4.30, 95% CI –7.76 to –0.84) but not at 16 weeks, and there was no significant difference in global function score at either follow-up time.
Steroid combined with physiotherapy versus steroid
Two studies compared steroid injection followed by physiotherapy with steroid alone. One study reported a SPADI total score and SPADI 8-item disability subscale score;35 another study reported SDQ and global function score. 41
There was no significant difference between treatment groups in SPADI total or SPADI 8-item disability subscale score at short-, medium- or long-term follow-up in the study by Carette et al. ,35 or between treatment groups in either SDQ or global function for short- or medium-term follow-up in the study by Ryans et al. (Table 10). 41
Range of movement
The range of movement measurements of interest in the review were external rotation, internal rotation and abduction (both passive and active); if internal rotation was not available but hand behind back was reported, this was used as a proxy measure. Five studies reported at least one of these measures (Table 11). Change from baseline data was available from three of these studies35,41,68 and final value data was available from one study. 66 A fifth study, by Rizk et al. ,42 reported only means with no measures of variance or p-values with which to impute SDs. Table 12 provides the between-group differences in means and 95% CIs. The outcome data for the individual groups in the included trials are available in Appendix 7.
| Study | ROM outcomes assesseda | Data reported and time of follow-up |
|---|---|---|
| Bal 200868 | Passive internal rotation (°), passive external rotation (°), passive abduction (°) | Change from baseline at 12 weeks |
| Calis 200666 | Passive abduction (°), passive external rotation (°) | Final value data at 15 days and 3 months (3-month data used in analysis) |
| Carette 200335 | Passive external rotation (°), passive abduction (°), hand behind back (cm) | Change from baseline at 6 weeks and 3, 6 and 12 monthsb |
| Rizk 199142 | Passive internal rotation (°), passive external rotation (°) and passive abduction (°) | Mean only (no measure of variance) for weeks 1–11, 15 weeks and 6 months (6 weeks, 11 weeks and 6 months used in analysis)c |
| Ryans 200541 | Passive external rotation (°) | Change from baseline at 6 and 16 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Steroid vs placebo | |||
| Passive internal rotation (°) | |||
| Bal 200868 | –28.50 (–41.56 to –15.44)a,b | ||
| Passive external rotation (°) | |||
| Bal 200868 | –3.80 (–13.86 to 6.26)a | ||
| Carette 200335 | 8.70 (–0.72 to 18.12)d | 0.40 (–9.02 to 9.82)e | |
| Ryans 200541 | 7.70 (–1.87 to 17.27)c | –3.10 (–17.76 to 11.56)f | |
| Passive abduction (°) | |||
| Bal 200868 | 3.60 (–10.90 to 18.10)a | ||
| Carette 200335 |
7.20 (–0.83 to 15.23)c 12.30 (4.27 to 20.33)a |
7.80 (–0.23 to 15.83)d | –1.20 (–9.23 to 6.83)e |
| Hand behind back (cm) | |||
| Carette 200335 |
–0.50 (–7.15 to 6.15)c –1.10 (–7.75 to 5.55)a |
–0.50 (–7.15 to 6.15)d | 2.80 (–3.85 to 9.4)e |
| Steroid vs home exercise | |||
| Passive external rotation (°) | |||
| Calis 200666 | 8.00 (2.48 to 13.52)a,b | ||
| Passive abduction (°) | |||
| Calis 200666 | 16.80 (6.60 to 27.00)a,b | ||
| Steroid vs physiotherapy | |||
| Passive external rotation (°) | |||
| Calis 200666 | –10.80 (–16.94 to –4.66)a,b | ||
| Passive abduction (°) | |||
| Calis 200666 | –8.10 (–19.07 to 2.87)a | ||
| Steroid vs physiotherapy + placebo | |||
| Passive external rotation (°) | |||
| Carette 200335 |
8.70 (–0.44 to 17.84)c 7.50 (–1.64 to 16.64)a |
6.50 (–2.64 to 15.64)d | –0.80 (–9.94 to 8.34)e |
| Ryans 200541 | –2.40 (–11.66 to 6.86)c | 1.10 (–11.39 to 13.59)f | |
| Passive abduction (°) | |||
| Carette 200335 |
3.20 (–4.57 to 10.97)c 6.40 (–1.37 to 14.17)a |
2.70 (–5.07 to 10.47)d | –1.90 (–9.67 to 5.87)e |
| Hand behind back (cm) | |||
| Carette 200335 |
–0.80 (–7.30 to 5.70)c 0.10 (–6.40 to 6.60)a |
0.70 (–5.80 to 7.20)d | 2.90 (–3.60 to 9.40)e |
| Steroid + physiotherapy vs placebo | |||
| Passive external rotation (°) | |||
| Carette 200335 | 13.30 (3.60 to 23.00)b,d | 7.60 (–2.10 to 17.30)e | |
| Ryans 200541 | –2.50 (–16.42 to 11.42)c | –2.50 (–16.42 to 11.42)f | |
| Passive abduction (°) | |||
| Carette 200335 | 13.00 (4.84 to 21.16)b,d | 5.70 (–2.46, 13.86)e | |
| Hand behind back (cm) | |||
| Carette 200335 | –5.90 (–12.70 to 0.90)d | –1.50 (–8.30 to 5.30)e | |
| Steroid + physiotherapy vs physiotherapy + placebo | |||
| Passive external rotation (°) | |||
| Carette 200335 | 11.10 (1.66 to 20.54)b,d | 6.40 (–3.04 to 15.84)e | |
| Ryans 200541 | 4.30 (–5.45 to 14.05)c | 1.70 (–9.91 to 13.31)f | |
| Passive abduction (°) | |||
| Carette 200335 | 7.90 (–0.00 to 15.80)d | 5.00 (–2.90 to 12.90)e | |
| Hand behind back (cm) | |||
| Carette 200335 | –4.70 (–11.36 to 1.96)d | –1.40 (–8.06 to 5.26)e | |
| Steroid + physiotherapy vs steroid | |||
| Passive external rotation (°) | |||
| Carette 200335 |
–8.20 (–17.90 to 1.50)c –5.50 (–15.20 to 4.20)a |
–4.60 (–14.30 to 5.1)d | –7.20 (–16.90 to 2.50)e |
| Ryans 200541 | –6.70 (–17.36 to 3.96)b,c | –0.60 (–14.62 to 13.42)f | |
| Passive abduction (°) | |||
| Carette 200335 |
–8.10 (–16.26 to 0.06)c –4.00 (–12.16 to 4.16)a |
–5.20 (–13.36 to 2.96)d | –6.90 (–15.06 to 1.26)e |
| Hand behind back (cm) | |||
| Carette 200335 |
6.00 (–0.80 to 12.80)a |
5.40 (–1.40 to 12.20)d | 4.30 (–2.50 to 11.10)e |
Steroid versus placebo
One study by Bal et al. 68 reported passive internal rotation at 12 weeks. There was a significantly greater improvement in the placebo group compared with steroid injection (MD –28.50°, 95% CI –41.56° to –15.44°). This result contradicts the original analysis in the paper, which reported that there was no statistically significant difference between the two groups, using the Mann–Whitney U-test.
Three studies reported passive external rotation at short-term follow-up, two at 6 weeks35,41 and two at 3 months. 35,68 Improvement in passive external rotation was significantly greater with steroid injection than with placebo injection at 6 weeks (pooled MD 9.48°, 95% CI 2.76° to 16.19°; two RCTs) (Figure 10). There was no significant heterogeneity between these studies (I2 = 0%)
FIGURE 10.
Passive external rotation at 6 weeks: steroid vs placebo.
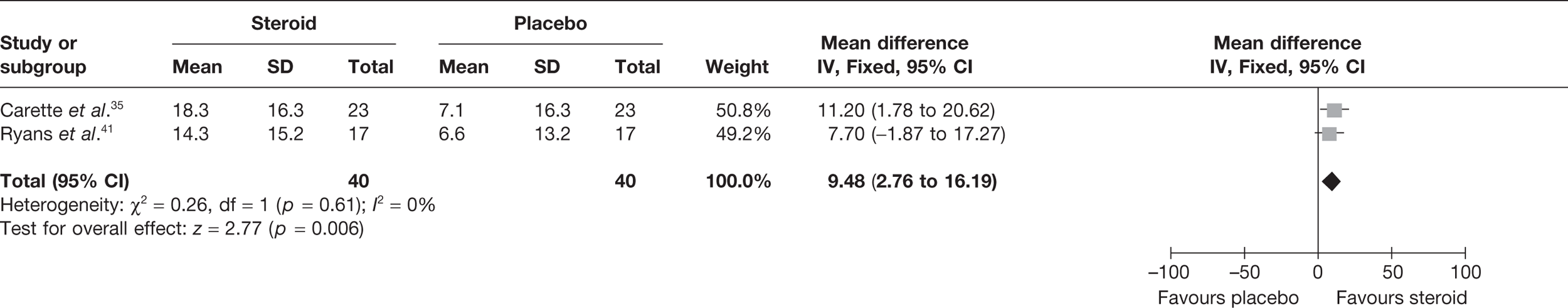
Meta-analysis of the two studies that reported passive external rotation at 3 months showed no significant difference between the steroid and the placebo groups (Figure 11)35,68 This pooled result was associated with substantial heterogeneity (I2 = 80%) and should therefore be treated with caution. The study by Carette et al. 35 showed a significant improvement in passive external rotation in the steroid group compared with placebo whereas the study by Bal et al. 68 showed no significant difference between groups (Table 11). There were several possible sources of heterogeneity, in particular differences in the risk of bias.
FIGURE 11.
Passive external rotation at 3 months: steroid vs placebo.
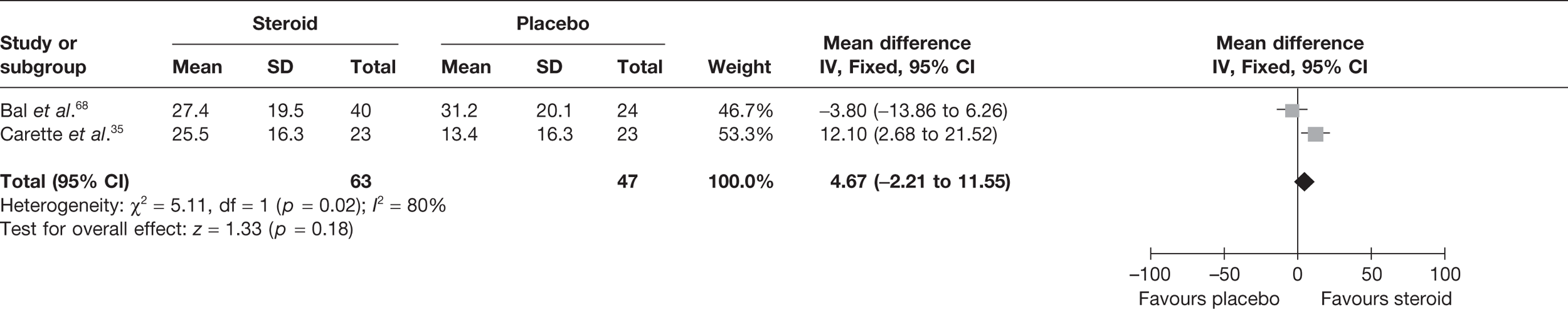
Two RCTs reported passive external rotation for medium-term follow-up: 16 weeks41 and 6 months. 35,41 There was no significant difference between steroid and placebo groups in passive external rotation (MD 4.67°, 95% CI –2.21° to 11.55°) (Figure 12). There was some evidence of moderate statistical heterogeneity (I2 = 43%). Both studies appeared clinically similar although the follow-up time points differed. Carette et al. 35 was the only study to examine passive external rotation at long-term follow-up and found no significant difference between steroid and placebo groups (Table 12). 35
FIGURE 12.
Passive external rotation at up to 6 months: steroid vs placebo.
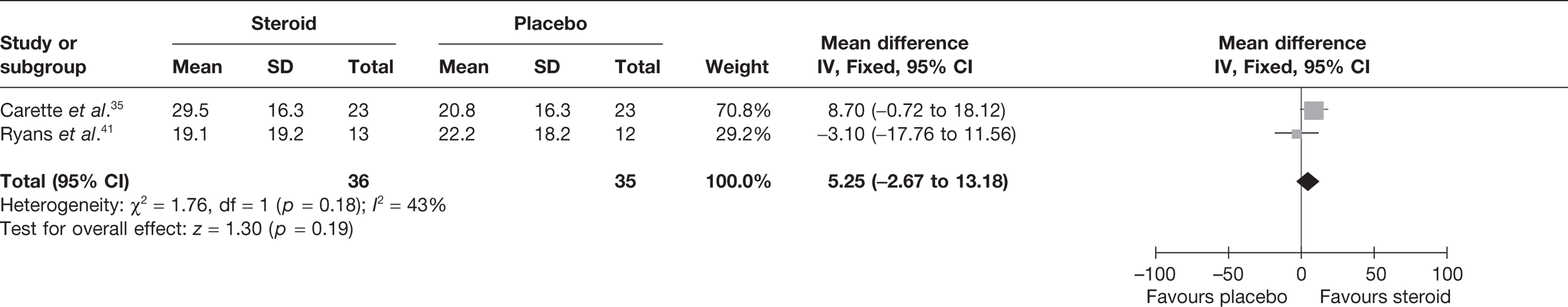
Carette et al35 reported passive abduction and hand behind back at 6 weeks and 3, 6 and 12 months. There was no significant difference between groups at any of the time points for either outcome (Table 11).
Steroid versus home exercise
Calis et al. 66 reported passive abduction and passive external rotation at 3 months. There was a significantly greater improvement in the steroid group compared with home exercise alone in both passive abduction (MD 16.80°, 95% CI 6.60° to 27.00°) and passive external rotation (MD 8.00°, 95% CI 2.48° to 13.52°).
Steroid versus physiotherapy
Calis et al. 66 reported passive external rotation and passive abduction at 3 months. There was significantly greater passive external rotation at 3 months with physiotherapy than with steroid (MD –10.80°, 95% CI –16.94° to –4.66°). There was no significant difference between treatment groups for passive abduction (Table 12).
Steroid versus physiotherapy combined with placebo
Three studies reported passive external rotation at time points up to 3 months. 35,41,66 Two of the studies involved physiotherapy sessions over 12 weeks and in both physiotherapy groups a placebo injection was also given. 35,41 Both studies reported data at 6 weeks and one35 reported data at 3 months; 6-week data were pooled. There was no significant difference in passive external rotation between groups at 6 weeks (Figure 13). There was substantial statistical heterogeneity between the studies (I2 = 64%).
FIGURE 13.
Passive external rotation at 6 weeks: steroid vs physiotherapy combined with placebo.
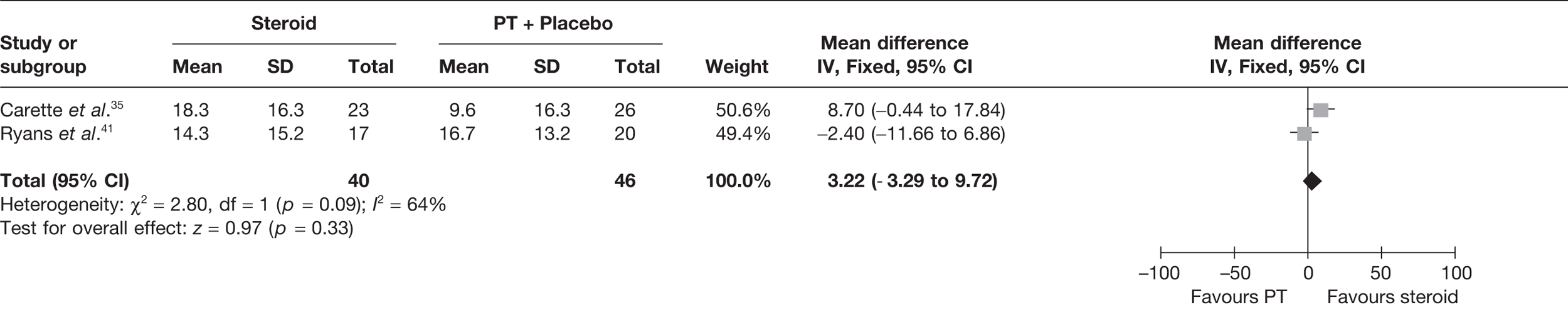
Carette et al. 35 reported passive external rotation, and passive abduction at 3 months. There was no significant difference between treatment groups for both outcomes (Table 12).
Medium-term follow-up data were available for the studies by Carette et al. 35 and Ryans et al. 41 Carette et al. 35 reported passive external rotation at 6 months and Ryans et al. 41 at 16 weeks. There was no significant difference between groups (Figure 14). There was no significant difference between groups in passive external rotation at 12 months in the Carette et al. study. 35 Similarly, the same study showed no significant difference between treatment groups in passive abduction or hand behind back at medium- or long-term follow-up (Table 12).
FIGURE 14.
Passive external rotation at up to 6 months: steroid vs physiotherapy combined with placebo.
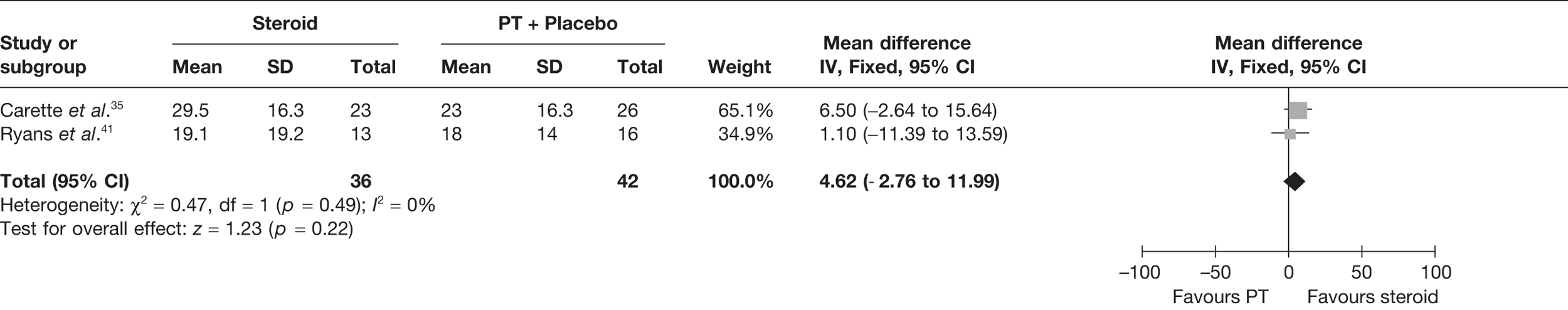
Steroid combined with physiotherapy versus placebo
Two studies reported passive external rotation at time points up to 3 months. 35,41 Both studies reported data at 6 weeks and one35 reported data at 3 months. Data at 6 weeks were pooled. The meta-analysis (Figure 15) indicated that passive external rotation was significantly greater with steroid injection plus physiotherapy than with placebo injection at 6 weeks (pooled MD 16.99°, 95% CI 10.01° to 23.97°).
FIGURE 15.
Passive external rotation at 6 weeks: steroid combined with physiotherapy vs placebo.
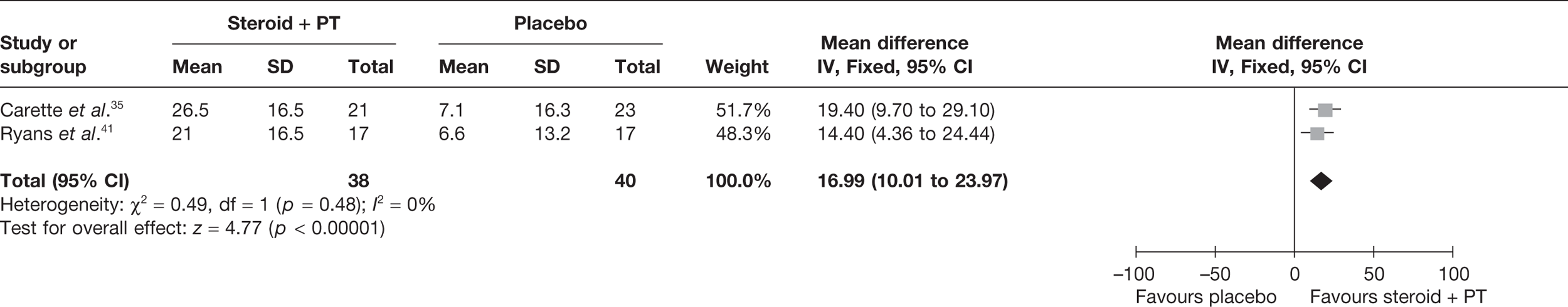
Passive external rotation was significantly greater with steroid injection plus physiotherapy than with placebo injection at 3 months (MD 17.60°, 95% CI 7.90° to 27.30°) in the study by Carette et al. 35
Medium-term follow-up data were also available up to 6 months: Carette et al. 35 reported passive external rotation at 6 months and Ryans et al. 41 at 16 weeks. The pooled estimate showed that passive external rotation was significantly greater with steroid injection plus physiotherapy than with placebo injection at medium-term follow-up (pooled MD 8.14°, 95% CI 0.18° to 16.09°); however, there was substantial statistical heterogeneity (I2 = 70%) (Figure 16). There were a number of possible sources of heterogeneity including the dose of steroid used, different length of follow-up and the proportion of participants with diabetes. There was no significant difference between groups at 12 months in the single study reporting long-term follow-up (Table 12). 35
FIGURE 16.
Passive external rotation at up to 6 months: steroid combined with physiotherapy vs placebo.
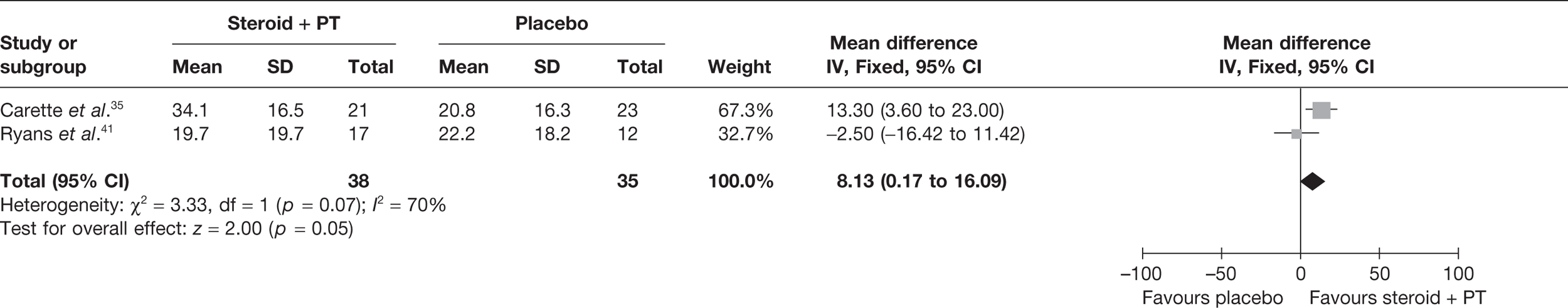
Carette et al. 35 reported passive abduction at 6 weeks and 3, 6 and 12 months. Improvement of passive abduction was significantly greater in the steroid plus physiotherapy group than with placebo injection at 6 weeks (MD 15.30°, 95% CI 7.14° to 23.46°), 3 months (MD 16.30°, 95% CI 8.14° to 24.46°) and 6 months (MD 13.00°, 95% CI 4.84° to 21.16°). There was no significant difference between treatment groups at 12 months (Table 12).
In the same study there was a significantly greater improvement in hand behind back for the steroid with physiotherapy group than with placebo injection at 6 weeks (MD –9.00 cm, 95% CI –15.80 cm to –2.20 cm) and 3 months (MD –7.10 cm, 95% CI –13.90 cm to –0.30 cm), but not at any of the other time points (Table 12).
Steroid combined with physiotherapy versus physiotherapy combined with placebo
Three studies reported passive external rotation at time points up to 3 months. 35,41,42 All studies reported data at 6 weeks35,41,42 and two also reported data at 3 months. 35,42 Because of SDs not being available for one study,42 data were pooled from two of the studies for 6-week follow-up. 35,41 The increase in passive external rotation was significantly greater with steroid injection plus physiotherapy than with physiotherapy (plus placebo injection) at 6 weeks (pooled MD 10.80°, 95% CI 4.02° to 17.58°) (Figure 17). There was moderate to high heterogeneity between the studies (I2 = 70%); the results of this analysis should therefore be considered with caution.
FIGURE 17.
Passive external rotation at 6 weeks: steroid combined with physiotherapy vs physiotherapy combined with placebo.
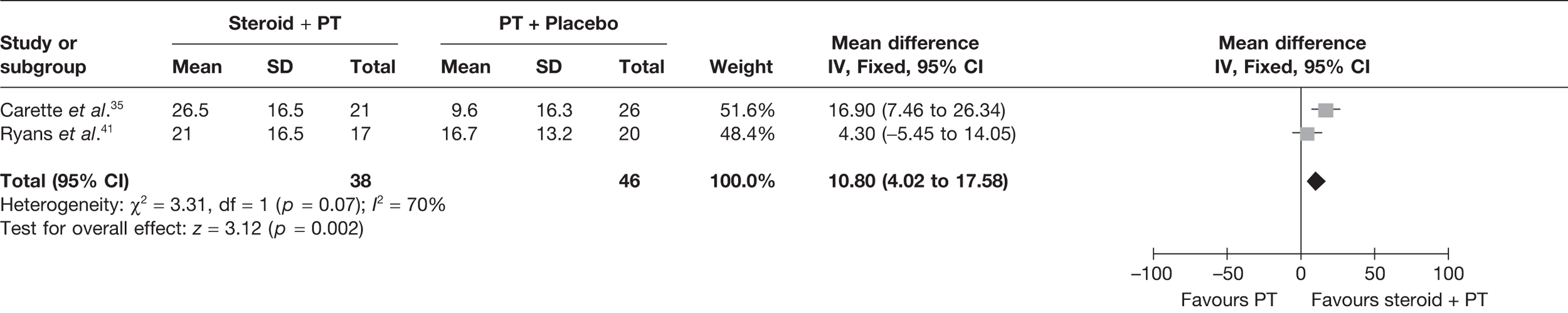
The study by Carette et al. 35 also showed that the increase in passive external rotation was significantly greater with steroid injection plus physiotherapy than with physiotherapy at 3 months (MD 13.00°, 95% CI 3.56° to 22.44°).
Medium-term data were also available from Carette et al. 35 and Ryans et al. 41 The pooled estimate (Figure 18) showed that passive external rotation was significantly greater with steroid injection plus physiotherapy than with physiotherapy at up to 6 months (pooled MD 7.36°, 95% CI 0.04° to 14.68°). The study by Carette et al. 35 showed no significant difference between groups at 12 months (Table 12).
FIGURE 18.
Passive external rotation at up to 6 months: steroid combined with physiotherapy versus physical therapy combined with placebo.
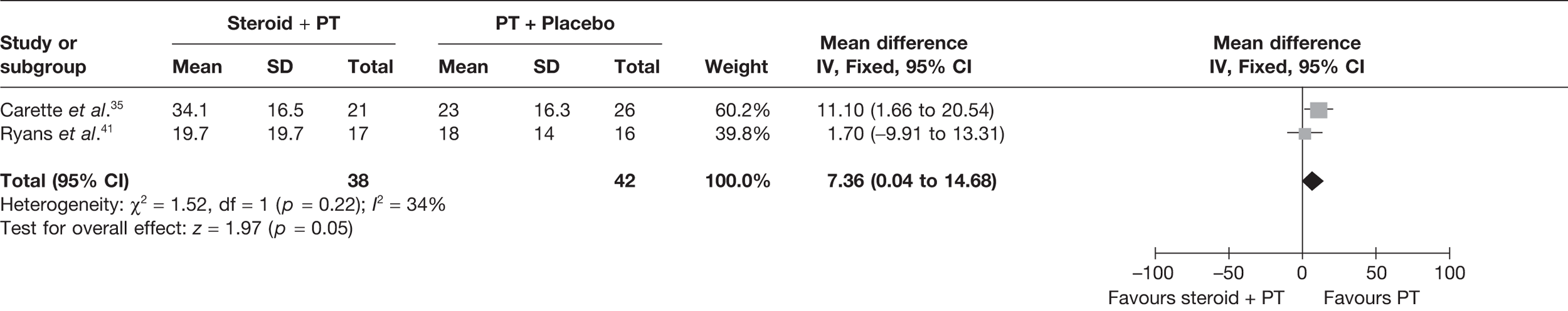
Carette et al. 35 reported passive abduction at 6 weeks and 3, 6 and 12 months. Improvement of passive abduction was significantly greater in the steroid plus physiotherapy group than with physiotherapy at 6 weeks (MD 11.30°, 95% CI 3.40° to 19.20°), 3 months (MD 10.40°, 95% CI 2.50° to 18.30°) and 6 months (MD 7.90°, 95% CI –0.00° to 15.80°). There was no significant difference between treatment groups at 12 months (Table 12).
Steroid versus steroid and physiotherapy
Two studies reported passive external rotation for short-term follow-up;35,41 both studies reported data at 6 weeks and one35 reported data at 3 months; the 6-week data were pooled. Passive external rotation was significantly greater with steroid injection plus physiotherapy than with steroid alone at 6 weeks (pooled MD –7.52°, 95% CI –14.70° to –0.34°). This analysis was not associated with statistical heterogeneity (I2 = 0%) (Figure 19). The study by Carette et al. 35 showed no significant difference between treatment groups at 3 months (Table 12).
FIGURE 19.
Passive external rotation at 6 weeks: steroid versus steroid and physiotherapy.
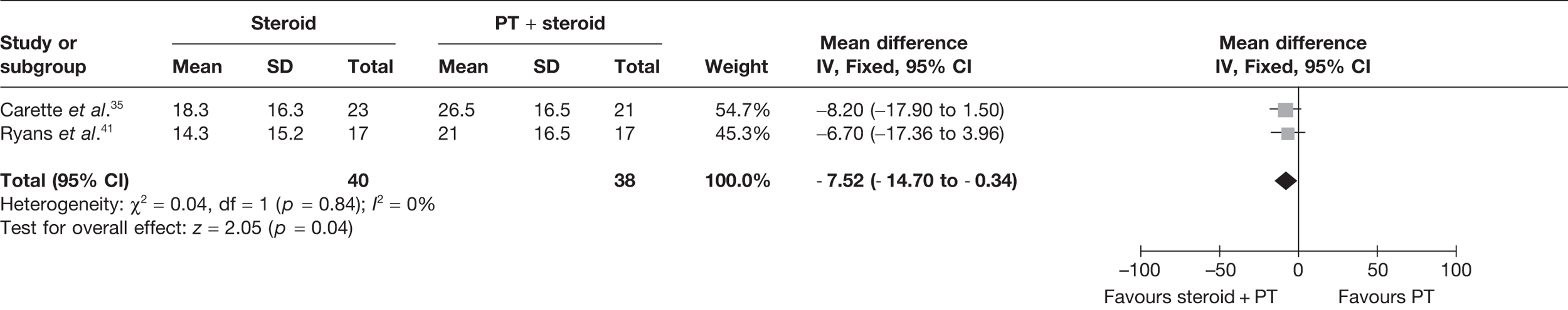
Data were also available for medium-term follow-up for the same two studies. The pooled analysis showed no significant difference between groups and the analysis was not associated with statistical heterogeneity (I2 = 0%) (Figure 20). The study by Carette et al. 35 showed no significant difference between treatment groups at long-term follow-up (Table 12).
FIGURE 20.
Passive external rotation at up to 6 months: steroid versus steroid and physiotherapy.
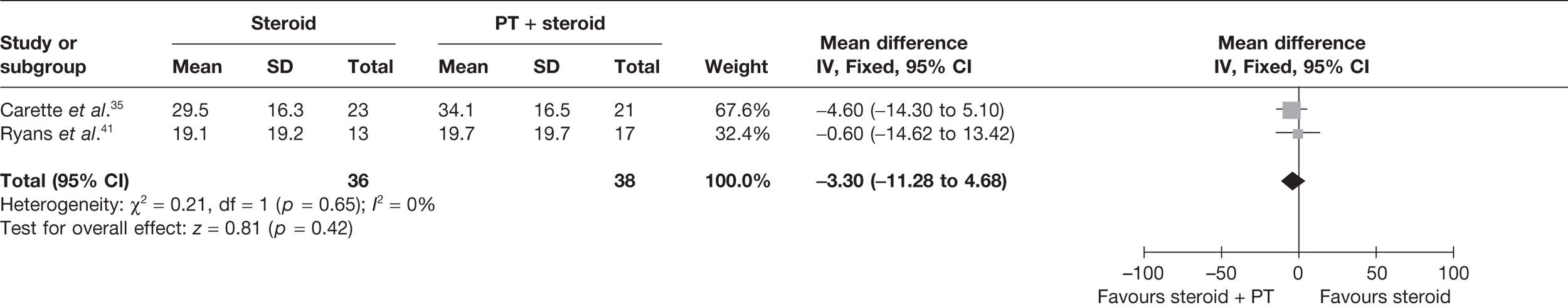
Carette et al. 35 reported passive abduction at 6 weeks and 3, 6 and 12 months. At 6 weeks improvement of passive abduction was significantly greater in the steroid plus physiotherapy group than with steroid alone (MD –8.10, 95% CI –16.26 to 0.06); there were no significant differences between treatment groups at later follow-up times (Table 12).
Similarly, for hand behind back, there was a significantly better improvement in the steroid with physiotherapy group than with steroid alone at 6 weeks (MD 8.50, 95% CI 1.70 to 15.30) but not at later follow-up (Table 12).
Quality of life
Only Carette et al. 35 reported quality of life data following treatment (Table 13). 35 Table 14 provides between-group differences in means and associated 95% CIs. Further outcome data can be found in Appendix 7.
| Study | QoL outcomes assessed | Data reported and time point before and closest to 3 and 6 months |
|---|---|---|
| Carette 200335 |
SF-36 physical component SF-36 mental component |
Change from baseline at 6 weeks and 3, 6 and 12 monthsa |
| Ryans 200541 | SF-36 scores: physical function, role limitation physical, role limitation mental, social functioning, mental health, vitality, bodily pain, health perception, change in health | Baseline data only reported |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Steroid vs placebo | |||
| SF-36 physical component | |||
| Carette 200335 |
1.90 (–3.36 to 7.16)a 5.10 (–0.16 to 10.36)b |
3.30 (–1.96 to 8.56)c | 1.00 (–4.26 to 6.26)d |
| SF-36 mental component | |||
| Carette 200335 |
–1.10 (–7.23 to 5.03)a 1.00 (–5.13 to 7.13)b |
1.70 (–5.34 to 8.74)c | 0.30 (–5.83 to 6.43)d |
| Steroid vs physiotherapy | |||
| SF-36 physical component | |||
| Carette 200335 |
3.30 (–1.97 to 8.57)a 3.70 (–1.57 to 8.97)b |
1.00 (–4.27 to 6.27)c | 1.70 (–3.57 to 6.97)d |
| SF-36 mental component | |||
| Carette 200335 |
–0.50 (–6.61 to 5.61)a –1.60 (–7.71 to 4.51)b |
2.80 (–3.31 to 8.91)c | 1.10 (–5.01 to 7.21)d |
| Steroid combined with physiotherapy vs placebo | |||
| SF-36 physical component | |||
| Carette 200335 |
3.90 (–1.36 to 9.16)a 3.60 (–1.66 to 8.86)b |
2.00 (–3.26 to 7.26)c | 1.40 (–3.86 to 6.66)d |
| SF-36 mental component | |||
| Carette 200335 |
3.10 (–3.14 to 9.34)a 5.40 (–0.84 to 11.64)b |
7.10 (0.86 to 13.34)c,e | 6.10 (–0.14 to 12.34)d |
| Steroid combined with physiotherapy vs physiotherapy | |||
| SF-36 physical component | |||
| Carette 200335 |
2.20 (–3.07 to 7.47)b |
–0.30 (–5.57 to 4.97)c | 2.10 (–3.17 to 7.37)d |
| SF-36 mental component | |||
| Carette 200335 |
3.70 (–2.52 to 9.92)a 2.80 (–3.42 to 9.02)b |
8.20 (1.98 to 14.42)c,e | 6.90 (0.68 to 13.12)d,e |
| Steroid combined with physiotherapy vs steroid | |||
| SF-36 physical component | |||
| Carette 200335 |
–2.00 (–7.26 to 3.26)a 1.50 (–3.76 to 6.76)b |
1.30 (–3.96 to 6.56)c | –0.40 (–5.66 to 4.86)d |
| SF-36 mental component | |||
| Carette 200335 |
–4.20 (–10.44 to 2.04)a –4.40 (–10.64 to 1.84)b |
–5.40 (–11.64 to 0.84)c | –5.80 (–12.04 to 0.44)d |
There was a statistically significant improvement in the physical component of the SF-36 with steroid plus physiotherapy compared with placebo at 6 months (MD 7.10, 95% CI 0.86 to 13.34). There was no statistically significant difference between these two groups at any of the other time points evaluated.
There was a statistically significant improvement with steroid plus physiotherapy compared with physiotherapy (plus placebo injection) in the physical component at 6 weeks (MD 5.30, 95% CI 0.03 to 10.57) and in the mental component at 6 months (MD 8.20, 95% CI 1.98 to 14.42) and 12 months (MD 6.90, 95% CI 0.68 to 13.12). There was no statistically significant difference between these two treatments groups at any of the other time points evaluated.
For the comparisons steroid compared with placebo, steroid compared with physiotherapy (plus placebo injection) and steroid compared with physiotherapy plus steroid, the study by Carette et al. 35 showed no statistically significant difference between treatment groups at any of the time points evaluated.
Adverse events
Three of the studies that investigated steroids reported adverse events. 42,67,68 There was a limited amount of information reported regarding adverse events in the included studies (Table 15). None of the studies reported any details regarding how adverse event data were collected or recorded; therefore, the data should be treated with caution. Two reported that no adverse effects were reported in any of the treatment groups67,68 and one reported that there were no withdrawals due to adverse effects. 42
Summary
The six included steroid injection studies were diverse in terms of the intervention: two used a single steroid injection of 20 mg, three a single injection of 40 mg and one used three injections of 40 mg administered over 3 weeks. Five of the studies reported using an intra-articular injection. There was also some variation between studies in the physiotherapy provided, although most of the studies used a programme of < 6 weeks’ duration that included mobilisation and home exercise for all groups. Five of the studies were multi-armed trials. Steroid injection was compared with placebo in three studies, home exercise in one study and physiotherapy alone in four studies (with or without placebo). The effect of steroid injection followed by physiotherapy was also assessed. This combined intervention was compared with placebo, steroid alone and physiotherapy (with placebo) alone.
Information about previous treatments that participants had received and stage of frozen shoulder was limited. Outcomes were not reported by stage of frozen shoulder or by whether or not participants had diabetes. As a result, it was not possible to explore in the synthesis the effect of these variables on outcome. It was unclear whether four of the studies were truly randomised and therefore these studies had a potentially high risk of bias. 42,66–68 The fifth study was of adequate quality,35 whereas the sixth was of reasonable quality but may have a risk of bias. 41
For most of the comparisons, data were available from only one or two studies, which may have been underpowered to detect an effect. There were sufficient data to pool the two best-quality studies for some of the outcomes, although in some instances statistical heterogeneity was high. In addition, although both studies used a single intra-articular injection, one evaluated a guided injection of 40 mg of steroid35 and the other used a 20-mg dose and the injection appeared to be unguided. 41 For most outcomes and comparisons it was necessary to undertake a narrative synthesis.
Steroid alone
There was evidence of short-term benefit with steroid injection compared with placebo for pain, function and disability, and range of movement but not quality of life. The evidence on function and disability was mixed and this may reflect differences in study quality and/or clinical differences. There was also evidence of short-term benefit with steroid injection compared with home exercise alone for function and disability and two measures of range of movement from one study with a potentially high risk of bias. There was no consistent evidence across outcomes for a benefit with steroid injection compared with physical therapy.
Steroid followed by physiotherapy
There was consistent evidence across all outcomes (pain, function and disability, range of movement and quality of life) of a short-term benefit with steroid injection followed by physiotherapy compared with placebo or physiotherapy; however, this was based on a small number of studies. There was also evidence of better quality of life with the combined treatment in the medium term. When steroid injection plus physiotherapy was compared with steroid alone there was a short-term beneficial reduction in pain and a better range of movement at the 6-week follow-up with the combined intervention but not at 3 months or later and there was no benefit for any other outcomes.
In conclusion, a small number of diverse studies were identified, four of which had a potentially high risk of bias. Based on the best available evidence, there was a benefit, mainly short term, with a single steroid. There was an added benefit with providing physiotherapy in addition to steroid injection compared with home exercise alone and physiotherapy alone for several outcomes. There was also benefit with the combined intervention compared with steroid alone for pain and for range of movement at 6 weeks but not the other outcomes. There was insufficient evidence to conclude with reasonable certainty in what clinical situations steroid injection, with or without physiotherapy, is most likely to be effective for primary frozen shoulder.
Sodium hyaluronate
Study characteristics
Three RCTs investigated sodium hyaluronate in the treatment of primary frozen shoulder. A summary of the study characteristics is reported in Table 16. All were full papers of studies conducted in Turkey,66 Italy69 and Japan70 between 1996 and 2006. There were two two-armed trials69,70 and one four-armed trial. 66
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Calis 200666 Four-arm RCT |
n = 90 (95 shoulders) Sodium hyaluronate injection: 24 (27) Steroid injection: 25 (26) PT: 21 (22) No intervention (home exercise only): 20 (20) |
Sodium hyaluronate: 30 mg, one injection once weekly for 2 weeks Steroid: triamcinolone acetonide, 40 mg, one injection PT: 10 daily sessions at least 45 minutes |
History of pain for at least 1 month; limited active and passive shoulder movement; decreased passive ROM of ≥ 20%, in at least three movements, according to the American Medical Association guide for the evaluation of permanent impairment |
|
Rovetta 199869 Two-arm RCT |
n = 30 Sodium hyaluronate + steroid injection + PT: 16 Steroid injections + PT: 14 |
Sodium hyaluronate: 20 mg at 15-day intervals in the first month, then monthly for 6 months Steroid: triamcinolone acetonide at 15-day intervals in the first month, then monthly for 6 months PT: for 4–12 weeks |
Clinical history of spontaneous shoulder pain; glenohumeral abduction and forward flexion < 90°, external rotation < 20°; clinical absence of signs of rotator cuff interruption; cervical examination excluding dysfunction in this area; plain radiographs in standard views; sonographic examination showing shrinking of the joint capsule with increased capsular echogenicity |
|
Takagishi 199670 Two-arm RCT |
n = 20 Sodium hyaluronate: 10 Steroid injection: 10 |
Sodium hyaluronate: 2 mg, one injection once weekly for 5 weeks Steroid: dexamethasone, 2 mg, one injection once weekly for 5 weeks |
Patients with frozen shoulder who have < 120° of shoulder joint flexion and those who suffer from pain on exercise of > 4 on the VAS and suffer from pain during night-time and daytime |
Calis et al. 66 used a 30-mg injection once weekly for 2 weeks, Takagishi et al. 70 used 2 mg injected once weekly for 5 weeks and Rovetta et al. 69 used 20 mg of sodium hyaluronate coadministered with 20 mg of steroid (triamcinolone acetonide) injected at 15-day intervals for the first month then monthly for 6 months and physical therapy (mobilisation or exercises) for 4–12 weeks. The comparators were steroid injection,66,70 steroid injection and physiotherapy,69 physiotherapy66 and no intervention except for home exercise. 66
Two of the three studies included physiotherapy. 66,69 Both studies differed in the duration and content of the regimen, but each contained some form of active exercise or mobilisation. Calis et al. 66 had a regimen of 10 daily sessions consisting of a heat pack applied for 20 minutes, ultrasonic therapy for 5 minutes (1.5 W/cm2 intensity), TENS for 20 minutes at the patient’s level of tolerance, and stretching exercises. In comparison, Rovetta et al. 69 had a 4- to 12-week regimen of the most appropriate therapy for the individual (passive mobilisation, active exercises and facilitation exercises), which was chosen by a physiotherapist. Only Calis et al. 66 reported that participants had a home exercise programme, which consisted of stretching and Codman exercises, but did not provide any further details.
Two studies reported that patients received concomitant treatments. 69,70 Rovetta et al. 69 allowed paracetamol (dose unspecified) to be taken if necessary, whereas Takagishi et al. 70 stated that participants who were using topical NSAIDs prior to commencing the study were allowed to continue to do so. Calis et al. 66 allowed paracetamol to be taken if necessary.
The inclusion criteria varied between the three studies. Rovetta et al. 69 reported that none of the participants had diabetes, whereas Calis et al. 66 and Takagishi et al. 69 did not report whether or not any of the participants had diabetes. Frozen shoulder was diagnosed through physical examination, laboratory tests, subacromial impingement test, clinical history, radiography or ultrasound when described. The method of diagnosis was unclear in one study. 70 The participants’ stage of frozen shoulder at baseline was not reported in any of the studies and it was unclear whether participants had received any previous treatments. The mean age of participants ranged from 48 years to 65.8 years. The proportion of women ranged from 63% to 75%.
Quality assessment
None of the studies reported the method of randomisation or allocation concealment, and only one stated that blinded outcome assessment was performed; therefore, all three studies have a potentially high risk of bias. The study by Calis et al. 66 was of higher quality and fulfilled five criteria: the number of participants randomised, treatment groups that were comparable at baseline, blinded outcome assessors, use of intention-to-treat analysis and no unexpected imbalances in dropouts. Rovetta et al. 69 fulfilled two criteria, stating the number of participants randomised and also having treatment groups that were comparable at baseline, whereas Takagishi et al. 70 fulfilled only one criterion, which was stating the number of patients randomised. Full details of study quality are reported in Appendix 8.
Pain
All three studies evaluating sodium hyaluronate assessed pain; however, the study by Calis et al. 66 reported pain in graphical form and therefore could not be included in the analysis. Calis et al. 66 did, however, report that there was significant improvement in pain severity at 3 months within each of the treatment groups, including the no intervention group (p < 0.001). Rovetta et al. 69 and Takagishi et al. 70 both used a VAS but assessed different types of pain at different time points (Table 17); therefore, it was inappropriate to pool the two studies. Table 18 provides the SMDs and 95% CIs. The outcome data for the individual groups in the included trials are available in Appendix 7.
| Study | Pain outcome assessed | Scale used | Data reported and time of follow-up |
|---|---|---|---|
| Calis 200666 | Pain severity | VAS | Data reported graphically only |
| Rovetta 199869 | Pain on passive joint motion | VAS 0–10 cm | Final value at 6 months |
| Takagishi 199670 | Pain on activity | VAS 0–10 | Final value at 5 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Sodium hyaluronate combined with steroid and physiotherapy vs steroid combined with physiotherapy | |||
| Rovetta 199869 | –0.78 (–1.50 to –0.06)a,b | ||
| Sodium hyaluronate vs steroid | |||
| Takagishi 199670 | –0.21 (–1.09 to 0.67)c | ||
Sodium hyaluronate versus steroid
Takagishi et al. 70 found no significant difference in pain at short-term follow-up with sodium hyaluronate compared with steroid (see Table 18).
Sodium hyaluronate combined with steroid and physiotherapy versus steroid combined with physiotherapy
Rovetta et al. 69 found a significant decrease in pain at 6 months with sodium hyaluronate combined with steroid and physiotherapy compared with steroid combined with physiotherapy (SMD –0.78, 95% CI –1.50 to –0.06).
Function and disability
Calis et al. 66 and Takagishi et al. 70 both assessed function and disability. Calis et al. assessed function and disability using the Constant score (Table 19), whereas Takagishi et al. 70 used a Japanese-specific activities of daily living questionnaire. However, Takagishi et al. reported baseline values and a p-value for the within-group change from baseline only and therefore could not be included in the analysis. Table 20 provides the MD and 95% CIs. The outcome data for the individual groups in the included trial are available in Appendix 7.
| Study | Function and disability outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Calis 200666 | Constant score | Final values at 3 months |
| Takagishi 199670 | Activities of daily living questionnaire (Japanese specific) | Baseline data and p-value for within-group change from baseline only reported |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Sodium hyaluronate vs no treatment | |||
| Constant score | |||
| Calis 200666 | 8.90 (2.62 to 15.18)a,b | ||
| Sodium hyaluronate vs physiotherapy | |||
| Constant score | |||
| Calis 200666 | –6.00 (–12.16 to 0.16)a | ||
| Sodium hyaluronate vs steroid | |||
| Constant score | |||
| Calis 200666 | –0.20 (–5.86 to 5.46)a | ||
At 3 months Calis et al. 66 found a significantly greater improvement in the Constant score with sodium hyaluronate compared with no treatment (MD 8.90, 95% CI 2.62 to 15.18), but no significant difference in Constant score between sodium hyaluronate and physiotherapy (MD –6.00, 95% CI –12.16 to 0.16) or between sodium hyaluronate and steroid injection (MD –0.20, 95% CI –5.86 to 5.46).
Range of movement
The range of movement measurements of interest were external rotation, internal rotation and abduction (both passive and active); if internal rotation was not available but hand behind back was reported, this was used as a proxy measure of active internal rotation. All three studies that evaluated sodium hyaluronate reported at least one of these measurements (Table 21). Table 22 provides the between-group difference in means and 95% CIs. The outcome data for the individual groups in the included trials are available in Appendix 7.
| Study | ROM outcomes assessed (°) | Data reported at time points up to 3 and 6 months |
|---|---|---|
| Calis 200666 | Passive external rotation, passive abduction | Final values at 15 days and 3 months |
| Rovetta 199869 | Internal rotation, external rotation, abduction | Final values at 6 months |
| Takagishi 199670 | External rotation | Final values at 5 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Sodium hyaluronate vs no treatment | |||
| Passive external rotation (°) | |||
| Calis 200666 | 8.30 (2.52 to 14.08)a,b | ||
| Passive abduction (°) | |||
| Calis 200666 | 12.40 (0.14 to 24.66)a,b | ||
| Sodium hyaluronate vs physiotherapy | |||
| Passive external rotation (°) | |||
| Calis 200666 | –10.50 (–16.87 to –4.13)a,b | ||
| Passive abduction (°) | |||
| Calis 200666 | –12.50 (–23.98 to –1.02)a,b | ||
| Sodium hyaluronate vs steroid | |||
| Passive external rotation (°) | |||
| Calis 200666 | 0.30 (–5.92 to 6.52)a | ||
| Passive abduction (°) | |||
| Calis 200666 | –4.40 (–15.78 to 6.98)a | ||
| External rotation (°) | |||
| Takagischi 199670 | –2.00 (–21.25 to 17.28)c | ||
| Sodium hyaluronate + steroid + physiotherapy vs steroid + physiotherapy | |||
| Internal rotation (°) | |||
| Rovetta 199869 | 23.70 (13.44 to 33.96)b,d | ||
| External rotation (°) | |||
| Rovetta 199869 | –0.70 (–15.11 to 13.71)d | ||
| Abduction (°) | |||
| Rovetta 199869 | 3.20 (–18.70 to 25.10)d | ||
Sodium hyaluronate versus no treatment
Calis et al. 66 reported passive external rotation and passive abduction at 3 months. There was a significantly greater improvement in the sodium hyaluronate group than in the no treatment group in both passive external rotation (MD 8.30°, 95% CI 2.52° to 14.08°) and passive abduction (MD 12.40°, 95% CI 0.14° to 24.66°).
Sodium hyaluronate versus physiotherapy
Calis et al. 66 reported passive external rotation and passive abduction at 3 months. There was significantly greater improvement in the physiotherapy group than in the sodium hyaluronate group in both passive external rotation (MD –10.50°, 95% CI –16.87° to –4.13°) and passive abduction (MD –12.50°, 95% CI –23.98° to –1.02°).
Sodium hyaluronate versus steroid
Calis et al. 66 reported passive external rotation and passive abduction at 3 months. There was no significant difference in passive external rotation or passive abduction between groups (see Table 22). Takagishi et al. 70 reported external rotation at 5 weeks. There was no significant difference between groups (see Table 22).
Sodium hyaluronate with steroid and physiotherapy versus steroid with physiotherapy
One study, by Rovetta et al. ,69 reported internal rotation, external rotation and abduction at 6 months only. There was a significantly greater improvement in internal rotation with sodium hyaluronate combined with steroid and physiotherapy than with steroid combined with physiotherapy (MD 23.70°, 95% CI 13.44° to 33.96°). There was no significant difference in mean external rotation or mean abduction between groups (see Table 22).
Quality of life
None of the three studies that evaluated sodium hyaluronate assessed quality of life.
Adverse events
None of the three studies evaluating sodium hyaluronate reported whether or not participants experienced any adverse events.
Summary
The three included studies investigating sodium hyaluronate used variable doses and in one study sodium hyaluronate was combined with steroid injection and physiotherapy. Because of variations in outcomes reported and comparators used it was not possible to pool data in a meta-analysis. For most outcome measures data were available from a single study only. The studies did not report participants’ stage of frozen shoulder or previous treatments received.
All of the studies had a potentially high risk of bias; none reported the method of randomisation and therefore it was unclear whether they were truly randomised. Because it was unclear whether all studies had enough participants to detect a statistically significant treatment effect, it should be kept in mind that where studies did not report a statistically significant benefit (i.e. the CI crossed the line of no difference) this should not be interpreted as evidence of no difference between the groups.
One study reported a medium-term benefit in pain outcome when sodium hyaluronate was added to steroid and physiotherapy. The same study also reported a significant benefit at medium-term follow-up for internal rotation but not for external rotation or abduction. Another study reported a benefit for function and disability with sodium hyaluronate compared with home exercise but not with physiotherapy or steroid. The same study reported significantly better short-term passive external rotation and abduction with sodium hyaluronate than with home exercise and physiotherapy but not steroid injection. A second study reported no evidence of a benefit for external rotation with sodium hyaluronate compared with steroid.
In conclusion, a small number of diverse studies of sodium hyaluronate were identified, all of which may have had a high risk of bias. There was insufficient evidence to make conclusions with any certainty about the effectiveness of sodium hyaluronate for primary frozen shoulder and in what situations it is likely to be effective.
Physical therapy
Study characteristics
Eleven RCTs and one controlled trial investigated various types of physical therapy in the treatment of primary frozen shoulder. A summary of the study characteristics is reported in Table 23, with further details available in Appendix 6. Eleven were full papers and one71 was available in abstract form only. One was a Chinese-language paper. 72 The studies were published in the Netherlands,40,73 Turkey,51,66,74 Hong Kong,75 Singapore,76 Thailand,77 Greece,16 Taiwan,78 China72 and the UK71 between 1994 and 2009. There were nine two-armed trials,16,40,51,72–74,76–78 two three-armed trials71,75 and one four-armed trial. 66
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Calis 200666 Four-arm RCT |
n = 90 (95 shoulders) Sodium hyaluronate injection: 24 (27) Steroid injection: 25 (26); PT: 21 (22); No intervention (home exercise only): 20 (20) |
Sodium hyaluronate: 30 mg, one injection once weekly for 2 weeks Steroid: triamcinolone acetonide, 40 mg, one injection PT: 10 daily sessions: heat pack applied for 20 minutes; ultrasonic therapy for 5 minutes (1.5 W/cm2 intensity); TENS for 20 minutes; and stretching exercises |
History of pain for at least 1 month; limited active and passive shoulder movement; decreased passive ROM of ≥ 20%, in at least three movements, according to the American Medical Association guide for the evaluation of permanent impairment |
|
Diercks 200473 Two-arm controlled trial |
n = 77 PT: 32 Supervised neglect: 45 |
PT: active PT; duration and frequency not reported Supervised neglect: education and home exercise within painless range |
Idiopathic frozen shoulder using Lundberg criteria: > 50% motion restriction of the glenohumeral joint in all directions for at least 3 months |
|
Dogru 200851 Two-arm RCT |
n = 50 Ultrasound + PT: 25 Sham + PT: 25 |
Ultrasound + PT: active PT; 10 × 50-minute sessions over 2 weeks of superficial heat (heat packs) (20 minutes), ultrasound (10 minutes) and exercise programme (20 minutes) Sham ultrasound + PT: same as above except that the ultrasound machine was not switched to ‘on’ |
Shoulder pain of at least 3 months’ duration; at least 25% loss of shoulder motion in all planes; pain on motion of at least 40 mm on VAS |
|
Dundar 200974 Two-arm RCT |
n = 57 Continuous passive motion: 29 PT: 28 |
Continuous passive motion: 20 × 1-hour sessions over 4 weeks using an external motorised device Conventional PT: active PT, 20 × 1-hour sessions over 4 weeks |
Frozen shoulder patients with gradually increasing shoulder pain and stiffness (painful phase and stiff phase) |
|
Leung 200875 Three-arm RCT |
n = 30 SWD + stretching: 10 Heat pack + stretching: 10 No intervention: 10 |
SWD + stretching: three 20-minute sessions per week for 4 weeks, machine frequency of 27.12 MHz, followed by stretching exercises Heat pack + stretching: superficial heat (63°C) with electrical heat pack; stretching exercises as above No intervention: home exercise only |
Idiopathic frozen shoulder with shoulder pain and limited shoulder movement for at least 8 weeks |
|
Maricar 199976 Two-arm RCT |
n = 54 PT + exercise: 16a Exercise only: 16a |
PT + exercise: active PT, eight weekly sessions and from week 5 a 15-minute exercise circuit of nine exercises Exercise only: 15-minute exercise circuit of nine exercises |
Major complaint of limitation in shoulder range of motion with secondary complaint of pain; able to place arms behind head and back reaching vertebral column; 90-degree abduction (late stage 2 to stage 3) |
|
Pajareya 200477 Two-arm RCT |
n = 122 PT: 61 No intervention: 61 |
PT: SWD (20 minutes) + active PT three times a week for 3 weeks No intervention: information only |
Shoulder pain and limitation of a passive range of shoulder motion in all directions that interfered with activities of daily living |
|
Stergioulas 200816 Two-arm RCT |
n = 74 Laser therapy: 37 Placebo laser: 37 |
Laser therapy: 12 sessions over 8 weeks: two per week in the first 4 weeks, then one per week Placebo laser: regimen as above |
Painful and limited passive glenohumeral mobility; more restricted lateral rotation (< 8%) relative to abduction and medial rotation |
|
Vermeulen 200640 Two-arm RCT |
n = 100 HGMT: 49 LGMT: 51 |
HGMT: 30-minute session twice weekly for a maximum of 12 weeks consisting of 5-minute assessment of ROM in order to apply the mobilisation technique into the stiffness zone. Mobilisation techniques were applied with intensities according to Maitland grades III and IV LGMT: 30-minute session twice weekly for a maximum of 12 weeks consisting of 5-minute assessment of ROM in order to apply the mobilisation technique within the pain-free zone. Mobilisation techniques were performed according to Maitland grades I and II |
≥ 50% loss of passive movement of shoulder joint relative to non-affected side, in one or more of three movement directions (abduction in frontal plane, forward flexion or external rotation); duration of complaints ≥ 3 months |
|
Wies 200371 Three-arm RCT |
n = 30 Osteopathy: 10 PT: 10 Control: 10 |
Osteopathy: the Niel-Asher technique, consisting of a progression of deep tissue manipulation, for 9 weeks PT: manual therapy and therapeutic exercise for 9 weeks Control: breathing exercises + massage + ROM exercises for 9 weeks |
Patients with primary frozen shoulder |
|
Yang 200778 Two-arm RCT |
n = 30 (28 started) ERM + MRM: 14 MWM + MRM: 14 |
ERM + MRM: 30 minutes twice per week for 3 weeks each MWM + MRM: 30 minutes twice per week for 3 weeks each |
Painful stiff shoulder for at least 3 months; ROM losses of at least 25% compared with the non-involved shoulder in at least two of glenohumeral flexion, abduction or medial or lateral rotation |
|
Yan 200572 Two-arm RCT |
n = 54 Dumb-bell gymnastics: 26 Barehanded exercises: 28 |
Dumb-bell gymnastics: 5–10 minutes, two to three times a day for 3 months using dumb-bells weighing 2–5 kg Barehanded exercises: performed for 3 months |
‘Standard shoulder periarthritis diagnostic criteria’ |
A variety of physical therapy regimens were used, most of which were physiotherapy modalities. These included various combinations of exercises (up to and beyond the pain threshold), stretching, manipulation of the glenohumeral joint, ultrasound, superficial heat, SWD, laser therapy, Codman’s exercises, wall-climbing exercises, continuous passive motion, manual therapy, dumb-bell gymnastics and massage. For the purposes of the synthesis, the interventions were grouped based on whether or not at least one component involved mobilisation (of any type) under the supervision of a therapist. Therapies that involved some form of mobilisation (with or without other physical therapies) are described as active therapies. Studies using interventions such as laser therapy or TENS without any supervised mobilisation or exercise were classified as therapy without mobilisation.
Seven compared at least two forms of physical therapy. 40,71,72,74–76,78 One of the studies had what was described as a ‘supervised neglect’ intervention. 73 This study by Diercks and Stevens was the only study identified that explicitly used a supervised neglect intervention; therefore, this study was included although it was not a RCT. 73 Undertaken in the Netherlands, a physiotherapy intervention of unspecified duration was used that involved a standardised protocol, carried out by a therapist, of active exercises up to and beyond the pain threshold. The supervised neglect comparator involved providing patients with an explanation of the natural course of the disease and instructions not to exercise in excess of their pain threshold, to undertake pendulum exercises and active exercises within the painless range and to resume all activities that were tolerated. In the Calis et al. study, physiotherapy was compared with sodium hyaluronate and steroid injection;66 and in the Pajareya et al. study, physiotherapy was compared with no intervention. 77
A single study did not have an arm with an active mobilisation component. This study by Stergioulas16 compared laser therapy with placebo laser (with home exercise). The remaining studies compared multiple active physical therapies. One compared ultrasound and physiotherapy with sham ultrasound and physiotherapy;51 one compared 1-hour sessions of continuous passive motion with physiotherapy;74 one compared osteopathy with physiotherapy and with breathing exercises, massage and range of movement exercises;71 one compared end-range mobilisation (ERM) and mid-range mobilisation (MRM) with mobilisation with movement (MWM) and MRM;78 one compared passive joint mobilisations and exercises with exercises only;76 one compared SWD plus stretching with heat pack plus stretching;75 one compared dumb-bell gymnastics with barehanded exercises;72 and one compared high-grade mobilisation techniques (HGMT) with low-grade mobilisation techniques (LGMT) (both followed by Codman pendular exercises). 40 All of the physiotherapy arms included an active mobilisation component. The intervention duration ranged from 2 weeks to 3 months.
Six studies reported that home exercise was used as part of the intervention, although details were limited in some studies. Where specified the home exercise routines included daily Codman and stretching exercises,66 Codman, stretching and active range of movement exercises,51 daily passive range of movement and pendulum exercises,74 pendulum and pain-free exercises16 and daily stretches. 75 Home exercise was discouraged in one study. 78 Analgesic use also differed between studies where reported. NSAIDs or other analgesics were prescribed when necessary in one study,73 paracetamol was taken when necessary in another,66 paracetamol was allowed to a maximum of 1000 mg daily in another,51 400 mg of ibuprofen was given daily for 3 weeks in one study77 and prescribed and non-prescribed pain medication was allowed in one study. 40 No NSAIDs or other analgesics were allowed in one RCT, with a week washout period before the study commenced. 74
The inclusion criteria varied between the studies. All included patients with some limitation of movement, although the extent of this varied where reported. Primary frozen shoulder was diagnosed through range of movement, biochemical analyses, radiography and clinical diagnosis where reported. Four studies did not report how frozen shoulder was diagnosed. 40,71,77,78 Three included participants with diabetes: 37% in the study by Dogru et al. ,51 16% in the study by Vermeulen et al. 40 and 33% in the study by Pajareya et al. 77 Separate data were not presented for this subgroup. The mean duration of frozen shoulder at baseline ranged from 3 months to 6.8 years. The stage of frozen shoulder included was reported by few studies: stage 1 or 2 in the study by Dundar et al. ,74 ‘stiff stage’ in the studies by Yan72 and Leung and Cheing75 and late stage 2 or 3 in the study by Maricar and Chok. 76 Outcome was not reported separately by stage of frozen shoulder. Only one study, Vermeulen et al. ,40 reported on whether participants had received any previous treatment for frozen shoulder. In this study 81% of participants had previously received physical therapy, 61% steroid injections and 6% surgery. The proportion of female participants ranged from 20% to 86% and the mean age of participants ranged from 50 to 62.5 years.
Quality assessment
The quality of the studies was generally poor. Only one was of satisfactory quality, reporting the number randomised and both an appropriate method of randomisation and adequate allocation concealment. 78 This study by Yang et al. also met most of the remaining criteria including comparability at baseline, blinding of outcome assessors and intention-to-treat analysis; however, the study may not be adequately powered to detect significant differences between groups and there was a large number of dropouts (23%). The main limitation of this study was the study design for the purposes of our review; only limited data were suitable for inclusion in our analysis as different sequences of the three same mobilisations were compared at end of treatment (see Appendix 6). 78
Leung and Cheing,75 Stergioulas16 and Vermeulen et al. 40 reported an appropriate method of randomisation; Leung and Cheing75 and Vermeulen et al. 40 did not report whether an adequate method of allocation concealment was used; and Stergioulas16 reported that allocation was performed using opaque envelopes but it was unclear whether the envelopes were numbered sequentially. Although these three studies fulfilled other criteria, such as comparability at baseline and blinding of outcome assessors, there was still a possible risk of bias. Leung and Cheing75 reported that there was no loss to follow-up and loss to follow-up was 15% in the Stergioulas study16 (the majority in the laser group) and 4% in the Vermeulen study40 (with equal numbers from each group). Vermeulen et al. 40 also used intention-to-treat analysis and was adequately powered for one outcome.
Calis et al. ,66 Dogru et al. ,51 Dundar et al. ,74 Pajareya et al. 77 and Yan72 reported the number of participants randomised. It was unclear whether the method of assignment was truly random in any of these studies and allocation concealment was not reported; therefore, they potentially have high risk of bias. These studies also fulfilled few of the remaining criteria. Dogru et al. 51 and Calis et al. 66 reported blinding of outcome assessment. Dropouts were relatively high in the Pajareya study77 (12%, the majority in the control group) and the reporting of satisfaction results was also unclear. There were no or very few dropouts in the Calis et al. ,66 Dogru et al. ,51 Yan72 and Dundar et al. 74 studies. Calis et al. 66 also used intention-to-treat analysis.
The number of participants randomised was inconsistently reported in the study by Maricar and Chok. 76 Neither Maricar and Chok76 or Wies et al. 71 reported the method of randomisation or allocation concealment, so were at high risk of bias. Few of the remaining criteria were met. Dropouts were high in the study by Maricar and Chok (41%)76 and not reported by Wies et al. 71
Allocation to groups was not randomised in the study by Diercks and Stevens73 and met none of the quality criteria (including blinding of outcome assessment) except comparability at baseline (there were no significant differences in age, sex or duration of disease). This study was quasi-experimental with a successive cohort as the physical therapy group. This study was therefore at a high risk of bias. The paper reported that there was no loss to follow-up.
Pain
Six studies assessed pain (Table 24). The study by Pajareya et al. 77 reported analgesic use at 3 weeks only and was therefore excluded from the synthesis. Calis et al. 66 reported data in graphical form only; consequently, the SMD could not be calculated and this study could not be included in a meta-analysis. Calis et al. 66 did, however, report that there was significant improvement in pain severity at 3 months within both the physical therapy group and the no intervention group (p < 0.001).
| Study | Pain outcome assessed | Scale used | Data reported and time of follow-up |
|---|---|---|---|
| Calis 200666 | Pain severity | VAS | Data reported graphically only |
| Dogru 200851 | Pain overall | SPADI 5-item pain subscale | Final mean values at 2 weeks and 3 months |
| Dundar 200974 | Pain overall | SPADI 5-item pain subscale | Final mean values at 4 weeks and 12 weeks |
| Pajareya 200477 | Analgesic use | No. of tablets | Median at 3 weeks |
| Stergioulas 200816 | Pain overall | VAS 0–100 mm | Final mean values at 4, 8 and 12 weeks |
| Vermeulen 200640 | Pain at night | VAS 0–100 mm | Mean change at 3, 6 and 12 months (baseline median and IQR reported only) |
The remaining four studies each evaluated different physical therapies using different comparators; therefore, it was not appropriate to pool the studies in a meta-analysis. 16,40,51,74
Table 25 provides the SMDs and 95% CIs. The outcome data for individual groups in the included trials are available in Appendix 7.
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Physical therapy vs physical therapy | |||
| Dundar 200974 | –0.31 (–0.83 to 0.21)a | ||
| Dogru 200851 | 0.28 (–0.28 to 0.85)b | ||
| Vermeulen 200640 | –0.15 (–0.55 to 0.24)b | –0.27 (–0.67 to 0.12)c | –0.33 (–0.73 to 0.06)d |
| Physical therapy without mobilisation vs control | |||
| Stergioulas 200816 | –1.71 (–2.29 to –1.12)e,f | –1.89 (–2.49 to –1.29)f,g | |
Physical therapy versus physical therapy
There was no significant difference in pain between continuous passive motion and conventional physiotherapy at short-term follow-up (12 weeks) (Table 25). 74 There was no significant benefit from adding ultrasound to a 20-minute supervised exercise programme of Codman’s exercises, wall-climbing and joint stretching for patient’s tolerance at short-term follow-up (3 months) (Table 25). 51 There was no significant difference in pain between HGMT and LGMT at any time point (Table 25). 40
Function and disability
Ten of the twelve studies that assessed physical therapies reported function and disability outcomes (Table 26). SPADI score16,51,71,74,77 and Constant score66,73,74 were common to several studies; however, the data could not be pooled in meta-analysis as the comparisons varied substantially. Short- and medium-term data were available from these studies. Final value data were available from six studies. 16,51,66,73–75 Pajareya et al. 77 included only baseline data, so this study was not included in the analysis. Change from baseline data was available from the study by Vermeulen et al. 40 Yang et al. 78 reported mean percentage of change only. Table 27 provides between-group differences and 95% CIs. Because of differences between interventions and comparators it was not appropriate to pool these studies. Outcome data for individual studies are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Calis 200666 | Constant score | Final value data at 15 days and 3 months |
| Diercks 200473 | Constant score (≥ 80 points indicates a normal functioning shoulder) | Final value data at 3, 6, 9, 12, 15, 18, 21 and 24 months reported |
| Dogru 200851 |
SPADI total score (0–100)a SPADI 8-item disability subscale (0–100)a |
Final value data at 2 weeks and 3 monthsb,c |
| Dundar 200974 |
SPADI total score (0–100)a Constant score |
Final value data at 4 weeks and 12 weeks |
| Leung 200875 | ASES assessment (score out of 100) | Final value data reported at 2, 4 and 8 weeks |
| Pajareya 200477 |
SPADI total score (0–100)a Global rating of pain and disability |
3-week data only (not included) Baseline data only |
| Stergioulas 200816 | SPADI total score (0–100), Croft score (out of 22), DASH (%) and HAQ (two or three items scored 0–3) | Final value data reported at 4, 8 and 16 weeks |
| Vermeulen 200640 |
Shoulder rating questionnaire (Dutch-language version) (17–100) SDQ (Dutch-language version) (0–100)a 5-point Likert scale (1 = much worse; 5 = much better) |
Change from baseline at 3, 6 and 12 months Percentage of participants reporting ‘much worse or no change’ and ‘better or much better’ at 3, 6 and 12 months |
| Wies 200371 | SPADI (0–100)a | Change from baseline at 9 weeks |
| Yang 200778 | FLEX-SF (1–50) | Mean percentage of change from baseline at 6 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Physical therapy vs control | |||
| Constant score | |||
| Calis 200666 | 14.90 (8.32 to 21.48)a,b | ||
| Diercks 200473 | –16.37 (–21.46 to –11.28)a,b | –15.40 (–20.50 to –10.30)b,c | |
| Leung 200875 |
SWD vs home exercise: 17.50 (1.76 to 33.24)b,g HP vs home exercise: 4.00 (–10.38 to 18.38)g |
||
| Physical therapy vs physical therapy | |||
| SPADI total score | |||
| Dogru 200851 | 4.50 (–6.36 to 15.36)a | ||
| Dundar 200974 | 0.17 (–0.73 to 1.07)a | ||
| Wies 200371 |
PT vs osteopathy: –19.90 (–40.11 to 0.31)h PT vs control: –4.00 (–22.47 to 14.47)h Osteopathy vs control: 15.90 (–2.04 to 33.84)h |
||
| SPADI 8-item subscale | |||
| Dogru 200851 | 3.10 (–8.44 to 14.64)a | ||
| Constant score | |||
| Dundar 200974 | –3.37 (–8.28 to 1.54)a | ||
| FLEX-SF | |||
| Yang 200778 |
ERM + MRM: 19.9% (SD 8.1%)i MWM + MRM: 17.25% (SD 12.2%)i |
||
| ASES | |||
| Leung 200875 | HP vs SWD: 13.50 (–2.16 to 29.16)g | ||
| Shoulder rating questionnaire | |||
| Vermeulen 200640 | 2.40 (–4.00 to 8.80)a | 4.50 (–2.39 to 11.39)c | 6.60 (–0.63 to 13.83)d |
| SDQ | |||
| Vermeulen 200640 | –5.20 (–14.95 to 4.55)a | –5.70 (–17.42 to 6.02)c | –11.20 (–22.60 to 0.20)d |
| Physical therapy without mobilisation vs control | |||
| SPADI | |||
| Stergioulas 200816 | –14.11 (–19.50 to –8.72)b,g | –13.83 (–18.88 to –8.78)b,j | |
| Croft score | |||
| Stergioulas 200816 | –4.34 (–6.34 to –2.34)b,g | –7.13 (–9.18 to –5.08)b,j | |
| DASH score | |||
| Stergioulas 200816 | –9.24 (–14.35 to –4.13)b,g | –10.51 (–15.37 to –5.65)b,j | |
| HAQ score | |||
| Stergioulas 200816 | –0.75 (–1.09 to –0.41)b,g | –0.31 (–0.64 to 0.02)j | |
Physical therapy versus control
Diercks and Stevens73 reported a statistically significant improvement in Constant score with supervised neglect compared with physiotherapy at 3 (MD –16.37, 95% CI –21.46 to –11.28), 6 (MD –15.40 95% CI –20.50 to –10.30), 12 (MD –17.74, 95% CI –22.75 to –12.73), 18 (MD –16.13, 95% CI –22.20 to –10.06) and 24 (MD 9.22, 95% CI –15.69 to –2.75) months’ follow-up. It is worth noting that there were several factors that could have biased this result. The physiotherapy group were prescribed exercises up to and beyond the pain threshold whereas the supervised neglect group were advised to exercise within the painless range. Also, as a successive cohort was recruited after 2 years (the physiotherapy group), treatment protocols may have changed during the time that elapsed (although the authors did report that a standardised treatment protocol was used). Although the baseline characteristics reported were similar between groups it is possible that there may have been unmeasured differences between groups.
Leung and Cheing75 found significant improvement in American Shoulder and Elbow Surgeons (ASES) score with SWD plus stretching compared with home exercise (MD 17.50, 95% CI 1.76 to 33.24). There was no significant difference between heat pack plus stretching and home exercise (Table 27).
Calis et al. 66 reported that Constant score was significantly improved with physiotherapy plus home exercise compared with home exercise alone (MD 14.90, 95% CI 8.32 to 21.48).
Physical therapy versus physical therapy
The study by Dogru et al. ,51 which compared ultrasound and physiotherapy with sham ultrasound and physiotherapy, found no significant difference between treatment groups in SPADI total score or the SPADI 8-item disability subscale at 3 months (Table 27).
The intention in the study by Yang et al. 78 was to compare two different sequences of three types of mobilisation (each type of mobilisation was delivered over a 3-week period). The two groups received the three types of mobilisation but in a different order over 12 weeks (see Appendix 6). Because at the end of the 12 weeks both groups had received the same treatments, only 6-week data were extracted for ERM and MRM versus MWM and MRM. The change in FLEX-SF score was similar at 6 weeks for ERM and MRM (19.9%, SD 8.1%) and MWM and MRM (17.25%, SD12.2%)
The study by Dundar et al. ,74 which compared continuous passive motion with physiotherapy, found no significant difference between treatment groups in SPADI total score or the Constant score (Table 27).
Leung and Cheing75 found no significant difference between SWD and heat pack groups in terms of ASES score (Table 27).
In the study by Vermeulen et al. 40 there was no significant difference between HGMT and LGMT in the shoulder rating questionnaire score (Table 27). There was also no significant improvement in SDQ score with HGMT compared with LGMT at any time point (Table 27); however, it should be noted that the 95% CI marginally crossed the line of no effect at 12 months (MD –11.20, 95% CI –22.60 to 0.20). There was no significant difference in the proportions of patients who reported ‘(much) worse or no change’ at 3 months (13% vs 12%), 6 months (13% vs 10%) or 12 months (9% vs 18%) and ‘better or much better’ at 3 months (87% vs 88%) 6 months (87% vs 90%) or 12 months (91% vs 82%).
There was no significant difference in SPADI score with physiotherapy compared with osteopathy in the study by Wies et al. ;71 however, it should be noted that the 95% CI marginally crossed the line of no effect (MD –19.90, 95% CI –40.11 to 0.31). There was no significant difference between either the physiotherapy or osteopathy group and the control group (breathing exercises, range of movement exercises and massage) (Table 27).
Physical therapy without mobilisation versus control
Stergioulas16 reported several measures of function and disability. At 8 weeks laser treatment was associated with significantly improved SPADI score (MD –14.11, 95% CI –19.50 to –8.72), Croft score (MD –4.34, 95% CI –6.34 to –2.34) Disabilities of the Arm, Shoulder and Hand (DASH) score (MD –9.24, 95% CI –14.35 to –4.13) and Health Assessment Questionnaire (HAQ) score (MD –0.75, 95% CI –1.09 to –0.41) compared with placebo laser. At 16 weeks laser treatment was associated with significantly improved SPADI score (MD –13.83, 95% CI –18.88 to –8.78), Croft score (MD –7.13, 95% CI –9.18 to –5.08) and DASH score (MD –10.51, 95% CI –15.37 to –5.65). There was no significant difference in HAQ score between groups at 16 weeks (Table 27).
Range of movement
Eleven of the included studies that investigated physical therapy reported a range of movement measure of interest (Table 28). All of the studies except two reported short- or medium-term follow-up. One presented data at 24 months73 and one at 12 months. 40 Six studies reported final value data and one77 presented data that were not included in the analysis as follow-up was l < 4 weeks. One study reported change from baseline data. 40 Maricar and Chok76 reported only graphs and p-values, presented narratively. Because of the differences between the interventions and comparators meta-analyses were not performed. Table 29 provides between-group differences and 95% CIs. Outcome data for individual studies are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Calis 200666 | Passive abduction, passive external rotation (°) | Final value data at 15 days and 3 months |
| Diercks 200473 | External rotation, internal rotation (ROM part of Constant score, number of points out of 10 correspond to extent of movement) | Final value data at 24 monthsa |
| Dogru 200851 | External rotation, internal rotation, passive abduction (°) | Final value data at 2 weeks and 3 monthsb |
| Dundar 200974 | Passive external rotation, passive internal rotation, passive abduction (°) | Final value data at 4 weeks and 12 weeks |
| Leung 200875 | External rotation (arm by side) (°), external rotation (arm at 90 (°)), hand behind back (cm) | Final value data at 2, 4 and 8 weeks |
| Maricar 199976 | External rotation (°), internal rotation (°), hand behind back (cm) | Graphs and p-values only reported at 3, 5, 7 and 8 weeks |
| Pajareya 200477 | External rotation, internal rotation | 3-week data only (not included in analysis) |
| Stergioulas 200816 | Active abduction, external rotation (in neutral abduction) (°) | Final value data at 4, 8 and 16 weeks |
| Vermeulen 200640 | Active abduction, active external rotation, passive abduction, passive external rotation (°) | Change from baseline at 3, 6 and 12 months |
| Wies 200371 | Active abduction (°) | Change from baseline at 9 weeks |
| Yang 200778 | Humeral lateral rotation (external rotation), humeral medial rotation (internal rotation) (°) | Mean percentage of change from baseline at 6 weeks |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Physical therapy vs control | |||
| External rotation | |||
| Diercks 200473 |
PT: median = 8 pointsa Supervised neglect: median = 10 |
||
| Leung 200875 |
SWD vs home exercise, arm by side: 21.00° (4.95° to 37.05°)b,c SWD vs home exercise, arm at 90°: 11.60° (–6.58° to 29.78°)b HP vs home exercise, arm by side: –8.50° (–28.19° to 11.19°)b HP vs home exercise, arm at 90°: –18.50° (–41.15° to 4.15°)b |
||
| Internal rotation | |||
| Diercks 200473 |
PT: median = 8 pointsa Supervised neglect: median = 8 |
||
| Hand behind back | |||
| Leung 200875 |
SWD + stretching vs home exercise: –7.00 cm (–13.14 cm to –0.86 cm)b,c HP + stretching vs home exercise: 5.30 cm (–0.93 cm to 11.53 cm)b |
||
| Passive external rotation | |||
| Calis 200666 | 18.80° (13.11° to 24.49°)c,d | ||
| Passive abduction | |||
| Calis 200666 | 24.90° (14.59° to 35.21°)c,d | ||
| Physical therapy vs physical therapy | |||
| External rotation | |||
| Dogru 200851 | –9.70° (–19.51° to 0.11°)d | ||
| Dundar 200974 | 0.76° (–7.40° to 8.92°)d | ||
| Yang 200778 |
ERM + MRM: 36.4% (SD 24.3%)e MWM + MRM: 34.2% (SD 13.3%)e |
||
| Leung 200875 |
HP vs SWD, arm by side: 29.50° (14.28° to 44.72°)b,c HP vs SWD, 90 degree abduction: 30.10° (13.51° to 46.69°)b,c |
||
| Internal rotation | |||
| Dogru 200851 | –3.50° (–11.67° to 4.67°)d | ||
| Yang 200778 |
ERM + MRM: 20.5% (SD 24.4%)e MWM + MRM: 45.6% (SD 38.5%)e |
||
| Passive internal rotation | |||
| Dundar 200974 | 0.92° (–8.34° to 10.18°)d | ||
| Passive abduction | |||
| Dogru 200851 | –0.20° (–16.06° to 15.66°)d | ||
| Dundar 200974 | –4.42° (–11.83° to 2.99°)d | ||
| Vermeulen 200440 | 13.10° (1.62° to 24.58°)c,d | 11.00° (–1.59° to 23.59°)f | 12.50° (1.03° to 23.97°)b,g |
| Active abduction | |||
| Vermeulen, 200440 | 10.00° (–2.16° to 22.16°)d | 8.90° (–4.67° to 22.47°)f | 12.60° (0.06° to 25.14°)c,g |
| Wies 200371 |
Physical therapy vs osteopathy: –6.40° (–32.77° to 19.97°)h |
||
| Active external rotation | |||
| Vermeulen 200440 | 2.30° (–2.81° to 7.41°)d | 2.70° (–2.36° to 7.76°)f | 4.90° (–0.67° to 10.47°)g |
| Passive external rotation | |||
| Vermeulen 200440 | 1.40° (–3.48° to 6.28°)d | 4.10° (–0.97° to 9.17°)f | 6.50° (0.27° to 12.73°)c,g |
| Hand behind back | |||
| Leung 200875 | Heat pack vs SWD: –12.30 cm (–18.79 cm to –5.81 cm)b,c | ||
| Physical therapy without mobilisation vs control | |||
| External rotation | |||
| Stergioulas 200816 | 2.05° (–2.75° to 6.85°)b | 4.19° (–0.74° to 9.12°)i | |
| Active abduction | |||
| Stergioulas 200816 | 5.47° (–0.40° to 11.34°)b | 5.20° (–1.60° to 12.00°)i | |
Physical therapy versus control
In the study by Calis et al. 66 there was a significant improvement in passive abduction (MD 24.90°, 95% CI 14.59° to 35.21°) and passive external rotation (MD 18.80°, 95% CI 13.11° to 24.49°) in the physiotherapy group compared with home exercise alone.
In the study by Leung and Cheing75 there was a significant improvement in external rotation with arm by side (MD 21.00°, 95% CI 4.95° to 37.05°) with SWD plus stretching compared with home exercise. Hand behind back was also significantly greater in the SWD plus stretching group compared with the home exercise group (MD –7.00 cm, 95% CI –13.14 cm to –0.86 cm). There was no significant difference between either SWD or heat pack (both with stretching exercises) and home exercise in external rotation with arm at 90°, and no significant difference between heat pack and home exercise in external rotation with arm by side or hand behind back (Table 29).
The study by Diercks and Stevens73 reported that at 24 months the physiotherapy group had a median of 8 points for the Constant score for external rotation, compared with 10 points in the supervised neglect group, although no measure of variance was provided. This corresponds to a greater range of motion in the supervised neglect group in this plane. For internal rotation, both groups reported a median of 8 points, corresponding to the position dorsum of the 12th dorsal vertebra.
Physical therapy versus physical therapy
Dogru et al. 51 found no significant difference between ultrasound and sham ultrasound in external rotation, internal rotation and abduction (Table 29).
There was also no significant difference in the study by Dundar et al. 74 between continuous passive motion and physical therapy in passive external rotation, passive internal rotation and passive abduction (Table 29).
The study by Wies et al. 71 showed a significantly greater improvement in active abduction in the osteopathy group (MD 45.20°, 95% CI 16.87° to 73.53°) and physiotherapy groups (MD 38.80°, 95% CI 5.76° to 71.84°) compared with control (breathing exercises, massage and exercise). There was no significant difference between the physiotherapy and osteopathy groups (Table 29).
Yang et al. 78 reported that the mean percentage of change in external rotation was similar between ERM plus MRM (mean 36.4%, SD 24.3%) and MWM plus MRM (mean 34.2%, SD 13.3%). However, there was a greater change in internal rotation with MWM + MRM (45.6%, SD 38.5%) compared with ERM and MRM (20.5%, SD 24.4%) (Table 29).
Leung and Cheing75 found that there was a significant improvement in external rotation both with arm by side (MD 29.50°, 95% CI 14.28° to 44.72°) and with arm at 90° abduction (MD 30.10°, 95% CI 13.51° to 46.69°) with SWD compared with heat pack. There was also a significant improvement in hand behind back with SWD compared with heat pack (MD –12.30 cm, 95% CI –18.79 cm to –5.81 cm) (Table 29).
Maricar and Chok76 compared passive joint mobilisations and exercises with exercises only and found no significant difference in external rotation or internal rotation between treatment groups at weeks five, seven or eight.
The study by Vermeulen et al. 40 showed a significant improvement in active abduction at 12 months with HGMT compared with LGMT (MD 12.60°, 95% CI 0.06° to 25.14°). There was also a significant improvement with HGMT in passive abduction at 3 months (MD 13.10°, 95% CI 1.62° to 24.58°) and 12 months (MD 12.50°, 95% CI 1.03° to 23.97°), and in passive external rotation at 12 months (MD 6.50°, 95% CI 0.27° to 12.73°). There was no significant difference between groups in active external rotation at any of the time points, or in passive external rotation, active abduction or passive abduction at the remaining time points (Table 29).
Quality of life
Two studies, one by Dogru et al. 51 and the other by Vermeulen et al. ,40 reported quality of life using the physical and mental components of the SF-36 (Table 30). Data at 3 months’ follow-up were available (Table 31). As the interventions varied substantially these studies were not pooled. Outcome data for the individual groups are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Dogru 200851 |
SF-36 physical component SF-36 mental component |
Final value data at 2 weeks and 3 monthsa (3-month data included in analysis) |
| Vermeulen 200440 |
SF-36 physical component SF-36 mental component |
Change from baseline at 3, 6 and 12 months |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Physical therapy vs physical therapy | |||
| SF-36 physical component | |||
| Dogru 200851 | –0.40 (–5.22 to 4.42)a | ||
| Vermeulen 200440 | 0.60 (–6.03 to 7.23)a | 2.10 (–5.08 to 9.28)b | 0.40 (–7.78 to 8.58)c |
| SF-36 mental component | |||
| Dogru 200851 | 1.00 (–5.19 to 7.19)a | ||
| Vermeulen 200440 | 4.10 (–3.91 to 12.11)a | 0.30 (–7.95 to 8.55)b | –2.50 (–8.36 to 3.36)c |
Physical therapy versus physical therapy
There was no significant difference between ultrasound and sham ultrasound groups in the SF-36 physical or mental components at 3 months’ follow-up in the study by Dogru et al. (Table 31). 51
There was no significant difference between HGMT and LGMT groups in the SF-36 physical or mental components at any time point in the study by Vermeulen et al. (Table 31). 40
Other
Two studies reported other outcomes of interest (Table 32). Pajareya et al. 77 reported satisfaction with treatment and percentage of patients reporting treatment success. Yan72 also reported the percentage of patients describing their rate of improvement as bad, average, good and excellent at 3 months. These data are summarised in Table 33 and further information for the individual studies is available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Pajareya 200477 | Satisfaction (number of patients reporting that they were ‘very satisfied’, ‘moderately satisfied’, ‘unsatisfied’ or ‘very unsatisfied’) | Data at 3 weeks |
| Treatment success (% success) | Treatment success at 3, 6, 12 and 24 weeks | |
| Yan 200572 | Rate of improvement (%) | Rate of improvement (excellent, good, average and bad) at 3 months |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Physical therapy vs control | |||
| Treatment success | |||
| Pajareya 200477 |
% of patients Physical therapy: 61.4%,a control: 60.8% Physical therapy: 76.8%,b control: 60.8% |
% of patientsc Physical therapy: 80.4%, control: 82.4% |
|
| Satisfaction | |||
| Pajareya 200477 |
No. of patientsd ‘very satisfied’: physical therapy 5, control 1 ‘moderately satisfied’: physical therapy 7, control 1 ‘unsatisfied’: physical therapy 24, control 13 ‘very unsatisfied’: physical therapy 23, control 45 |
||
| Physical therapy vs physical therapy | |||
| Rate of improvement | |||
| Yan 200572 |
No. of patientsb Bad: dumb-bell exercises 0, barehanded 7 Average: dumb-bell exercises 0, barehanded 16 Good: dumb-bell exercises 2, barehanded 5 Excellent: dumb-bell exercises 24, barehanded 0 |
||
Physical therapy versus control
In the study by Pajareya et al. 77 similar proportions of each group reported that they had successful treatment at 6 weeks (61.4% vs 60.8%). At 12 weeks, 76.8% in the physiotherapy compared with 60.8% in the control group reported successful treatment, and at 24 weeks the proportions were 80.4% in the physiotherapy group and 82.4% in the control group.
In the same study the number of patients ‘very satisfied’ at 3 weeks was five in the physical therapy group versus one in the control group; the numbers ‘moderately satisfied’, ‘unsatisfied’ and ‘very unsatisfied’ were 7 versus 1, 24 versus 13 and 23 versus 45 in the physical therapy group versus the control group respectively. 77
Physical therapy versus physical therapy
Yan72 compared dumb-bell exercises with barehanded exercises and found that a greater number of patients performing dumb-bell exercises than those performing barehanded exercises reported an excellent rate of improvement at 3 months (24 vs 0).
Adverse events
Two studies reported adverse events. 74,77 In the study by Pajareya et al. 77 patients in the physiotherapy group were asked whether they had pain that persisted for > 2 hours after treatment or more disability the next morning. There were 10 episodes of pain in the physiotherapy group (in four patients) that persisted for > 2 hours after treatment. Patients were also asked whether the trial drugs and/or treatment programme upset them in any way and were examined for signs of echymosis or burn during range of movement evaluation. Side effects of NSAIDs were reported but it was unclear which treatment groups these applied to (see Appendix 6). Dundar et al. 74 stated that no side effects were observed during the study. No further information regarding how adverse events were measured or assessed was reported. The study by Stergioulas16 reported that there were no complications. Adverse events did not appear to have been assessed in the remaining nine studies.
Summary
Twelve studies were included that investigated various types of physical therapy, without use of steroid injection. They were very diverse in the physical therapies evaluated, although most evaluated physiotherapy modalities. The comparators were another physical therapy or control. With the exception of one study,16 at least one component of the intervention involved active mobilisation or exercise. Half the studies reported that participants were advised to undertake home exercise.
One study of satisfactory quality was identified and three studies were of reasonable quality but had some risk of bias. The other studies had a potentially high risk of bias. Because of the considerable variability of the interventions investigated in the studies and different outcomes measures used it was not appropriate to pool any studies in a meta-analysis. The studies did not report outcome by stage of frozen shoulder or the presence of diabetes and information was very limited regarding previous treatments received.
Physical therapy versus control
There was evidence from one study (with some risk of bias) that there was a significant short-term benefit for function and disability with SWD in conjunction with stretching compared with home exercise and for some but not all measures of range of movement. A further study with some risk of bias reported significant short- and medium-term benefit with laser therapy (plus home exercise) compared with home exercise alone for pain and function and disability but not range of movement. Another study with a potentially high risk of bias reported significant short-term benefit with physiotherapy (plus home exercise) compared with home exercise alone for function and disability and two ranges of movement. One study reported significant short-, medium- and long-term benefit for function and disability with supervised neglect compared with physiotherapy; however, this study was at high risk of bias.
Comparing physical therapies
The majority of studies comparing two physical therapies reported no significant difference in outcome between therapies; however, with the exception of three studies,40,75,78 these all had a potentially high risk of bias. A single study with some risk of bias found a significant benefit of SWD compared with a heat pack (both with stretching) for three ranges of movement; however, in this same study there was no significant difference in benefit for function and disability. Another study with some risk of bias found a significant improvement in one measure of function and disability and in three ranges of movement with HGMT compared with LGMT, but no benefit for pain. A single satisfactory study found no benefit from providing ERM followed by MRM compared with MWM followed by MRM. Based on single studies with a high risk of bias there was no significant difference between continuous passive motion and conventional physical therapy, between mobilisation with and without ultrasound or between joint mobilisation and exercise. A single study with a high risk of bias reported that daily exercises using dumb-bells were significantly more effective than exercises without dumb-bells. One study available only as an abstract and at high risk of bias reported a significant benefit of physiotherapy and osteopathy compared with breathing and range of movement exercises combined with massage in one measure of range of movement. There was no significant difference between physiotherapy and osteopathy techniques.
In conclusion, the studies identified were diverse and mainly of poor quality. With the exception of physiotherapy including SWD, only single studies were identified of any one physical approach. Based on the best available evidence there may be benefit from SWD and stretching and from HGMT in patients who have already had physiotherapy or a steroid injection. There was insufficient evidence to make conclusions with any certainty about the best mode of physical therapy for primary frozen shoulder.
Acupuncture
Study characteristics
Three RCTs investigated acupuncture in the treatment of primary frozen shoulder. A summary of study characteristics is reported in Table 34. All were full papers of studies conducted in Hong Kong,79 China80 and Taiwan81 between 2006 and 2008. There was one two-armed trial80 and two three-armed trials. 79,81 Sample sizes ranged from 74 to 360.
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Cheing 200879 Three-arm RCT |
n = 74 Electroacupuncture: 25 Interferential electrotherapy: 24 Control: 25 |
Electroacupuncture: 10 sessions, two to three times per week over a 4-week period. Three points needled (one trigger point, LI15 and ST38) with a needle of 0.30 × 40 mm diameter Interferential electrotherapy: 10 × 20-minute sessions over 4 weeks at 80–120 Hz. Four suction-type electrodes were placed around the shoulder region in a coplanar arrangement Control: no treatment for 4 weeks |
Localised pain over one shoulder, night pain and restricted active and passive shoulder motion |
|
Fang 200680 Two-arm RCT |
n = 360 Electroacupuncture: 174 TENS: 186 |
Electroacupuncture: 10 × 40-minute sessions every other day. Four acupuncture points [Waiguan (SJ5) and Hegu (LI4) and, alternating each session, Jianliao (SJ14) and Jian qian (Ex-UE) or Jianyu (LI15) and Naoshu (SI10)]; 0.30 × 40 mm needles inserted to depth of 20–25 mm TENS: 10 × 40-minute sessions every other day. Electrodes placed at four acupuncture points [Waiguan (SJ5) and Hegu (LI4) and, alternating each session, Jianliao (SJ14) and Jian qian (Ex-UE) or Jianyu (LI15) and Naoshu (SI10)]. High frequency (100 Hz) for 10 minutes and low frequency (2 Hz) for 30 minutes. Intensity of current 10 ± 2 mA |
Used ‘recognised criteria’ (no further details provided |
|
Ma 2006 81 Three-arm RCT |
n = 75 Acupuncture: 30 Acupuncture + PT: 15 PT: 30 |
Acupuncture: 15-minute session twice a week for 4 weeks. Each session consisted of therapeutic principles in promoting flow of qi and blood, driving out the wind and cold, removing dampness and activating meridians; therapeutic methods on three yang meridians of the hand; and prescriptions with jianjiao, jianyu (LI15), fengchi (GB20), hegu (LI4) and yanglingquan (GB34) PT: five sessions per week for 4 weeks |
Shoulder pain for at least 3 months, could not lift arms more than 135° |
One study used acupuncture81 and two studies used electroacupuncture,79,80 The number of acupuncture points needled ranged from three to five. Although needle point prescriptions varied between studies, two studies both included Hegu (LI4) and Jianyu (LI15) needle points. 80,81 In the acupuncture study, acupuncture was administered in a course of eight sessions. In both studies of electroacupuncture, the intervention was administered in a course of 10 sessions, with acupuncture points needled to a depth of 15–25 mm and electricity administered when ‘De qi’ sensation was felt. Further details of the electrical stimulation used were reported in one study only79 and consisted of 2–100-Hz electrical stimulation at a pulse of 100–400 microseconds for 20 minutes. The comparators were interferential electrotherapy,79 waiting list control,79 TENS,80 physiotherapy81 and acupuncture with physiotherapy. 81
One study included a physiotherapy regimen. 81 This consisted of a heat pack for 15 minutes, joint mobilisation for 5–10 minutes and active shoulder exercises for 5–10 minutes, five times per week for 4 weeks. One study reported concomitant treatment of a home exercise programme of mobilisation exercises five times a day over 6 months. 79 Two studies did not report whether participants received any concomitant treatment. 80,81
The inclusion criteria varied between the three studies. It was unclear in all studies whether they included participants with diabetes. Frozen shoulder was diagnosed by an orthopaedic surgeon, through clinical examination and through clinical history. The participants’ stage of frozen shoulder was not reported in two studies. 79,81 In the other study participants were reported to have either stage 1 (described by the authors as pre-adhesive) frozen shoulder, that is, they had shoulder pain that increased at night with range of motion normal or only slightly affected, or stage 2 (described by the authors as adhesive) frozen shoulder, that is, a reduction in pain but severely affected range of motion. 80 None of the studies reported whether participants had received previous treatment for frozen shoulder. Where reported, the mean age of participants ranged from 51.5 to 56.4 years and the proportion of women from 52% to 55%.
Quality assessment
None of the studies reported the method of randomisation or allocation concealment; therefore, these three studies are potentially at high risk of bias. The study by Cheing et al. 79 was of the highest quality and fulfilled five criteria: the number randomised, the comparability of treatment groups at baseline, double blinded, blinding of outcome assessors to treatment allocation and no unexpected imbalances in dropouts between groups. This study, however, did not use intention-to-treat analysis. Fang et al. 80 fulfilled two criteria: the number of participants randomised and the blinding of participants. Ma et al. 81 also fulfilled two criteria: the number of participants randomised and the comparability of treatment groups at baseline for important prognostic factors.
The studies by Fang et al. 80 and Ma et al. 81 reported outcome measures at ≤ 4 weeks from baseline and are therefore excluded from the subsequent synthesis. Furthermore, the waiting list control arm for the study by Cheing et al. 79 reported outcomes at 4 weeks from baseline only. The following synthesis is therefore based on the study by Cheing et al. 79 and compares electroacupuncture with interferential therapy only. This study reported on the outcomes of pain and of function and disability only.
Pain
Cheing et al. 79 reported results of ‘pain at the moment’ at approximately 2, 4 and 7 months using a 0–10 cm VAS. There was no significant difference in pain between the electroacupuncture and interferential therapy groups at 2 months (SMD 0.35, 95% CI –0.23 to 0.93), 4 months (SMD 0.21, 95% CI –0.37 to 0.78) or 7 months (SMD 0.21, 95% CI –0.37 to 0.78).
Function and disability
Cheing et al. 79 reported results of the Constant score at 2, 4 and 7 months. There was no significant difference in Constant score between the electroacupuncture and interferential therapy groups at 2 months (MD –2.80, 95% CI –5.88 to 0.28), 4 months (MD 3.10, 95% CI –1.53 to 7.73) or 7 months (MD –1.70, 95% CI –4.76 to 1.36).
Adverse events
None of the three studies that evaluated acupuncture or electroacupuncture reported on whether or not participants experienced any adverse events.
Summary
Three studies compared acupuncture with another treatment, although only one provided data beyond 4 weeks’ follow-up. This study had a potentially high risk of bias and it was unclear whether it had enough participants to detect a difference between groups. Based on a single study, there was no statistically significant difference between electroacupuncture and inferential electrotherapy in pain or function and disability at short-, medium- or long-term follow-up. There was insufficient evidence to make conclusions with any certainty about the effectiveness of acupuncture for primary frozen shoulder and in what situations it is likely to be effective.
Manipulation under anaesthesia
Study characteristics
Four RCTs investigated MUA in the treatment of primary frozen shoulder. A summary of study characteristics is available in Table 35 and full details are provided in Appendix 6. All were full papers of studies conducted in Pakistan,82 the UK38,83 and Finland39 between 2007 and 2009. All were two-arm trials and sample sizes ranged from 3638 to 125. 39
The MUA procedure was described in three of the RCTs38,39,83 and was performed by a physician39 or, in the two UK studies, by an orthopaedic surgeon. 38,83 In two studies participants received a steroid injection (triamcinolone) in conjunction with MUA, 30 mg38 in one of the UK studies and 80 mg82 in the study from Pakistan.
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Amir-us-Saqlain 200782 Two-arm RCT |
n = 43 MUA + steroid injection + manipulated extremity + PT: 23 MUA + steroid injection + PT: 20 |
MUA: no further details Steroid: 80 mg Kenacort (triamcinolone) with 2% xylocaine Manipulated extremity: kept in 160° of abduction with 90° of external rotation for 24 hours PT: daily 30-minute sessions for 3 weeks |
History of pain and stiffness of the shoulder joint with no identifiable cause, restriction of glenohumeral motion < 50% of abduction and < 50% of external rotation compared with the contralateral shoulder joint and normal radiography. All symptoms had to be of 3 weeks’ duration |
|
Jacobs 200983 Two-arm RCT |
n = 53 MUA: 28 Steroid + distension: 25 |
MUA: manipulation of arm into full adduction and forward flexion, full external rotation, full internal rotation and full abduction Steroid + distension: three steroid and distension treatments at 6-week intervals: triamcinolone, 40 mg, 1 ml; 2% lidocaine, 5 ml; 0.25% bupivacaine, 10 ml; air, 5 ml |
Patients with primary frozen shoulder |
|
Kivimaki 200739 Two-arm RCT |
n = 125 MUA: 65 Home exercise: 60 |
MUA: manipulation of arm into flexion and abduction and rotation into internal and external rotation Home exercise: PT advice in two sessions and written instructions for a daily training programme |
Gradually increasing shoulder pain and stiffness, shoulder mobility of ≤ 140° in elevation and 30° in external rotation |
|
Quraishi 200738 Two -arm RCT |
n = 36 MUA + steroid: 17 (18 shoulders) Arthrographic distension: 19 (20) |
MUA: a specific protocol was followed to ensure safe breakage of adhesions by using a short lever arm Steroid: triamcinolone acetonide, 30 mg and 2% lidocaine, 2ml Distension: normal saline (10–55 ml) injected to progressively distend capsule rupture |
Stage 2 adhesive capsulitis, global loss of active and passive shoulder movement, restriction of rotation < 50% of normal, and normal anteroposterior and axillary lateral radiography of glenohumeral joint |
A single study, by Amir-us-Saqlain et al. ,82 reported use of a physiotherapy programme following MUA. The other three studies advised participants in the intervention and control groups to undertake home exercises. Kivimaki et al. 39 appeared to have the most intensive home exercise programme, although details were scanty in all the studies; participants in this study received physiotherapy advice in two sessions and written instructions for a daily training programme including pendulum and stretching exercises. Quraishi et al. 38 reported a home exercise programme of pendulum exercises and wall-climbing movements; specific details were not provided by Jacobs et al. 83
Each of the studies had a different comparator. In the study by Amir-us-Saqlain et al. 82 from Pakistan, the intervention being assessed was the effect of keeping the manipulated extremity in abduction and external rotation for 24 hours following MUA. Both groups received MUA, including a steroid injection, followed by physical therapy and the experimental group also had the extremity kept in a fixed position following MUA. In the two UK studies, Jacobs et al. 83 compared MUA with distension in combination with steroid injection; and Quraishi et al. 38 compared MUA, including a steroid injection, with arthrographic distension. Kivimaki et al. 39 compared MUA (plus home exercise) with home exercise alone, which involved physiotherapy advice in two sessions and written instructions for home exercise.
The inclusion criteria varied between studies in terms of extent of restriction of movement. 38,39,82 Jacobs et al. 83 did not specify the symptomatic inclusion criteria other than that patients with primary frozen shoulder were included. Quraishi et al. 38 included only patients with stage 2 frozen shoulder. The stage of frozen shoulder was described as painful and stiff or in the ‘freezing’ phase in two RCTs,39,83 and the majority of participants reported pain and stiffness in one RCT (the remainder had either pain or stiffness). 82 At baseline the duration of frozen shoulder ranged from a mean or median of 10 weeks to 40 weeks. Two studies reported on whether participants had received previous treatment for frozen shoulder. Quraishi et al. 38 reported that 44% had previously received physical therapy, 61% a steroid injection and 31% physical therapy combined with steroid injections. Kivimaki et al. 39 alluded to participants having received previous treatment by reporting that in the 3 months prior to randomisation there was no difference between treatment groups in terms of physical therapy, massage or chiropractic manipulations received. The mean or median age of the participants ranged from 53 to 57 years and the proportion of women ranged from 58% to 68%. It was unclear whether participants with diabetes were included in one study,82 one study reported that there were no participants with diabetes83 and in the remaining two studies the proportions with diabetes were 14%39 and 17%. 38
Quality assessment
The included RCTs generally varied in quality. All of the studies reported the number of participants randomised and comparability at baseline, although one reported only comparability for range of movement. 82 The Amir-us-Saqlain et al. 82 and Jacobs et al. 83 studies did not report the method of randomisation and so it was unclear whether these studies were truly randomised, and they did not report allocation concealment or whether the outcome assessors were blinded, although this would not have been possible in the study by Amir-us-Saqlain et al. 82 These studies therefore had a potentially high risk of bias. Additionally, Jacobs et al. 83 was not adequately powered, loss to follow-up was high (19%) and it was unclear whether imbalances in dropouts were adjusted for. Although the authors stated that intention-to-treat analysis was performed, not all patients were included in the analysis. It was unclear whether the study by Amir-us-Saqlain et al. 82 was adequately powered and loss to follow-up was high (23%).
Both Kivimaki et al. 39 and Quraishi et al. 38 reported appropriate randomisation methods; however, only Kivimaki et al. 39 reported allocation concealment and blinding of outcome assessors and was therefore classified as of adequate quality. Loss to follow-up was high in this study (34% at 6 months) and was greater in the MUA group than in the control group; the analysis did not appear to be intention to treat. The study did not appear to be adequately powered at the 6- and 12-month follow-ups. Although Quraishi et al. 38 reported that the investigator was blinded, it was not clear whether outcome assessment was blinded or whether there was allocation concealment and therefore this study may be at risk of bias. Loss to follow-up was relatively low in this study (8%) although it was unclear whether the study was adequately powered. Full details of study quality are reported in Appendix 8.
Manipulation under anaesthesia versus home exercise
Kivimaki et al. 39 compared MUA plus home exercise with home exercise alone. MDs and 95% CIs were reported by the authors for pain, function and disability, passive abduction, passive internal rotation, external rotation and working ability at the follow-up points of 6 weeks and 3, 6 and 12 months from baseline (Table 36). The outcome data for the individual groups are available in Appendix 7.
| Outcomes assessed | Data reported and time of follow-up |
|---|---|
| Pain intensity: Likert scale (0–10)a | MD at 6 weeks and 3, 6 and 12 months (converted to SMD)b |
| Function and disability: modified version of SDQ (14 activities of daily living in previous 24 hours, scored out of 28)a | MD at 6 weeks and 3, 6 and 12 months |
| ROM: passive abduction (°), passive internal rotation (cm), external rotation (°) (and flexion, not included in analysis) | MD at 6 weeks and 3, 6 and 12 months |
| Working ability (0–10) | MD at 6 weeks and 3, 6 and 12 months |
| Adverse events | Narrative summary |
Pain
There was no significant difference in pain intensity between MUA and home exercise at 6 weeks or 3, 6 or 12 months (Table 37).
| Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) | |
|---|---|---|---|
| Pain intensity (SMD and 95% CI) | |||
|
0.2 (–0.64 to 1.02)a 0.07 (–0.32 to 0.46)b |
–0.35 (–0.78 to 0.09)c | –0.28 (–0.73 to 0.16)d | |
| Function and disability | |||
|
4 (–3.8 to 11.8)a 0.3 (–2.69 to 2.75)b |
–1.7 (–5.3 to 1.9)c | 0 (–3.2 to 3.2)d | |
| ROM | |||
| Passive abduction (°) |
10 (–3.2 to 23.2)a 9 (–6 to 24)b |
9 (–4 to 22)c | 7 (–5 to 19)d |
| Passive internal rotation (cm) |
4 (–9 to 9)a –3 (–7.4 to 2.4)b |
–2 (–7.4 to 3.4)c | –1 (–4.1 to 6.1)d |
| External rotation (°) |
5 (–2 to 12)a 6 (–3 to 15)b |
6 (–2 to 14)c | 4 (–4.1 to 12.2)d |
| Working ability | |||
|
0.4 (–4.2 to 1.8)a 0 (–0.8 to 0.8)b |
0.5 (–0.6 to 1.6)c | 0.1 (–0.8 to 1.0)d | |
Function and disability
There was no significant difference in Constant score between treatment groups at 6 weeks or 3, 6 or 12 months (Table 37).
Range of movement
There was no significant difference in passive abduction, passive internal rotation or external rotation between treatment groups at 6 weeks or 3, 6 or 12 months (Table 37).
Working ability
There was no significant difference in the change in working ability between treatment groups at 6 weeks or 3, 6 or 12 months (Table 37).
Adverse events
The authors reported that there were no major complications during manipulation.
Manipulation under anaesthesia versus distension
Two studies compared MUA with distension;38,83 however, both MUA and distension differed between studies and so the data for pain and function and disability that were available could not be considered for pooling.
One RCT by Jacobs et al. 83 compared MUA with steroid injection in combination with distension treatment. Only limited data were available for pain, function and disability and quality of life, which were presented as mean regression coefficients up to 16 weeks (and 24 months for quality of life) (Table 38). The other RCT by Quraishi et al. 38 compared MUA plus steroid injection with arthrographic distension. This study reported means and ranges for pain and function and disability at 2 and 6 months. The percentage of patients who were satisfied with treatment was also reported (Table 38). The outcome data for the individual groups are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Jacobs 200983 | Pain (not specified): VAS (1–100)a | Mean regression coefficient (mean of 2, 6, 12 and 16 weeks) |
| Quraishi 200738 | Pain outcome overall: VAS (0–10)a | Mean and range at 2 and 6 months (no SDs reported)b |
| Jacobs 200983 | Function and disability: Constant score | Mean regression coefficient (mean of 2, 6, 12 and 16 weeks) |
| Quraishi 200738 | Function and disability: Constant score | Mean and range at 2 and 6 months (no SDs reported)b |
| Quraishi 200738 | ROM: external rotation, internal rotation, abduction (and forward elevation, not included in analysis) (°) | Between-group p-values at 6 months (mean values available only on graph) |
| Jacobs 200983 | Quality of life: SF-36, all components | Scores at 24 months presented in graphs and summarised narratively in text |
| Quraishi 200738 | Satisfaction | % satisfied at 6 months |
Pain
The study by Jacobs et al. 83 showed no significant difference in pain intensity between treatment groups up to 16 weeks. The mean regression coefficient (SE) was –2.77 (0.33) for MUA and –2.75 (0.42) for distension combined with steroid injection (95% CI –1.11 to 1.15).
In the study by Quraishi et al. 38 at 2 months’ follow-up the mean VAS score was 4.7 (range 0 to 8.5) in the MUA plus steroid arm and 2.4 (range 0 to 8) in the arthrographic distension arm (p-value not reported). At 6 months there was a significantly greater improvement in VAS in the arthrographic distension group than in the MUA group (SMD 1.52, 95% CI 0.75 to 2.30) (Table 39).
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Pain | |||
| Jacobs 200983 |
Mean regression coefficient (SE) MUA: –2.77 (0.33)a Distension: –2.75 (0.42)a SMD (95% CI) |
||
| Quraishi 200738 | 1.52 (0.75 to 2.30)b,c | ||
| Function and disability | |||
| Jacobs 200983 |
Mean regression coefficient (SE) MUA: 3.13 (0.24)a Distension: 3.23 (0.42)a SMD (95% CI) |
||
| Quraishi 200738 | –6.40 (–11.51 to –1.29)b,d | ||
Function and disability
The study by Jacobs et al. 83 found no significant difference between treatment groups in the Constant score up to 16 weeks: mean regression coefficient (SE) 3.13 (0.24) for MUA and 3.23 (0.42) for distension plus steroid injection (95% CI –1.09 to 0.81).
In the study by Quraishi et al. 38 at 2 months’ follow-up the mean Constant score was 58.5 (range 24 to 90) in the MUA plus steroid arm and 57.4 (range 17 to 80) in the arthrographic distension arm. At 6 months the Constant score was significantly improved in the arthrographic distension group compared with the MUA group (–6.40, 95% CI –6.56 to –6.24) (Table 39).
Range of movement
Quraishi et al. 38 reported no statistically significant difference between groups in external rotation (p = 0.13), internal rotation (p = 0.48) and abduction at 6 months’ follow-up (p = 0.62) (data available only in a graph).
Quality of life
Jacobs et al. 83 reported that all components of the SF-36 improved for all patients during the course of treatment, with the greatest improvements shown in the physical role and bodily pain components. There was no statistically significant difference with respect to change in SF-36 scores between the treatment groups (data available only in graph format, no p-values reported).
Satisfaction
Quraishi et al. 38 reported that at final follow-up 81% of MUA patients were satisfied or very satisfied compared with 94% of patients who received arthrographic distension. All of the patients with diabetes were satisfied with their outcome (three in both the MUA and arthrographic distension groups).
Adverse events
The study by Jacobs et al. 83 reported that no systemic or local complications were noted in either treatment group. Quraishi et al. 38 did not report any information on adverse events.
Manipulation under anaesthesia followed by manipulated extremity kept in fixed position
Amir-us-Saqlain et al. 82 assessed the effect of keeping the manipulated extremity in abduction and external rotation for 24 hours following MUA compared with MUA not using the fixed position. Both groups also received a steroid injection and physical therapy. Only measures of range of movement were assessed 12 weeks post treatment (Table 40). Between-group differences in means and 95% CIs are presented in Table 41. Outcome data for the individual groups are presented in Appendix 7.
| Outcomes assessed | Data reported and time of follow-up |
|---|---|
| ROM: passive abduction, passive internal rotation, passive external rotation, active abduction, active external rotation, active internal rotation (and active and passive forward flexion, not included in analysis)a | ROM 12 weeks post treatment |
| Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) | |
|---|---|---|---|
| ROM | |||
| Passive abduction (°) | 22.49 (13.14 to 31.84)a | ||
| Passive internal rotation (cm)b | 1.00 (0.71 to 1.29)a | ||
| Passive external rotation (°) | 4.84 (–2.26 to 11.94) | ||
| Active abduction (°) | 28.99 (17.07 to 40.91)a | ||
| Active internal rotation (cm) | 0.91 (0.49 to 1.33)a | ||
| Active external rotation (°) | 6.62 (–3.32 to 16.56) | ||
Range of movement
Passive abduction (MD 22.49°, 95% CI 13.14° to 31.84°), passive internal rotation (MD 1.00 cm, 95% CI 0.71 cm to 1.29 cm), active abduction (MD 28.99°, 95% CI 17.07° to 40.91°) and active internal rotation (MD 0.91 cm, 95% CI 0.49 cm to 1.33 cm) were significantly greater at 12 weeks post treatment in the MUA combined with steroid injection plus manipulated extremity and physiotherapy group than in the MUA combined with steroid injection plus physiotherapy group. There was no significant difference in passive or active external rotation between treatment groups.
Summary
The four included studies of MUA were diverse in terms of the intervention and the comparator. Some studies used a steroid injection (triamcinolone) in conjunction with MUA. The studies were not considered clinically similar enough to be pooled in a meta-analysis. Across all of the studies information about previous treatments that participants had received and stage of frozen shoulder was limited. It was not possible to explore variation between those with and without diabetes.
Because it was unclear whether all studies had enough participants to detect a statistically significant treatment effect, it should be kept in mind that where studies did not report a significant benefit (i.e. the CI crossed the line of no difference) this should not be interpreted as evidence of no difference between the groups. A single study of adequate quality reported no statistically significant difference between MUA (and home exercise) and home exercise alone in pain, function and disability, range of movement or working ability at 6 weeks and 3, 6 and 12 months. 39 Follow-up at 6 months and beyond in this study should be treated with some caution because of high loss to follow-up, which was somewhat higher in the MUA group, and also the lack of intention-to-treat analysis. Two studies comparing MUA with distension had mixed findings. One, with a potentially high risk of bias, found no significant difference in pain or function and disability between MUA and distension (which included steroid) up to 16 weeks post treatment. 83 The second, which had some risk of bias, found a significantly greater improvement in pain and function and disability at 6 months with arthrographic distension than with MUA in conjunction with steroid. 38 The fourth study, with a potentially high risk of bias, reported that keeping the manipulated extremity in abduction and external rotation for 24 hours following MUA was superior to MUA without this additional process for passive and active abduction and internal rotation at 12 weeks. 82
In conclusion, there was very little evidence available for MUA and most of the studies identified had limitations. The single adequate study found no evidence of benefit of MUA over home exercise alone. Generalisability is somewhat unclear because of the limited information about previous interventions that participants had received and stage of frozen shoulder.
Distension
Study characteristics
Three RCTs investigated distension with steroid injection in the treatment of primary frozen shoulder. A summary of study characteristics is reported in Table 42, with further details available in Appendix 6. All were full papers of studies conducted in Australia,43 Denmark84 and Norway36 and published between 1998 and 2008. All were two-armed trials.
| Study details and study design | Participants randomised (total n and by group) | Trial treatments | Condition-related inclusion criteria |
|---|---|---|---|
|
Buchbinder 200443 Two-arm RCT |
n = 46 Arthrographic distension + steroid: 25 Placebo: 21 |
Arthrogram: the needle was positioned, connected to the connector tap and tube and 0.5–1 ml of contrast injected and a radiographic image taken. Steroid: methylprednisolone acetate, 40 mg, 1 ml and up to 82 ml of normal saline was then injected (total volume 30–90 ml) Distension: the end point of the procedure was filling of the subscapular bursa, capsular rupture, injection of the total volume of liquid or the participant requesting termination of the procedure Placebo: arthrogram only |
Pain and stiffness in predominantly one shoulder for ≥ 3 months, restriction of passive ROM of > 30° in two or more planes of movements, measured to onset of pain |
|
Gam 199884 Two-arm RCT |
n = 22 Distension + steroid: 13 Steroid: 9 |
Distension + steroid: intra-articular injection, confirmed by ultrasound Steroid: triamcinolone hexacetonide, 20 mg, once per week for a maximum of 6 weeks or until no symptoms Distension: an additional 19 ml of lidocaine 0.5% |
Frozen shoulder of more than 6 weeks’ duration, nocturnal accentuation of pain, passive range of shoulder external rotation < 50% of opposite shoulder |
|
Tveita 200836 Two-arm RCT |
n = 76 Arthrographic distension + steroid: 37 Steroid: 39 |
Arthrogram: Kaye–Schneider technique was used Steroid: triamcinolone acetonide, 20 mg, 2 ml and 3–4 ml of bupivacaine hydrochloride (5 mg/ml). Three injections at 2-week intervals Distension: an additional 10 ml of saline. Three injections at 2-week intervals were given. The capsule would usually rupture in the wall of the subscapular recess or sometimes in the wall of the bicipital or axillary recesses, which was recorded as a loss of resistance, and contrast leakage was identified by fluoroscopy. If rupture had not occurred, more contrast and local anaesthetic was injected until rupture |
Limitation of passive movement in the glenohumeral joint compared with the unaffected side, > 30° for at least two of forward flexion, abduction or external rotation; pain in predominantly one shoulder lasting > 3 months and < 2 years |
The two most recent studies by Buchbinder et al. 43 and Tvetia et al. 36 used arthrographic distension. One injected 40 mg of steroid (in 1 ml) and up to 82 ml of saline on a single occasion43 and the other injected 20 mg of steroid (2 ml), 4 ml of local anaesthetic and 10 ml of saline on three occasions at 2-week intervals,36 along with contrast medium in the arthrographic distension groups. Both of these studies stated that the procedure continued until rupture occurred. Buchbinder et al. 43 had the additional end points of patient termination of the procedure and a maximum of 90 ml having been injected. The older study by Gam et al. 84 stated that the injection was confirmed by ultrasound and participants were injected with 20 mg of steroid with 19 ml of lidocaine once per week for up to 6 weeks. Details were not provided in this study as to whether distension occurred or how this was established. The two studies of arthrographic distension used different comparators: Buchbinder et al. 43 used a placebo comparator of arthrogram only, whereas Tveita et al. 36 used steroid injection (with local anaesthetic).
Home exercise of pendular exercises and scapular setting were given in the study by Buchbinder et al. 43 and no manual treatment (e.g. physiotherapy, massage) or other medical interventions were allowed. In contrast, in the Tvetia et al. study36 patients were allowed to continue with their current physiotherapy programme; no patients were prescribed new physiotherapy programmes during the study. The study of non-arthrographic distension did not report any information regarding home exercise. 84
There was some variability in concomitant treatments. Buchbinder et al. 43 allowed paracetamol and codeine preparations but NSAIDs were not allowed, Tveita et al. 36 allowed pain medication organised by the patients’ primary care physicians and Gam et al. 84 reported that analgesics were permitted.
The inclusion criteria varied between studies; however, the two most recent studies of arthrographic distension required similar extent of restriction in shoulder movement. 36,43 The duration of frozen shoulder at baseline ranged from a median or mean of 114 days (approximately 4 months) to 7 months, although the stage at baseline was not reported by any of the studies. Diagnosis was by measuring passive range of movement to onset of pain,43 clinical examination, blood samples and radiography or ultrasound,84 and clinical history and radiography. 36
A substantial proportion of patients (28%) had diabetes in the Buchbinder study;43 the remaining two studies had no patients with diabetes. Buchbinder et al. 43 also included a small proportion (2.2%) with secondary frozen shoulder. Buchbinder et al. 43 and Tveita et al. 36 also reported that patients had undergone treatment for frozen shoulder before the study: in the Buchbinder study 28% of patients had received corticosteroid injections whereas in the Tveita study 17% had undergone physiotherapy. The ages of patients ranged from a mean or median of 47 years to 57.3 years and the proportion of women was either 59% or 80%.
Quality assessment
Buchbinder et al. 43 was considered of satisfactory quality as the risk of bias was minimised through computerised randomisation and allocation concealment by a biostatistician who kept the assignment scheme. An intention-to-treat analysis was also performed. Patients were blinded to treatment and outcome assessors were also blinded. The treatment groups were also comparable at baseline. The main limitation of this study is that it was not sufficiently powered after dropouts (9%). The intention-to-treat analysis used in the study was adequately powered based on the authors’ power calculation (the sample size to detect a difference in SPADI scores).
Both Gam et al. 84 and Tveita et al. 36 reported a reliable method of randomisation; however, it was not clear whether an adequate method of allocation concealment had been used. Tveita et al. 36 reported that patients were not informed of their actual assignment until the first injection was to be given, and no information regarding allocation concealment was reported by Gam et al. 84 Therefore, these studies are potentially at risk of bias. Gam et al. 84 reported that outcome assessors were blinded and that the groups were comparable at baseline. In the study by Tveita et al. 36 there appeared to be a higher proportion with previous shoulder problems and who were on sick leave in the steroid injection group. Tveita et al. 36 also reported using statistical adjustment to control for baseline differences for the function and disability SPADI outcome but not for range of movement. Tveita et al. 36 used intention-to-treat analysis and both studies reported the same rate of loss to follow-up (9%), with similar numbers in each arm.
Pain
Two studies, one by Buchbinder et al. 43 and one by Gam et al. ,84 assessed pain. Buchbinder et al. 43 reported change from baseline data at 3, 6 and 12 weeks using a 10-point Likert scale (Table 43). However, the study by Gam et al. 84 reported data for VAS pain scores at rest and VAS pain scores on function in graphical form only. This study was described narratively.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Buchbinder 200443 | Likert scale, pain overall (0–10)a | Change from baseline at 3, 6 and 12 weeks (6 and 12 weeks included in analysis) |
| Gam 199884 |
VAS pain score at rest (0–10)a VAS pain score on function (0–10)a Analgesic consumption |
Data presented graphically at 3, 6 and 12 weeks, p-values only |
There was no significant difference in pain between arthrographic distension with steroid and placebo (arthrogram) at 6 weeks (SMD –0.40, 95% CI –0.99 to 0.19) or 12 weeks (SMD –0.15, 95% CI –0.73 to 0.44) in the study by Buchbinder et al. 43
In the study by Gam et al. 84 there was no significant difference between the steroid and distension groups for VAS pain score on function (p = 0.1) or VAS pain score at rest (p = 0.1) (time points unclear). The authors reported that there was a tendency toward less pain in the distension group. Additionally, the authors reported that analgesic consumption was significantly lower at week 11 in the distension group than in the steroid group (p = 0.008). These results were presented in graphical form only.
Function and disability
Two of the three included studies assessed function and disability. 36,43 Both reported SPADI total score and one43 reported a problem elicitation technique score, a patient preference disability measure in which patients identify their own problems related to the disease that they would like to see improve as a result of treatment. Change from baseline data were available from Buchbinder et al. 43 and final value data were available from Tveita et al. 36 (Table 44). These were not pooled as the comparator arms were different between studies: Buchbinder et al. 43 compared arthrographic distension with placebo whereas Tveita et al. 36 used steroid injection as a comparator. Table 45 provides the between-group MDs and 95% CIs. The outcome data for individual groups are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Buchbinder 200443 |
SPADI total score (0–100)a Problem elicitation technique scorea |
Change from baseline at 3, 6 and 12 weeks (6 and 12 weeks included in analysis) |
| Tveita 200836 | SPADI total score (0–100)a | Final value at 6 weeks after last injectionb |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Distension vs placebo | |||
| SPADI total score | |||
| Buchbinder 200443 |
5.00 (–7.85 to 17.85)a 4.40 (–10.08 to 18.88)b |
||
| Problem elicitation technique score | |||
| Buchbinder, 200443 | |||
| Distension vs steroid | |||
| SPADI total score | |||
| Tveita 200836 | –6.00 (–14.12 to 2.12)d | ||
Distension versus placebo
There was no significant difference between arthrographic distension and placebo in SPADI total score at 6 or 12 weeks in the study by Buchbinder et al. 43 (Table 45). In contrast, there was a significant improvement in the problem elicitation technique score with distension compared with placebo at 6 weeks (MD –46.00, 95% CI –80.99 to –11.01) and 12 weeks (MD 54.40, 95% CI 12.57 to 96.23) in the same study.
Range of movement
The range of movements of interest in this review that were reported by the studies of distension were passive and active abduction, passive and unspecified external rotation, passive internal rotation and hand behind back. Change from baseline data were available from Buchbinder et al. 43 and final value data were available from Tveita et al. 36 (Table 46). It was considered inappropriate to pool the two studies as one had a placebo comparator and one a steroid injection comparator. The study by Gam et al. 84 provided limited data for extraction and these were summarised narratively. Table 47 summarises the between-group MDs and 95% CIs. The outcome data for individual groups are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Buchbinder 200443 | Active abduction, external rotation in neutral, hand behind back (°) | Change from baseline at 3, 6 and 12 weeks |
| Gam 199884 | Passive abduction, passive external rotation (°) | Data presented graphically (number achieving four levels of improvement) at 3, 6 and 12 weeks, p-values only |
| Tveita 200836 | Passive abduction, passive external rotation, passive internal rotation, active abduction, active external rotation, active internal rotation (°) | Final value at 6 weeks after last injection |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Distension vs placebo | |||
| Active abduction (°) | |||
| Buchbinder 200443 |
8.90 (–8.55 to 26.35)a 4.50 (–15.16 to 24.16)b |
||
| External rotation (°) | |||
| Buchbinder 200443 |
–6.50 (–24.62 to 11.62)a 2.10 (–14.50 to 18.70)b |
||
| Hand behind back (°) | |||
| Buchbinder 200443 |
1.90 (–0.76 to 4.56)a 0.80 (–1.95 to 3.55)b |
||
| Distension vs steroid | |||
| Active abduction (°) | |||
| Tveita 200836 | 3.00 (–13.00 to 19.00)c | ||
| Active external rotation (°) | |||
| Tveita 200836 | 2.00 (–6.33 to 10.33)c | ||
| Active internal rotation (°) | |||
| Tveita 200836 | 2.00 (–5.88 to 9.88)c | ||
| Passive abduction (°) | |||
| Tveita 200836 | –34.00 (–40.78 to –27.22)c,d | ||
| Passive external rotation (°) | |||
| Tveita 200836 | –2.00 (–9.42 to 5.42)c | ||
| Passive internal rotation (°) | |||
| Tveita 200836 | –3.00 (–9.13 to 3.13)c | ||
Distension versus placebo
There was no significant difference between groups in the outcomes active abduction, external rotation or hand behind back at 6 or 12 weeks’ follow-up in the study by Buchbinder et al. 43 (Table 47).
Distension versus steroid
The study by Tveita et al. 36 found that there was a significant improvement in passive abduction with steroid compared with distension at 6 weeks after the last injection (MD –34.00°, 95% CI –40.78° to –27.22°). There was no significant difference between groups for active abduction, external or internal rotation, or passive internal or external rotation (Table 47).
The study by Gam et al. 84 reported that there was a significant improvement with distension compared with steroid in external rotation (p = 0.0007) but no statistically significant difference in abduction (time point unclear).
Adverse events
All of the studies investigating distension reported adverse events. These were reported in detail in the study by Buchbinder et al. 43 and to a lesser extent by Gam et al. 84 and Tveita. 36 The adverse events reported by all studies are summarised in Table 48.
| Study | Adverse events reported |
|---|---|
| Buchbinder 200443 |
Total reported adverse events: arthrographic distension (n = 25): 9 (36%), placebo (n = 21): 1 (5%) Pain associated with procedure: arthrographic distension (n = 25): 4 (16%), placebo (n = 21): 1 (5%) Increased pain for up to 48 hours after procedure: arthrographic distension (n = 25): 3 (12%), placebo (n = 21): 1 (5%) Claustrophobia at time of procedure: arthrographic distension (n = 25): 1 (4%), placebo (n = 21): 1 (5%) Unsettled, anxious and hot: arthrographic distension (n = 25): 0 (0%), placebo (n = 21): 1 (5%) Shoulder noisy (i.e. fluid noises): arthrographic distension (n = 25): 1 (4%), placebo (n = 21): 0 (0%) |
| Gam 199884 | Two cases of unacceptable pain after injection (one in each treatment group) |
| Tveita 200836 |
Injections reported as very painful: distension: 5, steroid: 6 Other possible side effects reported: distension: 14, steroid: 20 Complaints of flushing or disturbances in heat regulation: distension: 9, steroid: 13 Minor loss of sensation and motor control loss in affected arm: distension: 2, steroid: 2 Glenohumeral joint infection: treatment group unspecified: 1 |
Buchbinder et al. 43 reported that open-ended questions were used to assess the extent of adverse events. More patients in the arthrographic distension group than in the placebo group reported pain associated with the procedure (16% vs 5%) and pain lasting up to 48 hours (12% vs 5%). None of the adverse events was reported as serious and a number of additional adverse events were reported (Table 48).
Gam et al. 84 reported that the number and type of side effects were recorded. The only adverse events reported in this study were two cases of unacceptable pain after injection (one in each treatment group); these patients dropped out of the study.
In the study by Tveita et al. 36 the patients recorded pain intensity related to the injection procedures. Six patients in the steroid group and five in the distension group felt that the injections were very painful. Flushing and disturbances in heat regulation were also common in both groups and a number of other non-serious adverse events were reported (Table 48).
Summary
Three studies were included that investigated the effects of distension with steroid injection. Two of these studies investigated arthrographic distension36,43 and the other investigated non-arthrographic distension. 84 The comparators evaluated were steroid alone36,84 and placebo (arthrogram only). 43 Only one study was judged to be of satisfactory quality,43 whereas the remainder were potentially at risk of bias. Because of the variability in the interventions and comparators it was not considered appropriate to pool any of the studies. Stage of frozen shoulder was not reported in any of these studies; however, previous treatments were described in two of the studies. 36,43
Distension compared with placebo
One study of satisfactory quality compared distension with placebo and reported benefit with distension in the short term in one of two measures of function and disability. There was no difference in three measures of range of movement or in pain at function at short-term follow-up. None of the adverse events reported was considered serious.
Distension compared with steroid
Two studies that were potentially at risk of bias compared distension with steroid. One study that compared arthrographic distension with steroid reported no difference between groups in one measure of function and disability, but reported a benefit in one range of movement in the short term. Some patients in both groups considered the injections as being very painful and one case of glenohumeral joint infection (septic arthritis) was reported. The study that compared non-arthrographic distension with steroid reported no difference between groups in pain at function or at rest, but a significant benefit with distension in one measure of range of movement but not another at short-term follow-up. One patient from each group dropped out as a result of unacceptable pain.
In conclusion, few studies of distension were identified and only single studies of different comparisons were available. Based on one study of satisfactory quality there is a little evidence of potential benefit with distension compared with placebo. There is insufficient evidence to draw conclusions about the efficacy of distension (arthrographic or non-arthrographic) for frozen shoulder.
Capsular release
Two case series of more than 50 participants were identified that investigated capsular release in the treatment of primary frozen shoulder. A summary of the study characteristics is reported in Table 49, with further details available in Appendix 7. One study was published in Norway in 2007 by Austgulen et al. ;85 the other in Taiwan in 2002 by Chen et al. 86
| Study details and study design | Participants | Treatment | Condition-related inclusion criteria |
|---|---|---|---|
|
Austgulen 200785 Case series |
n = 66 (70 shoulders) |
Arthroscopic capsular and ligament release + PT Surgery was performed under general anaesthesia in beach chair position. Shoulder arthroscopy was performed using normal technique. Access to the shoulder joint was from behind and surgical instruments entered from in front in the rotator interval. The rotator interval was cleaned out and the frontal capsule and glenohumeral ligament and coracohumeral ligament were split from biceps tendon to 6 o’clock. Both capsule and ligaments were split with an Acufex Upbiter Scissor punch. The subacromial space was inspected and adherences were loosened. The space and displacement were evaluated. Where spaces were narrow subacromial decompression was performed until the shoulder could move at maximal outward rotation and to 180° in abduction. All patients received aggressive rehabilitation with a physiotherapist from the first day after surgery |
Patients with primary frozen shoulder. Diagnosis of frozen shoulder was confirmed during anaesthesia: limited outward rotation of < 20° and < 45° in abduction with a fixed scapula |
|
Chen 200286 Case series |
n = 183 cases | Arthroscopic brisement (distension, debride, release) followed by gentle manipulation + PT (further details provided), then the arm was kept in abduction–external rotation position for 2 days during which the patient was confined to bed. Passive and active exercise of the shoulder was then allowed, with a rehabilitation programme at the hospital rehabilitation facility | Basic criteria for definition of idiopathic frozen shoulder |
The capsular release procedures varied between the two studies. Only a summary of the procedures are provided here; fuller descriptions are available in the original papers. In the study by Chen et al. ,86 the authors stated that the intervention involved distension and debridement, release and manipulation. The degree of joint movement was confirmed arthroscopically (after distension by water and the syringe rebound), a synovectomy was performed using an arthroscopic shaver or vaporisation, capsular scar tissue was described by the authors as being debrided and then gentle manipulation was performed. This involved arm elevation in the scapular plane (which was usually associated with audible popping of the contracted capsule) and external rotation followed by internal rotation at varying degrees of abduction. This was done with gradual pressure and stopped if unyielding resistance was met. The authors stated that repetition of these steps led to tearing of the capsular structures. The arm was kept in abduction–external rotation for 2 days following the procedure.
The Austgulen et al. study85 reported that shoulder arthroscopy was performed using normal technique, and the frontal capsule, glenohumeral ligament and coracohumeral ligament were split from the bicep tendon to 6 o’clock. The authors stated that the subacromial space was inspected and adherences loosened. Where the spaces were narrow subacromial decompression was performed until the shoulder could move at maximal outward rotation and at 180° in abduction.
Both studies included a programme of physiotherapy following the procedure. Austgulen et al. 85 specifically stated that the patients received aggressive rehabilitation from the first day after surgery (no further details reported). This study also reported that participants were asked to perform home exercises (including stretches) every day. In addition, concomitant NSAIDs were given as needed and postoperative oxycodone was given occasionally. 85 Chen et al. 86 did not state whether a home exercise routine was undertaken or whether concomitant medication was given.
The inclusion criteria differed between the studies, with Chen et al. 86 reporting limited criteria (i.e. basic criteria for diagnosis of idiopathic frozen shoulder). Additionally, Austgulen et al. 85 required that participants had already tried physiotherapy but without a satisfactory result. The nature and intensity of the previous physiotherapy was not reported. The diagnostic criteria also varied between studies. In the Austgulen study85 diagnosis was based around limited range of movement and confirmed under anaesthesia. Chen et al. 86 used clinical history, physical examination, radiography and arthrography. Neither study reported the stage of frozen shoulder; the average duration of frozen shoulder at baseline was 13 months (range 3 to 60 months) in the Austgulen study85 and 8 months (range not reported) in the Chen study86 (it was unclear whether this was mean or median). A total of 17% of the patients in the Austgulen study had diabetes85 but this information was not reported in the study by Chen et al. 86 In both studies participants had received previous treatment for frozen shoulder. Austgulen et al. 85 reported that participants had received physical therapy, whereas Chen et al. 86 reported that none of the participants had responded to conservative treatment of at least 12 weeks’ duration. The average age (mean or median not specified) of participants was 53 years in the Austgulen study,85 whereas Chen et al. 86 reported that ages ranged from 32 years to 79 years. The percentage of women was similar in both studies: 67%85 and 75%. 86
Quality assessment
Both studies were case series, which are inherently at high risk of bias because of the lack of a control group. The Bradford Hill criteria (a group of minimal conditions necessary to provide adequate evidence of a causal relationship between an incidence and a consequence) state that there must be a temporal relationship between the incidence of the factor (in this case surgery) and consequence (recovery). For a condition such as frozen shoulder, which for most people will resolve over a 1- to 3-year period, this relationship cannot be ascertained without a control group. 87 Austgulen et al. 85 was considered better quality as this study reported inclusion criteria, the population appeared representative of the frozen shoulder population, SDs were reported and patients were recruited prospectively, although the extent of this was unclear. The number of dropouts was unclear. Chen et al. 86 met only one criterion (reporting of inclusion criteria); the remainder were unclear or not met. There were no dropouts in this study. Further details of quality assessment are available in Appendix 8.
Outcomes available
Data were sparse for all outcomes assessed (Table 50). The effect on short-term pain was described narratively in Chen et al. 86 Function and disability and relevant measures of range of movement (external rotation, internal rotation and abduction; it was not specified whether these were active or passive) were reported at baseline and follow-up and as average gain in range of movement by Chen et al. (no further details available). 85,86 Additionally, Austgulen et al. 85 reported data for ability to work, physical activity, sleep at night and satisfaction. Both studies reported adverse events. Numerical results are summarised in Table 51. The complete outcome data for these studies are available in Appendix 7.
| Study | Outcomes assessed | Data reported and time of follow-up |
|---|---|---|
| Chen 200286 | Pain (reduction of pain): pain component of ASES (modified version)a | Pain described narratively at ‘a few weeks’ and 1 and 3 months (3-month data used in analysis) |
| Austgulen 200785 | Function and disability: Oxford Shoulder score (12, best outcome; 60, worst) | Mean and SD postoperatively (average follow-up 10 months, range 3 to 29 months) |
| Function and disability: telephone questionnaire assessing ability to work, physical activity, sleep at night (all 0, most complaints; 10, least complaints) and patient satisfaction (0, worst; 10, best) | Mean and SD postoperatively (average follow-up 10 months, range 3 to 29 months) | |
| Chen 200286 | Function and disability: modified version of ASES (0–100)a | Final value mean and SDb at time of follow-up (average follow-up 23 months, range 6 months to 5 years) |
| Austgulen 200785 | ROM: external rotation, abduction (both degrees) (and flexion, not included in analysis) | Mean and SD postoperatively (average follow-up 10 months, range 3 to 29 months) |
| Chen 200286 | ROM: external rotation, internal rotation (both degrees) (and elevation, not included in analysis) | Average gain at latest follow-up (not specified) |
| Study | Short term (≤ 3 months) | Medium term (> 3 and ≤ 6 months) | Long term (> 6 and ≤ 12 months) |
|---|---|---|---|
| Function and disability | |||
| Oxford Shoulder score | |||
| Austgulen 200785 |
Baseline: mean 41.0 (SD 7.5) Postoperatively: mean 18.4 (SD 7.3) (p < 0.001) |
||
| Modified ASES | |||
| Chen 200286 |
Baseline: mean 41 (SD 13) Follow-up: mean 87 (SD 11)a (p < 0.005) |
||
| Range of movement | |||
| Internal rotation (°) | |||
| Chen 200286 | Average gain: 35 | ||
| External rotation (°) | |||
| Austgulen 200785 |
Baseline: mean 3 (SD 5) Postoperatively: mean 39 (SD 23) (p < 0.001) |
||
| Chen 200286 | Average gain: 30 | ||
| Abduction (°) | |||
| Austgulen 200785 |
Baseline: mean 34 (SD 8) Postoperatively: mean 154 (SD 37) (p < 0.001) |
||
| Other | |||
| Working ability | |||
| Austgulen 200785 |
Baseline: mean 2.4 (SD 2.6) Postoperatively: mean 7.4 (SD 2.5) (p < 0.001) |
||
| Physical activity | |||
| Austgulen 200785 |
Baseline: mean 2.3 (SD 2.5) Postoperatively: mean 7.4 (SD 2.4) (p < 0.001) |
||
| Sleep at night | |||
| Austgulen 200785 |
Baseline: mean 1.7 (SD 2.5) Postoperatively: mean 7.2 (SD 2.6) (p < 0.001) |
||
| Satisfaction | |||
| Austgulen 200785 | Mean (SD) 8.6 (1.6) | ||
Pain
Only one study reported any data regarding pain. Chen et al. 86 reported that two-thirds of patients complained of pain exacerbation after arthroscopic brisement; 5% had pain duration of longer than a few weeks. Half of patients had pain relief within 1 month whether in motion or not. After 3 months all except eight shoulders were pain free in any direction of shoulder movement (data were not reported).
Function and disability
Austgulen et al. 85 reported a significant improvement in the Oxford Shoulder score from a baseline mean of 41.0 (SD 7.5) to 18.4 (SD 7.3) postoperatively (p < 0.001), a scale for which the best possible outcome is 12 and worst possible outcome is 60 (Table 51).
Similarly, Chen et al. 86 reported that an improvement in the mean modified ASES score from a baseline score of 41 (SD 13) to 87 (SD 11) was significant (p < 0.005) at follow-up (Table 51).
Austgulen et al. 85 reported significant improvements from baseline to follow-up (p < 0.001) on a 10-point scale in mean working ability (baseline 2.4, SD 2.6; follow-up 7.4, SD 2.5), mean physical activity (baseline 2.3, SD 2.5; follow-up 7.4, SD 2.4) and sleep at night (baseline 1.7, SD 2.5; follow-up 7.2, SD 2.6). This study also reported high satisfaction postoperatively (8.6, SD 1.6 on a 10-point scale) (Table 51).
Range of movement
Austgulen et al. 85 reported that there was a significant improvement in mean external rotation from 3° (SD 5°) to 39° (SD 23°) (p < 0.001) and in mean abduction from 34° (SD 8°) to 154° (SD 37°) (p < 0.001) postoperatively.
Chen et al. 86 reported a similar average gain in external rotation of 35°. The gain in internal rotation was 30°.
Adverse events
Austgulen et al. 85 reported that two patients had frozen shoulder again and had repeat surgery. No deep infections, nerve damage or other complications were reported. Chen et al. 86 found that only one patient experienced complications (superficial wound infection). No further information regarding how adverse events had been assessed was reported in either study.
Summary
Two studies investigated capsular release. Both were case series and therefore inherently at high risk of bias. One of the studies stated that debrisement was a component of the capsular release procedure and both reported that physiotherapy was undertaken post procedure.
One case series reported that the majority of patients were pain free at movement at short-term follow-up. Statistical significance was not reported. There was evidence of benefit for function and disability from both studies. Each study reported a significant improvement in one function and disability scale. Additionally, one study reported significant benefits in working ability, physical activity, sleep at night and satisfaction with capsular release. One study reported evidence of significant benefit in two measures of range of movement with capsular release; the other study reported an improvement in two measures of range of movement, but did not report whether or not these were significant.
In conclusion, although the evidence available suggested potential benefit from capsular release, these studies were at high risk of bias and cannot be used to draw conclusions regarding the efficacy of this treatment for frozen shoulder.
Mixed-treatment comparison results
A MTC was performed for pain only. As previously stated in the report, this was because pain was the only outcome for which a network was available.
Analyses were planned for pain recorded at ≤ 3 months (excluding pain recorded at ≤ 4 weeks from the start of treatment), > 3 months and ≤ 6 months, and > 6 months and ≤ 12 months. However, data were only available for ≤ 3 months.
Four networks were evaluated:
-
Network 1: studies of any intervention (i.e. conservative and invasive) and any quality.
-
Network 2: studies of any intervention that were at least of satisfactory quality (i.e. method of randomisation was adequate and outcome assessment was blinded). The overall quality of the available trials for the MTC was poor, with most not reporting on the a priori quality criterion of allocation concealment. So that a network of trials could be formed, method of randomisation rather than allocation concealment was used as a quality criterion in the MTC.
-
Network 3: studies of non-invasive treatments of any quality, excluding sodium hyaluronate (because it is rarely used in the NHS). 18
-
Network 4: studies of non-invasive treatments that were at least of satisfactory quality (i.e. method of randomisation was adequate and outcome assessment was blinded), excluding sodium hyaluronate.
Interpretation of standardised mean differences
Reduction in pain recorded as a SMD can be transformed back into the units of one or more of the pain measurement instruments. The decision was made to back transform to a VAS 0–100 mm as this measure had more substantial information than other pain measures. This included one study performed in a shoulder-specific population. 50 Transformation from SMD to VAS was made using a representative pooled SD at baseline from one of the included trials. 51 No data could be located on the MCID in pain for individuals with frozen shoulder. However, it is suggested that a difference of 14 mm on a 100-mm VAS scale is the MCID for individuals with rotator cuff disease, a shoulder disorder also characterised by pain and restricted range of movement. 50 Given that the between-group MCID is thought to approximate 40% of that within individuals6,53 the MCID between groups for rotator cuff disorder can be estimated to be approximately 5.6 mm. This value is therefore used as a proxy for the between-group MCID for frozen shoulder. 6 The reduction in pain recorded as a SMD can be interpreted on a VAS 0–100 mm scale using Table 52.
| Reduction in pain score | |||||
|---|---|---|---|---|---|
| SMD | –0.3 | –0.5 | –1.0 | –1.5 | –2.0 |
| VAS 0–100 mm | –5.6 | –9.15 | –18.3 | –27.45 | –36.6 |
From the 31 included trials, the total number of trials potentially available for analysis was nine. The reasons for trials being excluded from the analysis are presented in Table 53.
| Reason for exclusion from the MTC | No. of studies excluded |
|---|---|
| Trial did not report pain as an outcome | 9 |
| Trial did not report pain data | 6 |
| Trial reported pain at ≤ 4 weeks | 2 |
| Trial reported pain at > 3 months only | 1 |
| Trial reported mean change and baseline medians only | 2 |
| SDs could not be imputed | 1 |
| Trial compared the same class of intervention with each other | 1 |
The nine trials and the 10 interventions they evaluated are shown in Table 54. The placebo used varied between studies and included saline injections, lidocaine injection, placebo laser, sham distension and sham ultrasound. The four studies of physical therapy consisted of physiotherapy with an active mobilisation component. Physical therapies without an active component consisted of laser therapy and interferential therapy. Four studies included steroid injections; these were triamcinolone hexacetonide (40 mg once, 20 mg once), methylprednisolone (40 mg once for 3 weeks) and dexamethasone (2 mg once weekly for 5 weeks). Steroid injections were either given alone (three studies35,41,70) or combined with physiotherapy (three studies35,41,42). Seven of the nine studies included in the review had a home exercise component. 16,35,39,41–43,51
| Study | Placebo | Steroid injection | Steroid + active physical therapy | Sodium hyaluronate | Active physical therapy | Active physical therapy + placebo | Electroacupuncture | Physical therapy without mobilisation | Arthrographic distension + steroid | MUA + active physical therapy |
|---|---|---|---|---|---|---|---|---|---|---|
| Buchbinder 200443 | ✓a | ✓ | ||||||||
| Carette 200335 | ✓b | ✓ | ✓ | ✓b | ||||||
| Cheing 200879 | ✓ | ✓c | ||||||||
| Dogru 200851 | ✓ | ✓d | ||||||||
| Kivimaki 200739 | ✓ | ✓ | ||||||||
| Rizk 199142 | ✓✓e | ✓f | ||||||||
| Ryans 200541 | ✓b | ✓ | ✓ | ✓b | ||||||
| Stergioulas 200816 | ✓g | ✓h | ||||||||
| Takagishi 199670 | ✓ | ✓ |
In these nine trials, a variety of pain outcomes and pain scales were reported (Table 55). If a trial reported more than one pain outcome or used more than one scale, only the pain outcome/scale prioritised for use in the analysis (see Chapter 2, Mixed-treatment comparison) is reported.
| Study | Type of pain reported | Type of pain scale used | Time point used | At least satisfactory qualitya |
|---|---|---|---|---|
| Buchbinder 200443 | Pain overall | Likert scale 0–9 | 12 weeks | ✓ |
| Carette 200335 | Pain overall | SPADI 5-item pain subscale | 3 months | ✓ |
| Cheing 200879 | Pain at the moment | VAS 0–10 cm | Approx. 2 months | |
| Dogru 200851 | Pain overall | SPADI 5-item pain subscale | 3 months | |
| Kivimaki 200739 | Pain intensity | Likert scale 0–10 | 3 months | ✓ |
| Rizk 199142 | Pain overall | Likert scale 0–5 | 11 weeks | |
| Ryans 200541 | Daytime pain at rest | VAS 0–100 mm | 6 weeks | ✓ |
| Stergioulas 200816 | Pain overall | VAS 0–100 mm | 8 weeks | ✓ |
| Takagishi 199670 | Pain on activity | VAS 0–10 | 5 weeks |
To check that the non-informative priors used in the analysis were truly non-informative a sensitivity analysis was performed for each network by changing the range of the uniform prior distribution on the SD from 0–2 to 0–0.8, 0–5, 0–10 and, as a final check for stability for network 1, 0–15. An additional sensitivity analysis was also performed by repeating each analysis using a burn-in of 30,000 iterations.
All four networks were evaluated; however, the lack of evidence available to inform networks 2, 3 and 4 led to a lack of stability in the models, particularly a lack of stability in between-study variance. For completeness these analyses have been presented in Appendix 10 although no conclusions can be drawn from the results presented. Only the results for the network of any intervention and of any quality (network 1) are presented here. The slightly higher number of trials included in this network allows the model to achieve stability.
Network 1: studies of any intervention and of any quality
There were nine trials with data that could be used in the analysis of pain at or close to 3 months. These nine trials formed a connected network of nine interventions compared with placebo. The network is presented in Figure 21.
FIGURE 21.
Network diagram of studies of any intervention and any quality. PT, physical therapy.
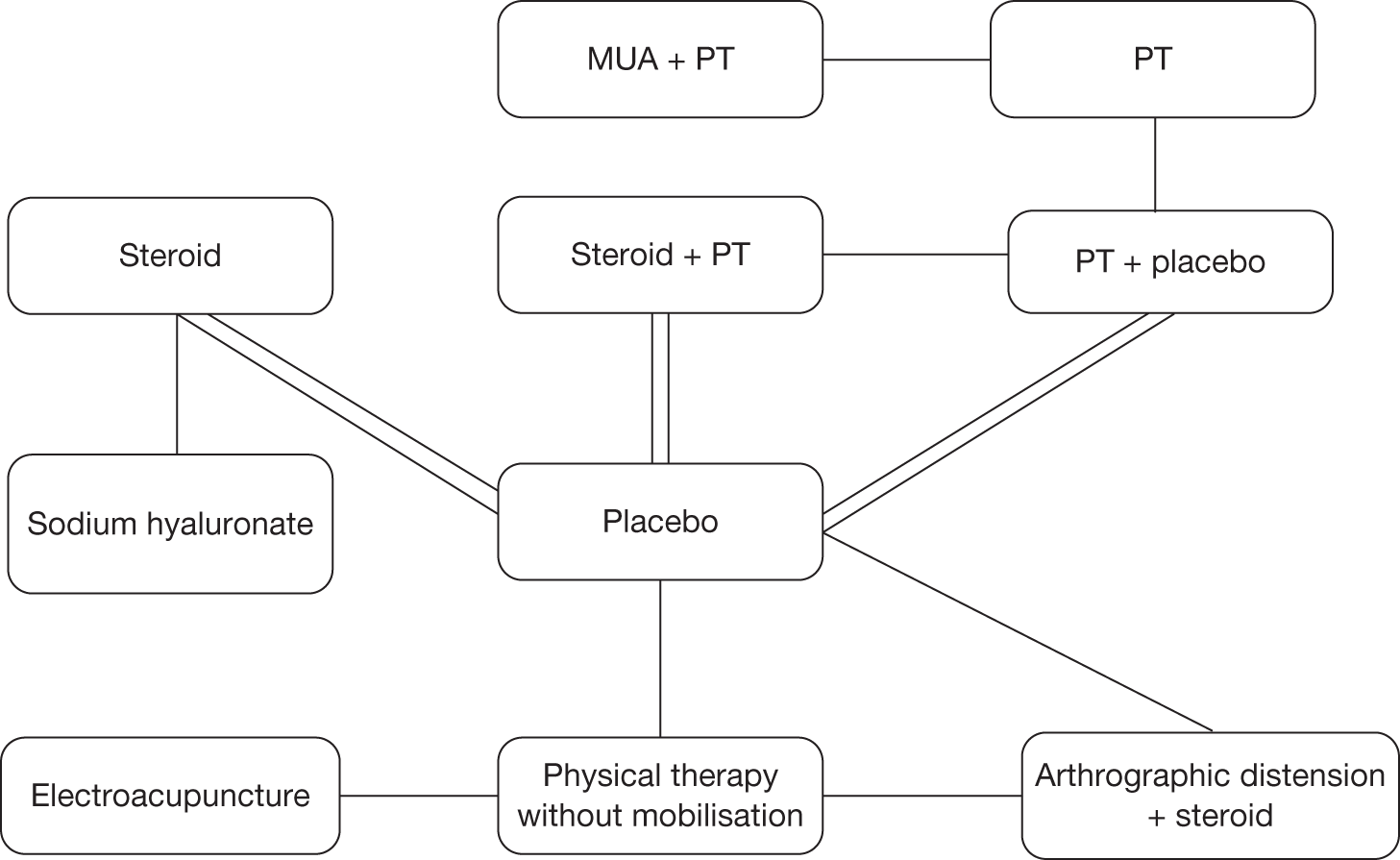
Each line represents one comparison. For example, there were two studies available for the comparison of steroid with placebo. Where there is no line there were no studies available, for example physical therapy without mobilisation versus arthrographic distension and steroid.
The models were run for 100,000 iterations and were not sensitive to the length of burn-in. Stable estimates of treatment effect, CrIs and between-study heterogeneity were produced by the models using a uniform prior of (0, 5), (0, 10) and (0, 15). Convergence, determined by graphical traces and the Brooks–Gelman–Rubin statistic, was achieved. The results of the uniform prior (0, 5) are presented here. Results for uniform priors (0, 10) and (0, 15) are available in Appendix 10.
The model was a good fit as the residual deviance was close to the number of data points. Table 56 presents the treatment effects compared with placebo in order of mean effectiveness. Steroid combined with physiotherapy showed a beneficial treatment effect compared with placebo with 95% CrIs that did not cross the line of no effect. The 95% CrI of the treatment effect of physical therapy without mobilisation and physiotherapy combined with placebo marginally crossed the line of no effect. The remaining interventions (electroacupuncture, steroid injection, physiotherapy, MUA plus physiotherapy, sodium hyaluronate and arthrographic distension) all crossed the line of no effect. There was no clear difference between the interventions in treatment effects, that is, the CrIs overlapped. These results are also displayed graphically in Figure 22.
| Prior: uniform (0, 5) | |||
| Comparator: placebo | |||
| Treatment | Node number | SMD (95% CrI) | VAS 0–100 mm, MD (95% CrI) |
|---|---|---|---|
| Physical therapy without mobilisation | 4 | –1.71 (–3.46 to 0.04) | –31.3 (–63.3 to 0.7) |
| Steroid + physiotherapy | 6 | –1.58 (–2.96 to –0.42)a | –28.9 (–54.1 to –7.7) |
| Electroacupuncture | 7 | –1.36 (–3.84 to 1.14) | –24.9 (–70.3 to 20.9) |
| Physiotherapy + placebo | 2 | –1.22 (–2.42 to 0.07) | –22.3 (–44.3 to 1.2) |
| Steroid | 5 | –1.15 (–2.46 to 0.19) | –21.0 (–45.0 to 3.5) |
| Physiotherapy | 3 | –0.95 (–3.09 to 1.25) | –17.4 (–56.5 to 22.9) |
| MUA + physiotherapy | 8 | –0.88 (–3.60 to 1.90) | –16.1 (–65.9 to 34.8) |
| Sodium hyaluronate | 10 | –0.21 (–1.90 to 1.47) | –3.8 (–34.8 to 26.9) |
| Arthrographic distension + steroid | 9 | –0.14 (–1.89 to 1.62) | –2.6 (–34.6 to 29.6) |
FIGURE 22.
Caterpillar plot of the SMD estimates and their 95% CrIs compared with placebo. The treatments associated with each number can be identified from Table 56.
Steroid combined with physiotherapy showed a clinically significant as well as statistically significant reduction in pain, with a 95% CrI that corresponded to a clinically significant reduction in pain only: –28.9 mm (95% CrI –54.1 to –7.7 mm). Although the MD was clinically significant for most of the other comparisons, the 95% CrIs contained values that were clinically insignificant. For example, steroid injection had a MD of –21.0 mm (95% CrI –45.0 to 3.5 mm).
Opportunities for comparison with standard meta-analysis are limited as most of the studies were not pooled in a quantitative synthesis. It was possible to compare with the standard meta-analyses for steroid plus physiotherapy versus placebo and steroid versus placebo. The results of the MTC are consistent with the standard meta-analysis of steroid combined with physical therapy versus placebo presented earlier in this report. This standard meta-analysis contained the same two studies included in the MTC. The results of the meta-analysis also showed that steroid combined with physiotherapy resulted in both a statistically and a clinically significant reduction in pain compared with placebo (pooled SMD –0.98, 95% CI –1.43 to –0.52; back transformed MD 17.93 mm, 95% CI –26.2 to –9.5 mm).
Assumptions
In a MTC there are three key assumptions: homogeneity, consistency and methodological similarity.
Homogeneity
The assumption of homogeneity is that trials are sufficiently homogeneous to be quantitatively combined. In the analysis presented, placebo included sham distension, saline injections and placebo laser. Although we believed that it was appropriate to pool different types of placebo, it could be argued that these are not similar enough because of the differing degree of invasiveness. Furthermore, there were differences in the populations of the included studies. For all studies of conservative treatments, it was not reported whether participants had received any previous treatment for frozen shoulder. However, for both studies of invasive treatments (MUA combined with physical therapy, and arthrographic distension with steroid) it was reported that at least some participants had undergone previous treatments for frozen shoulder including corticosteroid injections or some form of physical therapy. This would suggest that the populations of the invasive studies had a longer duration and possibly more treatment-resistant forms of frozen shoulder than those in the conservative treatment studies.
Consistency
The assumption of consistency is that had treatment C been included in a trial comparing interventions A and B then the treatment effect dAC would be equivalent to that obtained from a trial of interventions A and C. 33 Assuming consistency, the treatment effect dAC is the sum of the treatment effects dAB and dBC. However, this assumption requires that the treatments involved belong to closed loops in the network of evidence, such as that seen in a connected polygonal structure. 88 Given that the structure of the network presented here was of a radiating star, with only one loop of evidence, it was not feasible to assess consistency.
Methodological similarity
Of the nine studies included in the analysis, five were of at least satisfactory quality (method of randomisation was adequate and outcome assessment was blinded)16,35,39,41,43 and four were not. 42,51,70,79 To assess the effect of quality on the estimates, a network of studies that were at least of satisfactory quality was evaluated (network 2). However, this model was unstable and, although presented in Appendix 10, the reliability of the data included in the analysis presented is uncertain and does not allow a robust assessment of the impact of quality.
Summary
Nine interventions formed part of a connected network with placebo and the evidence was informed by all nine trials. Twenty-two studies had to be excluded from the MTC for a range of reasons; therefore, the network was a small subset of the available evidence. Steroid combined with physiotherapy showed a beneficial treatment effect compared with placebo with 95% CrIs that did not cross the line of no effect. Furthermore, this treatment, when back transformed to a VAS 0–100 mm scale, showed a clinically significant reduction in pain with 95% CrIs that excluded clinically insignificant values. The remaining treatments had 95% CrIs that crossed the line of no effect and which contained clinically insignificant values. Overall, there was no clear difference in the treatment effects of any of the interventions, that is, the CrIs overlapped. Although the model was a good fit and gave stable estimates, given the arguable heterogeneity of the placebos and of study populations, and the inclusion of poor-quality studies, the reliability of the results of the MTC is uncertain and they should be interpreted with caution.
Patients’ views of interventions for frozen shoulder
Searches of three databases (CINAHL, MEDLINE and PsycINFO) identified 1067 potentially relevant references (Figure 23). Nine full papers were ordered on the basis of screening titles and abstracts. Of these, two papers could not be obtained within the time frame required; however, it is unlikely that these would have met the inclusion criteria. One paper appeared to focus on the clinical effectiveness of steroid injections89 and the remaining paper appeared to examine the quality of life of patients with frozen shoulder but did not appear to address treatments. 90 None of the remaining six papers met the inclusion criteria; therefore, no data were available on patients’ views regarding interventions for frozen shoulder.
FIGURE 23.
Study selection.
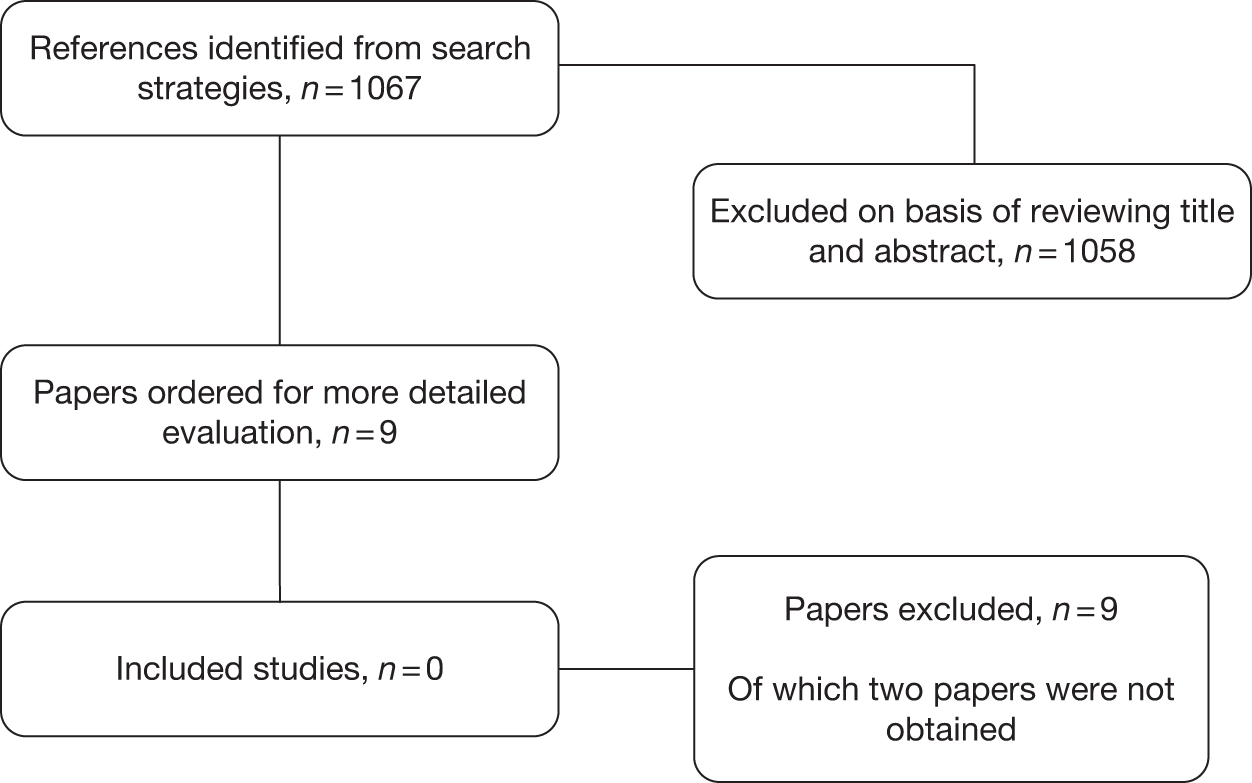
Economic analyses
Previous economic evaluations
We identified one full economic evaluation, conducted in the Netherlands, that met our inclusion criteria as outlined in Chapter 2 (see Inclusion and exclusion criteria). 91 The included economic evaluation, a cost–utility analysis, was conducted as part of a clinical study40 comparing HGMT with LGMT in a Dutch frozen shoulder population. The clinical study, reported in a separate paper, also met the inclusion criteria for the effectiveness review. 40 In summary, the clinical study randomised 100 patients to either HGMT or LGMT. Patients were treated twice weekly for 30 minutes during a period of 12 weeks and were encouraged to attend all treatment sessions. From 6 weeks onward, treatment could be reduced in frequency or stopped if a normal range of movement was noticed by the therapist. The study assessed a number of clinical outcomes including active and passive range of movement, shoulder disability (measured by a shoulder rating questionnaire and the SDQ), pain and quality of life (measured by the SF-36). For a full assessment of the study details and quality see Physical therapy. The study was considered to be of reasonable quality, with some risk of bias.
The aim of the economic evaluation component of the study was to compare the two techniques in terms of costs and QALYs from a societal perspective. In addition, the authors also estimated the impact of frozen shoulder on costs and health. Costs were assessed over the 12-month follow-up period and presented as undiscounted annual costs in euros for the price year 2004. All resource-use information was collected from the patients using quarterly cost questionnaires. Wherever possible Dutch standard prices, which were designed to reflect social costs and standardise economic evaluations, were used. Where standard prices were not available, charges were used. Costs included treatment sessions, alternative medicine sessions, hospitalisations (MUA, acromioplasty), home nursing care, medication, travel costs and non-health-care costs (labour and domestic help). All appropriate costs appear to have been included for a societal perspective.
The measure of benefit used in the analysis was QALYS; these were based on the Short Form questionnaire-6 Dimensions (SF-6D) utility index values, which were estimated using SF-36 data collected alongside the study. The SF-6D provides a means for using the SF-36 data to estimate a preference-based single index measure for health using general population values, thereby allowing the authors to obtain QALYs.
Differences in outcome measures (SF-6D and costs) were tested using double-sided non-parametric bootstrapping. The average estimated QALYs were 0.695 for HGMT and 0.702 for LGMT. The difference of 0.007 in favour of LGMT was reported to be not statistically significant (p = 0.71, 95% CI –0.32 to 0.049). The total reported average annual societal costs were €8809 for HGMT and €6911 for LGMT (a cost difference of €1898 in favour of LGMT). The difference in costs was also reported to be not statistically significant (p = 0.37, 95% CI –€2551 to €5711). The authors concluded that the economic analysis does not allow for evidence-based recommendation regarding the preferred treatment.
A full assessment of the quality of this economic evaluation based on the Drummond checklist and the study data extraction/summary are presented in Appendices 11 and 12 respectively. There were a number of limitations to the study including the lack of an incremental analysis, which would not have resulted in an incremental cost-effectiveness ratio (ICER) but would have informed us that LGMT dominates HGMT (i.e. has higher effectiveness and lower costs). The authors dismiss the difference in QALYs and costs based on statistical inference, which is inappropriate. Further probabilistic sensitivity analysis would have allowed parameter uncertainty to be fully characterised and the expected value of further research to be considered. Overall, despite the limitations the analysis was of reasonable quality and the results, although uncertain, provide us with an indication that LGMT may be a more cost-effective option than HGMT.
Although this one economic evaluation provides us with some information regarding the cost-effectiveness of these two physiotherapy techniques, it does not fully address the question posed. The study was conducted in the Netherlands, was limited to two active physiotherapy techniques, failed to appropriately deal with uncertainty and presented utilities using SF-6D, which cannot easily be compared with the preferred EQ-5D measure because of differences in the descriptive systems and values applied to health states. EQ-5D has become the instrument of choice for many agencies including NICE. 64 The use of one instrument allows more comparability across projects undertaken by these agencies. The small amount of evidence that we are able to glean from this analysis is useful but, given the lack of economic evaluations for other relevant interventions, to further develop and inform decision-making in the UK it was felt that it was essential for us to undertake some additional work.
Decision model
Based on the preliminary examination of the clinical effectiveness data it was anticipated that any modelling undertaken was likely to be in the form of a simple decision tree. As a tool for modelling decision trees provide an effective method for structuring a problem and combining data from various sources. Possible treatment pathways are laid out in a linear manner and appropriate costs and effects are assigned. The final outcome of interest, in this instance QALYs, is attached to the end of the tree. The proportion of participants achieving particular end points is calculated to give a measure of effect. Costs can be attached to both the end points and the treatments/events within the tree. Total costs for each intervention are calculated by summing the costs associated with each pathway. At each point at which a choice is available there is a chance node; a probability conditional on the previous event is attached to each chance node. This node determines the proportion of individuals who progress down each unique pathway in the tree. A simple example is shown in Figure 24.
FIGURE 24.
Example decision tree.
We reviewed the literature to identify existing models in a similar population and sequences or order of treatments, to explore whether treatments (or sequences of treatments) were influenced by phase of condition and to identify any potential source data (e.g. unit costs, resources or utilities) to help populate the model.
As outlined earlier, we identified one economic evaluation as part of a clinical study; we did not identify any modelling studies in this area. Further, very few of the trials in the review of effectiveness consider phase of frozen shoulder or previous treatment history of participants. Because of the limited data available, it was not possible in the clinical synthesis to investigate the impact of phase of frozen shoulder on treatment effectiveness. The only potential ‘proxy’ for phase of condition was duration of frozen shoulder at baseline. The uncertainty associated with such an assumption would need to be carefully considered when drawing conclusions as there is a considerable individual variability in the duration of each phase: it is suggested that the painful freezing stage may last from 2 to 9 months, the adhesive phase from 4 to 12 months and the resolution phase from 12 to 42 months. It was felt that the poor reporting in many of the clinical trials made it difficult to support the assumption that mean duration of frozen shoulder at baseline could be used as a proxy for stage of condition.
Supplementary examination of the available literature failed to provide any clear indication as to potential treatment pathways or treatment sequences. The recent survey of UK health-care professionals suggested that there is a fairly constant view that treatments should depend on the phase of the condition and/or that a step-up approach should be adopted in terms of the degree of treatment invasiveness. 18 With this in mind the clinical advisory group were presented with two alternative modelling scenarios that were thought to be potentially feasible. These were discussed in depth at a group meeting. Option one involved comparing all possible treatments for each phase of the condition (painful, adhesive and resolution) as mutually exclusive options, ignoring any treatment sequencing and phase of condition. This was considered unrepresentative of clinical practice where treatments would often be given simultaneously or in sequence. The results of this type of model would be uninformative and misleading. Option one was rejected as a viable modelling scenario. Option two involved sequencing treatments dependent on their invasiveness. This would potentially allow treatments such as physiotherapy followed by steroid injections followed by MUA to be evaluated. In essence, all treatments, in all possible sequence order, would be considered. This modelling option required an assumption that the clinical effectiveness of each treatment, as obtained from the included clinical trials, would remain the same regardless of where it was placed in a sequence of treatments or what treatments had been received before it. Again, it was the view of the advisory group that this was not a realistic assumption and in fact the effectiveness of treatments is likely to be linked to the position of the treatment in the sequence. The general consensus within the advisory group surrounding this issue was that the assumptions that would be required were, from a clinical perspective, inappropriate.
Given the lack of relevant data from the trials included in the review, the consensus from the advisory group that the required assumptions were clinically inappropriate and the confirmatory position regarding the lack of consensus on recommended treatment pathways in recently published articles,18 it was decided that it was not feasible or useful in this instance to construct a decision-analytic model. In fact, it was perceived that any attempt to undertake modelling would provide hugely uncertain results, which would not allow any conclusions to be made.
Given the lack of UK data on physiotherapy from the identified economic evaluation91 and the lack of economic evaluations of the other relevant interventions we opted to elicit from our clinicians on the advisory group details of the resource use associated with the modalities identified in the review for a frozen shoulder population. The available evidence was then used to obtain utility valuations, which we present as exploratory cost-effectiveness analyses. Our intention, had a de novo model been developed, was to undertake the analysis from an NHS perspective. This perspective has been maintained for the investigative costing exercise. Resources estimated have been valued using national average unit costs (pounds sterling) at 2009–10 prices where possible. Resource use, unit costs and total cost are presented separately, where possible, for each individual treatment. The estimation of resource use gives us only part of the evidence requirements for an economic analysis. The presentation of cost data alone should allow some conjecture about the economic implications of alternative treatments, but does not provide the information required to inform a decision on the cost-effectiveness of these alternatives. To help inform such decisions, the cost estimates obtained from this exercise will be combined with our derived health-related quality of life (HRQoL) estimates (see Mapping from the pain VAS and SF-36 onto the EQ-5D) and presented as ICERs. An ICER is calculated as the difference in the expected cost of two interventions, divided by the difference in the expected QALYs produced by the two interventions. To inform which therapies are of greatest clinical as well as economic value, cost-effectiveness results are usually compared against an acceptable threshold of cost-effectiveness to determine whether a health-care intervention is cost-effective, and a good use of resources, or one that represents poor value for money. The cost-effectiveness threshold for interventions in the English and Welsh NHS is suggested by NICE to be £30,000 per additional QALY; therefore, we have used this threshold. 92
The following sections will outline:
-
resource-use estimation and costing
-
mapping from clinical outcomes to EQ-5D
-
estimation of QALYs and ICERs.
Resource-use estimation
Steroid injections
Six RCTs investigated steroid injections in the treatment of primary frozen shoulder, either alone or in combination with physical therapy. 35,41,42,66–68 Steroid injections can be delivered by a number of different practitioners using different approaches. We have broken these down into a guided injection delivered by an orthopaedic surgeon, a rheumatologist or a radiologist in a hospital setting; unguided injection delivered by a physiotherapist or a GP in a non-hospital setting; and unguided injection delivered by a physiotherapist, an orthopaedic surgeon or a rheumatologist in a hospital setting. Given the nature of the condition and the individual nature of treatments, it was not possible to be precise about the number of injections that an average individual may receive. It was also suggested by our advisory group that the number of patients receiving guided injection within the NHS was likely to be low.
Unguided injections
The clinical experts suggested that it would be rare for individuals to receive more than three injections and on average they would receive two injections spread over various durations of time. Table 57 provides the unit costs of the resources that are used when delivering a steroid injection outside of a hospital setting. We have not included the cost of consumables as these costs are likely to be negligible. The six trials evaluating injections identified in the review of effectiveness assess the use of dexamethasone alone or in conjunction with lidocaine, or triamcinolone alone or in conjunction with lidocaine. We have elected to present costs for both types of steroid although both of the UK trials identified in the review evaluated triamcinolone. 41,67
| Unguided injection | Unit cost | Source |
|---|---|---|
| GP | £35 per consultation | Curtis 200993 |
| Physiotherapist | £39 per hour | Curtis 200993 |
| Dexamethasone 40 mg/lidocaine10 mg | £3.28 per vial | BNF94 |
| Dexamethasone 40 mg | £1.00 per vial | BNF94 |
| Triamcinolone 20 mg | £1.49 per vial | BNF94 |
| Lidocaine 5 ml | 35p | BNF94 |
Alternative scenarios for the delivery of unguided steroid injections were considered and full details of these are presented in Appendix 13. The base case considered three sessions, an initial session of 20 minutes’ duration when an assessment would be undertaken plus two injection sessions of 10–20 minutes’ duration. Using an average of 15 minutes for the two injection sessions, and assuming a combined injection of triamcinolone and lidocaine, the total cost if delivered by a physiotherapist is £36.18. The total cost if delivered by a GP is £108.68. The choice of steroid injection for the base case was based on the UK trials identified as part of the effectiveness review. 41,67 The use of a premixed ready-to-inject vial of dexamethasone plus lidocaine would increase the total cost to £39.06 if delivered by a physiotherapist and £111.56 if delivered by a GP.
Unguided injections are also delivered within a hospital setting. We further estimated that the costs associated with unguided injections in the hospital setting would be £27.17 if delivered in a hospital physiotherapy unit, £88.01 if delivered in an orthopaedic setting and £138.51 if delivered in a rheumatology setting.
Guided injections
Guided injections were assumed to be delivered in a hospital setting by key personnel including an orthopaedic surgeon, a rheumatologist or a radiologist. We estimated that the same number of sessions would be required, using the same type of steroid. The variation in the cost of the guided injections is driven by who delivers the injection. For the base case we have again used the cost of a combined injection of triamcinolone and lidocaine, but present the cost of the premixed dexamethasone plus lidocaine to allow a range to be expressed. We have also assumed that on average two injections will be given over three visits. Alternative scenarios showing varying resource-use levels (suggested alternative numbers of sessions/injections) are presented in Appendix 13. Unit costs for guided injections are presented in Table 58.
| Guided injection | Unit cost | Source |
|---|---|---|
| Orthopaedic outpatient visit, first | £124 | Department of Health95 |
| Orthopaedic outpatient visit, follow-up | £86 | Department of Health95 |
| Rheumatology outpatient visit, first | £211 | Department of Health95 |
| Rheumatology outpatient visit, follow-up | £129 | Department of Health95 |
| Dexamethasone 40 mg/lidocaine 10 mg | £3.28 per vial | BNF94 |
| Dexamethasone 40 mg | £1.00 per vial | BNF94 |
| Physiotherapist outpatient attendance, first | £38 per hour | Department of Health95 |
| Physiotherapist outpatient attendance, follow-up | £29 per hour | Department of Health95 |
| Triamcinolone 20 mg | £1.49 per vial | BNF94 |
| Lidocaine 5 ml | 35p | BNF94 |
The total cost of injections delivered within the orthopaedic setting ranges from £299.68 to £302.56 depending on the steroid injected. Injections delivered in the rheumatology setting have a total cost ranging from £472.68 to £475.56. These alternative delivery settings have been presented as it is believed that these are all viable options depending on available NHS services within a local area.
Steroid injection plus physiotherapy
The resource use associated with the combined treatment of steroid injection plus physiotherapy is again related to the setting in which the treatments are given and the method of steroid delivery. A number of scenarios are presented (see Table 59 for relevant unit costs):
-
Scenario one: six physiotherapy sessions with an active mobilisation component and two unguided steroid injections all delivered by a physiotherapist. It has been assumed that no additional time was required and the total cost is equal to the total cost of active physiotherapy plus the steroid injections. The total cost ranges from £130.43 to £133.31 if delivered by a community physiotherapist, or from £121.43 to £126.81 if delivered by a hospital-based physiotherapist, depending on choice of steroid.
-
Scenario two: six physiotherapy sessions with an active mobilisation component delivered by a community-based physiotherapist, plus two unguided steroid injections delivered by a GP over three visits. The total cost of this scenario ranges from £235.75 to £238.75, depending on choice of steroid.
-
Scenario three: six physiotherapy sessions with a mobilisation component delivered by a hospital-based physiotherapist, plus two unguided steroid injections delivered by a GP over three visits. The total cost of this scenario ranges from £226.75 to £238, depending on choice of steroid.
-
Scenario four: six physiotherapy sessions with a mobilisation component delivered by a community-based physiotherapist, plus two guided steroid injections delivered within the orthopaedic setting. The total cost ranges from £420.23 to £423.31, depending on choice of steroid.
-
Scenario five: six physiotherapy sessions with a mobilisation component delivered by a community-based physiotherapist, plus two guided steroid injections delivered within the rheumatology setting. The total cost ranges from £605.43 to £607.31, depending on choice of steroid.
-
Scenario six: six physiotherapy sessions with an active mobilisation component, plus two guided steroid injections delivered by a hospital-based physiotherapist. The total cost ranges from £160.43 to 165.81, depending on choice of steroid.
| Staff delivering treatment | Unit cost | Source |
|---|---|---|
| Community physiotherapist | £39 per hour | Curtis 200993 |
| Physiotherapist outpatient attendance, first | £38 per hour | Department of Health95 |
| Physiotherapist outpatient attendance, follow-up | £29 per hour | Department of Health95 |
Alternative scenarios could be estimated by varying the number of injections, delivery method and setting. However, the base-case scenarios presented reflect what we believe are current options in the NHS.
Sodium hyaluronate injection
Three trials evaluating sodium hyaluronate were identified in the effectiveness review. 66,69,70 In a recent UK-based survey of treatment recommendations by health-care professionals involved in managing frozen shoulder, sodium hyaluronate injection was recommended by < 1% for treating the early ‘painful’ phase or ‘resolution’ phase of frozen shoulder. 18 It was further suggested by the clinical advisory group that one injection would typically be delivered over two sessions in a hospital setting. The injection was likely to be delivered by a consultant. However, the costs were not available for sodium hyaluronate for provision under the NHS. It was felt that this intervention was likely to be delivered in a hospital setting, but we were unable to ascertain any costs associated to its delivery that would be borne by the NHS.
Physical therapy
The broader range of physical therapies are not available through the NHS and therefore have not been costed. Two categories of physiotherapy were costed, physiotherapy with mobilisation (active) and physiotherapy without mobilisation. It was felt by the advisory group that physiotherapy without mobilisation would not be delivered on its own in an NHS setting, but may be used to complement another therapy. Therefore, for the purpose of resource-use estimation, physiotherapy without mobilisation has been considered only as an add-on delivered alongside other therapies.
Active physiotherapy
It was estimated by the advisory group that physiotherapy with a mobilisation component would on average comprise six sessions each of 30 minutes’ duration, followed by a final review session of 15 minutes’ duration. Full details of all estimates of resource use and any plausible ranges are presented in Appendix 13.
As can be seen from Table 59, the cost of physiotherapy is dependent on the setting in which the treatment is delivered. We have used all variations in hourly cost to calculate a plausible range of total cost for the delivery of active physical therapy. Within a community setting, active physical therapy is estimated to cost a total of £126.75. Within a hospital setting the same treatment is estimated to have an average total cost of £98.75.
Physiotherapy without mobilisation
The review of effectiveness identified one trial that was classified as ‘physiotherapy without mobilisation’. 16 The trial evaluated laser versus placebo laser (plus home exercise). Given that it is unlikely that laser therapy would be delivered without some form of mobilisation (active physiotherapy) within the NHS, we have assumed that laser therapy would be provided in conjunction with an active mobilisation, adding an additional 15 minutes to a 20- to 30-minute session. The additional cost reflects what we believe is current practice, that is, the delivery of both active mobilisation and other passive modalities such as heat treatment in the same treatment session. The additional cost would be either £49.38 or £63.38 depending on the setting.
Acupuncture
Three trials evaluating acupuncture were identified in the effectiveness review. 79–81
It was felt by the clinical advisory group that acupuncture, like other physiotherapies, would in clinical practice rarely be given in isolation of other treatments. We have therefore assumed that acupuncture would be delivered in physiotherapy sessions, in either a hospital or a community setting, over the same duration as active physiotherapy, that is, six sessions of 20–30 minutes’ duration at a total cost ranging from £117.75 to £126.75.
Manipulation under anaesthesia
The review identified four RCTs investigating MUA in the treatment of primary frozen shoulder. 38,39,82,83 The resource implications of MUA were difficult to firmly establish. The actual cost of MUA has been estimated at £424 (range £191–500) based on the HD24C code, which was obtained from the NHS Reference Costs. 95 In addition, there is a need for extensive rehabilitation physiotherapy, which was estimated on average to consist of two 30-minute sessions per week for 4 months, plus a final review, which would take place with the orthopaedic surgeon. Therefore, in total, the estimated cost of MUA is £1446. If steroid injections are also used this cost would increase accordingly.
Arthrographic distension
Three trials evaluating arthrographic distension with steroid injection were identitied in the effectiveness review. 36,43,84 Arthrographic distension is assumed to be delivered in the hospital setting by a radiologist or an orthopaedic surgeon and involves the use of radiological imaging (e.g. fluoroscopy) to ensure that the injection is accurately placed. The procedure was assumed to require around 15 minutes based on the clinical literature. Because there are no UK trials in the clinical review it is unclear how many visits for injections might be needed. We have therefore conservatively estimated the cost as involving one hospital outpatient visit with injection. The estimated cost of this procedure has been derived from the NHS Reference Costs95 at £113 (range £78–133) based on code RA16Z, plus the cost of a combined injection of triamcinolone and lidocaine (£1.49 + £0.35), giving a total of £114.845 (range £79.84–134.84).
Capsular release
Two studies evaluating capsular release were identified in the effectiveness review. 85,86 Capsular release is performed in the hospital setting and delivered by a surgical team, which is likely to comprise anaesthetist, orthopaedic surgeon, anaesthetic nurse, scrub nurse and assistant and recovery nurse. The procedure is likely to take 30–45 minutes, followed by a further 15 minutes for recovery from anaesthetic. The estimated cost of this procedure has been derived from the NHS Reference Costs95 at £1182 (range £787–1489) based on code HB62C. Like MUA, capsular release requires a follow-up of intensive physiotherapy. We have estimated that the physiotherapy following capsular release would be the same as that following MUA. On average this would consist of two 30-minute sessions per week for 4 months, plus a final review that would take place with the orthopaedic surgeon. Therefore, in total, the estimated cost of capsular release is £2204 (range £1809–2511).
Summary
Average costs were estimated for the interventions included in the review. These ranged from £36.18 for an unguided steroid injection to £2204 for capsular release. Where possible, these estimated average costs will be used to inform tentative cost-effectiveness ratios, which will be presented later in the report. They will be combined with HRQoL data estimated by means of a mapping exercise, which is presented in the following section. The aim is to estimate HRQoL by mapping from three clinical outcomes to EQ-5D using an available data set. The main focus of the mapping is to explore the nature and direction of the relationship between different clinical and HRQoL outcome measures. We will then use the EQ-5D estimates to obtain HRQoL outcomes for a subset of clinical trials identified in the review that have presented data on the three relevant outcomes.
Mapping from the pain VAS and SF-36 onto the EQ-5D
The clinical trials identified in the effectiveness review report outcomes using a variety of measures including pain (overall, at rest, on movement, at night), range of movement (e.g. internal and external rotation, elevation), function and disability, quality of life, time to recovery and return to work and recreation, and adverse events. Of the selection of measures used in the trials, none was designed to generate health state utility values, which are required to calculate QALYs, which can then be used to assess cost-effectiveness. Outcomes from treatments affect two basic components – the quantity and quality of life – both of which are captured in the QALY. There are a variety of standardised ‘off-the-shelf’ preference-based measures available to derive estimates of HRQoL, such as the EQ-5D, SF-6D, Health Utilities Index (HUI) and 15-dimension utility measure (15D). 65,96,97,98 These HRQoL values can subsequently be incorporated in economic models for performing cost-effectiveness analyses. They are the preferred benefit measure when undertaking any cost-effectiveness analysis.
An additional search of MEDLINE was conducted to identify health quality of life outcomes that could be used to inform any modelling. The lack of use of generic preference-based measures in existing frozen shoulder clinical studies is a potential current barrier to populating economic models with the best evidence on effectiveness and to thereby allow broader comparisons across different diseases.
The included economic evaluation presented HRQoL in the form of the SF-6D, but it was felt that given the general preference for EQ-5D and the possibility of bias in the clinical study further investigation was warranted. The EQ-5D is the recommended instrument of choice for use in cost-effectiveness analyses by NICE and is the most commonly used instrument.
In the context of this clinical review, it is not clear why HRQoL has not been included in the battery of outcome measures in the clinical trials in frozen shoulder populations to date. Possible reasons why such instruments have not been administered may be because it is not practical or they are too time-consuming for patients to complete in trials in addition to all of the other forms/questionnaires that are used – often across multiple time points – or even because studies were not initially designed to focus on economic questions. It may be that the general perception is that these generic instruments are not sensitive enough.
In a situation such as this, in which such instruments have not been used in clinical studies, a pragmatic alternative is to apply a mapping function to convert non-preference-based health data into one of the generic preference-based measures. Mapping essentially provides a statistical equation that allows the estimation of health utilities and the subsequent calculation of QALYs in clinical studies that do not use any preference-based HRQoL instruments. In the published literature, the EQ-5D was recently reported as being the most popular target measure used for mapping. 96
On the other side of the mapping equation, the most widely used generic measures/instruments that have been mapped include the SF-36 and SF-12 health surveys, which offer the option of presenting scores across eight dimensions or two summary measures derived from the eight dimensions. These two summary measures are the physical component and the mental component. 99,100 Also, a number of shoulder-specific questionnaires (scores) have been shown to be significantly correlated with the EQ-5D in the literature, in particular the SDQ. 55 Of particular importance in the current clinical effectiveness review were patient-assessed pain intensity, function and disability, quality of life and range of movement. Pain was the most common outcome measure in the included clinical studies and, for instance, in the MTC analysis, pain was analysed based on the following preference hierarchy: overall pain, pain at night, pain on activity, any other type of pain (excluding pain at rest) and pain at rest. Furthermore, and within the context of the current clinical effectiveness review, three of the studies reported the SF-36 as an outcome measure. As mentioned earlier in the clinical sections of the report, two studies meeting the full inclusion criteria reported the mean physical and mental component summary scores. 31,47 One additional study, which did not meet the analysis criteria, reported the means for the eight dimensions of the SF-36. 81 The outcomes were reported over the time horizon of 4 weeks and as a result the study was excluded from further analysis. Therefore, in line with the decision made as part of the clinical review we elected not to map from this evidence base, but rather to utilise the potentially more reliable component summary scores that had been reported in the other two trials. 35,51
In a recent review of mapping studies, simple additive models that employed a utility index score as the dependent variable and the main effects of either total or instrument dimension scores as independent variables performed almost as well as more complex models comprising many variables. 99 With this in mind, and as the main focus of this analysis was to explore the nature and direction of the relationship between different outcome measures, we chose to follow a simple approach and use mean pain VAS score (as it was a main outcome in the clinical review) and mean SF-36 physical and mental component summary scores (as it is one of the most widely used generic quality-of-life measures in mapping studies). These were important outcomes in the clinical effectiveness review.
To undertake an exploratory mapping analysis of this nature we required a data set in which the two measures being mapped had been administered specifically to a frozen shoulder population. We were unable to obtain a data set of this nature for a frozen shoulder population; however, we obtained a UK data set involving patients with rotator cuff disease from the SAPPHIRE RCT. 101 Both measures chosen for the exploratory mapping analyses were already calculated and readily available in the SAPPHIRE data set. 102
The selection of outcomes on which to map was a pragmatic decision based on the data available in the SAPPHIRE data set. The relationship is estimated using a range of techniques and statistical specifications. Furthermore, we compare the mapping approaches tested in terms of their predictive performance in the SAPPHIRE trial population. Within the context of the current review, we conducted an analysis to explore whether or not relationships between the pain VAS and SF-36 and the EQ-5D exist and the nature of those relationships. In doing so, we considered that we might be able to demonstrate the need for the collection of utility EQ-5D data in future clinical trials and offer a potential alternative approach for frozen shoulder populations for whom there is currently a lack of HRQoL data.
The SAPPHIRE trial compared the efficacy of corticsosteroid (40 mg/ml triamcinolone acetonide) versus local anaesthetic (1% lidocaine) injection in subjects presenting to GPs with (acute) shoulder pain (in one or both shoulders) persisting no longer than 12 months, and with diagnosis of rotator cuff tendonitis. 102 The trial evaluated the impact of giving GPs additional training in diagnosing rotator cuff pathologies and in performing shoulder injections, or no training. Trial participants were followed up at 1 and 3 months, with a final follow-up at 6–12 months post randomisation. The outcome measures were the SF-36, EQ-5D, British SDQ103 and VAS to assess the extent of shoulder pain (at night, in daytime at rest, in daytime on movement).
The population characteristics for the two studies included in the clinical review that reported mean overall pain VAS scores,16,51 using a 0- to 100-mm scale, and the two studies included in the clinical review that reported mean SF-36 physical and mental component summary scores35,51 are reported in Table 60. The population characteristics of the participants in the SAPPHIRE trial102,104 are also shown in Table 60.
| Study | Treatment arms | Duration of shoulder conditiona | Patient characteristics | EQ-5D (mean) | SF-36 PCS (mean) | SF-36 MCS (mean) | Pain VAS (mean) (0–100 mm) | |
|---|---|---|---|---|---|---|---|---|
| Age (years) | % female | |||||||
| Dogru (Turkey)51 | 2 | Shoulder pain of at least 3 months’ duration | 53.9–56.8 (55.4 total) | 57 (total) | – | 36.6–38.9 | 42–43.5 | 78–80.8 |
| Stergioulas (Greece)16 | 2 | Duration of frozen shoulder at baseline of 26.5–27.1 weeks | 55.5–56.8 | 37 | – | – | – | 67.03–70.9 |
| Carette (Canada)35 | 4 | Adhesive capsulitis of < 1 year’s duration, mean duration of current episode of shoulder pain 20.3–22.1 weeks | 54.2–56.5 | 46.2–66.7 | – | 35.2–37.6 | 43.1–49.8 | – |
| SAPPHIRE trial (UK)102,104 | 2 | Shoulder pain persisting for ≤ 12 months, median duration of shoulder pain 6.5–8 weeks | 55–59 | 51–55 | 0.453–0.512 | 35.96–38.89 | 44.64–46.54 | 68.43b |
The three studies included in the clinical effectiveness review reporting mean pain VAS scores and/or mean SF-36 physical component summary (PCS) and mental component summary (MCS) scores and the SAPPHIRE trial include populations with broadly similar proportions of men and women and of a similar age and duration of shoulder condition or pain and with similar mean pain VAS and SF-36 PCS and MCS scores.
The mapping presented here is an exploratory exercise, based on a patient population with very similar baseline characteristics as the frozen shoulder population. We examine the relationship between the EQ-5D and pain as measured by a VAS 0–100 mm and the EQ-5D and SF-36 (specifically the SF-36 PCS and MCS) health surveys.
Methods
The following sections present the methods used in estimating HRQoL data to inform our tentative analysis and potentially inform other economic evaluations.
Instruments
Pain was measured on a scale from 0 to 100 mm in both the studies included in the clinical review (see Table 60) and the SAPPHIRE data set. Values anchored on the VAS at 0 generally represent no pain whereas values anchored at 100 generally represent the worst pain possible. Both studies have data available as mean values. When more than one type of pain was measured in any single study, data were selected for the mapping exercise based on the pain hierarchy as reported earlier in the clinical sections of this report.
The SF-36 questionnaire covers different aspects of HRQoL and the items can be pooled in eight different subscales: physical functioning, role – physical, body pain, general health, vitality, social functioning, role – emotional, and mental health. 107 Patient scores are rated on a 0 (worst)–100 (best) scale for each subscale. These eight subscales can be pooled in two different clusters: the PCS and the MCS. The calculation of the SF-36 summary scores (PCS and MCS) is based on a published algorithm which uses a standardised scoring system with mean = 50 and SD = 10. The two component summary scores are used for the current regression analyses.
The EQ-5D is a five-item questionnaire encompassing the domains of mobility, self-care, usual activities, pain/discomfort and anxiety/depression. Respondents are asked to indicate whether they have no problems, some problems or major problems in each domain, with responses scored as 1, 2 and 3 respectively. To produce a combined utility health score, in which 1 is full health and 0 is equivalent to being dead, individuals’ answers about the extent of their problems in each of the five health domains are then weighted. The weights, based on a large UK study, are used to take into consideration population preferences for each health domain. 65 The resulting descriptive system defines 243 health states. The worst-valued health state is –0.594 (worse than death), whereas the best-valued health state is 1.0 (perfect health).
Data set
We used the SAPPHIRE trial data set and measurements at 3 months for the primary (base-case) analysis. The number of participants with complete outcomes data at 3 months was (1) 141 for the EQ-5D and pain VAS and (2) 133 for the EQ-5D and SF-36 PCS and MCS. The time point of 3 months was chosen as it represented a clinically relevant time point in the frozen shoulder trials included in this review. The data sets for (1) and (2) were then each divided into two by taking a random sample of 60% of the data and separating that data from the remaining 40% to provide an estimation data set and a validation data set respectively. In the case of (1), the EQ-5D and pain VAS, 85 measurements were available for the estimation data set and 56 measurements for validation. For (2), the EQ-5D and SF-36 PCS and MCS, 80 measurements were available for the estimation data set and 53 measurements for validation.
It should be noted that the collection of new data to validate a model’s predictions is the preferred approach. 108 However, in many situations this is neither practical nor possible and an alternative approach is to split the data in hand into two parts. The first part is used to estimate the model coefficients (estimation sample) and the second part is used to assess the prediction accurary of the model (validation sample). Thus, data splitting provides a data set to measure the ‘in-use’ (or within-sample) prediction accuracy of the model and simulates the complete or partial replication of the data set, in this case the SAPPHIRE trial data set.
Statistical analysis
Ordinary least squares (OLS) regression was used as the main statistical technique in the analysis. The target outcome (dependent variable) was the overall EQ-5D index score, and the explanatory (independent) variables were (1) the pain VAS and (2) the SF-36 PCS and MCS scores. Based on the theoretical limitations of OLS modelling approaches reported in the empirical econometric literature,100,109,110 the current exploratory investigation estimates the EQ-5D index scores also using TOBIT (an ecometric model named after its developer James Tobin,111 which allows for upper censoring of the dependent variable, EQ-5D, at 1.0; a caveat is that it produces biased estimates in the presence of heteroscedasticity or non-normality) and CLAD (censored least absolute deviations, which produces consistent estimates in the presence of heteroscedasticity or non-normality, whereas also allowing for censoring at either the upper or the lower bound).
All regressions were conducted in Stata version 11 (StataCorp LP, College Station, TX, USA) and specifically included the estimation of the following models and included independent variables. The general regression models for (1) the EQ-5D and pain VAS and (2) the EQ-5D and SF-36 PCS and MCS were defined as:
where EQ-5D represents the EQ-5D health state index value, i = 1,2,. . ., n represents individual respondents, painVAS represents the pain score, α is the intercept and ε is the error term, and
where EQ-5D represents the EQ-5D health state index value, i = 1,2,. . ., n represents individual respondents, PCS represents the SF-36 PCS score, MCS represents the SF-36 MCS score, α is the intercept and ε is the error term. Variations of the first model were explored by including squared terms. Variations of the second model were explored by including squared and interaction terms.
We conducted three sets of analyses using alternative approaches relating to the choice of estimation and validation samples from the main SAPPHIRE data set:
-
Regression models using main effects with and without squared terms and interaction term (using individual-level data at 3 months):
-
– for (1), EQ-5D and pain VAS, total n = 141, estimation data set = 85 (60%), validation data set = 56 (40%)
-
– for (2), EQ-5D and SF-36 PCS and MCS, total n = 133, estimation data set = 80 (60%), validation data set = 53 (40%).
-
-
Regression models using main effects with and without squared terms and interaction term (using individual-level data at 1, 3 and 12 months):
-
– for (1), EQ-5D and pain VAS, total n = 491, estimation data set = 295 (60%), validation data set = 196
-
– for (2), EQ-5D and SF-36 PCS and MCS, total n = 467, estimation data set = 280 (60%), validation data set = 187.
-
Data for all time points (1, 3 and 12 months) were pooled to increase the sample size and the statistical precision of the estimates with correction for the clustering of several responses per patient by using the option ‘cluster’ in the regress command in Stata version 11. 112
-
Prediction models using the main effects with and without squared terms and interaction term (using models estimated from 3-month data and used to predict EQ-5D scores at 12 months):
-
– for (1), EQ-5D and pain VAS, validation data set = 171
-
– for (2), EQ-5D and SF-36 PCS and MCS, validation data set = 163.
-
Model fit (explanatory performance) and predictive/mapping performance were based on assessments of the adjusted R2, mean error (ME), mean absolute error (MAE) and root-mean-square error (RMSE), as well as the percentage of individual predictions within 0.10, 0.05 and 0.01 of their actual EQ-5D utility values (arbitrary cut-off values). Descriptive statistics and plots of actual versus predicted EQ-5D scores were also generated to examine the pattern of errors across EQ-5D scale, for example to assess whether they were evenly distributed across poor/better health states.
Results from the mapping exercise
Pattern of EQ-5D, pain visual analogue scale and SF-36 PCS and MCS scores
The mean EQ-5D scores (ordered according to health state severity) in the SAPPHIRE population vary over a wide range (e.g. from negative utility values to ‘perfect health’ as indicated by utility values of 1.0; Table 61 and Figure 25). In contrast, the SAPPHIRE trial mean pain VAS scores vary over a narrower range (e.g. highest mean VAS score = 60, when a maximum pain VAS of 100 is possible). An increasing EQ-5D score (i.e. better health status) appears to be generally accompanied by decreasing pain VAS scores (i.e. lower scores = less pain).
| EQ-5D grouping | n | EQ-5D (mean) | Pain VAS (mean) |
|---|---|---|---|
| < 0 | 8 | –0.05 | 18.13 |
| 0–0.249 | 15 | 0.08 | 60 |
| 0.25–0.499 | 1 | 0.26 | 50 |
| 0.5–0.699 | 52 | 0.63 | 41.87 |
| 0.7–0.799 | 24 | 0.75 | 40.17 |
| 0.8–0.899 | 26 | 0.83 | 38.88 |
| 0.9–1.0a | 15 | 1.0 | 34.67 |
| Full index | 15 | 1.0 | 34.67 |
| Overall | 141 | 0.63 | 37.63 |
FIGURE 25.
Pattern of EQ-5D scores and pain VAS scores within the SAPPHIRE data set (for complete responses) at 3 months’ follow-up (base case).
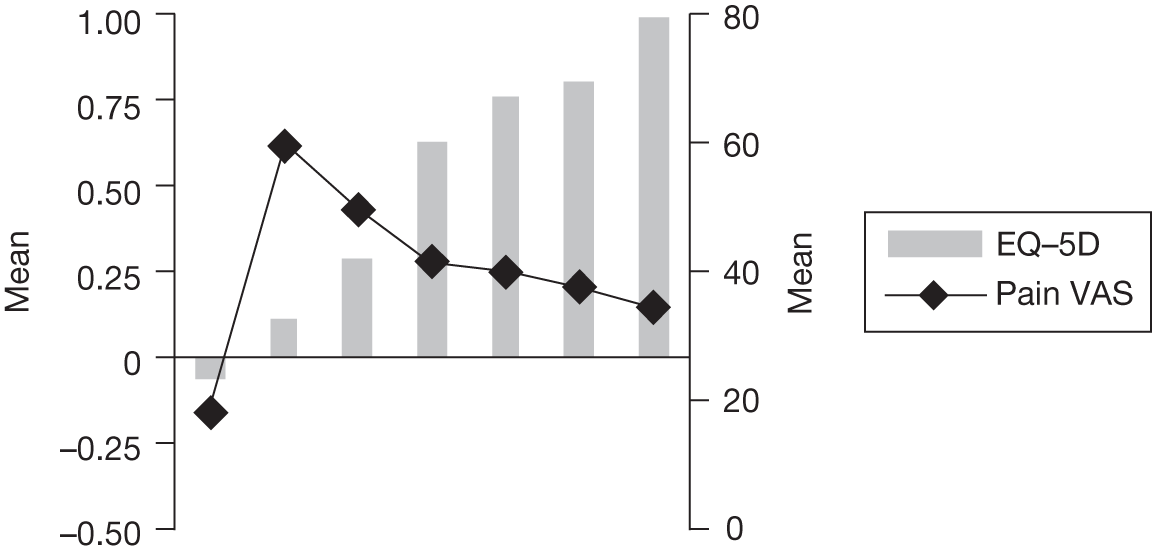
Figure 26 and Table 62 show that mean EQ-5D scores (ordered according to health state severity) in the SAPPHIRE population vary over a wide range. In contrast, the SAPPHIRE trial mean SF-36 PCS and MCS scores vary over a relatively narrow range. An increasing EQ-5D score (i.e. better health status) appears to be generally accompanied by increasing SF-36 PCS and MCS scores (i.e. higher scores = a better quality of life).
FIGURE 26.
Pattern of EQ-5D scores and SF-36 PCS and MCS scores within the SAPPHIRE data set (for complete responses) at 3 months’ follow-up (base case).
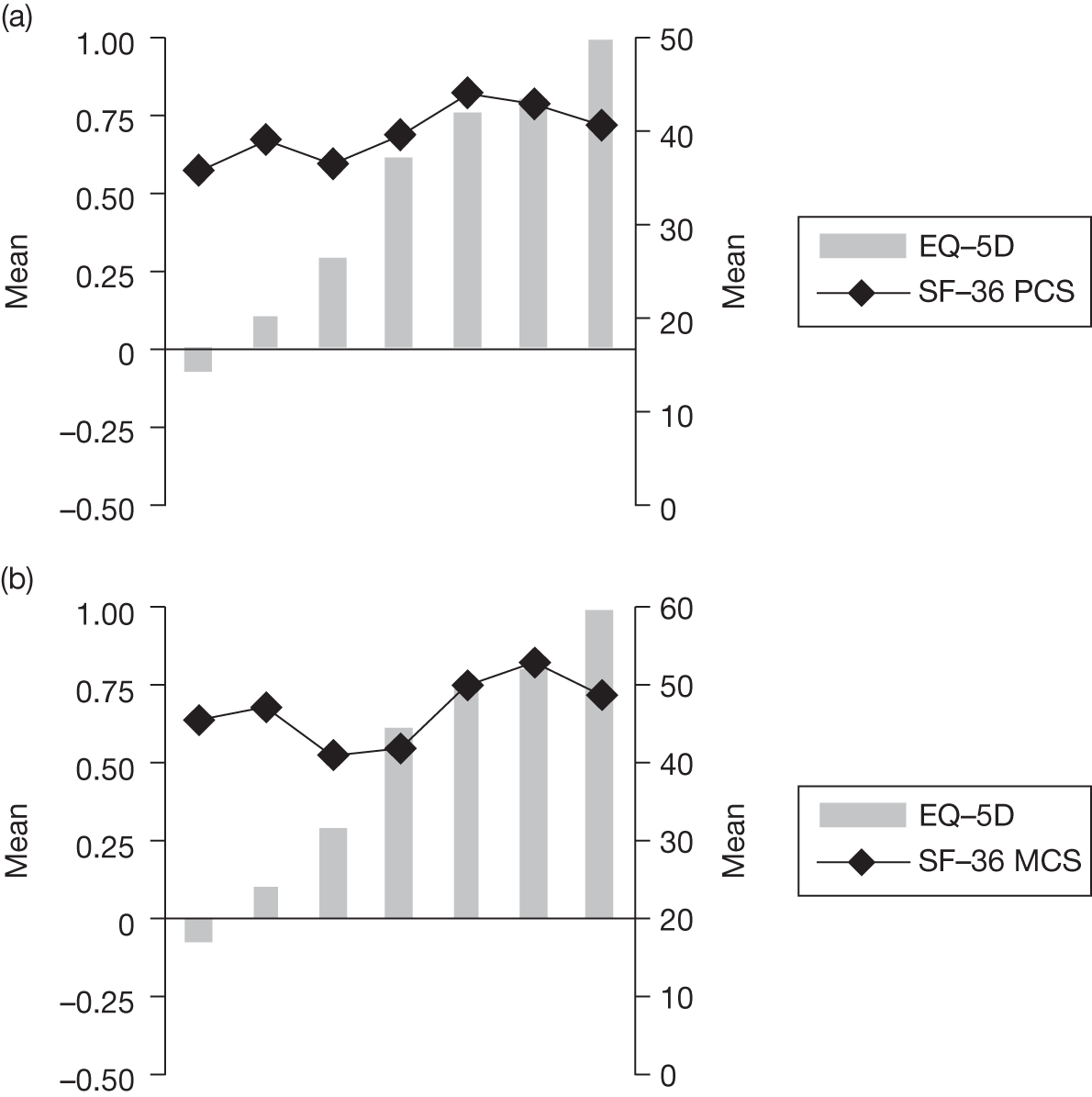
| EQ-5D grouping | n | EQ-5D (mean) | SF-36 PCS (mean) | SF-36 MCS (mean) |
|---|---|---|---|---|
| < 0 | 10 | –0.07 | 35.96 | 45.70 |
| 0–0.249 | 18 | 0.11 | 39.18 | 47.35 |
| 0.25–0.499 | 1 | 0.26 | 36.64 | 41.14 |
| 0.5–0.699 | 44 | 0.63 | 39.68 | 42.01 |
| 0.7–0.799 | 21 | 0.75 | 44.07 | 50.23 |
| 0.8–0.899 | 28 | 0.82 | 42.91 | 53.2 |
| 0.9–1.0a | 0 | NA | NA | NA |
| Full index | 11 | 1.0 | 40.68 | 48.95 |
| Overall | 133 | 0.59 | 40.79 | 47.21 |
Regression analyses
Table 63 shows the results of the regression of the pain VAS scores onto the EQ-5D using the 3-month data set (base case). The explanatory power and fit of both OLS models was poor, with an adjusted R2 ranging from –0.0014 to 0.0081. The TOBIT and CLAD models including main effects and squared terms were still relatively poor although they produced the smallest MAEs (0.2158 and 0.1877 respectively) compared with the two OLS models. However, all models predicted only between 16% and 23% of scores to within 0.05 of actual EQ-5D utility values.
| Model 1 | Model 2 | Model 3 | Model 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLS | OLS | TOBIT | CLAD | ||||
| Independent variables | Pain VAS night | Pain VAS night, squared terms | Pain VAS night, squared terms | Pain VAS night, squared terms | ||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | 0.5540896 | 0.0524505 | 0.5353785 | 0.0663995 | 0.5413921 | 0.0725511 | 0.6872609 | 0.0621687 |
| pVASnight | 0.0013853 | 0.0010672 | 0.0033283 | 0.0043293 | 0.0042145 | 0.0047531 | 0.0009522 | 0.0036473 |
| pVASnight*pVASnight | –0.000023 | 0.0000497 | –0.0000312 | 0.0000546 | –0.000013 | 0.0000385 | ||
| Adjusted R2 | 0.0081 | –0.0014 | – | – | ||||
| ME | 0.03958 | 0.0402 | 0.0206 | –0.0479 | ||||
| MAE | 0.21718 | 0.2218 | 0.2158 | 0.1877 | ||||
| RMSE | 0.276951 | 0.280038 | 0.280351 | 0.279724 | ||||
| |Difference|a | ||||||||
| |∆|≤ 0.10 | 14 | 25% | 14 | 25% | 16 | 28% | 28 | 49% |
| |∆|≤ 0.05 | 9 | 16% | 10 | 18% | 9 | 16% | 13 | 23% |
| |∆|≤ 0.01 | 3 | 5% | 1 | 2% | 2 | 4% | 8 | 14% |
| Actual | Predict | Predict | Predict | Predict | ||||
| Mean | 0.64429 | 0.60470 | 0.60413 | 0.62368 | 0.69218 | |||
| SD | 0.279102 | 0.043380 | 0.048668 | 0.057483 | 0.011428 | |||
| Min. | –0.140 | 0.554 | 0.535 | 0.541 | 0.652 | |||
| Max. | 1.000 | 0.693 | 0.656 | 0.684 | 0.705 | |||
| Range | 1.140 | 0.139 | 0.120 | 0.142 | 0.69218 | |||
Appendix 14 shows the results of the regression of the pain VAS scores onto the the EQ-5D using individual-level data at 1, 3 and 12 months. Compared with using data from 3 months only, the explanatory power and fit improves but only slightly. The two OLS models generated an adjusted R2 of 0.1009. Errors were generally similar across all models with RMSEs ranging from 0.26403 to 0.26689. Models 1–4 were able to predict between 20% and 26% of the scores to within 0.05 of actual EQ-5D utility values. The predictive performances of all models were generally similar.
Appendix 14 also shows the results of the regression of the pain VAS scores onto the EQ-5D using models estimated from 3-month data and used to predict EQ-5D scores at 12 months. Predicting EQ-5D scores at 12 months based on the mapping functions derived from the 3-month data set did not perform as well as the analyses using data from all time points. Models 1–4 were able to predict between 11% and 21% of the scores to within 0.05 of actual EQ-5D utility values.
Table 64 shows the results of the regression of the SF-36 PCS and MCS scores onto the EQ-5D using the 3-month data set (base case). The explanatory power and fit of the three OLS models was poor, with an adjusted R2 ranging from 0.0147 to 0.0830. The simplest OLS model including only main effects produced the smallest MAEs (0.21135) compared with the other models. However, models 1–5 were only able to predict between 13% and 25% of the scores to within 0.05 of actual EQ-5D utility values.
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLS | OLS | OLS | TOBIT | CLAD | |||||
| Independent variables | PCS MCS | PCS MCS, squared terms | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | |||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | 0.2842682 | 0.1811317 | –0.6233533 | 0.5770837 | –1.552434 | 0.749881 | –1.814838 | 0.7887539 | –0.6770489 | 1.035821 |
| PCS | 0.004559 | 0.003425 | 0.0018081 | 0.0201721 | 0.0249701 | 0.0232969 | 0.0286508 | 0.0244527 | 0.0157259 | 0.0273862 |
| MCS | 0.0026323 | 0.0030736 | 0.0516204 | 0.0221907 | 0.073418 | 0.0246642 | 0.0831682 | 0.0259928 | 0.0399689 | 0.0316185 |
| PCS*PCS | 0.0000186 | 0.000258 | 0.0000464 | 0.0002541 | 0.000041 | 0.0002683 | 0.0000152 | 0.0002815 | ||
| MCS*MCS | –0.0005561 | 0.0002495 | –0.0005877 | 0.0002459 | –0.0006711 | 0.0002584 | –0.0002875 | 0.0002772 | ||
| PCS*MCS | –0.00052 | 0.0002744 | –0.0005816 | 0.0002883 | –0.0002641 | 0.0003168 | ||||
| Adjusted R2 | 0.0147 | 0.0513 | 0.0830 | – | – | |||||
| ME | 0.00025 | –0.00220 | 0.01147 | –0.00645 | –0.07296 | |||||
| MAE | 0.21135 | 0.23023 | 0.242658 | 0.24848 | 0.215912 | |||||
| RMSE | 0.27294 | 0.301496 | 0.314054 | 0.324165 | 0.300666 | |||||
| |Difference|a | ||||||||||
| |∆|≤ 0.10 | 17 | 32% | 19 | 36% | 19 | 36% | 18 | 34% | 24 | 45% |
| |∆|≤ 0.05 | 7 | 13% | 12 | 21% | 11 | 21% | 9 | 17% | 13 | 25% |
| |∆|≤ 0.01 | 2 | 4% | 3 | 6% | 2 | 4% | 2 | 4% | 2 | 4% |
| Actual | Predict | Predict | Predict | Predict | Predict | |||||
| Mean | 0.59623 | 0.59598 | 0.59842 | 0.58476 | 0.60267 | 0.66918 | ||||
| SD | 0.274200 | 0.070105 | 0.119652 | 0.150510 | 0.169153 | 0.118474 | ||||
| Min. | –0.020 | 0.444 | 0.128 | 0.044 | –0.03 | 0.258 | ||||
| Max. | 1.000 | 0.700 | 0.734 | 0.799 | 0.843 | 0.799 | ||||
| Range | 1.020 | 0.256 | 0.607 | 0.755 | 0.846 | 0.541 | ||||
Appendix 14 shows the results of the regression of the SF-36 PCS and MCS scores onto the EQ-5D using individual-level data at 1, 3 and 12 months. Compared with using data only from 3 months, the explanatory power and fit of all models improves. The three OLS models generated an adjusted R2 ranging from 0.3840 to 0.4284. Errors are smaller, with the smallest MAE and RMSE for model 5 (CLAD) of 0.1814 and model 1 (OLS) of 0.1889 respectively. Models 1–5 were able to predict between 70% and 78% of the scores to within 0.05 of actual EQ-5D utility values but only 41–55% to within 0.01 of actual EQ-5D utility values. The predictive performances of all models are generally similar.
Appendix 14 also shows the results of the regression of the SF-36 PCS and MCS scores onto the EQ-5D using models estimated from 3-month data and used to predict EQ-5D scores at 12 months. Predicting EQ-5D scores at 12 months based on the mapping functions derived from the 3-month data set was somewhat less accurate than the analyses using data from all time points. Models 1–5 were able to predict between 31% and 58% of the scores to within 0.05 of actual EQ-5D utility values.
Finally, the data suggest that all models (whether they are based on pain VAS or SF-36 PCS and MCS scores) predict better for less severe health states, but overpredict the value of more severe EQ-5D states.
Potential implications for application to frozen shoulder populations
The population used in mapping studies should cover the range of clinical and demographic characteristics of the population on which the mapping function is ultimately to be applied. The two populations should be sufficiently homogeneous that relationships between outcomes in one population can be used to infer relationships about outcomes in the other population. The current exploratory analyses were undertaken on a UK data set of patients with rotator cuff disease and not on the specific population of interest in the current review, that is, frozen shoulder patients. However, we considered the patients in the SAPPHIRE trial data set to exhibit similar enough characteristics to patients with frozen shoulder (see Table 60) to be potentially useful for drawing inferences for this population from the mapping analyses. Our findings indicated that, of the models estimated, the simple OLS model including only the main effects of (1) pain VAS mapped onto the EQ-5D and (2) SF-36 PCS and MCS mapped onto the EQ-5D performed just as well as some of the more complex models/approaches. If we apply this mapping function to the two studies in the review that did report data for (1) mean pain VAS16,51 and (2) mean SF-36 PCS and MCS scores35,51 then hypothetically it becomes possible to derive utilities based on the EQ-5D, which in principal can then be used in performing assessments of the cost-effectiveness (cost–utility) of two or more treatments in patients with frozen shoulder.
Table 65 illustrates the results of this process using data from the two frozen shoulder trials in the clinical effectiveness review that reported mean pain VAS scores16,51 (see Appendix 7 for full details and data extraction tables for these studies). Using the OLS1 model function/equations we mapped the reported mean pain VAS at baseline and after 3 months of follow-up onto the EQ-5D. The corresponding EQ-5D scores can then be predicted at baseline and after 3 months of follow-up.
| Study | Intervention | EQ-5D estimated from mapping | Mean QALYs over 3 months | Estimated cost per patient | QALY difference | Cost difference | ICER | |
|---|---|---|---|---|---|---|---|---|
| Baseline | 3 months | |||||||
| Dogru (Turkey)51 | Ultrasound + physiotherapy | 0.518 | 0.695 | 0.15163 | £148.13 | –0.00163 | 0 | |
| Sham ultrasound + physiotherapy | 0.527 | 0.699 | 0.15325 | £148.13 | Benefit favours | |||
| Stergioulas (Greece)16 | Physical therapy (laser) | 0.55 | 0.687 | 0.15463 | £49.38 | 0.00350 | 0 | Benefit favours |
| Control (placebo laser) | 0.562 | 0.647 | 0.15113 | £49.38 | ||||
Table 66 illustrates the same using data from the two frozen shoulder trials in the clinical effectiveness review that reported the SF-36 PCS and MCS35,51 (see Appendix 7 for full details and data extraction tables for these studies).
| Study | Intervention | EQ-5D estimated from mapping | Mean QALYs over 3 months | Estimated cost per patient | QALY difference | Cost difference | ICER | |
|---|---|---|---|---|---|---|---|---|
| Baseline | 3 months | |||||||
| Carette (Canada)35 | Steroid injection + physiotherapy | 0.51 | 0.673 | 0.14788 | £121.43 | Dominated | ||
| Physiotherapy + placebo injection | 0.587 | 0.702 | 0.16113 | £117.75 | Dominated | |||
| Steroid injection | 0.583 | 0.739 | 0.16525 | £36.18 | 0.01275 | £3.68 | £288.63 | |
| Placebo injection | 0.571 | 0.649 | 0.15250 | £32.50 | ||||
| Dogru (Turkey)51 | Ultrasound + physiotherapy | 0.564 | 0.647 | 0.15138 | £148.13 | 0.00537 | 0 | Benefit favours |
| Sham ultrasound + physiotherapy | 0.522 | 0.646 | 0.14600 | £148.13 | ||||
Based on these EQ-5D estimates QALYs have been approximated by applying the area under the curve (AUC) method, which is implemented by summing the areas of the geometrical shapes obtained by linearly interpolating between utility scores over the study period. Using the UK estimated costs obtained from our resource-use assessment we have presented tentative cost-effectiveness results, which may allow some inference about the cost-effectiveness of some of these interventions.
These calculations are based on the assumption that no further downstream costs will be incurred by the patient. Also, because of the methods we are using we have only been able to compare interventions within trials, not across trials. Any future evaluation should consider all relevant interventions within the same analysis.
Tables 65 and 66 show the baseline and 3-month EQ-5D scores that have been obtained using the estimates provided by the mapping and the QALYs obtained based on these estimates. The costs presented in these tables represent the cheapest UK scenarios presented in Resource-use estimation. In some instances, because of interventions requiring the same resource and therefore costing the same, no ICERs have been presented. For the study evaluating interventions that incur different costs,35 the incremental results suggest that physiotherapy (with placebo injection) and steroid injection with physiotherapy were both dominated (less effective and more costly) by steroid injection alone. Steroid injection has an ICER of £288.63, which is well below the widely used threshold of £30,000.
These results are tentative. Their purpose is to try to help inform further research, not to directly inform decision-making. Actual QALY gains, and incremental cost per QALY estimates, can differ based on how the utilities were derived: directly from preference-based measures or predicted on the basis of mapping. 113 This is neatly demonstrated by the results of the Dogru study, which, depending on whether mapping from the VAS or SF-36, suggest QALY gains in favour of alternative interventions. 51 Furthermore, the choice of direct measure used in cost–utility analyses matters (e.g. EQ-5D, SF-6D, HUI, 15D) and may, in an economic evaluation, lead to different treatment options being considered more or less cost-effective relative to one another. 114 These are all issues outside the scope of this research.
Summary
There were no studies identified in the clinical review that provided utilities data needed for generating cost per QALY estimates in assessments of the cost-effectiveness of alternative treatment options for frozen shoulder.
-
The exploratory analysis presents an alternative approach to deriving utility estimates by mapping from outcomes measured in a trial onto a preference-based measure of QoL.
-
For mapping to be of any value, a degree of overlap is required between the descriptive systems of the measures being used. The results of the exploratory analysis suggest that a statistical association between the EQ-5D and pain VAS and the EQ-5D and the SF-36 PCS and MCS measures potentially exists, specifically that a decreasing pain VAS score (less pain) is accompanied by an increasing (better) EQ-5D score and similarly that increasing (better) SF-36 PCS and MCS scores are accompanied by an increasing (better) EQ-5D score.
-
There is insufficient evidence available to make conclusions about the effectiveness of treatments for frozen shoulder in terms of QoL outcomes based on patient preference measures.
Chapter 4 Discussion
The aims of the project were to undertake a systematic review of the clinical effectiveness and cost-effectiveness of treatments used for the management of idiopathic frozen shoulder, by stage of condition, in order to make recommendations for clinical practice and identify gaps in the evidence. The project also aimed to develop a decision-analytic model to estimate the cost-effectiveness of the alternative treatment options and undertake value of information analysis to assess the potential value of future research on interventions for frozen shoulder.
Both conservative and more invasive treatment options were included in the review. Conservative treatments are generally considered to be education about the condition with advice for self-management, physiotherapy and corticosteroid injection, either alone or alongside physiotherapy. Physiotherapy and steroid injection are delivered in a range of NHS settings including primary care, delivered mainly by physiotherapists and GPs, secondary tier specialist musculoskeletal clinics and a hospital setting. More invasive treatments that were included in the review were distension, MUA and capsular release, which are delivered in a hospital setting. Sodium hyaluronate does not appear to be commonly used in the NHS but there appeared to be a growing literature on its use and it was therefore included. This was classified with the more invasive treatments, mainly because it would be delivered in a hospital setting.
Principal findings
Clinical effectiveness
Data from studies with a low risk of bias were sparse, in particular for the more invasive treatments. For most comparisons the necessary data were not available or it was inappropriate to statistically pool studies because of their variability. Primarily a narrative synthesis was undertaken. For the majority of studies it was unclear whether they had sufficient power to detect a statistically significant difference between groups; therefore, where there is no evidence of an effect it cannot be assumed that there is no effect. Information on outcome by stage of condition was not available. Based on the duration of frozen shoulder and the inclusion criteria, the trials included mixed populations with idiopathic frozen shoulder, probably mainly people in the first painful phase and the second stiffening stage. In addition, only five of the studies were undertaken in a UK NHS setting. RCTs were available for all of the interventions except capsular release, for which case series only were available, and watchful waiting or ‘supervised neglect’, for which a quasi-experimental study was available. However, some of the RCTs may in fact have been quasi-experimental. Across most trials there did not appear to be systematic methods for recording of adverse events.
Conservative interventions
Although several of the studies had a home exercise only control group, the information provided was often limited in terms of the extent of educational advice provided. The single study of ‘supervised neglect’, perhaps more appropriately called watchful waiting, was the only study to formally assess a broader intervention of self-management. However, as with the reporting of many complex interventions, there was insufficient information for replication in a clinical setting. 73 The finding from this study from the Netherlands, that supervised neglect was associated with a better outcome than physiotherapy (of unspecified duration), should be treated with considerable caution because of limitations in the study design. The lack of high-quality research on watchful waiting was surprising given that in a recent survey of UK professionals it was a commonly used treatment in the painful phase of frozen shoulder. 18
A further 11 studies evaluated physical therapy, mainly physiotherapy with active mobilisation. The comparators were a control or another form of physical therapy. Because of the diversity of the interventions and comparators only single studies were available for individual therapies and most of the studies had a high risk of bias. Based on a single study there was evidence of a statistically significant short-term benefit of SWD plus stretching (12 sessions over 4 weeks) compared with home exercise only and compared with a heat pack plus stretching for some outcomes. 75 There was also evidence from a single study that laser therapy (12 sessions over 8 weeks) had benefit over home exercise alone in the short and medium term. 16 Both studies had some risk of bias and the content, intensity and quality of the home exercise comparator was unclear. A further study, also at some risk of bias and in a population in which the majority had already received physiotherapy and/or a steroid injection, found marginally significant improvements for function and disability and improvement in range of movement with HGMT compared with LGMT. 40 None of these studies was undertaken in a UK setting.
Only studies comparing acupuncture with another of the treatments of interest were included, not studies comparing different methods of acupuncture. Only three studies were identified, none of which was from the UK, and all had a potentially high risk of bias. 79–81 There was insufficient evidence to make conclusions about the effectiveness of acupuncture for primary frozen shoulder.
Six studies evaluated steroid injections, although the majority of the available data were from two multi-arm studies that were of satisfactory quality, one with some risk of bias. 35,41 Both studies evaluated a single intra-articular steroid injection in patients with frozen shoulder of < 6 months’ duration. The comparators were home exercise alone, physiotherapy alone (both with placebo injection) and steroid injection followed by physiotherapy. One used a guided injection and the other appeared to use an unguided approach. The physiotherapy was of 4 weeks’ duration in both studies, with one including twelve sessions and the other eight. The actual content of the sessions varied between the studies, although both used mobilisation techniques. All groups in both studies were also given a home exercise programme.
Given that steroid injection is administered to reduce inflammation and provide pain relief, pain outcome is of particular interest for this intervention. For pain there was a short-term statistically significant benefit (up to 3 months) with steroid injection compared with home exercise alone but not compared with 4 weeks of physiotherapy. When steroid injection was provided in conjunction with physiotherapy, there was an added benefit for pain over physical therapy alone and over steroid injection alone. This was broadly consistent with the evidence for function and disability and range of movement. Only one of these studies used a QoL outcome measure (SF-36). There was no statistically significant benefit for QoL with a steroid injection alone compared with home exercise or physiotherapy alone. However, there was a benefit for QoL when physiotherapy was added to steroid injection over either home exercise or physiotherapy alone but not steroid injection alone. Overall, the evidence suggests that there is some evidence of a short-term benefit from adding steroid injection to physiotherapy alone; however, there remains some uncertainty as to whether this adds any substantial clinical benefit over steroid alone (with home exercise).
Invasive interventions
The data from high-quality RCTs of invasive interventions were even more limited than for conservative interventions and only two were undertaken in a UK setting. MUA, distension and capsular release are most commonly used when conservative treatments have failed, although for most of these studies it was unclear what treatments participants had already received, if any. In addition, there was considerable variability between the studies in how the procedures were delivered, making comparability between studies difficult.
There was insufficient evidence on all three interventions to make any robust recommendations regarding their use. Based on a single, satisfactory quality study, there was no evidence, across several outcomes, that MUA was better than a conservative intervention involving provision of advice in two sessions from a physical therapist plus written instructions for a daily training programme. 39 A study from the UK, with some risk of bias, reported greater improvement across outcomes with arthrographic distension than with MUA in participants with second stage (stiffening or ‘freezing’ stage) primary frozen shoulder. 38
There was no consistent evidence that arthrographic distension was associated with a better outcome than home exercise only or steroid injection. A single satisfactory study comparing arthrographic distension including steroid with placebo arthrography (with home exercise) reported a significant improvement with distension in function and disability measured using a patient-defined scale, but not in another function measure, range of movement or pain. 43 A second study with some risk of bias reported a benefit with arthrographic distension including steroid compared with steroid alone but only for a single range of movement measure at 6 weeks and not for other range of movement outcomes or a measure of function and disability. 36
Only case series were available for capsular release. The lack of a control group presents particular problems in assessing the effectiveness of an intervention in a condition such as frozen shoulder in which the condition normally resolves within a 3-year period. Based on the best-quality case series, undertaken in Norway with the procedure performed by a single surgeon, there was a significant improvement in several outcomes based on an average length of follow-up of 10 months (range 3 to 29 months). 85 All patients had tried physiotherapy previously without a satisfactory result.
Based on three studies of sodium hyaluronate, all at high risk of bias, there was insufficient evidence to recommend use of this treatment in preference to steroid or physical therapy. The best-quality study reported a benefit with two injections of sodium hyaluronate compared with home exercise but there was no consistent evidence across outcomes of a benefit over 10 daily sessions of physical therapy or single steroid injection. 66
Mixed-treatment comparison
A MTC was undertaken for the outcome of pain with the aim of bringing together both direct and indirect evidence in a connected network, thereby allowing inferences about the relative efficacy of all treatments to be made. Nine interventions formed part of a connected network with placebo and the evidence was informed by nine trials. A total of 22 studies had to be excluded from the MTC for a range of reasons; therefore, the network was a small subset of the available evidence. Of the nine interventions compared with placebo (plus home exercise) in the MTC, only steroid combined with physiotherapy and steroid alone were compared with placebo in the direct evidence synthesis (i.e. standard meta-analysis). The results from the MTC were consistent with the direct evidence synthesis of steroid combined with physiotherapy versus placebo. In both analyses the combined intervention showed a beneficial and clinically significant effect. In contrast, the results of the MTC were inconsistent with the direct evidence synthesis of steroid compared with placebo. In the MTC the 95% CrIs crossed the line of no effect. In the direct evidence synthesis, steroid showed both a statistically and a clinically significant beneficial effect compared with placebo. The remaining interventions included in the MTC (physiotherapy, physiotherapy combined with placebo, physiotherapy without mobilisation, electroacupuncture, sodium hyaluronate, MUA combined with physiotherapy, and arthrographic distension with steroid) had 95% CrIs that crossed the line of no effect.
Gaps in the clinical effectiveness evidence
There were several gaps in the evidence regarding conservative treatment options for primary frozen shoulder. Watchful waiting is the most conservative treatment option. However, it remains unclear what constitutes watchful waiting. It would seem inappropriate to do absolutely nothing and this is unlikely to happen in clinical practice. The intervention of supervised neglect evaluated by Diercks and Stevens73 included provision of education about the condition. This included an explanation of the natural course of the disease and advice not to exercise in excess of the pain threshold and to carry out pendulum exercises and active exercises within this painless range, with resumption of all activities that were tolerated. In addition, NSAIDs and analgesics were prescribed where necessary. Details were not provided about how the information was delivered and any methods used to maximise compliance with the home exercise programme and the extent of any monitoring, for example in the form of primary care review appointments, were unclear. In our view, education about the condition and guidance for self-management including home exercises are appropriate elements of a standard care package. Methods of maximising compliance with any advice may also be important. Several of the studies used a home exercise control group but it was often unclear what this consisted of and the extent and quality of education provided. Therefore, it was unclear in most studies whether the control group were receiving a high-quality standard care package or whether they were simply provided with a leaflet of exercises. In our view, the former is the most appropriate comparator to use in trials to establish any additional benefit from any more intensive interventions such as a package of physiotherapy treatment or steroid injection. The benefit of a physiotherapy intervention over a structured high-quality conservative intervention of education and home exercise using methods to maximise compliance has not been adequately addressed. Physiotherapy is the most commonly used NHS physical therapy and is therefore of particular interest.
It also remains unclear, based on the research evidence available, what constitutes an optimal physiotherapy intervention in terms of the essential components, number and length of sessions and overall duration and the type of physiotherapy. It also remains unclear at what stage and severity of frozen shoulder such an intervention would be of greatest benefit and whether any effect is similar in people with and without diabetes.
In clinical practice, patients can receive a second or third steroid injection when there has been no benefit from the first injection or when symptoms deteriorate again. There was no evidence available on the effectiveness of a second or third injection provided in a way that reflects NHS clinical practice. One US study evaluated the effect of once-weekly injections for 3 weeks rather than on the basis of the patients’ response to the first injection. 42
The role of the more invasive interventions in the treatment pathway for frozen shoulder remains unclear and further research is required to establish their effectiveness. There was considerable overlap in the duration of symptoms between the participants in the trials of invasive and conservative treatments. This suggests that the populations may have been similar across the more conservative and invasive interventions. In most studies it was unclear whether participants had received conservative treatments first that had not been effective, whether they were presenting for treatment having tried self-management or whether their preference was for an intervention that involved a shorter duration of treatment.
Comparison with other effectiveness research
We are aware of three systematic reviews that have been published since our assessment started: one of corticosteroid injections compared with physiotherapy for frozen shoulder;115 one of sodium hyaluronate injection for chronic painful shoulder;116 and one of conservative and surgical interventions for frozen shoulder. 117 A guideline based on a systematic review has also recently been published focusing on diagnosis, assessment and physiotherapy management of frozen shoulder. 6 Our conclusions regarding steroid injections are similar to those of Blanchard and colleagues115 who concluded that corticosteroid injections have a greater effect in the short term than physiotherapy but that the results of the review should be interpreted with caution because of the limited data available and variability between the studies. The recent guideline also comes to similar conclusions to the Blanchard review and our review regarding steroids compared with physiotherapy. 6
The review of conservative and surgical interventions, undertaken by Favejee et al. ,117 highlighted similar limitations in the literature to our review. They identified the lack of an unambiguous definition for frozen shoulder and the lack of information on phase of condition as well as a need for further research. However, they made somewhat stronger conclusions about the effectiveness of some interventions than our review has done or those of Blanchard et al. 115 and Hanchard et al. 6 have done. They concluded that there was strong evidence for the short-term effectiveness of steroid injections and laser therapy and moderate evidence for the effectiveness of mobilisation techniques in the short and long term as well as evidence for the effectiveness of arthrographic distension alone and in addition to active physiotherapy in the short term. The Favejee et al. 117 review used a broad definition of frozen shoulder that did not appear to be restricted to primary frozen shoulder. For example, the conclusion about strong evidence for the effectiveness of laser therapy is based on three trials only, one of which was of a primary frozen shoulder population. The other two trials were excluded from our review. The review of sodium hyaluronate is not directly comparable with our review as a range of chronic shoulder conditions were included, as well as dose-ranging studies. The review concluded that sodium hyaluronate injection was modestly more effective than steroid injection for function and there was an improvement in pain and function compared with placebo. 116
Patient views
We were unable to identify any papers that explored patients’ views or experiences of treatments for frozen shoulder. As treatment for frozen shoulder can vary substantially and is often guided by patient preference, this was considered an important area for investigation; however, the lack of available evidence did not allow us to explore this further. Although no papers exploring patients’ views were found, there were limitations to this review because of time constraints. Our searches were restricted to searches of three databases for English-language studies and unpublished studies may have been missed. Nonetheless, there does appear to be a clear need for research in this area.
Cost-effectiveness review
A single Dutch cost-effectiveness study met the inclusion criteria for the systematic review. 91 This was conducted as part of a trial that was included in the clinical effectiveness review. 40 The trial reported statistically significant improvement in range of movement with HGMT compared with LGMT in a population in which the majority of patients had already received physiotherapy and/or steroid injection. There was no significant difference in QoL, although the CIs were wide. There was also no significant difference in pain or function and disability, although for one of the function and disability scales the CIs just crossed the line of no effect. The economic analysis was of reasonable quality with some risk of bias and was conducted outside of the UK (the Netherlands), which may limit generalisability. It showed that LGMT may be a cost-effective option compared with HGMT.
Our searches failed to identify any modelling studies conducted in the relevant population or similar populations/conditions. We did identify a limited number of economic evaluations conducted alongside clinical trials in patients with unilateral shoulder pain, chronic shoulder complaints and acute and subacute shoulder complaints. 91,118–121 We considered these populations to be similar enough to allow some comparisons to be made, although these populations were not the focus of our research and we did not undertake a systematic review in these patient populations. The economic evaluations did not meet our inclusion criteria but we anticipated that they would provide some useful information to inform our research. One of these studies, James et al. ,119 was conducted in the UK and allowed some comparison of the cost estimates obtained in the clinical trial and the estimates obtained from our advisory group for the frozen shoulder population. Because of the limited evidence available to us through the review, alternative methods of obtaining evidence were pursued. The clinical advisors on the project were utilised and primary data from a clinical trial in a similar population identified.
Cost estimates
The estimated resource use provided by our clinical advisory group allowed the calculation of average treatment costs from the perspective of the UK NHS for the interventions identified in the systematic review. The estimated average costs for therapies involving administration of steroid injections ranged from £36.18 to £138.51 for unguided injections and from £299.68 to £475.56 for guided injections. Variations in costs were attributable to the type of health-care professionals involved and the delivery setting. The costs of therapies involving active physiotherapy alongside steroids were estimated to vary over a wide range, from £121.43 to £607.31, and reflected practice variations in regard to number of physiotherapy sessions, delivery setting and the steroid used.
Physiotherapy alone, involving some form of mobilisation as a component, was estimated to range from £98.75 to £126.75. Acupuncture was estimated to cost from £117.75 to £126.75. Difference in costs was mainly due to community versus hospital-based setting.
The estimated average costs associated with surgical interventions ranged from £1213 to £1522 for MUA, from £79.84 to £134.84 for arthrographic distension and from £1809 to £2511 for capsular release. The difference in costs was driven by the difference in the hospital resources utilised in performing MUA arthrographic distension and capsular release. This is reflected in the different NHS reference cost codes under which these interventions are reported by hospitals and subsequently costed.
Comparison with costs of other research
The validation of these estimates is difficult given the lack of evidence available. James et al. 119 conducted a cost–consequence analysis comparing local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in UK primary care. The analysis collected resource-use data from patients enrolled in the trial from entry until 6 months post randomisation. The study reports an average cost of £114.60 for those patients receiving the physiotherapy intervention. This is very similar to our estimate of an average cost of £112.75 for physiotherapy (average based on community/hospital setting). They also report an average cost of £71.28 for the steroid group, which compares favourably with our average estimate of £73.87 for an unguided injection (average based on alternative delivery setting and choice of steroid). Although these costs appear to be close, the uncertainty remains on how closely either set of costings would reflect clinical practice. Patients throughout the James et al. study had access to additional treatments that were included in the resource-use figures. So the average cost for the physiotherapy group also included costs for resources such as radiography, non-trial injections, etc. In addition, costs were presented for 2003 and we have not inflated them to match our 2009 price year. This may suggest that our cost estimates are a little on the high side given the difference in price years and the fact that our costs consider only resources for one specific treatment and not the additional resources that a patient may consume by choosing to access alternative treatments or specialists alongside the primary prescribed treatment.
As with the single included economic evaluation for frozen shoulder, the majority of economic evaluations identified were conducted in the Netherlands. Although some provided useful data for the comparability of utility estimates they did not provide us with a clear picture of the resource utilisation of the alternative treatments that we have considered in this review. Van den Hout et al. ,91 which met the inclusion criteria, presented societal costs of €8809 for HGMT and €6911 for LGMT. Enough data were presented to allow costs from the perspective of the health-care provider to be estimated and these range from €2552 for HGMT to €2293 for LGMT. Because of differences in health-care systems the cost analysis used charges, which are relevant in the Netherlands, rather than costs, which are relevant for the NHS setting. So even if we convert euros into British pounds a like-for-like comparison would be difficult. In our view, the estimates obtained from our clinical advisory group represent the most reliable resource estimates and, although we acknowledge that there is some uncertainty surrounding the estimates because of the lack of evidence available, it was not possible to investigate this further. The need for a clearer picture of the resources used as patients progress through their treatment pathway is evident and is lacking in the current evidence base.
Quality of life and utility data
To fully evaluate HRQOL it is necessary to consider all of the ways that the condition being treated and the intervention being delivered impact on patient health outcomes. This means measuring the effect of the condition and the treatment on both duration and quality of life. The use of QALYs allows both of these concerns to be captured and reflected in one outcome. The QALY integrates mortality and morbidity to convey health status in terms of equivalent well years of life. A key component of the QALY is the utility values, which can be measured using a variety of methods. The measurement of utilities is an area of ongoing debate and research, which is beyond the scope of this discussion.
The clinical trials identified in the effectiveness review reported a variety of outcomes, mainly pain, range of movement and function and disability, with some measuring QoL, time to recovery and return to work and recreation, and adverse events. There seems little agreement across the trials regarding the tools with which these outcomes should be measured or which is the primary outcome. This may be related to the changing, progressive nature of the condition. In the early painful stages those outcomes that capture pain are likely to be more important; however, as the condition progresses the function and disability outcomes may become more important. When faced with a plethora of outcomes such as this it raises the question of why HRQoL, which may capture all of these outcomes, has not been routinely measured alongside those outcomes more traditionally considered clinical outcomes. We found only one study reporting HRQoL (utility outcomes) in the frozen shoulder population91 and only scant data on HRQoL in patient populations considered similar.
It is unclear why these data have not been collected in the clinical trials identified in the effectiveness review. Possible reasons may be because it was not practical or was too time-consuming for patients to complete in addition to other measures or possibly because the studies were not designed to focus on economic questions and therefore utilities were not thought to be important. These reasons are only conjecture; further research is required to establish why HRQoL does not appear to be considered a relevant outcome in these types of patients. However, this lack of utility data hinders any decision analysis being undertaken. When outcome measures used in trials are limited to condition-specific outcomes the ability to evaluate the cost-effectiveness of one treatment against another treatment in a different population is lost.
Because of the potential for bias in the one study presenting HRQoL data and the use of the less popular SF-6D tool,91 we undertook a mapping exercise. Our aim was to try and establish a link between the outcomes considered in the clinical trials and HRQoL. The EQ-5D is the most popular instrument of choice for the measurement of utilities and is recommended in the NICE reference case. 64 The decision was made to use this instrument in a mapping exercise. The mapping investigated the relationship between QoL, as measured by two components of the SF-36, and HRQoL, as measured by the EQ-5D, and pain, as measured by a VAS 0–100 mm, and EQ-5D. The SF-36 was used as this is a widely used and accepted tool for measuring QoL and was used in two of the included studies in the effectiveness review: one compared ultrasound and placebo;51 the other compared steroid injection, steroid injection plus physiotherapy, physiotherapy alone and placebo. 35 A VAS 0–100 mm was used as it was readily available in the data set used for mapping and was a scale used in the clinical trials. The mapping was based on data from the SAPPHIRE trial. 40 The complete data set was made available by the authors of the trial. The findings of the mapping suggest that there is a positive relationship between outcomes: a decreasing pain VAS score (less pain) is accompanied by an increasing (better) EQ-5D score; an increasing EQ-5D score (i.e. better health status) appears to be generally accompanied by increasing SF-36 scores (i.e. higher scores = a better quality of life). Two trials included in the review of effectiveness, Dogru et al. 51 and Stergioulas,16 used pain VAS (0–100 mm). Using the prediction equation obtained from mapping pain VAS and the results of the two trials we were able to estimated incremental QALY improvements, which ranged from –0.00163 to 0.00350. Two trials included in the review, Carette et al. 35 and Dogru et al. ,51 used SF-36 PCS and MCS as an outcome measure. Using the prediction equation obtained from the mapping and the results of the two trials we were able to estimate incremental QALY improvements, which ranged from 0.00537 to 0.01275.
Although the mapping was conducted on data from a similar population, primary collection of utility data would be required to validate that the positive relationship remains in the frozen shoulder population.
Those economic evaluations in similar populations that have reported EQ-5D data118,120,121 have demonstrated improvements from baseline ranging from 0.06 to 0.16, which are comparable to those in the SAPPHIRE trial. These trials included a variety of interventions including an education and activation programme, manipulative therapy, which comprised specific manipulation and mobilisation techniques, and graded exercise therapy delivered by a physiotherapist. All were compared with usual care, which was variable in its content and the depth of its description. The populations in these trials had a higher baseline EQ-5D value (range 0.51 to 0.587 for pain VAS; 0.518–0.562 for SF-36 PCS/MCS) than the population from the SAPPHIRE trial (range 0.453 to 0.512). 102 This may suggest that the population of the SAPPHIRE trial, each of whom had a confirmed diagnosis of rotator cuff tendonitis, differs from populations with other shoulder complaints. It is likely that this population may be comparable to a population of frozen shoulder patients in the painful phase of the condition; however, we have no evidence to support this other than the comparability of the SF-36 scores across the SAPPHIRE population and the frozen shoulder populations in Carette et al. 35 and Dogru et al. 51 These data suggest that the populations have very similar QoL scores at baseline. Further investigation of these issues is warranted but is outside the scope of this research.
None of those economic evaluations presenting EQ-5D data presented QALYs. It is not clear why QALYs were not derived. One study provided justification, which was that the minimal changes in clinical effectiveness of the EQ-5D outcome provided no additional information when analysed in a cost-effectiveness analysis. 113 It is not clear that this is valid justification for not deriving QALY outcomes.
Strengths and limitations of the review
To our knowledge this is the first systematic review that has included all NHS-relevant interventions in a single synthesis. A total of 19 electronic databases were searched including potential sources of unpublished data. Unlike several previous reviews there were no language restrictions. Full papers of 67 studies in 12 languages other than English were screened for eligibility, of which four were subsequently included in the review. Risk of bias was assessed and taken into consideration in the synthesis. Whenever possible, the treatment effect for individual studies was reported with a 95% CI, even when quantitative synthesis was not undertaken.
The key limitation of this technology assessment was the lack of data available. This was despite comprehensive searches without language restrictions across a range of sources of studies. There was insufficient evidence to make robust conclusions about the effectiveness of any of the interventions. Because of the lack of evidence on stage of frozen shoulder in the included trials, it was not possible to undertake the planned synthesis exploring the influence of stage of frozen shoulder or the presence of diabetes on the effectiveness of the various interventions. Even where there were several trials of a single intervention, there was such diversity in the intervention and comparators used and such poor reporting of outcome data that there were few instances in which the planned quantitative synthesis was possible or appropriate. Given that most of the included studies had a small number of participants and may have been underpowered, this was unfortunate.
Unlike some other reviews that have looked at shoulder complaints across the board as a single condition, we attempted to reduce variability due to the nature of the shoulder condition by including only patients with primary frozen shoulder. However, despite this, it is likely that the population included is diverse because of variability in how the included studies defined frozen shoulder. Together with the lack of information on impact of stage, this means that even for those interventions for which there is some evidence of effectiveness, there is some uncertainty as to what primary frozen shoulder populations this applies to.
Because the studies were often poorly reported it was difficult in some instances to be certain that the population included those with primary frozen shoulder and not those with secondary frozen shoulder. As a result we may have excluded studies that did in fact include a primary frozen shoulder population. Comparing the results of our review with those of other recent reviews it is unlikely that the inclusion of such studies would have altered our conclusions.
The variability in the outcome measures used was a further limitation of the data available. Pain, and function and disability were the most commonly used end points in clinical trials for frozen shoulder and there was considerably variability in the measures used to assess these three outcomes. Studies included in the review measured pain used the verbal numeric rating scales or VAS scales (including the SPADI 5-item subscale), which have been shown to be highly correlated with one another. 122 The decision to standardise results of these studies using the SMD enabling pooling of studies was therefore appropriate. In direct contrast the outcome of function and disability was measured by a number of different shoulder-specific scales, which have been shown to have low to moderate correlation with one another. This indicates that different shoulder-specific function and disability scales may not be measuring the same thing. 54,55,123,124 On the basis of this we did not use a standardised score for function and disability outcomes, which meant that a quantitative pooling of these data, for the most part, was not possible.
To aid interpretation, pooled SMDs for the outcome of pain were back transformed to a 0–100 mm VAS. As no between-group, or, for that matter, within-group, MCID on the 0–100 mm VAS was available for frozen shoulder, a proxy was derived from the within-group MCID of a rotator cuff disease population. Although this proxy represents the best available between-group MCID for frozen shoulder at this time and has been used elsewhere,6 any implied clinical significance should be seen as tentative rather than definitive. In addition, such an approach was appropriate only for pooled estimates.
Several studies that were included in the review were not included in the synthesis because the period of follow-up was ≤ 4 weeks. Such short follow-up periods are unlikely to capture meaningful changes in outcome measures.
Mixed-treatment comparison
A key factor to consider in evaluating the strengths and limitations of the MTC undertaken is whether the assumption that the studies are sufficiently homogeneous to be quantitatively combined has been met. Arguable sources of heterogeneity in the MTC were the fact that the placebos were not identical between studies, given the differing nature of the interventions; and that there was differential reporting of the use of previous treatments for frozen shoulder associated with the invasiveness of treatment, which may indicate that populations between trials varied in the treatment resistance of frozen shoulder. Furthermore, as in the main synthesis, there was a paucity of high-quality studies. However, the model was a good fit and gave stable estimates and it is interesting that there was no clear difference between the interventions in effectiveness and no single intervention was substantially better than the others. Given the possible heterogeneity of placebos and of study populations, and the inclusion of poor-quality studies, there is some uncertainty regarding the results of the MTC, as there is with the main synthesis.
Economic evidence
The lack of available data made the development of a decision-analytic model impossible. We found little evidence on treatment related to stage of condition, treatment pathways, the impact on quality of life and associated resource use and no information on utilities. Without making a number of questionable assumptions, modelling was not possible. Following clinical advice it was decided that insufficient evidence was available to inform the structure of a model or to guide and justify our assumptions. Although it was not possible to build a decision model, the assessment has estimated the costs for the various treatments included in the review and investigated the link between QoL and utilities where possible. We have not been able to quantify the uncertainty in our cost estimates and the use of a small group of clinical experts to derive the estimates is a limitation. Despite this limitation we believe that the estimates obtained may provide useful information as input to (future) cost-effectiveness assessments of treatments in frozen shoulder populations.
The findings of the exploratory mapping analysis suggest that there is likely to be a relationship between the SF-36 and the EQ-5D, and VAS and the EQ-5D in frozen shoulder populations. Caution is needed, however, as further data on QoL outcomes using both non-preference-based and preference-based measures are now needed. This will allow full validation of the nature and direction of the relationship between the instruments and investigation of whether utilities might be a useful outcome measure in frozen shoulder populations. It is clear from the results obtained that the relationship between the two clinical outcomes and EQ-5D is not the same. This is demonstrated in the Dogru et al. study51 by the different direction of QALY benefit, which favours the intervention over the control when VAS is mapped to EQ-5D and the control over the intervention when SF-36 is mapped. The choice of clinical outcome to be included in the mapping is obviously very important and how this choice is made warrants further research. It is also unclear whether the use of more complex statistical methods would alter the predictive performance of the SF-36 PCS and MCS mapping onto the EQ-5D utilities, compared with the models tested in the exploratory analysis, for example whether including all eight dimensions of the SF-36 profile in mapping onto the EQ-5D changes the nature of the relationship. These methodological investigations are beyond the scope of this report.
Mapping is a substitute for primary measurement of utilities and is subject to a number of potential limitations. In this instance the main limitations are likely to be that it was not performed directly using individual-level patient data drawn from frozen shoulder trials but used a UK data set of patients with rotator cuff disease (the SAPPHIRE trial); and the small study sample (n = 141 for pain VAS, and n = 133 for SF-36 PCS/MCS in the base case presented). It would be possible to further validate the apparent relationship between the SF-36/VAS and the EQ-5D through the use of an external data set (i.e. an alternative trial). However, to fully measure the apparent relationship between these two outcomes the collection of primary data will be required, which will have greater validity.
Improving the evidence on the effectiveness and cost-effectiveness
This assessment highlighted considerable gaps in the evidence for the effectiveness and cost-effectiveness of all of the interventions investigated for frozen shoulder. Given the range of treatment options a multi-arm trial would be the most appropriate way forward. This raises the question of which interventions should be given priority for future research on treatments for frozen shoulder and where resources would best be spent in undertaking future research. Given the lack of an economic model we were not in a position to characterise the decision uncertainty and present a value of information analysis to support recommendations about where future resources for research should focus.
Given that clinically there is a fairly consistent view that a stepped-up approach should be used in terms of invasiveness of treatment, most patients are likely to receive only the conservative treatments. It could be argued, therefore, that this is the most important area on which to focus the research effort. However, given the lack of consensus across professional groups regarding the most appropriate care pathway and the apparent overlap in patients receiving treatments of varying invasiveness, it may be inappropriate to put research effort into conservative treatments at the expense of the invasive treatments. The costing exercise we undertook found that the invasive treatments may be substantially more expensive than the conservative options. It would therefore be important to establish whether these interventions produce benefits for the patient and are cost-effective choices.
In a recent survey of 303 health-care professionals involved in the management of frozen shoulder, 88% of respondents, including GPs, physiotherapists and orthopaedic surgeons, considered that there was a need for a RCT comparing treatments, particularly in the early painful phase. 18 Several interventions were supported for future research: watchful waiting and education (classified as conservative), physical therapy, injections and surgery (encompassing MUA and capsular release). Overall preferences for interventions requiring further research differed between the professional groups. The interventions that received least support were arthrographic distension, sodium hyaluronate injection, oral painkillers and oral steroids. There was support for research on physiotherapy and mobilisation techniques but much less so for individual therapies such as acupuncture, ultrasound, heat, laser therapy and electroacupuncture. A recently published guideline from the Chartered Society for Physiotherapy has also suggested that there may not be much to be gained from evaluating specific modalities as stand-alone treatments. 6
Any future trials should give more serious attention to the control group used. Home exercise (with or without a placebo) was the most commonly reported control in the included studies. However, in general the content of this control intervention was poorly reported. There is a large gap in the evidence about the effectiveness of a high-quality conservative intervention of education, home exercise and self-management of pain with support and monitoring. Any future trial should pay careful attention to the control condition to clearly establish what additional benefit other interventions provide. In addition, the maximum length of follow-up in most studies was 3 months. Future trials should therefore have follow-up periods that are of sufficient length to allow determination of whether interventions are effective in the medium and long term.
There is a view clinically that stage of frozen shoulder is an important aspect to consider when making treatment choices. This is not reflected in how most trials have been undertaken to date, although it is unclear why not. Obviously it makes recruitment to trials easier if the population is not restricted in this way. Stratification within trials may be a more pragmatic approach. Another possible factor explaining the lack of consideration for stage is the wide range of countries in which the trials have been undertaken. It is clear, however, that any future research should not neglect consideration of stage. Because of the lack of evidence on treatment effects in patients with diabetes, this will be an important subgroup. This reflects the recent survey that highlighted those with diabetes as an important subgroup. 18
Populations included in the review were diverse because of considerable variation in how frozen shoulder was defined in the individual studies. This is unsurprising given that there is a noted absence of a standard definition and classification for frozen shoulder. 2,3 The variations in study populations together with the lack of information on the stage of frozen shoulder mean that, even for those interventions for which there is some evidence of effectiveness, there is some uncertainty as to which frozen shoulder populations these apply to. Although potential classifications for primary and secondary frozen shoulder have been put forward,3 a consensus on the definition of frozen shoulder is sorely needed to enable the generalisability of further research to be assessed.
Given that studies of clinimetric properties (i.e. validity, reproducibility, responsiveness and ease of scoring) of various shoulder-specific disability scores have conflicting findings, it is difficult to suggest the most appropriate score to use. 125,126 Further research is therefore needed to determine the clinimetric properties of shoulder-specific function and disability scores. This should conclude with a consensus on the most appropriate tools to assess shoulder function, both in research and in clinical practice. For assessing the clinical meaningfulness of between-group differences in future trials, consideration should be given to the recommendations of the Initiative on Methods, Measurement and Pain Assessment in Clinical Trials (IMPACT). 127 This group suggests that the clinical meaningfulness of group differences in trials, in terms of pain, should be established using a multifactorial evaluation of factors such as statistically significant treatment effects, responder analysis, effect size compared with that of other established treatments, safety and tolerability, and results for other end points.
Based on the available evidence we were unable to structure a simple decision tree, but, given the complex nature of the treatment pathways, future decision-analytic modelling undertaken in this area will need to take into account the nature of any sequential decision-making processes and treatments rather than just a single therapy. This will require tangible evidence of current treatment patterns and resource use in frozen shoulder populations. There may be benefit in undertaking an elicitation exercise with a range of health-care professionals to prioritise the interventions to be compared and underpin the design of any future trial.
There is a current lack of studies providing data on HRQoL specific to frozen shoulder populations. This information is required to enable assessments of cost–utility to be undertaken. The inclusion of preference-based QoL measures alongside clinical trials in frozen shoulder populations is a necessity. Whether the EQ-5D as an instrument is considered to be the most appropriate for use in frozen shoulder populations has yet to be determined and further research is required to address this and other questions on the use of HRQoL measures in clinical studies. Mapping is always a second-best solution to using a preference-based (generic or condition-specific) measure in the first place, but it is often the approach followed for pragmatic reasons and so this remains an important area of research. Primary data collection in frozen shoulder populations is now needed.
Chapter 5 Conclusions
Implications for service provision
-
There may be short-term benefit for patients with frozen shoulder of < 6 months’ duration of adding a single intra-articular steroid injection to home exercise. There is also short-term benefit for the same population of adding physiotherapy to a single intra-articular steroid injection. This is based on two studies that varied in steroid dose and in whether the injection was guided, making it difficult to determine how the steroid intervention should be delivered. Although neither study explicitly provided information on stage of frozen shoulder, only patients with frozen shoulder of < 6 months’ duration were included, suggesting that these patients were likely to be at the initial painful stage. Based on two studies of physiotherapy that included mobilisation in 8–12 sessions over a 4-week period, it is unclear what is the most effective physiotherapy to provide with injection in terms of content and duration.
-
Based on a single study, and for some outcomes only, there may be benefit from adding SWD to passive mobilisation and home exercise.
-
Based on a single study, HGMT may be more effective for some outcomes, but not others, than LGMT in a population that has already received physiotherapy and/or a steroid injection. In the study, high-grade mobilisation consisted of twice-weekly, 30-minute sessions for a maximum of 12 weeks during which Maitland grade III and IV mobilisation was applied into the stiffness zone. Low-grade mobilisation was delivered according to the same schedule but consisted of Maitland grade I and II mobilisation applied within the pain-free zone. LGMT may be more cost-effective than HGMT.
-
Given the paucity of economic evidence no conclusions can be made about the cost-effectiveness of the different interventions.
Suggested research priorities
There are large gaps in the evidence for the effectiveness and cost-effectiveness of all of the interventions investigated. Taking into account the gaps in the evidence, the views of health-care professionals in a recent survey18 and the interventions that are most commonly used in the NHS, we suggest that the following should be given priority in future high-quality RCTs assessing effectiveness and cost-effectiveness:
-
A ‘standard care’ package of high-quality conservative management. This should be fully specified in any future trial and we suggest that it should involve a structured protocol of high-quality education, advice, home exercise and monitoring/support to encourage completion of the home exercises.
-
Steroid injection, in particular an investigation of the effectiveness and cost-effectiveness of multiple injections and whether there is any added benefit from providing physiotherapy after steroid injection over and above that obtained with steroid injection plus high-quality conservative management.
-
Physical therapies, specifically physiotherapy interventions that have a component which involves mobilisation or exercises. Research is required to establish whether there is any benefit from having physical therapy alone over and above that of high-quality conservative management.
-
Intensive interventions, specifically arthrographic distension, MUA and arthroscopic capsular release. Although there is also a gap in the evidence regarding the effectiveness of distension, in the recent survey of health-care professionals this intervention did not receive as much support as a topic for future research as the other intensive interventions.
The large number of treatment options for frozen shoulder and the limited evidence for their effectiveness and cost-effectiveness makes prioritisation of these difficult and they are not listed in order of importance. We suggest that an appropriate starting point would be a multi-arm trial that compares the effectiveness and cost-effectiveness of interventions of differing intensity and costs: high-quality conservative management, steroid injection (possibly in conjunction with arthrographic distension) and surgical management (MUA and capsular release).
Important subgroups in any future RCTs are phase of frozen shoulder and patients with diabetes. These trials should collect data on resource use and use a utility measure to allow assessment of cost-effectiveness. A systematic approach to recording adverse events will also be important.
Acknowledgements
Many thanks to all members of the advisory group for the many helpful contributions.
Many thanks to Jonathan Minton for the help provided with study selection.
Contribution of authors
Emma Maund contributed to writing the protocol, study selection, data extraction, quality assessment, data analysis and report writing.
Dawn Craig contributed to writing the protocol, undertaking the economic analysis and report writing.
Sara Suekarran contributed to writing the protocol, study selection, data extraction, quality assessment, data analysis and report writing.
Aileen Rae Nielson was involved in the economic analysis and report writing.
Kath Wright developed the search strategies, undertook the searches and wrote the literature search methods section.
Stephen Brealey, Laura Dennis, Lorna Goodchild, Nigel Hanchard, Amar Rangan, Gerry Richardson and James Robertson provided clinical and/or methodological input and advice throughout the project and commented on the protocol and drafts of the report.
Catriona McDaid was responsible for writing the protocol, contributed to study selection, data extraction, quality assessment, data analysis and report writing and had overall responsibility for co-ordinating the project.
Disclaimers
The views expressed in this publication are those of the authors and not necessarily those of the HTA programme or the Department of Health.
References
- Codman E. Rupture of the supraspinatus tendon and other lesions in or about the subacromial bursa. Malabar, FL: Krieger; 1965.
- Schellingerhout JM, Verhagen AP, Thomas S, Koes BW. Lack of uniformity in diagnostic labeling of shoulder pain: time for a different approach. Man Therap 2008;13:478-83.
- Zuckerman J, Rokito A. Frozen shoulder: a consensus definition. J Shoulder Elbow Surg 2010;20:322-5.
- de Jong BA. The Painful Stiff Shoulder 1991.
- Dias R, Cutts S, Massoud S. Frozen shoulder. BMJ 2005;331:1453-6.
- Hanchard N, Goodchild L, Thompson J, O’Brien T, Davison D, Richardson C, et al. Evidence-based clinical guidelines for the diagnosis, assessment and physiotherapy management of contracted (frozen) shoulder. London: Chartered Society of Physiotherapy; 2011.
- Bunker TD. Frozen shoulder: unravelling the enigma. Ann R Coll Surg Engl 1997;79:210-13.
- Bunker TD. Time for a new name for frozen shoulder – contracture of the shoulder. Shoulder Elbow 2009;1:4-9.
- Hand GCR, Athanasou NA, Matthews T, Carr AJ. The pathology of frozen shoulder. J Bone Joint Surg Br 2007;89:928-32.
- van der Windt DA, Koes BW, de Jong BA, Bouter LM. Shoulder disorders in general practice: incidence, patient characteristics, and management. Ann Rheum Dis 1995;54:959-64.
- Walker-Bone K, Palmer KT, Reading I, Coggon D, Cooper C. Prevalence and impact of musculoskeletal disorders of the upper limb in the general population. Arthritis Care Res 2004;51:642-51.
- Dawson J, Shepperd S, Carr A. An overview of factors relevant to undertaking research and reviews on the effectiveness of treatment for frozen shoulder. Shoulder Elbow 2010;2:232-7.
- Hand C, Clipsham K, Rees JL, Carr AJ. Long-term outcome of frozen shoulder. J Shoulder Elbow Surg 2008;17:231-6.
- New Zealand Guidelines Group . The Diagnosis and Management of Soft Tissue Shoulder Injuries and Related Disorders 2004. www.nzgg.org.nz (accessed 9 March 2009).
- Linsell L, Dawson J, Zondervan K, Rose P, Randall T, Fitzpatrick R, et al. Prevalence and incidence of adults consulting for shoulder conditions in UK primary care; patterns of diagnosis and referral. Rheumatology 2006;45:215-21.
- Stergioulas A. Low-power laser treatment in patients with frozen shoulder: preliminary results. Photomed Laser Surg 2008;26:99-105.
- NHS Choices . Health A–Z – Conditions and Treatments n.d. www.nhs.uk/Conditions/Pages/hub.aspx (accessed 11 March 2011).
- Dennis L, Brealey S, Rangan A, Rookmoneea M, Watson J. Managing idiopathic frozen shoulder: a survey of health professionals’ current practice and research priorities. Shoulder Elbow 2010;2:294-300.
- Manske RC, Prohaska D. Clinical commentary and literature review: diagnosis, conservative and surgical management of adhesive capsulitis. Shoulder Elbow 2010;2:238-54.
- Buchbinder R, Green S, Youd JM, Johnston Renea V. Oral steroids for adhesive capsulitis. Cochrane Database Syst Rev 2006.
- Buchbinder R, Green S, Youd JM. Corticosteroid injections for shoulder pain. Cochrane Database Syst Rev 2003.
- Shah N, Lewis M. Shoulder adhesive capsulitis: systematic review of randomised trials using multiple corticosteroid injections. Br J Gen Pract 2007;57:662-7.
- Arroll B, Goodyear-Smith F. Corticosteroid injections for painful shoulder: a meta-analysis. Br J Gen Pract 2005;55:224-8.
- Green S, Buchbinder R, Hetrick SE. Physiotherapy interventions for shoulder pain. Cochrane Database Syst Rev 2003.
- Cleland J, Durall CJ. Physical therapy for adhesive capsulitis: systematic review. Physiotherapy 2002;88:450-7.
- Alvado A, Pelissier J, Benaim C, Petiot S, Herisson C. Physical therapy of frozen shoulder: literature review. Ann Readapt Med Phys 2001;44:59-71.
- Green S, Buchbinder R, Hetrick SE. Acupuncture for shoulder pain. Cochrane Database Syst Rev 2005.
- Buchbinder R, Green S, Youd JM, Johnston Renea V, Cumpston M. Arthrographic distension for adhesive capsulitis (frozen shoulder). Cochrane Database Syst Rev 2008.
- Green S, Buchbinder R, Glazier R, Forbes A. Systematic review of randomised controlled trials of interventions for painful shoulder: selection criteria, outcome assessment, and efficacy. BMJ 1998;316:354-60.
- Rookmoneea M, Dennis L, Brealey S, Rangan A, White B, McDaid C, et al. The effectiveness of interventions in the management of patients with primary frozen shoulder. J Bone Joint Surg Br 2010;92B:1267-72.
- Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med 2004;23:3105-24.
- Caldwell DM, Ades AE, Higgins JP. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ 2005;331:879-900.
- Sutton A, Ades A, Cooper N, Abrams K. Use of indirect and mixed treatment comparisons for technology assessments. Pharmacoeconomics 2008;26:753-67.
- Centre for Reviews and Dissemination . Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care 2009.
- Carette S, Moffet H, Tardif J, Bessette L, Morin F, Fremont P, et al. Intraarticular corticosteroids, supervised physiotherapy, or a combination of the two in the treatment of adhesive capsulitis of the shoulder: a placebo-controlled trial. Arthritis Rheum 2003;48:829-38.
- Tveita EK, Tariq R, Sesseng S, Juel NG, Bautz-Holter E. Hydrodilatation, corticosteroids and adhesive capsulitis: a randomized controlled trial. BMC Musculoskelet Disord 2008;9.
- Higgins J, Deeks JJ, Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions 2008. www.cochrane-handbook.org (accessed 9 March 2009).
- Quraishi NA, Johnston P, Bayer J, Crowe M, Chakrabarti AJ. Thawing the frozen shoulder. A randomised trial comparing manipulation under anaesthesia with hydrodilatation. J Bone Joint Surg Br 2007;89:1197-200.
- Kivimaki J, Pohjolainen T, Malmivaara A, Kannisto M, Guillaume J, Seitsalo S, et al. Manipulation under anesthesia with home exercises versus home exercises alone in the treatment of frozen shoulder: a randomized, controlled trial with 125 patients. J Shoulder Elbow Surg 2007;16:722-6.
- Vermeulen HM, Rozing PM, Obermann WR, le Cessie S, Vliet Vlieland TPM. Comparison of high-grade and low-grade mobilization techniques in the management of adhesive capsulitis of the shoulder: randomized controlled trial. Phys Ther 2006;86:355-68.
- Ryans I, Montgomery A, Galway R, Kernohan WG, McKane R. A randomized controlled trial of intra-articular triamcinolone and/or physiotherapy in shoulder capsulitis. Rheumatology 2005;44:529-35.
- Rizk TE, Pinals RS, Talaiver AS. Corticosteroid injections in adhesive capsulitis: investigation of their value and site. Arch Phys Med Rehabil 1991;72:20-2.
- Buchbinder R, Green S, Forbes A, Hall S, Lawler G. Arthrographic joint distension with saline and steroid improves function and reduces pain in patients with painful stiff shoulder: results of a randomised, double blind, placebo controlled trial. Ann Rheum Dis 2004;63:302-9.
- Rodgers M, McKenna C, Palmer S, Chambers D, van Hout S, Golder S, et al. Curative catheter ablation in atrial fibrillation and typical atrial flutter: systematic review and economic evaluation. Health Technol Assess 2008;12.
- Drummond MF. Methods for the economic evaluation of healthcare programmes. Oxford: Oxford University Press; 2005.
- Review Manager (RevMan) [computer program.] 2008.
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analysis. BMJ 2003;327:557-60.
- Deeks JJ, Higgins JPT, Altman DG, Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions. The Cochrane Collaboration; 2008.
- Borenstein M, Hedges LV, Higgins JPT, Rothstein HR. Introduction to meta-analysis. Oxford: John Wiley; 2009.
- Tashjian RZ, Deloacha J, Porucznik CA, Powell AP. Minimal clinically important differences (MCID) and patient acceptable symptomatic state (PASS) for visual analog scales (VAS) measuring pain in patients treated for rotator cuff disease. J Shoulder Elbow Surg 2009;18:927-32.
- Dogru H, Basaran S, Sarpel T. Effectiveness of therapeutic ultrasound in adhesive capsulitis. Joint Bone Spine 2008;75:445-50.
- Scholten R, de Beurs E, Bouter L. Is the standardised mean difference a suitable measure of treatment effect? In: The best evidence for health care: the role of The Cochrane Collaboration Università S. Tommaso d’Aquino, Rome, Italy; 1999.
- Finch E, Brooks D, Stratford PW, Mayo NE. Physical rehabilitation outcome measures: a guide to enhanced clinical decision making. Toronto: Lippincott, Williams and Wilkins; 2002.
- Placzek J, Lukens S, Badalanmenti S, Roubal P, Freeman D, Walleman K, et al. Shoulder outcome measures: a comparison of 6 functional tests. Am J Sports Med 2004;32:1270-7.
- Paul A, Lewis M, Shadforth M, Croft P, van der Windt D, Hay E. A comparison of four shoulder-specific questionnaires in primary care. Ann Rheum Dis 2004;63:1293-9.
- Romeo A, Bach B, O’Halloran K. Scoring systems for shoulder conditions. Am J Sports Med 1996;24:472-6.
- Ginn K, Cohen M, Herbert R. Does hand-behind-back range of motion accurately reflect shoulder internal rotation?. J Shoulder Elbow Surg 2006;15:311-4.
- Higgins JPT, Deeks JJ, Altman DG, Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions. The Cochrane Collaboration; 2008.
- Song F, Harvey I, Lilford R. Adjusted indirect comparison may be less biased than direct comparison for evaluating new pharmaceutical interventions. J Clin Epidemiol 2008;61:455-63.
- Lunn DJ, Thomas A, Best N, Spiegelhalter D. WinBUGS – a Bayesian modelling framework: concepts, structure, and extensibility. Stat Comput 2000;10:325-37.
- Lambert PC, Sutton AJ, Burton PR, Abrams KR, Jones DR. How vague is vague? A simulation study of the impact of the use of vague prior distributions in MCMC using WinBUGS. Stat Med 2005;24:2401-28.
- Flemming K, Briggs M. Electronic searching to locate qualitative research: evaluation of three strategies. J Adv Nurs 2007;57:95-100.
- Hawker S, Payne S, Kerr C, Hardey M, Powell J. Appraising the evidence: reviewing disparate data systematically. Qual Health Res 2002;12:1284-99.
- National Institute for Health and Clinical Excellence . Guide to the Methods of Technology Appraisal 2008.
- Dolan P. Modeling valuations for EuroQol health states. Med Care 1997;35:1095-108.
- Calis M, Demir H, Ulker S, Kirnap M, Duygulu F, Calis HT. Is intraarticular sodium hyaluronate injection an alternative treatment in patients with adhesive capsulitis?. Rheumatol Int 2006;26:536-40.
- Dacre JE, Beeney N, Scott DL. Injections and physiotherapy for the painful stiff shoulder. Ann Rheum Dis 1989;48:322-5.
- Bal A, Eksioglu E, Gulec B, Aydog E, Gurcay E, Cakci A. Effectiveness of corticosteroid injection in adhesive capsulitis. Clin Rehabil 2008;22:503-12.
- Rovetta G, Monteforte P. Intraarticular injection of sodium hyaluronate plus steroid versus steroid in adhesive capsulitis of the shoulder. Int J Tissue React 1998;20:125-30.
- Takagishi K, Saito A, Segawa K, Takahira N, Itomitu S. Evaluation of intra-articular injection in patients with so-called Gojyukata: comparison hyaluronate and steroid. Jpn J Med Pharm Sci 1996;35:377-81.
- Wies J. A pilot randomised placebo controlled trial of osteopathic and physiotherapy treatment for frozen shoulder. J Osteopath Med 2003;6.
- Yan F. Comparison of dumbbell gymnastics and bare-handed exercise in ameliorating the symptoms of shoulder periarthritis. Chin J Clin Rehab 2005;9:187-9.
- Diercks RL, Stevens M. Gentle thawing of the frozen shoulder: a prospective study of supervised neglect versus intensive physical therapy in seventy-seven patients with frozen shoulder syndrome followed up for two years. J Shoulder Elbow Surg 2004;13:499-502.
- Dundar U, Toktas H, Cakir T, Evcik D, Kavuncu V. Continuous passive motion provides good pain control in patients with adhesive capsulitis. Int J Rehabil Res 2009;32:193-8.
- Leung MSF, Cheing GLY. Effects of deep and superficial heating in the management of frozen shoulder. J Rehabil Med 2008;40:145-50.
- Maricar NN, Chok B. A comparison of the effect of manual therapy with exercise therapy and exercise therapy alone for stiff shoulders. Physiother Singapore 1999;2:99-104.
- Pajareya K, Chadchavalpanichaya N, Painmanakit S, Kaidwan C, Puttaruksa P, Wongsaranuchit Y. Effectiveness of physical therapy for patients with adhesive capsulitis: a randomized controlled trial. J Med Assoc Thai 2004;87:473-80.
- Yang J-l, Chang C-W, Chen S-Y, Wang S-F, Lin J-J. Mobilization techniques in subjects with frozen shoulder syndrome: randomized multiple-treatment trial. Phys Ther 2007;87:1307-15.
- Cheing GLY, So EML, Chao CYL. Effectiveness of electroacupuncture and interferential eloctrotherapy in the management of frozen shoulder. J Rehabil Med 2008;40:166-70.
- Fang J-Q, Zhang Y, Xuan L-H, Liu K-Z, Chen L. Observation on clinical therapeutic effect of transcutaneous point electric stimulation on periarthritis of shoulder at different stages. Zhongguo Zhenjiu 2006;26:11-4.
- Ma T, Kao MJ, Lin IH, Chiu YL, Chien C, Ho TJ, et al. A study on the clinical effects of physical therapy and acupuncture to treat spontaneous frozen shoulder. Am J Chin Med 2006;34:759-75.
- Amir-us-Saqlain H, Zubairi A, Taufiq I. Functional outcome of frozen shoulder after manipulation under anaesthesia. J Pak Med Assoc 2007;57:181-5.
- Jacobs LG, Smith MG, Khan SA, Smith K, Joshi M. Manipulation or intra-articular steroids in the management of adhesive capsulitis of the shoulder? A prospective randomized trial. J Shoulder Elbow Surg 2009;18:348-53.
- Gam AN, Schydlowsky P, Rossel I, Remvig L, Jensen EM. Treatment of ‘frozen shoulder’ with distension and glucorticoid compared with glucorticoid alone. A randomised controlled trial. Scand J Rheumatol 1998;27:425-30.
- Austgulen OK, Oyen J, Hegna J, Solheim E. Arthroscopic capsular release in treatment of primary frozen shoulder. Tidsskr Nor Laegeforen 2007;127:1356-8.
- Chen S-K, Chien S-H, Fu Y-C, Huang P-J, Chou P-H. Idiopathic frozen shoulder treated by arthroscopic brisement. Kaohsiung J Med Sci 2002;18:289-94.
- Höfler M. The Bradford Hill considerations on causality: a counterfactual perspective. Emerg Themes Epidemiol 2005;2.
- Salanti G, Higgins J, Ades A, Ioannidis J. Evaluation of networks of randomized trials. Stat Methods 2008;17:279-301.
- Ucuncu F, Capkin E, Karkucak M, Ozden G, Cakirbay H, Tosun M, et al. A comparison of the effectiveness of lanmark-guided injections and ultrasonography-guided injections for shoulder pain. Clin J Pain 2009;25:786-9.
- Weber MD. Factors that predict change in health-related qualityof life of patients with adhesive capsulitis. Jackson, MS: University of Mississippi Medical Centre; 2001.
- van den Hout WB, Vermeulen HM, Rozing PM, Vliet Vlieland TPM. Impact of adhesive capsulitis and economic evaluation of high-grade and low-grade mobilisation techniques. Aust J Physiother 2005;51:141-9.
- National Institute for Health and Clinical Excellence . Guide to the Methods of Technology Appraisal 2004. www.nice.org.uk/aboutnice/howwework/devnicetech/technologyappraisalprocessguides/guide_to_the_methods_of_technology_appraisal_reference_n0515.jsp (accessed 9 March 2009).
- Curtis L. Unit costs of health and social care 2009. Canterbury: Personal Social Services Research Unit, University of Kent; 2010.
- British Medical Association and Royal Pharmaceutical Society of Great Britain . British National Formulary 2010.
- Department of Health . NHS Reference Costs 2008–2009 2010. www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/@dh/@en/@ps/documents/digitalasset/dh_118329.pdf (accessed 2 February 2011).
- Brazier J, Roberts J, Deverill M. The estimation of a preference-based measure of health from the SF-36. J Health Econ 2002;21:271-92.
- Feeny D, Furlong W, Torrance G, Goldsmith C, Zhu Z, DePaw S, et al. Multi-attribute and single-attribute utility functions for the Health Utilities Index Mark 3 system. Med Care 2002;40:113-28.
- Sintonen H. The 15D instrument of health-related quality of life: properties and applications. Ann Med 2001;33:328-36.
- Brazier JE, Yang Y, Tsuchiya A, Rowen DL. A review of studies mapping (or cross walking) non-preference based measures of health to generic preference-based measures. Eur J Health Econ 2010;11:215-25.
- Rowen D, Brazier J, Roberts J. Mapping SF-36 onto the EQ-5D index: how reliable is the relationship?. Health Qual Life Outcomes 2009;7.
- Watson J, Helliwell P, Morton V, Adebajo A, Dickson J, Russell I, et al. Shoulder acute pain in primary healthcare: is retraining effective for GP principals? SAPPHIRE – a randomized controlled trial. Rheumatology 2008;47:1795-802.
- Watson J, Helliwell P, Morton V, Adebajo A, Dickson J, Russell I, et al. Shoulder acute pain in primary healthcare: is retraining effective for GP principals? SAPPHIRE—a randomized controlled trial. Rheumatology 2008;48:558-63.
- Croft P, Pope D, Zonca M, O’Neill T, Silman A. Measurement of shoulder related disability: results of a validation study. Ann Rheum Dis 1994;53:525-8.
- McKenna C, Bojke L, Manca A, Adebajo A, Dickson J, Helliwell P, et al. Shoulder acute pain in primary health care: is retraining GPs effective? The SAPPHIRE randomized trial: a cost-effectiveness analysis. Rheumatology 2009;48:558-63.
- Hurvitz AP, Hodapp KL, Jadgchew J, Solomon DJ, Stolldorf HS, Provencher MT. Central serous chorioretinopathy resulting in altered vision and color perception after glenohumeral corticosteroid injection. Orthopedics 2009;32. http://orthosupersite.com/view.asprID=A1926 (accessed 22 March 2010).
- Lee KS. Musculoskeletal ultrasound: how to evaluate for Morton’s neuroma. AJR Am J Roentgenol 2009;193.
- Ware J, Sherbourne C. The MOS 36-item short-form health survey (SF-36): conceptual framework and item selection. Med Care 1992;30:473-81.
- Snee RD. Validation of regression models. Methods and examples. Technometrics 1977;19:415-28.
- Chay K, Powell J. Semiparametric censored regression models. J Econ Persp 2001;15:29-42.
- Sullivan PWG. Mapping the EQ-5D from the SF-12: US general population preferences in a nationally representative sample. Med Decis Making 2006;26:401-9.
- Tobin J. Estimation of relationships for limited dependent variables. Econometrica 1958;26:22-36.
- Crott R, Briggs A. Mapping the QLQ-C30 quality of life cancer questionnaire to EQ-5D patient preferences. Eur J Health Econ 2010;11:427-34.
- Barton G, Sach T, Jenkinson C, Avery A, Doherty M, Muir K. Do estimates of cost–utility based on the EQ-5D differ from those based on the mapping of utility scores?. Health Qual Life Outcomes 2008;6.
- Sach T, Barton G, Jenkinson C, Doherty M, Avery A, Muir K. Comparing cost–utility estimates: does the choice of EQ-5D or SF-6D matter?. Med Care 2009;47:889-94.
- Blanchard V, Barr S, Cerisola FL. The effectiveness of corticosteroid injections compared with physiotherapeutic interventions for adhesive capsulitis: a systematic review. Physiotherapy 2010;96:95-107.
- Saito S, Furuya T, Kotake S. Therapeutic effects of hyaluronate injections in patients with chronic painful shoulder: a meta-analysis of randomized controlled trials. Arthritis Care Res 2010;62:1009-18.
- Favejee MM, Huisstede BM, Koes BW. Frozen shoulder: the effectiveness of conservative and surgical interventions – systematic review. Br J Sports Med 2010;45:19-56.
- de Bruijn C, Goossens M, de Bie R, Ament A, Geraets J, Dinant GJ. Cost-effectiveness of an education and activation program for patients with acute and subacute shoulder complaints compared to usual care. Int J Technol Assess Health Care 2007;23:80-8.
- James M, Stokes EA, Thomas E, Dziedzic K, Hay EM. A cost consequences analysis of local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in primary care. Rheumatology 2005;44:1447-51.
- Geraets JJXR, Goossens MEJB, de Bruijn CPC, de Groot IJM, Koke AJS, Pelt RAGB, et al. Cost-effectiveness of a graded exercise therapy program for patients with chronic shoulder complaints. Int J Technol Assess Health Care 2006;22:76-83.
- Bergman GJ, Winter JC, van Tulder MW, Meyboom-De Jong B, Postema K, van der Heijden GJ. Manipulative therapy in addition to usual medical care accelerates recovery of shoulder complaints at higher costs: economic outcomes of a randomized trial. BMC Musculoskelet Disord 2010;11.
- Holdgate A, Asha S, Craig J, Thompson J. Comparison of a verbal numeric rating scale with the visual analogue scale for the measurement of acute pain. Emerg Med (Fremantle) 2003;15:441-6.
- Beaton D, Richards R. Assessing the reliability and responsiveness of 5 shoulder questionnaires. J Shoulder Elbow Surg 1998;7:565-72.
- Kirkley A, Griffin S, Dainty K. Scoring systems for the functional assessment of the shoulder. Arthroscopy 2003;19:1109-20.
- Bot S, Terwee C, van der Windt D, Bouter L, Dekker J, de Vet H. Clinimetric evaluation of shoulder disability questionnaires: a systematic review of the literature. Ann Rheum Dis 2004;63:335-41.
- Staples MP, Forbes A, Green S, Buchbinder R. Shoulder-specific disability measures showed acceptable construct validity and responsiveness. J Clin Epidemiol 2010;63:163-70.
- Dworkin RH, Turk DC, McDermott MP, Peirce-Sandner S, Burke LB, Cowan P, et al. Interpreting the clinical importance of group differences in chronic pain clinical trials: IMMPACT recommendations. Pain 2009;146:238-44.
- NCT00160784. Arthroscopy and Manipulation Vs a Home Therapy Program in the Treatment of Adhesive Capsulitis n.d. http://ClinicalTrials.gov/show/NCT00160784 (accessed 6 April 2010).
- NCT00840229. Capsular and Intra-Articular Corticosteroid Injection for Adhesive Capsulitis (frozen shoulder): A Randomised, Double Blind, Placebo Controlled Trial n.d. http://ClinicalTrials.gov/show/NCT00840229 (accessed 6 April 2010).
- NCT00679887. Chronic Shoulder Pain Treated by Pressures With the Thumbs on the Trigger Points n.d. http://ClinicalTrials.gov/show/NCT00679887 (accessed 6 April 2010).
- NCT00261196. Collagenase in the Treatment of Adhesive Capsulitis (frozen Shoulder) n.d. http://ClinicalTrials.gov/show/NCT00261196 (accessed 6 April 2010).
- NCT00873158. Dynamic Splinting for Patients With Adhesive Capsulitis n.d. http://ClinicalTrials.gov/show/NCT00873158 (accessed 6 April 2010).
- NCT01087229. Effectiveness of an Oxygen-Nitrous Oxide Mixture During Physical Therapy for Frozen Shoulder n.d. http://ClinicalTrials.gov/show/NCT01087229 (accessed 6 April 2010).
- NCT00884065. Effectiveness of Diacutaneous Fibrolysis in Painful Shoulder n.d. http://ClinicalTrials.gov/show/NCT00884065 (accessed 6 April 2010).
- NCT00415441. Effectiveness of Physiotherapy for Chronic Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00415441 (accessed 6 April 2010).
- NCT00742846. Effects of Intra-Articular Versus Subacromial Steroid Injections on Clinical Outcomes in Adhesive Capsulitis n.d. http://ClinicalTrials.gov/show/NCT00742846 (accessed 6 April 2010).
- NCT00694538. Efficacy of Interferential Laser Therapy in Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00694538 (accessed 6 April 2010).
- NCT00680472. HKT-500 in Adult Patients With Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00680472 (accessed 6 April 2010).
- NCT00377624. HUPS: Hyalgan Use in Painful Shoulder n.d. http://ClinicalTrials.gov/show/NCT00377624 (accessed 6 April 2010).
- NCT00211718. Intra-Articular Injection of Botulinum Toxin Type A for Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00211718 (accessed 6 April 2010).
- NCT00929305. Low Level Laser Light Therapy and Chronic Neck and Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00929305 (accessed 6 April 2010).
- NCT00172601. Mobilization Techniques in Patients With Frozen Shoulder: A Randomized Multiple-Treatment Trial n.d. http://ClinicalTrials.gov/show/NCT00172601 (accessed 6 April 2010).
- NCT00587626. Proposal to Evaluate the Efficacy of the InterX 5000 in the Treatment of Chronic Neck and Shoulder Pain n.d. http://ClinicalTrials.gov/show/NCT00587626 (accessed 6 April 2010).
- NCT00163124. A Randomized Controlled Trial of Best Approach to Care Compared to Diversified Chiropractic Adjustive Technique n.d. http://ClinicalTrials.gov/show/NCT00163124 (accessed 6 April 2010).
- NCT00992927. Should the Joint Capsule of the Painful Stiff Shoulder Be Ruptured During Intra-Articular Hydraulic Distension? n.d. http://ClinicalTrials.gov/show/NCT00992927 (accessed 6 April 2010).
- NCT00875862. Shoulder Adhesive Capsulitis and Ambulatory Continuous Interscalene Nerve Blocks n.d. http://ClinicalTrials.gov/show/NCT00875862 (accessed 6 April 2010).
- NCT01029600. Surgery or Capsular Distention With Steroid in the Treatment of Primary Frozen Shoulder? n.d. http://ClinicalTrials.gov/show/NCT01029600 (accessed 6 April 2010).
- Anonymous . Steroid injections vs PT for stiff shoulder. Joint Lett 1999;5.
- Van der Heijden GJM, . Anonymous. UK physio challenges Dutch findings . No effect of bipolar interferential electrotherapy and pulsed ultrasound for soft tissue shoulder disorders: a randomised controlled trial. Ann Rheum Dis 1999;58:530-40.
- Anonymous . Pearls. Phys Sportsmed 2001;29.
- Anonymous . Statistical reanalysis of four recent acupuncture trials. Acupunct Med 2004;22.
- Anonymous . Acupuncture for shoulder pain (n = 130). Acupunct Med 2005;23.
- Anonymous . Manipulative therapies. Focus Altern Complement Ther 2005;10.
- Anonymous . Application of fascial manipulation technique in chronic shoulder pain: anatomical basis and clinical implications. SportEX Dynamics 2009;22.
- Ahmad I, Askar Z, Durrani Z, Idrees M, Ayaz M, Hakim A, et al. Intraarticular injection of methylprednisolone for idiopathic frozen shoulder. J Med Sci 2009;17:16-8.
- Ahn K, Lee Y-J, Kim E-H, Yang S-M, Lim T-K, Kim Y-S, et al. Interventional microadhesiolysis: a new nonsurgical release technique for adhesive capsulitis of the shoulder. BMC Musculoskelet Disord 2008;9.
- Ainsworth R, Dziedzic K, Hiller L, Daniels J, Bruton A, Broadfield J. A prospective double blind placebo-controlled randomized trial of ultrasound in the physiotherapy treatment of shoulder pain. Rheumatology 2007;46:815-20.
- Alegre Marcet C. Trimethylacetate of prednisolone for intra-articular injection in therapy of several disorders of the locomotor apparatus. Rev Clin Esp 1959;74:32-5.
- Alexander LD, Gilman DRD, Brown DR, Brown JL, Houghton PE. Exposure to low amounts of ultrasound energy does not improve soft tissue shoulder pathology: a systematic review. Phys Ther 2010;90:14-25.
- Allano G, Katz D, Fournier-Boursier A. Physiotherapy of interscalenic blocks in painful or stiff shoulders. Kinesither Sci 2005;452:21-4.
- Altman RD, Moskowitz R, Jacobs S, Daley M, Udell J, Levin R, et al. A double-blind, randomized trial of intra-articular injection of sodium hyaluronate (hyalgan (R)) for the treatment of chronic shoulder pain. Arthritis Rheum 2005;52.
- Andersen NH, Sojbjerg JO, Johannsen HV, Sneppen O. Frozen shoulder: arthroscopy and manipulation under general anesthesia and early passive motion. J Shoulder Elbow Surg 1998;7:218-22.
- Andersen NH, Johannsen HV, Sneppen O, Sojbjerg JO. Frozen shoulder. Arthroscopy and manipulation in general anesthesia, followed by early passive mobilization. Ugeskr Laeger 1996;158:147-50.
- Andren L, Lundberg BJ. Treatment of rigid shoulders by joint distension during arthrography. Acta Orthop Scand 1965;36:45-53.
- Ankermann KJ, Gobisch F. The frozen shoulder: results of a 10-year study. Z Arztl Fortbild (Jena) 1986;80:975-80.
- Aoki T. Clinical study of tiaprofenic acid on frozen shoulder. Jpn Pharmacol Therapeut 1982;10:605-39.
- Aoki T, Yamamoto M, Kawaji W, Yamamoto R, Sugawara S, Ogawa R. Clinical evaluation of CN-100 on lumbago, cervicobrachial syndrome and periarthritis scapulohumeralis. Multicenter double-blind study in comparison with indomethacin. Jpn Pharmacol Therapeut 1991;19:197-222.
- Arias M, Alcala T. Frozen shoulder: a new approach to its treatment with local anaesthesia. Eur J Phys Rehabil Med 1992;2:11-4.
- Arslan S, Celiker R. Comparison of the efficacy of local corticosteroid injection and physical therapy for the treatment of adhesive capsulitis. Rheumatol Int 2001;21:20-3.
- Atra E, Sarmento P, Pizzanelli M. Comparative study between glucametacin and indomethacin in the treatment of acute periarthritis of the shoulder. Arq Bras Med 1986;60:423-30.
- Avetisova EG, Ovchinnikova IP, Yankovskaya AN. Physio therapeutic treatment of patients with scapulo humeral peri arthritis. Kazanskii Meditsinskii Zhurnal 1980;61:22-3.
- Awad T, Losada M, Losada A. Treatment of scapulo-humeral periarthritis with alpha-chymotrypsin and rehabilitation. Rev Med Chil 1967;95:372-6.
- Azevedo DC, de Carvalho SC, Leal EWPS, Damasceno SP, Ferreira ML. Influence of the range of motion (ROM) limitation on the shoulder flexibility improvement after a six-week training. Rev Bras Med Esporte 2008;14:119-21.
- Badalamente M, Ang W, Selby RM, Safran M. Enzymatic Capsulotomy for Adhesive Capsulitis n.d. www3.aaos.org/education/anmeet/anmt2006/podium/ppr06_22.cfm.
- Badalamente MA. inventor, Advance Biofactures Corporation, assignee . Methods for Treating Adhesive Capsulitis 2009.
- Bancheri C, Di Loreto C, Occhi P. Evaluation of the effectiveness of peri- and intraarterial somatostatin 14 in gonarthrosis and scapulo-humeral periarthritis. G Ital Ricerche Clin Terapeutiche 1993;14:93-7.
- Baslund B, Thomsen BS, Jensen EM. Humero-scapular periarthrosis. Ugeskr Laeger 1991;153:170-3.
- Batra YK, Chari P, Negi ON. Comparison of acupuncture and placebo in treatment of chronic shoulder pain. Am J Acupunct 1985;13:69-71.
- Battisti E, Bianciardi L, Albanese A, Piazza E, Rigato M, Galassi G, et al. The new magnetic therapy TAMMEF in the treatment of simple shoulder pain. Clin Ter 2007;158:397-401.
- Battisti E, Albanese A, Bianciardi L, Fiaschi AI, Rigato M, Vittoria A, et al. TAMMEF therapy in the treatment of shoulder periarthritis: efficacy and safety. Environmentalist 2009;29:190-5.
- Baumann F. Ventral capsular denervation: an operative treatment of periarthropathia humero-scapularis. Arch Orthop Trauma Surg 1981;98:13-7.
- Baumgartner H, Wagenhauser FJ. Results of mobilization under narcosis. Orthopade 1981;10:238-41.
- Baums MH, Spahn G, Nozaki M, Steckel H, Schultz W, Klinger HM. Functional outcome and general health status in patients after arthroscopic release in adhesive capsulitis. Knee Surg Sports Traumatol Arthrosc 2007;15:638-44.
- Beaufils P, Prevot N, Boyer T, Allard M, Dorfmann H, Frank A, et al. Gleno-humeral arthroscopic arthrolysis for shoulder stiffness. Apropos of 26 cases. Rev Chir Orthop Reparatrice Appar Mot 1996;82:608-14.
- Beaufils P, Prevot N, Boyer T, Allard M, Dorfmann H, Frank A, et al. Arthroscopic release of the glenohumeral joint in shoulder stiffness: a review of 26 cases. Arthroscopy 1999;15:49-55.
- Beckerman H, Bouter LM, van der Heijden GJ, de Bie RA, Koes BW. Efficacy of physiotherapy for musculoskeletal disorders: what can we learn from research?. Br J Gen Pract 1993;43:73-7.
- Bell S, Coghlan J, Richardson M. Hydrodilatation in the management of shoulder capsulitis. Australas Radiol 2003;47:247-51.
- Bellmann H, Zacharias J, Hasert V, Kretschmar KH, Weidenbach H. Use of hylase ‘Dessau’ in periarthritis humero-scapularis (Duplay syndrome). Zentralbl Chir 1969;94:1288-304.
- Bennett WF. Addressing glenohumeral stiffness while treating the painful and stiff shoulder arthroscopically. Arthroscopy 2000;16:142-50.
- Beres I, Staba R. Treatment of periarthritis humeroscapularis by acupuncture. Acta Chir Orthop Traumatol Cech 1979;46:436-40.
- Berger MR, Froimson AI. Frozen shoulder. Nurse Pract 1980;5:28-9.
- Berghs BM, Sole-Molins X, Bunker TD. Arthroscopic release of adhesive capsulitis. J Shoulder Elbow Surg 2004;13:180-5.
- Berglezov MA, Vial’ko VV. Complex treatment of brachio-scapular periarthrisis using laser therapy. Klin Khir 1986:31-2.
- Bergman GJD, Winters JC, van der Heijden GJMG, Postema K, Meyboom-de Jong B. Groningen Manipulation Study. The effect of manipulation of the structures of the shoulder girdle as additional treatment for symptom relief and for prevention of chronicity or recurrence of shoulder symptoms. Design of a randomized controlled trial within a comprehensive prognostic cohort study. J Manipulative Physiol Ther 2002;25:543-9.
- Bergman GJD, Winters JC, Groenier KH, Pool JJM, Meyboom-de Jong B, Postema K, et al. Manipulative therapy in addition to usual medical care for patients with shoulder dysfunction and pain: a randomized, controlled trial. Ann Intern Med 2004;141:432-9.
- Bergman G, Winters J, Groenier K, Pool J, Meyboom-de Jong B, Postema K, et al. Additional manual therapy in patients with shoulder complaints: a randomized effect study. Huisarts Wet 2005;48:438-43.
- Bergman GJ, Winters JC, Groenier KH, Meyboom-de Jong B, Postema K, van der Heijden GJ. Manipulative therapy in addition to usual care for patients with shoulder complaints: results of physical examination outcomes in a randomized controlled trial. J Manipulative Physiol Ther 2010;33:96-101.
- Bertoft ES. Painful shoulder disorders from a physiotherapeutic view: a review of literature. Crit Rev Phys Rehab Med 1999;11:229-77.
- Bettermann AA. Periarthritis humeroscapularis: alternative pain treatment with magnet plasters. Ther Ggw 1982;121:487-92.
- Bicer A, Ozisik S, Aksik SC, Erdogan C. Comparison of local corticosteroid injection and conventional physical therapy in management of the painful shoulder. Turkiye Klinikleri J Med Sci 2005;25:506-12.
- Bierner SM. Manipulation in the treatment of frozen shoulder. Orthopedics 1989;12.
- Bilgici A, Kuru O, Alayli G, Ulusoy H. Static stretching therapy with traction in adhesive capsulitis of the shoulder. J Rheumatol Med Rehabil 2002;13:102-7.
- Bingol U, Altan L, Yurtkuran M. Low-power laser treatment for shoulder pain. Photomed Laser Surg 2005;23:459-64.
- Biswas AK, Sur BN, Gupta CR. Treatment of periarthritis shoulder. J Indian Med Assoc 1979;72:276-7.
- Blaine T, Moskowitz R, Udell J, Skyhar M, Levin R, Friedlander J, et al. Treatment of persistent shoulder pain with sodium hyaluronate: a randomized, controlled trial. A multicenter study. J Bone Joint Surg Am 2008;90:970-9.
- Blanchard TK, Bearcroft PW, Maibaum A, Hazelman BL, Sharma S, Dixon AK. Magnetic resonance imaging or arthrography for shoulder problems: a randomised study. Eur J Radiol 1999;30:5-10.
- Blauth W. Diagnosis and therapy of frozen shoulder. Hefte Unfallheilkd 1989:68-81.
- Booi GA, Vanhorn JR, Vanrens TJG. Results and comparison of a Neer acromioplasty versus manipulation under anesthetics of the painful shoulder. Acta Orthop Scand 1986;57.
- Bosch Olives V, Llurba Llurba J, Peinado Vistuer A. Electrophysiotherapy in the treatment of a frozen shoulder. Rev Esp Reum Enferm Osteoartic 1967;12:149-56.
- Boyer T, Dorfmann H, Berquet R. Frozen shoulder. Rhumatologie 1994;46:111-16.
- Boylan M. Massage and exercise reduces pain and improves function in frozen shoulder. J Aus Tradit Med Soc 2005;11.
- Boylan M. Soft tissue massage improves range of motion, function and pain in shoulder pain. J Aus Tradit Med Soc 2005;11:177-8.
- Boyles RE, Flynn TW, Whitman JM. Manipulation following regional interscalene anesthetic block for shoulder adhesive capsulitis: a case series. Man Therap 2005;10:164-71.
- Bratanova KA. Medical exercises in the treatment of periarthritis of the shoulder. Med Sestra 1978;37:41-2.
- Brigo B, Campaccu R, Schinina V. Reflex therapy in functional rehabilitation: treatment of scapulohumeral periarthritis by needle puncture. Riabilitazione 1981;14:25-32.
- Brox JI. Regional musculoskeletal conditions: shoulder pain. Best Pract Res Clin Rheumatol 2003;17:33-56.
- Buchbinder R, Green S, Hall S, Lawler G, Forbes A. Efficacy of hydrodilatation for frozen shoulder (FS): results of a randomised double-blind placebo-controlled trial. Aust N Z J Med 2000;30.
- Buchbinder R, Green S, Youd JM. Corticosteroid injections for shoulder pain. Cochrane Database Syst Rev 2003.
- Buchbinder R, Hoving JL, Green S, Hall S, Forbes A, Nash P. Short course prednisolone for adhesive capsulitis (frozen shoulder or stiff painful shoulder): a randomised, double blind, placebo controlled trial. Ann Rheum Dis 2004;63:1460-9.
- Buchbinder R, Green S. Effect of arthrographic shoulder joint distension with saline and corticosteroid for adhesive capsulitis. Br J Sports Med 2004;38:384-5.
- Buchbinder R, Youd JM, Green S, Stein A, Bennell K, Harris A, et al. Physiotherapy (manual therapy and directed exercise) following arthrographic distension of the glenohumeral joint for adhesive capsulitis: a randomized double-blind placebo-controlled trial. Arthritis Rheum 2006;54.
- Buchbinder R, Youd JM, Green S, Stein A, Bennell K, Forbes A, et al. Physiotherapy following hydrodilatation for adhesive capsulitis: a randomised placebo-controlled double blind trial. Intern Med J 2006;36.
- Buchbinder R, Green S, Youd JM, Johnston RV. Oral steroids for adhesive capsulitis. Cochrane Database Syst Rev 2006.
- Buchbinder R, Youd JM, Green S, Stein A, Forbes A, Harris A, et al. Efficacy and cost-effectiveness of physiotherapy following glenohumeral joint distension for adhesive capsulitis: a randomized trial. Arthritis Rheum 2007;57:1027-37.
- Buchbinder R, Green S, Youd JM, Johnston RV, Cumpston M. Arthrographic distension for adhesive capsulitis (frozen shoulder). Cochrane Database Syst Rev 2008.
- Bulgen DY, Binder AI, Hazleman BL, Dutton J, Roberts S. Frozen shoulder: prospective clinical study with an evaluation of three treatment regimens. Ann Rheum Dis 1984;43:353-60.
- Bumin G, Can F. Effects of iontophoresis and phonophoresis methods on pain in cases with shoulder periarthritis. Pain Clinic 2001;13:159-62.
- Bunker TD. Frozen shoulder. Curr Orthop 1998;12:193-201.
- Calabro JJ. Indomethacin in acute painful shoulder bursitis and/or tendinitis. Semin Arthritis Rheum 1982;12:130-1.
- Caldwell KD, Nuys SR, Brown CL. Evaluation of mobilization in the treatment of frozen shoulder. Arthritis Rheum 1986;29.
- Callinan N, McPherson S, Cleaveland S, Voss DG, Rainville D, Tokar N. Effectiveness of hydroplasty and therapeutic exercise for treatment of frozen shoulder. J Hand Ther 2003;16:219-24.
- Camarinos J, Marinko L. Effectiveness of manual physical therapy for painful shoulder conditions: a systematic review. J Man Manip Ther 2009;17:206-15.
- Caniggia M, Carnevale I, Passarello F. Analgesic and antiflogistic properties of intra- and periarticular thymopentin. Riforma Med 1989;104:417-19.
- Capone M, Stancati MT, Tolla V, Chiatti R, Muscolo V, Pasquale M. Observations on the administration of sodium edetate in calcified scapulohumeral periarthritis. Ionophoresis and mesotherapy: comparison of two techniques. Ortop Traumatol Oggi 1994;14:163-8.
- Carette S, Moffet H, Tardif J, Bessette L, Fremont P, Blanchette C, et al. Intraarticular (IA) corticosteroids, physiotherapy or the combination of both in patients with adhesive capsulitis of the shoulder: a placebo controlled trial. J Rheumatol 2002;29.
- Carey TS. Adding single-point acupuncture to physiotherapy for painful shoulder improved function and reduced pain. ACP J Club 2008;149:JC4-12.
- Carter B. Clients’ experiences of frozen shoulder and its treatment with Bowen technique. Complement Ther Nurs Midwifery 2002;8:204-10.
- Casanova G. Crenotherapy and bone and joint surgery. Presse Therm Clim 1988;125:95-7.
- Castellarin G, Ricci M, Vedovi E, Vecchini E, Sembenini P, Marangon A, et al. Manipulation and arthroscopy under general anesthesia and early rehabilitative treatment for frozen shoulders. Arch Phys Med Rehabil 2004;85:1236-40.
- Castelli PG, Schneider L, Sartori G, Camisassa R, Grandi G. Release arthroscopy for post-traumatic frozen shoulder: a review of 19 cases. Minerva Ortop Traumatol 2006;57:399-404.
- Champion G. Frozen shoulder. Ann Rheum Dis 2005;64:795-6.
- Chang WH, Im SH, Ryu JA, Lee SC, Kim JS. The effects of scapulothoracic bursa injections in patients with scapular pain: a pilot study. Arch Phys Med Rehabil 2009;90:279-84.
- Chavero Carrasco V, Salguero Molpeceres O, Lopez Ruano P, Alvarez Montero S. Treatment of shoulder pain; non steroidal anti-inflammatory drug (NSAID) or intraarticular injection?. Rev Med Fam Comun 2002;12:341-3.
- Chavez-Lopez MA, Navarro-Soltero LA, Rosas-Cabral A, Gallaga A, Huerta-Yanez G. Methylprednisolone versus triamcinolone in painful shoulder using ultrasound-guided injection. Mod Rheumatol 2009;19:147-50.
- Checchia GA, Pezzoli R, Gorini L, Amadori L, Foresti A, Gazzi A. Electrotherapy in the treatment of shoulder pain. Riabilitazione 1991;24:121-7.
- Chen SK, Lin SY, Liao JS. Primary and secondary frozen shoulder. J Surg Ass Rep China 1985;18:121-8.
- Chen SK, Lin SY, Liao JS. An orthopaedic study of frozen shoulder. Gaoxiong Yi Xue Ke Xue Za Zhi 1988;4:1-9.
- Chen Y, Zheng K. New exploration of needling manipulations at point Tianzong. J Tradit Chin Med 2002;22:38-41.
- Chen ZS, Yang RK. Effects of alkalinizing administration of suprascapular nerve block on the periarthritis of shoulder-induced pain and the function of joint movement. Chin J Clin Rehab 2005;9:8-9.
- Chen L. Comparison of therapeutic effects between normal acupuncture and shallow needling with short-needle on periarthritis of shoulder. Zhongguo Zhenjiu 2006;26:647-8.
- Chen S-J, Li H, Zhang J-W. Control study of therapeutic effects of three phase acupuncture method and routine acupuncture method on periarthritis of shoulder. Zhongguo Zhenjiu 2006;26:421-3.
- Chen S-K, Chou P-H, Lue Y-J, Lu Y-M. Treatment for frozen shoulder combined with calcific tendinitis of the supraspinatus. Kaohsiung J Med Sci 2008;24:78-84.
- Chen JF, Ginn KA, Herbert RD. Passive mobilisation of shoulder region joints plus advice and exercise does not reduce pain and disability more than advice and exercise alone: a randomised trial. Aust J Physiother 2009;55:17-23.
- Cheng P, Shi WM, He J, Lang HT, Xi CL, Wu MJ. Efficacy and safety of infrared rays and Qingpeng paste in treating periarthritis of shoulder. J Clin Rehab Tissue Eng Res 2008;12:1353-6.
- Cherkashin VV, Burtseva NI. Exercise therapy in the complex treatment of scapulohumeral periarthritis. Vopr Kurortol Fizioter Lech Fiz Kult 1969;34:236-9.
- Cho S-H, Shin B-C, Kim I-H. Effects of myofascial-meridian stimulation therapy (MMST) on shoulder pain. Int J Neurosci 2005;115:1175-81.
- Ciapetti A, Salaffi F, Garofalo J, Stancati A, Filippucci E, Grassi W. Painful shoulder: short-term efficacy of a randomized controlled trial of local steroid injection versus physiotherapy. Ann Rheum Dis 2006;65.
- Cinar M, Akpinar S, Derincek A, Circi E, Uysal M. Comparison of arthroscopic capsular release in diabetic and idiopathic frozen shoulder patients. Arch Orthop Trauma Surg 2010;130:401-6.
- Cloke DJ, Watson H, Purdy S, Steen IN, Williams JR. A pilot randomized, controlled trial of treatment for painful arc of the shoulder. J Shoulder Elbow Surg 2008;17:17-21.
- Cohen NP, Levine WN, Marra G, Pollock RG, Flatow EL, Brown AR, et al. Indwelling interscalene catheter anesthesia in the surgical management of stiff shoulder: a report of 100 consecutive cases. J Shoulder Elbow Surg 2000;9:268-74.
- Compernolle P, Vansteenland H, Rosselle N. Adhesive capsulitis of the shoulder. Acta Belg Med Phys 1987;10:71-8.
- Connolly J, Regen E, Evans OB. The management of the painful, stiff shoulder. Clin Orthop Relat Res 1972;84:97-103.
- Connolly JF. Exercises to loosen the shoulder joint. J Musculoskeletal Med 1998;15.
- Coombes WN. Distension-manipulation for the treatment of adhesive capsulitis (frozen shoulder syndrome). Clin Orthop Relat Res 1984:309-10.
- Corazza G, Basaglia N. Rehabilitation of patients with periarthritis humeroscapularis. Riabilitazione 1982;15:185-91.
- Corbeil V, Dussault RG, Leduc BE, Fleury J. Adhesive capsulitis of the shoulder: a comparative study of arthrography with intra-articular corticotherapy and with or without capsular distension. Can Assoc Radiol J 1992;43:127-30.
- Cossu M, Conti B, Celestini M. EDTA ionophoresis in calcified periarthritis of the shoulder. New aspects. Riabilitazione 1993;26:19-23.
- Coudane H, Sommelet J, Mole D, Schmitt D. Arthroscopy of the shoulder in scapulo-humeral periarthritis. Rev Chir Orthop Reparatrice Appar Mot 1988;74.
- Crawshaw DP, Helliwell P, Hensor EM, Hay EM, Aldous SJ, Conaghan PG. Is there a window of opportunity for physiotherapy after steroid injections for shoulder pain? Results from a large randomised trial. Rheumatology 2009;48.
- Dacre JE, Beeney N, Scott DL. Physiotherapy or steroids for the painful stiff shoulder?. Br J Rheumatol 1987;26:1-13.
- Dahan TH, Fortin L, Pelletier M, Petit M, Vadeboncoeur R, Suissa S. Double blind randomized clinical trial examining the efficacy of bupivacaine suprascapular nerve blocks in frozen shoulder. J Rheumatol 2000;27:1464-9.
- Dal Conte G, Rivoltini P, Combi F. Pulsed magnetic field therapy for calcific periarthritis of the shoulder joint. Riabilitazione 1990;23:27-33.
- Danneskiold-Samsoe B. Manipulation of frozen shoulder?. Ugeskr Laeger 1996;158.
- de Jong BA, Dahmen R, Hogeweg JA, Marti RK. Intra-articular triamcinolone acetonide injection in patients with capsulitis of the shoulder: a comparative study of two dose regimens. Clin Rehabil 1998;12:211-15.
- de la Serna AR, Moreno MA, Pereira AA. Preliminary study to evaluate the efficacy and tolerance of joint lavage in shoulder pain vs intra-articular injection of hyaluronic acid. Osteoarthr Cartil 2004;12:S143-4.
- de Macedo JM, Goldstein RC, Marinho MASDO, Pena LW, Machado JKS. Frozen shoulder: results of the treatment with suprascapular nerve block. Rev Bras Ortop 2000;35:131-6.
- de Seze S, Debeyre J, Denis A. Surgical treatment of certain forms of periarthritis of the shoulder. Rev Rhum Mal Osteoartic 1950;17:172-5.
- Debeyre J. Surgical treatment of scapulohumeral periarthritis and of painful shoulder. Minerva Ortop 1971;22:371-3.
- Degen IL. Magnetotherapy of humeroscapular periarthritis. Ortop Travmatol Protez 1974;35:66-8.
- Denicolai F, Dettoni A, Piolatto G, Sibelli P. Assessment of the comparative efficacy of orgotein and betamethasone in scapulohumeral periarthritis and tendinopathies. Minerva Ortop 1986;37:79-86.
- Desproges-Gotteron R. Treatment of recalcitrant periarthritis of the shoulder. Rev Med Limoges 1980;11:1-2.
- Devitt M. Acupuncture and frozen shoulder. Treatment plus exercise better than exercise alone. Acupunct Today 2002;3.
- Deyle GD, Bang MD. Examination and treatment of the shoulder: a literature-based approach. Orthop Phys Ther Clin N Am 1999;8:83-115.
- DiMarcantonio T. Multiple collagenase injections effective, safe for treating ‘frozen shoulder’. Orthop Today 2006;26:62-3.
- Diwan DB, Murrell GAC. An evaluation of the effects of the extent of capsular release and of postoperative therapy on the temporal outcomes of adhesive capsulitis. Arthroscopy 2005;21:1105-13.
- Dodenhoff RM, Levy O, Wilson A, Copeland SA. Manipulation under anesthesia for primary frozen shoulder: effect on early recovery and return to activity. J Shoulder Elbow Surg 2000;9:23-6.
- Dogru H, Basaran S, Sarpel T. Effectiveness of therapeutic ultrasound in adhesive capsulitis. Rev Rhum Ed Fr 2008;75:625-31.
- Dorian R, Miletic M. Local cryotherapeutical or heat applications in patients with painful shoulder stiffness. Med Welt 1985;36:157-8.
- D’Orta S, Germani M, Parente F, Bigazzi G, Pietrogrande V. Double-blind study on acupuncture treatment of frozen shoulder. Ortop Traumatol Oggi 1985;5:37-44.
- Drakos MC, Green DM, Dodson CC, Allen AA, Warren RF. Shoulder dislocation after mobilization procedures for adhesive capsulitis. Orthopedics 2008;31.
- Duke O, Zecler E, Grahame R. Anti-inflammatory drugs in periarthritis of the shoulder: a double-blind, between-patient, study of naproxen versus indomethacin. Rheumatol Rehabil 1981;20:54-9.
- Duschatko DM. Certified pilates and gyrotonics trainer. J Bodyw Mov Ther 2000;4:13-9.
- Echternach JL. Audioanalgesia as an adjunct to mobilization of the chronic frozen shoulder. Phys Ther 1966;46:839-46.
- Ekelund AL, Rydell N. Combination treatment for adhesive capsulitis of the shoulder. Clin Orthop Relat Res 1992:105-9.
- Elleuch MH, Baklouti S, Gdoura H, Keskes H, Sellami S. The rehabilitation in the frozen shoulder syndrome. Rhumatologie 1994;46:77-80.
- Elleuch MH, Yahia A, Ghroubi S, Kharrat O, Mezghanni M, Ayedi K. The contribution of capsular distension to the treatment of primary adhesive capsulitis of the shoulder: a comparative study versus rehabilitation. Ann Readapt Med Phys 2008;51:722-8.
- Erlendsson F. Frozen shoulder. Ugeskr Laeger 1996;158:2139-40.
- Ernst E, Dong H. Encouraging long-term results for acupuncture for neck and shoulder pain. Focus Altern Complement Ther 2004;9:297-8.
- Ernst E. Physical therapy for shoulder pains?. MMW Fortschr Med 2009;151.
- Escalante Triay FJ. Combined use of cobamamide and thiocolchicoside in treatment of pain syndromes accompanied by muscle contracture. Invest Med Int 1981;8:272-8.
- Esposito S, Ragozzino A, Russo R, Minelli S, Tuccillo M. Arthrography in the diagnosis and treatment of idiopathic adhesive capsulitis. Radiol Med 1993;85:583-7.
- Eulert J, Apoil A, Dautry P. Pathogenesis and surgical treatment of ‘periarthritis humeroscapularis’. Z Orthop Ihre Grenzgeb 1981;119:25-30.
- Famaey JP, Ginsberg F. Treatment of periarthritis of the shoulder: a comparison of ibuprofen and diclofenac. J Int Med Res 1984;12:238-43.
- Famaey JP, Broux G, Cleppe D. Ionization with Voltaren. A multi-centre trial. J Belge Med Phys Rehabil 1982;5:55-60.
- Fan Z-Y, Huang Z-B, Lai S-H, Zhong W, Guo R-S. Clinical experience on the manipulative treatment of cervical frozen shoulder. Zhongguo Gushang 2008;21.
- Fareed DO, Gallivan WR. Office management of frozen shoulder syndrome. Treatment with hydraulic distension under local anesthesia. Clin Orthop Relat Res 1989:177-83.
- Farrell CM, Sperling JW, Cofield RH. Manipulation for frozen shoulder: long-term results. J Shoulder Elbow Surg 2005;14:480-4.
- Feng Zg. Two-hundred and ten cases of shoulder periarthritis treated by needling Lingxia and Sanjian. J Tradit Chin Med 2003;23:201-2.
- Filshie J. Acupuncture improves short- and long-term pain and disability in patients with shoulder pain compared with a non-penetrating sham treatment. Focus Altern Complement Ther 2005;10:124-5.
- Flannery O, Mullett H, Colville J. Adhesive shoulder capsulitis: does the timing of manipulation influence outcome?. Acta Orthop Belg 2007;73:21-5.
- Foster NE, Vas J. Trial suggests that adding single-point acupuncture to a package of physiotherapy is better for shoulder pain patients than adding mock TENS to the same physiotherapy package of care. Focus Altern Complement Ther 2008;13:276-8.
- Fuhr AW, Menke JM. Status of activator methods chiropractic technique, theory, and practice. J Manipulative Physiol Ther 2005;28:e1-e20.
- Fujiwara M, Tamura K, Odera K, Tada K, Takaya Y, Ikeda N. Injection therapy of high molecular sodium hyaluronate (ARTZ) for patients with periarthritis scapulohumeralis: evaluation of combined therapy with non-steroidal anti-inflammatory drugs. Jpn J Med Pharm Sci 1993;30:965-70.
- Fukuhara F. Clinical evaluation of the OTC product K98–01 (KINKAN) on the shoulder stiffness, backache, confusion, sprain. Jpn Pharmacol Therapeut 1999;27:255-62.
- Fura M, Lenartowski E, Soroka L. Methods and results of treatment of primary painful shoulder. Chir Narzadow Ruchu Ortop Pol 1981;46:65-8.
- Gabrhelik T, Adamus M, Michalek P, Pieran M. Invasive therapy for shoulder pain. Bolest 2009;12:101-6.
- Gado K, Emery P. Intra-articular guanethidine injection for resistant shoulder pain: a preliminary double blind study of a novel approach. Ann Rheum Dis 1996;55:199-201.
- Galarraga B. Adhesive capsulitis of the shoulder: a review article. CPD Rheumatology 2002;3:54-8.
- Galgano RC. Manipulative therapy for patients with shoulder symptoms. Ann Intern Med 2005;142.
- Garrido FV, Muñoz F, Monasterioguren XV, Ortiz AM, Orejón CO, Hernández PS, et al. Effectiveness of hyperthermia at 434 MHz after manipulation under anesthesia in the treatment of frozen shoulder. Fisoterapia 2009;31:203-12.
- Gaspar PD, Willis FB. Adhesive capsulitis and dynamic splinting: a controlled, cohort study. BMC Musculoskelet Disord 2009;10.
- Gavant ML, Rizk TE, Gold RE, Flick PA. Distention arthrography in the treatment of adhesive capsulitis of the shoulder. J Vasc Interv Radiol 1994;5:305-8.
- Geraets JJXR, Goossens MEJB, de Bruijn CPC, Koke AJA, de Bie RA, Pelt RAGB, et al. A behavioural treatment for chronic shoulder complaints: concepts, development, and study design. Aust J Physiother 2004;50:33-8.
- Geraets JJXR, Goossens MEJB, de Groot IJM, de Bruijn CPC, de Bie RA, Dinant G-J, et al. Effectiveness of a graded exercise therapy program for patients with chronic shoulder complaints. Aust J Physiother 2005;51:87-94.
- Gerber C, Espinosa N, Perren TG. Arthroscopic treatment of shoulder stiffness. Clin Orthop Relat Res 2001:119-28.
- Gilula LA, Schoenecker PL, Murphy WA. Shoulder arthrography as a treatment modality. AJR Am J Roentgenol 1978;131:1047-8.
- Ginn KA, Herbert R, Khouw W, Lee R. A Randomised Controlled Clinical Trial of the Effectiveness of a Physiotherapy Treatment for Shoulder Pain n.d.
- Ginn KA, Herbert RD, Khouw W, Lee R. A randomized, controlled clinical trial of a treatment for shoulder pain. Phys Ther 1997;77:802-9.
- Ginn K. Corticosteroid injections are more effective than joint mobilisations and exercise for managing painful stiff shoulders. Aust J Physiother 1999;45.
- Ginn KA, Cohen ML. Exercise therapy for shoulder pain aimed at restoring neuromuscular control: a comparative clinical trial. J Rehabil Med 2001;37:115-22.
- Ginn KA, Cohen ML. Conservative treatment for shoulder pain: prognostic indicators of outcome. Arch Phys Med Rehabil 2004;85:1231-5.
- Ginn KA, Cohen ML. Exercise therapy for shoulder pain aimed at restoring neuromuscular control: a randomized comparative clinical trial. J Rehabil Med 2005;37:115-22.
- Ginn K. Do passive mobiliztions [sic] applied to shoulder region joints improve the treatment outcome in patients with shoulder pain?. N Z J Physiother 2009;37.
- Ginsberg F, Famaey JP. Double-blind, randomized crossover study of the percutaneous efficacy and tolerability of a topical indomethacin spray versus placebo in the treatment of tendinitis. J Int Med Res 1991;19:131-6.
- Gobezie R, Pacheco IH, Petit CJ, Millett PJ. Dislocation and instability after arthroscopic capsular release for refractory frozen shoulder. Am J Orthop 2007;36:672-4.
- Goh GJ, Over KE, Daroszewska A, Whitehouse GH, Bucknall RC. The value of arthrography in steroid injection of the shoulder joint. Br J Rheumatol 1997;36:709-10.
- Gotte S, Spreitzer FO, Stratz T. Percutaneous treatment of periarthritis humeroscapularis with a new formulation of etofenamate. Z Allgemeinmed 1986;62:1198-201.
- Gotter G. Comparative evaluation of tenoxicam and piroxicam in the treatment of humeroscapular periarthritis. Eur J Rheumatol Inflamm 1987;9:95-7.
- Graber MA. Treating shoulder pain: a randomized trial. J Fam Pract 1997;45:103-4.
- Grabovoi AF, Grishko AI, Rodichkin VA, Rudenko AT, Gozhenko IS. Blockade of the suprascapular nerve in the complex treatment of humero-scapular periarthritis. Vestn Khir Im I I Grek 1986;136:65-6.
- Grammont P, Lelaurin G, Lemaire JP. Surgical treatment of chronic periarthritis of the shoulder joint. Acta Orthop Belg 1982;48:762-74.
- Green S, Buchbinder R, Hetrick S. Physiotherapy interventions for shoulder pain. Cochrane Database Syst Rev 2003.
- Green S. Physiotherapy and injection better than injection alone or physiotherapy alone for improving range of motion in adhesive capsulitis. Aust J Physiother 2003;49.
- Green S, Buchbinder R, Hetrick S. Acupuncture for shoulder pain. Cochrane Database Syst Rev 2005.
- Green S, Buchbinder R, Glazier R, Forbes A. Interventions for shoulder pain. Cochrane Database Syst Rev 2006.
- Green S, Alexander M. Australian Physiotherapy Association: Shoulder Pain Position Statement [with Systematic review.] n.d. www.physiotherapy.asn.au/ (accessed 19 April 2010).
- Grete J. Physical therapy in impaired mobility and pain in the shoulder. Therapiewoche 1981;31:6318-25.
- Griggs SM, Ahn A, Green A. Idiopathic adhesive capsulitis. A prospective functional outcome study of nonoperative treatment. J Bone Joint Surg Am 2000;82–A:1398-407.
- Grossi E, Monza GC, Pollavini S, Bona L. NSAID ionisation in the management of soft-tissue rheumatism: role played by the drug, electrical stimulation and suggestion. Clin Exp Rheumatol 1986;4:265-7.
- Grubbs N. Frozen shoulder syndrome: a review of literature. J Orthop Sports Phys Ther 1993;18:479-87.
- Gruehn F. On humeroscapular periarthritis. Hippokrates 1965;36:66-71.
- Gu J. The effect of audio-frequency electrotherapy and carbon dioxide laser treated 96 cases of periarthritis of shoulder. Acta Acad Med Hubei 1992;13:88-9.
- Gudushauri ON, Goguadze DM. Ambulatory treatment of humero-scapular periarthritis, humeral epicondylitis and radial styloiditis by local injections of hydrocortisone. Ortop Travmatol Protez 1975:24-6.
- Guerra de Hoyos JA, Andres Martin Mdel C, Bassas y Baena de Leon E, Vigara Lopez M, Molina Lopez T, Verdugo Morilla FA, et al. Randomised trial of long term effect of acupuncture for shoulder pain. Pain 2004;112:289-98.
- Guler-Uysal F, Kozanoglu E. Comparison of the early response to two methods of rehabilitation in adhesive capsulitis. Swiss Med Wkly 2004;134:353-8.
- Guo XY, Li YF. Etiological factor of periarthritis of shoulder and advantages of integrated traditional Chinese medicine and western medicine in treatment. Chin J Clin Rehab 2006;10:150-2.
- Guo C-Q, Zhang F-M, Sha Y, Ma G-H, Liu Y-X, Liu Y-G, et al. Randomized controlled trials of acupuncture at Tiaokou (ST 38) for treatment of periarthritis of shoulder. Zhongguo Zhenjiu 2006;26:544-6.
- Guo C-Q, Rui N, Liu Y-G, Liu Y-X, Sha Y, Ma G-H. Multi-central randomized controlled observation on clinical therapeutic effect of the new Bian-stone therapy on scapulohumeral periarthritis. Zhongguo Zhenjiu 2007;27:633-7.
- Gusarova SA. Massage in humeroscapular periarthosis. Med Sestra 1989;48:36-8.
- Gwilym SE, Mullett H, Levy O. The one-stop diagnostic clinic in shoulder surgery: costs and benefits. Clin Manag 2007;15:37-41.
- Habib GS. Systemic effects of intra-articular corticosteroids. Clin Rheumatol 2009;28:749-56.
- Haines JF, Hargadon EJ. Manipulation as the primary treatment of the frozen shoulder. J R Coll Surg Edinb 1982;27:271-5.
- Hall FM. Adhesive capsulitis. Radiology 1988;169.
- Hall FM. Frozen shoulder. Radiology 2005;235:713-14.
- Hamdan TA, Al-Essa KA. Manipulation under anaesthesia for the treatment of frozen shoulder. Int Orthop 2003;27:107-9.
- Hamer J, Kirk JA. Physiotherapy and the frozen shoulder: a comparative trial of ice and ultrasonic therapy. N Z Med J 1976;83:191-2.
- Han EJ. The Comparison of the Effectiveness in Auricular Acupuncture Between Acupuncture Points and Tender Points for Frozen Shoulder n.d.:205-8.
- Hando BR. Glenohumeral Joint Translational Manipulation under Anesthesia for the Treatment of Adhesive Capsulitis: A Case Series Study and Findings from Immediate Postmanipulation Arthroscopic Visualization 2010;40.
- Hannafin JA, Strickland SM. Frozen shoulder. Curr Opin Orthop 2000;11:271-5.
- Harryman DT. Shoulders: frozen and stiff. Instr Course Lect 1993;42:247-57.
- Harryman DT, Matsen FA, Sidles JA. Arthroscopic management of refractory shoulder stiffness. Arthroscopy 1997;13:133-47.
- Hart BF. Frozen shoulder treated with acupuncture and nutrition. Am J Acupunct 1976;4:365-7.
- Hauzeur JP. Conservative treatment of the painful shoulder. Review of the literature. Rev Med Brux 2004;25:A411-5.
- Hay EM, Thomas E, Paterson SM, Dziedzic K, Croft PR. A pragmatic randomised controlled trial of local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in primary care. Arthritis Rheum 2001;44.
- Hay EM, Thomas E, Paterson SM, Dziedzic K, Croft PR. A pragmatic randomised controlled trial of local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in primary care. Ann Rheum Dis 2003;62:394-9.
- He J, Chen X-L, Li Z-B, Zhang W, Fu W-Y, Gong J-D. Research of the clinical results of scapulohumeral periarthritis treated with comprehensive therapy. J Shanghai Med Univ 2000;27:141-6.
- Heber A, Eulert J. Conservative treatment of periarthritis humeroscapularis. Therapiewoche 1983;33:580-4.
- Helbig B, Wagner P, Dohler R. Mobilization of frozen shoulder under general anaesthesia. Acta Orthop Belg 1983;49:267-74.
- Heller B, Tarricone R. Oxaprozin versus diclofenac in NSAID-refractory periarthritis pain of the shoulder. Curr Med Res Opin 2004;20:1279-90.
- Hempel V. Suprascapular nerve blockade in treatment of periarthritis humeroscapularis. Therapiewoche 1983;33:584-6.
- Herold HZ. Frozen shoulder. Harefuah 1982;103:26-7.
- Heuleu JN, Courtillon A, Nahon E. Physiotherapy for a painful shoulder. Gaz Med Fr 1979;86:4087-2.
- Hieber F. Periarthritis stiffness of the shoulder and its therapy. Hippokrates 1967;38:888-94.
- Ho C-YC, Sole G, Munn J. The effectiveness of manual therapy in the management of musculoskeletal disorders of the shoulder: a systematic review. Man Therap 2009;14:463-74.
- Hoenle R. Radiotherapy for relief of pain in recalcitrant periarthritis humeroscapularis. Therapiewoche 1983;33:5269-75.
- Hollingworth GR, Ellis RM, Hattersley TS. Comparison of injection techniques for shoulder pain: results of a double blind, randomised study. BMJ 1983;287:1339-41.
- Hollis R, Lahav A, West HS. Manipulation of the shoulder using Codman’s paradox. Orthopedics 2006;29:971-3.
- Hong CZ, Lin JC, Bender LF. Magnetic necklace: its therapeutic effectiveness on neck and shoulder pain application of a static magnetic field as a physical therapy modality. Arch Phys Med Rehabil 1982;63:462-6.
- Hormusjee DN, Mehta SD. Our experience in manipulative therapy for periarthritis of shoulder. Indian J Surg 1980;42:297-300.
- Hossain S, Jacobs LGH, Hashmi R. The long-term effectiveness of steroid injections in primary acromioclavicular joint arthritis: a five-year prospective study. J Shoulder Elbow Surg 2008;17:535-8.
- Hosseini H, Agneskirchner JD, Lobenhoffer P. Arthroscopic capsular release in the management of refractory adhesive capsulitis. Technique and results. Unfallchirurg 2006;109:212-18.
- Hsu SY, Chan KM. Arthroscopic distension in the management of frozen shoulder. Int Orthop 1991;15:79-83.
- Hu XD. 63 cases of periarthritis of shoulder joint treated by scalp-acupuncture and body-needling. Shanghai J Acupunct Moxibustion 1993;12.
- Hu Y. Clinical application of the point Xuanzhong. J Tradit Chin Med 2003;23:275-7.
- Hu J, Yan J-T. Evaluation of clinical reports in treating periarthritis of shoulder by manipulation with the theory of evidence based medicine. Zhong Xi Yi Jie He Xue Bao 2004;2:185-8.
- Hu J. Acupuncture treatment of shoulder pain. J Tradit Chin Med 2006;26:78-9.
- Huang WEI. Analysis on therapeutic effects of 46 cases of periarthritis of shoulder treated by needling jiansanzhen with magnetic pole needle and massage. World J Acup-Moxi 1996;6:31-4.
- Hulstyn MJ, Weiss AP. Adhesive capsulitis of the shoulder. Orthop Rev 1993;22:425-33.
- Hummel-Berry KH. Assessment of Shoulder Function and Functional Impact of Clinic Physical Therapy Versus Home Exercise for Patients With Shoulder Stiffness: A Randomized Controlled Trial 2001.
- Ibrahim T, Rahbi H, Beiri A, Jeyapalan K, Taylor GJS. Adhesive capsulitis of the shoulder: the rate of manipulation following distension arthrogram. Rheumatol Int 2006;27:7-9.
- Ide J, Takagi K. Early and long-term results of arthroscopic treatment for shoulder stiffness. J Shoulder Elbow Surg 2004;13:174-9.
- Imai R, Kusaka Y, Sakakida K. Clinical studies on Voltaren suppository for stiff shoulder. Jpn Pharmacol Therapeut 1983;11:373-86.
- Indeck W. Office management of frozen shoulder syndrome. Clin Orthop Relat Res 1990:309-10.
- Ingram-Rice B. Occupational therapy perspective. J Bodyw Mov Ther 2000;4:20-6.
- Itel R. Exercise therapy and physical therapy measures in the treatment of shoulder and arm pain. Ther Umsch 1988;45:812-19.
- Itzkowitch D, Ginsberg F, Leon M, Bernard V, Appelboom T. Peri-articular injection of tenoxicam for painful shoulders: a double-blind, placebo controlled trial. Clin Rheumatol 1996;15:604-9.
- Ivanov VA, Iakovleva EA. Auricular reflexotherapy of humeroscapular periarthritis. Ortop Travmatol Protez 1986:44-5.
- Jacchia GE, Cosco Mazzuca R. Mobilization under anesthesia in periarthritis of the shoulder. Arch Putti Chir Organi Mov 1968;23:231-41.
- Jacobs LG, Barton MA, Wallace WA, Ferrousis J, Dunn NA, Bossingham DH. Intra-articular distension and steroids in the management of capsulitis of the shoulder. BMJ 1991;302:1498-501.
- Jacobs L, Barton M, Wallace W, Ferrousis J, Dunn NBD. Intra-articular distension and steroids in the management of capsulitis of the shoulder. J Orthop Med 1992;14:40-4.
- Jacobs LGH, Smith MG, Khan SA. Comparision of manipulation under anaesthetic and distension injections in the management of frozen shoulder. J Bone Joint Surg Br 2005;87–B:162-3.
- Jayson MI. Frozen shoulder: adhesive capsulitis. BMJ 1981;283:1005-6.
- Jensen I, Nygren A, Gamberale F, Goldie I, Westerholm P, Jonsson E. The role of the psychologist in multidisciplinary treatments for chronic neck and shoulder pain: a controlled cost-effectiveness study. Scand J Rehabil Med 1995;27:19-26.
- Ji XP. Shoulder pain. J Tradit Chin Med 1988;8:310-12.
- Jia H, Li Q. Treatment of periomarthritis with scalp acupuncture therapy: a report of 210 cases. J Tradit Chin Med 1993;13:199-201.
- Jia C-S, Li X-F, Ma X-S, Shi J, Liu E-J. Comparison of analgesic effects between subcutaneous penetration needling and straight needling of otopoints in the treatment of scapulohumeral periarthritis. Zhen Ci Yan Jiu 2008;33:339-42.
- Jiang YK. Frozen shoulder effectively treated by single needle penetration acupuncture. Am J Acupunct 1982;10.
- Jiang D, Liu W. Thick needle treatment for periomitis. J Tradit Chin Med 1991;11:112-14.
- Jin D, Li Z. Acupuncture and the elevation manipulation of massage for treatment of frozen shoulder. J Tradit Chin Med 2003;23:212-13.
- Johnson AJ, Godges JJ, Zimmerman GJ, Ounanian LL. The effect of anterior versus posterior glide joint mobilization on external rotation range of motion in patients with shoulder adhesive capsulitis. J Orthop Sports Phys Ther 2007;37:88-99.
- Jones DS, Chattopadhyay C. A randomised trial to compare suprascapular nerve block with intra-articular injections for the treatment of frozen shoulder in primary care. Br J Rheumatol 1997;36.
- Jones DS, Chattopadhyay C. Suprascapular nerve block for the treatment of frozen shoulder in primary care: a randomized trial. Br J Gen Pract 1999;49:39-41.
- Joshi GP, McCarroll SM. Intra-articular morphine for the management of frozen shoulder. Anaesthesia 1992;47.
- Judet H, Judet J. Surgery in the treatment of refractory forms of stiff and painful shoulder. Rev Chir Orthop Reparatrice Appar Mot 1985;71:75-7.
- Jurgel J, Rannama L, Gapeyeva H, Ereline J, Kolts I, Paasuke M. Shoulder function in patients with frozen shoulder before and after 4-week rehabilitation. Medicina (Kaunas) 2005;41:30-8.
- Kalke SJ, Perera SD, Dasgupta B. Ultrasound in the evaluation of shoulder pain. Arthritis Rheum 1999;42.
- Kanai S, Taniguchi N, Kawamoto M, Endo H, Higashino H. Effect of static magnetic field on pain associated with frozen shoulder. Pain Clinic 2004;16:173-9.
- Kanai S, Taniguchi N. Effect of polarity exchangeable permanent magnet on frozen shoulder pain. Pain Clinic 2006;18:37-45.
- Kaptelin AF, Larina LE. Complex conservative treatment of brachio-scapular periarthritis. Ortop Travmatol Protez 1976:60-4.
- Karatas GK, Meray J. Suprascapular nerve block for pain relief in adhesive capsulitis: comparison of 2 different techniques. Arch Phys Med Rehabil 2002;83:593-7.
- Karkan Z, Mikes K, Vurm F, Maresova B. Our experiences with the therapy of the stiffening shoulder. Rehabilitacia 1985;18:31-8.
- Katz JN, Solomon DH, Schaffer JL, Horsky J, Burdick E, Bates DW. Outcomes of care and resource utilization among patients with knee or shoulder disorders treated by general internists, rheumatologists, or orthopedic surgeons. Am J Med 2000;108:28-35.
- Kay NR. The clinical diagnosis and management of frozen shoulders. Practitioner 1981;225:164-7.
- Kay NRM, Eyres KS. The role of manipulation for the frozen shoulder. J Orthop Rheumatol 1990;3:203-12.
- Keilholz L, Seegenschmiedt MH, Kutzki D, Sauer R. Periarthritis humeroscapularis (PHS). Indications, technique and outcome of radiotherapy. Strahlenther Onkol 1995;171:379-84.
- Kent H. Physical medicine and rehabilitation of the painful shoulder. J Okla State Med Assoc 1961;54:433-7.
- Kent JM. Help for the stiff, sore shoulder. Patient Care 1985;19.
- Keyl W. Mobilization of the stiff shoulder under anesthesia – indication, technique and results. Z Orthop Ihre Grenzgeb 1982;120:574-5.
- Khan AA, Mowla A, Shakoor MA, Rahman MR. Arthrographic distension of the shoulder joint in the management of frozen shoulder. Mymensingh Med J 2005;14:67-70.
- Khan JA, Devkota P, Acharya BM, Pradhan NMS, Shreshtha SK, Singh M, et al. Manipulation under local anesthesia in idiopathic frozen shoulder – a new effective and simple technique. Nepal Med Coll J 2009;11:247-53.
- Khitrov NA. Periarthritis of the shoulder joint: variants of the course and treatment with artrofoon. Ter Arkh 2007;79:40-6.
- Kirillova RA. Diagnosis and treatment of humeroscapular periarthritis. Vrach Delo 1986:15-7.
- Kivimaki J, Seitsalo S. Treatment of adhesive capsulitis (frozen shoulder) with manipulation under anaesthesia. Eur J Phys Rehabil Med 1996;6:59-61.
- Kivimaki J, Pohjolainen T. Manipulation under anesthesia for frozen shoulder with and without steroid injection. Arch Phys Med Rehabil 2001;82:1188-90.
- Klinger H-M, Otte S, Baums MH, Haerer T. Early arthroscopic release in refractory shoulder stiffness. Arch Orthop Trauma Surg 2002;122:200-3.
- Knebl JA, Shores JH, Gamber RG, Gray WT, Herron KM. Improving functional ability in the elderly via the Spencer technique, an osteopathic manipulative treatment: a randomized, controlled trial. J Am Osteopath Assoc 2002;102:387-96.
- Kneer W, Eulert J. Operative treatment of periarthritis humeroscapularis. Therapiewoche 1983;33:588-94.
- Kneer W, Kuhnau S, Bias P, Haag RF. Dimethylsulfoxide (DMSO) gel for the treatment of acute tendopathy. A multicentric, placebo-controlled randomized study. Fortschr Med 1994;112:142-6.
- Knusel O. Die transkutane elektrische nervenstimulation beim weichteilrheumatismus: eine kontrollierte untersucherblinde studie an 60 patienten mit levator-scapulae-syndrom. Z Phys Med Bal Med Klim 1984;13:337-9.
- Koel G. Comment on: A prospective double-blind placebo-controlled randomized trial of ultrasound in the physiotherapy treatment of shoulder pain. Rheumatology 2008;47:229-30.
- Kong JC, Lee MS, Shin BC. Randomized clinical trials on acupuncture in Korean literature: a systematic review. Evid Based Complement Alternat Med 2009;6:41-8.
- Kostadinov D, Michailov S, Dimitrov M. Treatment of patients with periarthritis of the shoulder joint using the stimulator ‘Arfai’. Kurortol Fizioter 1980;17:16-20.
- Koubaa S, Ben Salah FZ, Lebib S, Miri I, Dziri C, Ben Hammouda M, et al. Capsular distension and physical therapy in treatment of adhesive capsulitis. Tunis Med 2006;84:621-5.
- Kovacs E. Comparative examinations with pelsonin and indometacin by double blind method in patients suffering from locomotor diseases. Ther Hung 1982;30:180-3.
- Kucukdeveci AA, Sahin M, Tuncer S, Gok H. Effectiveness of electrical stimulation in shoulder pain due to intrinsic mechanical shoulder problems. Ann Rheum Dis 2005;64.
- Kuijpers T, van Tulder MW, van der Heijden GJ, Bouter LM, van der Windt DA. Costs of shoulder pain in primary care consulters: a prospective cohort study in the Netherlands. BMC Musculoskelet Disord 2006;7.
- Kulenkampff HA, Reichelt A. Studies on conservative and operative treatment of the supraspinatus syndrome. Aktuel Rheumatol 1989;14:70-6.
- Kuptniratsaikul S, Kuptniratsaikul V, Tejapongvorachai T, Itiravivong P. A capsular dilatation facilitated shoulder manipulation for treating patients with frozen shoulder. J Med Assoc Thai 2002;85:163-9.
- Kurtais Gursel Y, Ulus Y, Bilgic A, Dincer G, van der Heijden GJMG. Adding ultrasound in the management of soft tissue disorders of the shoulder: a randomized placebo-controlled trial. Phys Ther 2004;84:336-43.
- Laidley S. Shiatsu massage for frozen shoulder. Positive Health 2004;101:52-4.
- Lanfranchi R, Volpi A. Results of the clinical trial of intra-articular thiocolchicoside in scapulo-humeral periarthritis. Minerva Med 1968;59:4678-81.
- Laroche M, Ighilahriz O, Moulinier L, Constantin A, Cantagrel A, Mazieres B. Adhesive capsulitis of the shoulder: an open study of 40 cases treated by joint distention during arthrography followed by an intraarticular corticosteroid injection and immediate physical therapy. Rev Rhum Engl Ed 1998;65:313-19.
- Laskowski I, Schuckmann W. Contribution to operative treatment of periarthritis humeroscapularis. Beitr Orthop Traumatol 1985;32:337-41.
- LaStayo P, Jaffe R. Assessment and management of shoulder stiffness: a biomechanical approach. J Hand Ther 1994;7:122-30.
- Latham BA. Injections and physiotherapy for the painful stiff shoulder. Ann Rheum Dis 1989;48.
- Laznicky J. Experiences with spa treatment in humeroscapular periarthropathy in spa Teplice. Rehabilitacia 1989;22:207-13.
- Lech O, Subbrack G, Valenzuela Neto C. Capsulite adesiva (‘ombro congelado’): abordagem multidisciplinar. Rev Bras Ortop 1993;28:617-24.
- Leclaire R, Bourgouin J. Electromagnetic treatment of shoulder periarthritis: a randomized controlled trial of the efficiency and tolerance of magnetotherapy. Arch Phys Med Rehabil 1991;72:284-7.
- Lee M, Haq AM, Wright V, Longton EB. Periarthritis of the shoulder: a controlled trial of physiotherapy. Physiotherapy 1973;59:312-15.
- Lee PN, Lee M, Haq AM, Longton EB, Wright V. Periarthritis of the shoulder: trial of treatments investigated by multivariate analysis. Ann Rheum Dis 1974;33:116-19.
- Lee WK, Maguire MG. Injection of the shoulder. Med J Aust 1986;144.
- Lee H, Hong K, Kim Y, Yim Y, Ahn T, Kang W. A clinical trial of acupuncture treatment for frozen shoulder. J Korean Acupunct Mox Soc 2006;23:165-77.
- Lee H-J, Lim K-B, Kim D-Y, Lee K-T. Randomized controlled trial for efficacy of intra-articular injection for adhesive capsulitis: ultrasonography-guided versus blind technique. Arch Phys Med Rehabil 2009;90:1997-2002.
- Lee MS, Choi TY, Kim JI, Choi SM. Using Guasha to treat musculoskeletal pain: a systematic review of controlled clinical trials. Chin Med 2010;5. www.ncbi.nlm.nih.gov/pmc/articles/PMC2827462/ (accessed 6 January 2010).
- Lehmann JF, Erickson DJ, Martin GM, Krusen FH. Comparison of ultrasonic and microwave diathermy in the physical treatment of periarthritis of the shoulder; study of the effects of ultrasonic and microwave diathermy when employed in conjunction with massage and exercise. Arch Phys Med Rehabil 1954;35:627-34.
- Levenets VN, Iurchenko AP, Gertsen GI. Humeroscapular periarthrosis and its treatment. Ortop Travmatol Protez 1982:24-7.
- Li ZM. 235 cases of frozen shoulder treated by manipulation and massage. J Tradit Chin Med 1984;4:213-15.
- Li H-M. Treatment of 84 cases of shoulder periarthritis by tuina plus acupuncture. J Acu Tuina Sci 2003;1:47-9.
- Liang HC, Lien IN. Comparative study in the management of frozen shoulder. Taiwan Yi Xue Hui Za Zhi 1973;72:243-7.
- Liao B-N, Zhang S, Zhang L-M. Clinical observation on ‘Jianying point’-injection combined with massage for treatment of periarthritis of shoulder. Zhongguo Zhenjiu 2007;27:261-3.
- Liaw SC. The effect and timing of physiotherapy on change in range of motion and function in frozen shoulder. Physiother Singapore 2000;3:82-6.
- Lidstrom A. The ‘frozen’ shoulder. Pathologic anatomy and treatment with mobilization. Nord Med 1963;69:125-9.
- Liebolt FL. Frozen shoulder. Passive exercises for treatment. N Y State J Med 1970;70.
- Liem D, Meier F, Thorwesten L, Marquardt B, Steinbeck J, Poetzl W. The influence of arthroscopic subscapularis tendon and capsule release on internal rotation strength in treatment of frozen shoulder. Am J Sports Med 2008;36:921-6.
- Liu X. Periarthritis of shoulder therapy by combination of drug hammer and exercise in 83 cases. Chin J Clin Rehab 2003;7.
- Liu HP, Wang C, Xu SF. Comparison of different treatments for relieving pain and improving joint movement disorder in patients with frozen shoulder. Chin J Clin Rehab 2004;8:3946-7.
- Lin JG, Chen CT, Lu TW, Lin YS, Chen HL, Chen YS. Quantitative evaluation of the motion of frozen shoulders treated with acupuncture by puncturing from Tiaokou (St. 38) towards Chengshan (U.B. 57). Biomed Eng 2005;17:31-7.
- Lin H-C, Li J-S, Lo S-F, Shih Y-F, Lo C-Y, Chen S-Y. Isokinetic characteristics of shoulder rotators in patients with adhesive capsulitis. J Rehabil Med 2009;41:563-8.
- Livinya BY. Osteoreflexotherapy of humerocapsular periarthritis. Latvijas Psr Zinatnu Akademijas Vestis 1989:121-3.
- Long J, Peer G, Cwik J. The triscapular block. A new application of therapy of periscapular pain and glenohumeral restriction. Reg Anesth 1987;12:70-5.
- Lorbach O, Anagnostakos K, Scherf C, Seil R, Kohn D, Pape D. Nonoperative management of adhesive capsulitis of the shoulder: oral cortisone application versus intra-articular cortisone injections. J Shoulder Elbow Surg 2010;19:172-9.
- Loyd JA, Loyd HM. Adhesive capsulitis of the shoulder: arthrographic diagnosis and treatment. South Med J 1983;76:879-83.
- Lu S. Scalp acupuncture therapy and its clinical application. J Tradit Chin Med 1991;11:272-80.
- Lu J, Sun J-H, Fu Z-H, Yuan J-H, Li J, Ji A-Q. Transient therapeutic effect and safety of superficial needling therapy for treatment of periarthritis of shoulder. Zhongguo Zhenjiu 2008;28:414-16.
- Ludwig J, Hedtmann DA. Orthopedic treatment of shoulder pain. Z Orthop Ihre Grenzgeb 1998;136:OA10-OA13.
- Lundberg BJ. Arthrography and manipulation in rigidity of shoulder joint. Acta Orthop Scand 1965;36:35-44.
- Luo F, Shen Y, Liu YQ, Wang EZ, Wang BG. Intraarticular injection of sodium hyaluronate plus nerve and pain block with betamethasone in treatment of periarthritis of shoulder. Chin J Clin Rehab 2005;9:266-7.
- Luziatelli S, Colagrosso B, Alaimo A. Clinical data on intraarticular administration of orgotein; comparison with a corticosteroid. G Med Mil 1984;134:490-3.
- Ma SH, Sun Y, Yan XM. Effect of suprascapular nerve block combined with triole nerve block for scapulohumeral periarthritis. Chin J Clin Rehab 2004;8:4698-9.
- Maiotti M, Massoni C, Basile A, Cedrone P. Role of arthroscopic arthrolysis in stiff shoulder. Eur J Sports Traumat Rel Res 2001;23:94-9.
- Maitland GD. Treatment of the glenohumeral joint by passive movement. Physiotherapy 1983;69:3-7.
- Mao YR, Huang DF, Ding JX, Xu GQ, Xu YL, Zhao M. Analysis of extracorporeal shock wave therapy in immediate treatment of musculoskeletal disorders. Chin J Clin Rehab 2003;7:3216-17.
- Marcus A. A combined treatment approach to shoulder pain using orthopaedic medicine and acupuncture technique. J Orthop Med 1994;16:75-9.
- Mardjuadi A, Dequeker J. Double-blind trial comparing bufexamac infiltrations with triamcinolone acetonide infiltrations in patients with periarthritis of the shoulder. Curr Med Res Opin 1978;5:401-5.
- Marx RG, Malizia RW, Kenter K, Wickiewicz TL, Hannafin JA. Intra-articular corticosteroid injection for the treatment of idiopathic adhesive capsulitis of the shoulder. HSS J 2007;3:202-7.
- Massoud SN, Pearse EO, Levy O, Copeland SA. Operative management of the frozen shoulder in patients with diabetes. J Shoulder Elbow Surg 2002;11:609-13.
- Mattara L, Trotta F, Biasi D, Cervetti R. Evaluation of the efficacy and tolerability of a new locally acting preparation of flurbiprofen in scapulohumeral periarthritis. Eur J Rheumatol Inflamm 1994;14:15-20.
- Mavrikakis ME, Sfikakis PP, Kontoyannis SA, Antoniades LG, Kontoyannis DA, Moulopoulou DS. Clinical and laboratory parameters in adult diabetics with and without calcific shoulder periarthritis. Calcif Tissue Int 1991;49:288-91.
- Mayerhofer H. Enhancement of effect of a high dosage salicylate ointment by iontophoresis. Z Allgemeinmed 1981;57:634-6.
- McClatchie L, Laprade J, Martin S, Jaglal SB, Richardson D, Agur A. Mobilizations of the asymptomatic cervical spine can reduce signs of shoulder dysfunction in adults. Man Therap 2009;14:369-74.
- McHardy A, Hoskins W, Pollard H, Onley R, Windsham R. Chiropractic treatment of upper extremity conditions: a systematic review. J Manipulative Physiol Ther 2008;31:146-59.
- McKeever DC. Thawing the frozen shoulder. Clin Orthop 1958;11:168-76.
- McQuay HJ, Moore RA, Eccleston C, Morley S, Williams AC. Systematic review of outpatient services for chronic pain control. Health Technol Assess 1997;1.
- Meijer EM, Sluiter JK, Heyma A, Sadiraj K, Frings-Dresen MH. Cost-effectiveness of multidisciplinary treatment in sick-listed patients with upper extremity musculoskeletal disorders: a randomized, controlled trial with one-year follow-up. Int Arch Occup Environ Health 2006;79:654-64.
- Melzer C, Wallny T, Wirth CJ, Hoffmann S. Frozen shoulder – treatment and results. Arch Orthop Trauma Surg 1995;114:87-91.
- Mencke M, Wieden TE, Hoppe M, Porschke W, Hoffmann O, Herget HF. Acupuncture of shoulder pain and low back pain. Two prospective double-blind studies. Akupunktur 1988;16:204-15.
- Menkes CJ. [Scapulo-humeral periarthritis: efficacy, safety and therapeutic benefit of etodolac (600 mg daily) versus piroxicam (40/20 mg daily)]. Rhumatologie 1990;42:195-200.
- Mert M, Atakanli LK, Temizel M. The comparison of treatment response of diabetic and non-diabetic adhesive capsulitis to physical therapy. Diabetes 2009;58.
- Meyer MP, Ter Waarbeek B. Humeroscapular periarthritis and exercise therapy. Ned Tijdschr Geneeskd 1952;96:2297-301.
- Miccoli G, Angiolini G, Vannini A. Association of a benzofurane derivative and kinesitherapy in the treatment of periarthritis of the shoulder. Clin Ter 1964;31:310-23.
- Michlovitz SL, Ann Harris B, Watkins MP. Therapy interventions for improving joint range of motion: a systematic review. J Hand Ther 2004;17:118-31.
- Miller MD, Rockwood CA, Wirth MA. Thawing the frozen shoulder – the ‘patient’ patient. South Med J 1991;84.
- Miller JS, Stanley I, Moore K. Videotaped exercise instruction: a randomised controlled trial in musculoskeletal physiotherapy. Physiother Theory Pract 2004;20:145-54.
- Miller JS, Litva A, Gabbay M. Motivating patients with shoulder and back pain to self-care: can a videotape of exercise support physiotherapy?. Physiotherapy 2009;95:29-35.
- Mior S. Manipulation and mobilization in the treatment of chronic pain. Clin J Pain 2001;17:70-6.
- Miszczyk L, Wozniak G, Walichiewicz P, Spindel J. The effectiveness of radiotherapy for painful humeroscapular periarthritis (PHS). Nowotwory 2005;55:49-53.
- Mizuno S. Frozen shoulder. Nippon Rinsho 1976;34:3552-4.
- Mohindra Y, Jain S, Sain A. R. compound (Alarsin) in periarthritis shoulder. Clinical trial. Med Surg 1987;27:9-10.
- Molsberger AF, Schneider T, Gotthardt H, Drabik A. German Randomized Acupuncture Trial for chronic shoulder pain (GRASP). A pragmatic, controlled, patient-blinded, multi-centre trial in an outpatient care environment. Pain 2010;151:146-54.
- Monreal González RJ, Díaz Ramos H, León Santana P, Paredtes González LF. Capsulitis adhesiva del hombro: tratamiento con distensión hidráulica y anestesia local. Rev Cuba Ortop Traumatol 2006;20. http://scielo.sld.cu/pdf/art/v20n2/ort0526.pdf (accessed 26 March 2010).
- Montemagni P, Carnazza G. Diagnosis and arthroscopic treatment of frozen shoulder. Minerva Ortop Traumatol 1989;40:85-90.
- Moore ME, Berk SN. Acupuncture for chronic shoulder pain. An experimental study with attention to the role of placebo and hypnotic susceptibility. Ann Intern Med 1976;84:381-4.
- Morgan B, Jones AR, Mulcahy KA, Finlay DB, Collett B. Transcutaneous electric nerve stimulation (TENS) during distension shoulder arthrography: a controlled trial. Pain 1995;64:265-7.
- Morris CR, Morris AJ. Intravenous pulse corticosteroids for the treatment of frozen shoulder. Arthritis Rheum 1994;37.
- Moutounet J, Chevrot A, Godefroy D. Radioguided puncture-filtration treatment of intractable calcifying periarthritis of the shoulder. J Radiol 1984;65:569-72.
- Mueller EE, Mead S, Schulz BF, Vaden MR. A placebo-controlled study of ultrasound treatment for periarthritis. Am J Phys Med 1954;33:31-5.
- Mund-Hoym WD. Degree of efficacy of salicylate therapy of periarthropathia humeroscapularis. Med Welt 1987;38:1500-2.
- Munting E. Ultrasonic therapy for painful shoulders. Physiotherapy 1978;64:180-1.
- Murnaghan GF, McIntosh D. Hydrocortisone in painful shoulder. A controlled trial. Lancet 1955:798-800.
- Musil D, Sadovsky P, Stehlik J, Filip L, Vodicka Z. Arthroscopic capsular release in frozen shoulder syndrome. Acta Chir Orthop Traumatol Cech 2009;76:98-103.
- Nabeta T. Effect of a tender point acupuncture therapy for shoulder stiffness: a randomized controlled trial. J Jpn Acupunct Moxibustion Soc 2000;50.
- Nabeta T, Kawakita K. Relief of chronic neck and shoulder pain by manual acupuncture to tender points: a sham-controlled randomized trial. Complement Ther Med 2002;10:217-22.
- Naglic N, Klopcic-Spevak M. The effect of TENS during kinesiotherapy on chronic shoulder pain. Int J Rehabil Res 2009;32:10-1.
- Naredo E, Cabero F, Beneyto P, Cruz A, Mondejar B, Uson J, et al. A randomized comparative study of short term response to blind injection versus sonographic-guided injection of local corticosteroids in patients with painful shoulder. J Rheumatol 2004;31:308-14.
- Narouze SN, Govil H, Guirguis M, Mekhail NA. Continuous cervical epidural analgesia for rehabilitation after shoulder surgery: a retrospective evaluation. Pain Physician 2009;12:189-94.
- Nash P, Hazleman BL. Frozen shoulder. Baillieres Clin Rheumatol 1989;3:551-66.
- Neviaser TJ, Mcginty JBE. Operative arthroscopy. New York, NY: Raven Press; 1991.
- Ng CY, Amin AK, Narborough S, McMullan L, Cook R, Brenkel IJ. Manipulation under anaesthesia and early physiotherapy facilitate recovery of patients with frozen shoulder syndrome. Scott Med J 2009;54:29-31.
- Nicholson GG. The effects of passive joint mobilization on pain and hypomobility associated with adhesive capsulitis of the shoulder. J Orthop Sports Phys Ther 1985;6:238-46.
- Nicholson GP. Arthroscopic capsular release for stiff shoulders: effect of etiology on outcomes. Arthroscopy 2003;19:40-9.
- Nicolova L. Efficiency of interferential current in scapulo-humeral periarthritis calcarea. J Radiol Electrol Arch Electr Med 1966;47.
- Nikolova L. Low-frequency treatment with interference current of patients with periarthritis humeroscapularis. MMW Munch Med Wochenschr 1970;112:472-6.
- Nobuhara K, Sugiyama D, Ikeda H, Makiura M. Contracture of the shoulder. Clin Orthop Relat Res 1990:105-10.
- Noel E. Treatment of calcific tendinitis and adhesive capsulitis of the shoulder. Rev Rhum Engl Ed 1997;64:619-28.
- Noskov SM, Fetelego OI, Krasivina IG, Dolgova LN. Alflutop in local therapy of shoulder periarthritis. Ter Arkh 2005;77:57-60.
- Nouijai A, Mahi M, El J, Ghazi M, Mounach A, Achemlal L, et al. Arthrographic joint distension with saline and steroid followed by continuous passive motion improves function in patients with frozen shoulder: an open study. Ann Rheum Dis 2006;65.
- Nykanen M. Pulsed ultrasound treatment of the painful shoulder a randomized, double-blind, placebo-controlled study. Scand J Rehabil Med 1995;27:105-8.
- Ogilvie-Harris DJ, Biggs DJ, Fitsialos DP, MacKay M. The resistant frozen shoulder. Manipulation versus arthroscopic release. Clin Orthop Relat Res 1995:238-48.
- Ogilvie-Harris DJ, Myerthall S. The diabetic frozen shoulder: arthroscopic release. Arthroscopy 1997;13:1-8.
- Ohshima Y. Intra-articular injection therapy of high molecular weight sodium hyaluronate (SPH) on periarthritis of the shoulder. Jpn Pharm Ther 1987;15:341-56.
- Ohta K, Yano T. Comparison study between superficial and deep acupuncture on the neck and shoulder stiffness – as index of muscle hardness, surface EMG and deep hemodynamics. J Jpn Assoc Phys Med Balneol Climatol 2005;68:122-33.
- Okamura K. Arthroscopic shoulder surgery. Hokkaido J Med Sci 1995;70:375-81.
- Olejarova M. Local nonsteroidal antiinflamatory drugs from the point of view of evidence-based medicine. Bolest 2004;7:93-9.
- Omari A, Bunker TD. Open surgical release for frozen shoulder: surgical findings and results of the release. J Shoulder Elbow Surg 2001;10:353-7.
- Orr B. Treating shoulder complaints in general practice. Study’s results must be viewed in context of Dutch, not British, physiotherapy practice. BMJ 1997;315.
- Ortolani M, Tarantello G, Cognolatto F, Faggin A. Manipulation by McMennel’s technique in treatment of noncalcified periarthritis of the shoulder. Riabilitazione 1985;18:155-65.
- Othman A, Taylor G. Manipulation under anaesthesia for frozen shoulder. Int Orthop 2002;26:268-70.
- Ou HC. An observation of four physiotherapies for 249 cases with shoulder periarthritis. Chin J Phys Therap 1989;12:32-3.
- Ozaki J, Nakagawa Y, Sakurai G, Tamai S. Recalcitrant chronic adhesive capsulitis of the shoulder. Role of contracture of the coracohumeral ligament and rotator interval in pathogenesis and treatment. J Bone Joint Surg Am 1989;71:1511-15.
- Ozaki J. Frozen shoulder – operative management and pathogenesis. Surg Shoulder 1995;1085:275-8.
- Ozaki J. Pathomechanics and operative management of chronic frozen shoulder. Ann Chir Gynaecol 1996;85:156-8.
- Pages IH, Herrmann H, Retter D. Magnetic field therapy with Poljus 1-first experimental and clinical results. Z Physiother 1982;34:205-7.
- Pap G, Liebau C, Meyer M, Merk H. Results of mobilization under anesthesia in adhesive capsulitis in relation to stage of the disease. Z Orthop Ihre Grenzgeb 1998;136:13-7.
- Parker RD, Froimson AI, Winsberg DD, Arsham NZ. Frozen shoulder. Part II: treatment by manipulation under anesthesia. Orthopedics 1989;12:989-90.
- Parker RD, Froimson AI, Winsberg DD, Arsham NZ. Frozen shoulder. Part I: chronology, pathogenesis, clinical picture, and treatment. Orthopedics 1989;12:869-73.
- Parsons JL, Shepard WL, Fosdick WM. DMSO an adjutant to physical therapy in the chronic frozen shoulder. Ann N Y Acad Sci 1967;141:569-71.
- Patel M, Gutzwiller F, Paccaud F, Marazzi A. A meta-analysis of acupuncture for chronic pain. Int J Epidemiol 1989;18:900-6.
- Patte D, Goutallier D. [‘Reparative’ and ‘delivering’ conceptions in treatment of the periarthritis of the shoulder. Acta Orthop Belg 1983;49:299-30.
- Pearsall AW, Osbahr DC, Speer KP. An arthroscopic technique for treating patients with frozen shoulder. Arthroscopy 1999;15:2-11.
- Peng AT, Behar S, Yue SJ. Long-term therapeutic effects of electro-acupuncture for chronic neck and shoulder pain: a double blind study. Acupunct Electrother Res 1987;12:37-44.
- Peng J-M, Lu H, Hu X-B. Observation on therapeutic effect of Shaoshanhuo needling method on periarthritis of shoulder. Zhongguo Zhenjiu 2006;26:581-2.
- Petri M, Dobrow R, Neiman R, Whiting-O’Keefe Q, Seaman WE. Randomized, double-blind, placebo-controlled study of the treatment of the painful shoulder. Arthritis Rheum 1987;30:1040-5.
- Petukhov IA. Poly clinical diagnosis and treatment of shoulder scapular periarthritis. Zdravookhr Beloruss 1979:53-5.
- Philadelphia Panel . Philadelphia Panel evidence-based clinical practice guidelines on selected rehabilitation interventions for shoulder pain. Phys Ther 2001;81:1719-30.
- Piotte F, Gravel D, Moffet H, Fliszar E, Roy A, Nadeau S, et al. Effects of repeated distension arthrographies combined with a home exercise program among adults with idiopathic adhesive capsulitis of the shoulder. Am J Phys Med Rehabil 2004;83:537-46.
- Pittler MH. Static magnets for reducing pain. Focus Altern Complement Ther 2008;13:5-10.
- Placzek JD, Roubal PJ, Freeman DC, Kulig K, Nasser S, Pagett BT. Long-term effectiveness of translational manipulation for adhesive capsulitis. Clin Orthop Relat Res 1998:181-91.
- Polimeni V, Panuccio A, Furfari P, Crupi D, Barreca G, Forgione C, et al. Preliminary study on the efficacy of various rehabilitation therapies for shoulder pain. Eura Medicophys 2003;39:59-63.
- Politano B, Jucopill AN, Gherardi GM, Loda M. Review of 11 cases of surgically treated Duplay’s disease. Minerva Ortop 1982;33:1025-8.
- Pollock RG, Duralde XA, Flatow EL, Bigliani LU. The use of arthroscopy in the treatment of resistant frozen shoulder. Clin Orthop Relat Res 1994:30-6.
- Singh Poonam, Singh PD. Manipulation for periarthritis of the shoulder in tribal population of Chotanagpur-Ranchi. Indian Med J 1990;84:247-8.
- Qiu Q-Z. Treatment of severe scapulohumeral periarthritis with combined use of suprascapular nerve blockade and manipulation therapy: a report of 49 cases. Zhong Xi Yi Jie He Xue Bao 2006;4:82-3.
- Quigley TB. Checkrein shoulder – a type of frozen shoulder – diagnosis and treatment by manipulation and ACTH or cortisone. N Engl J Med 1954;250:188-92.
- Quigley TB. Checkrein shoulder. A type of ‘frozen’ shoulder diagnosis and treatment by manipulation and ACTH or cortisone. Clin Orthop 1982;164:4-9.
- Quin CE. ‘Frozen shoulder’. Evaluation of treatment with hydrocortisone injections and exercises. Ann Phys Med 1965;10:22-5.
- Quraishi NA, Johnston P, Bayer J, Crowe M, Chakrabarti A. Thawing the frozen shoulder: a randomised controlled trial comparing manipulation under anaesthesia with hydrodilatation. J Bone Joint Surg Br 2006;89B:1197-200.
- Quraishi NA, Johnston P, Bayer J, Crowe M, Chakrabarti A. Thawing the frozen shoulder – a randomised controlled trial of manipulation under anaesthetic versus hydrodilatation. J Bone Joint Surg Br 2008;90B.
- Radaelli E, Veneziani GA, Rossi A. Manual medicine in treatment of adhesive capsulitis. Riabilitazione 1984;17:3-12.
- Rainbow DM, Weston JP, Brantingham JW, Globe G, Lee F. A prospective clinical trial comparing chiropractic manipulation and exercise therapy vs chiropractic mobilization and exercise therapy for treatment of patients suffering from adhesive capsulitis/frozen shoulder. J Am Chiropr Assoc 2008;45:12-28.
- Ramsey ML, Getz CL, Parsons BO. What’s new in shoulder and elbow surgery. J Bone Joint Surg Am 2009;91:1283-93.
- Refior HJ, Melzer C. Shoulder stiffness and mobilization under anesthesia. Hefte Unfallheilkd 1984;170:145-9.
- Reichmister JP, Friedman SL. Long-term functional results after manipulation of the frozen shoulder. Md Med J 1999;48:7-11.
- Ren YM, Shang JB, Wu XM, Chen YF, He XF. Treatment of periarthritis humeroscapularis with medical ozone injection. Chin J Interven Imag Therap 2006;3:341-3.
- Rendeiro DG, Majkowski GR, Lee IE, Gill NW III, Jensen DA, Deyle GS, et al. The effectiveness of translational manipulation under interscalene block for the treatment of adhesive capsulitis of the shoulder: a prospective clinical trial. J Man Manip Ther 2006;14.
- Revel M, Ghanem N. Adhesive capsulitis of the shoulder. Rev Prat 1999;49:1406-8.
- Rey B, Gerber NJ, Schlapbach P, Gerber NJ. Rheumatology, Vol. 14. Physiotherapy: controlled trials and facts. Basel, Switzerland: S. Karger Ag; 1991.
- Rhind V, Downie WW, Bird HA. Naproxen and indomethacin in periarthritis of the shoulder. Rheumatol Rehabil 1982;21:51-3.
- Rich FR, Waligora M. Chronic shoulder pain: evaluation and management. Physician Assist 1985;9:66-7.
- Richardson AT. Ernest Fletcher Lecture. The painful shoulder. Proc R Soc Med 1975;68:731-6.
- Rigato M, Battisti E, Fortunato M, Giordano N. Comparison between the analgesic and therapeutic effects of a musically modulated electromagnetic field (TAMMEF) and those of a 100 Hz electromagnetic field: blind experiment on patients suffering from cervical spondylosis or shoulder periarthritis. J Med Eng Technol 2002;26:253-8.
- Ritchie LD. A clinical evaluation of flurbiprofen LAT and piroxicam gel: a multicentre study in general practice. Clin Rheumatol 1996;15:243-7.
- Ritzmann P. [‘Frozen shoulder’: intraarticular corticosteroids lead to faster pain relief than physiotherapy. Praxis 1999;88:1369-70.
- Rizk TE, Christopher RP, Higgins AC. Adhesive capsulitis – a new therapeutic approach. Arch Phys Med Rehabil 1982;63.
- Rizk TE, Christopher RP, Pinals RS, Higgins AC, Frix R. Adhesive capsulitis (frozen shoulder): a new approach to its management. Arch Phys Med Rehabil 1983;64:29-33.
- Rizk TE, Gavant ML, Pinals RS. Treatment of adhesive capsulitis (frozen shoulder) with arthrographic capsular distension and rupture. Arch Phys Med Rehabil 1994;75:803-7.
- Roberts HJ. Local Injection of a preparation containing 2 betamethasone esters in the treatment of noninfectious musculoskeletal disorders. J Am Geriatr Soc 1965;13:275-91.
- Romoli M, van der Windt D, Giovanzana P, Masserano G, Vignali F, Quirico E, et al. International research project to devise a protocol to test the effectiveness of acupuncture on painful shoulder. J Altern Complement Med 2000;6:281-7.
- Rompe G. Remedial gymnastic treatment in degenerative affections of the shoulder. Krankengymnastik 1981;33:727-35.
- Roques CF, Railhac JJ, Arbus L. Arthrography and electromyography of shoulder periarthriti. Rhumatologie 1984;36:5-12.
- Roubal PJ, Dobritt D, Placzek JD. Glenohumeral gliding manipulation following interscalene brachial plexus block in patients with adhesive capsulitis. J Orthop Sports Phys Ther 1996;24:66-77.
- Rowlingson JC, Arasi R. The use of suprascapular nerve blocks in the management of shoulder pain. Reg Anesth 1986;11:156-9.
- Saadat Niaki A, Siaie M, Sadeghi H, Khatibi H. The use of steroids for the management of chronic shoulder pain by interventional techniques. Arch Iran Med 2005;8:127-30.
- Saadeh CK, Ramos LG, Malacara JM, Mitchell A, Shelton D, Christian N, et al. Ultrasound guided joint injection: comparison with outcomes in blinded injections. Ann Rheum Dis 2005;64.
- Saeidian SR, Hemmati AA, Haghighi MH. Pain relieving effect of short-course, pulse prednisolone in managing frozen shoulder. J Pain Palliat Care Pharmacother 2007;21:27-30.
- Saggini R, Zoppi M, Vecchiet F, Gatteschi L, Obletter G, Giamberardino MA. Comparison of electromotive drug administration with ketorolac or with placebo in patients with pain from rheumatic disease: a double-masked study. Clin Ther 1996;18:1169-74.
- Sakeni RA, Al-Nimer MSM. Comparison between intraarticular triamcinolone acetonide and methylprednisolone acetate injections in treatment of frozen shoulder. Saudi Med J 2007;28:707-12.
- Sanders WE, Hooper WE. Recalcitrant chronic adhesive capsulitis of the shoulder. J Bone Joint Surg Am 1990;72:1269-70.
- Sandor R. Adhesive capsulitis: optimal treatment of ‘frozen shoulder’. Phys Sportsmed 2000;28:23-5.
- Sandor R, Brone S. Exercising the frozen shoulder. Phys Sportsmed 2000;28:83-4.
- Santalena G, Favalli E, Ligniere GC. Local infiltrative therapy of the painful shoulder. Riv It Biol Med 1998;18:84-7.
- Sauvain MJ. Periarthritis of the shoulder. Ther Umsch 1985;42:695-8.
- Scendoni P. Therapy of periarthropathies. Clin Ter 1987;122:357-66.
- Scheef SE. The treatment of humero-scapular periarthritis. ZFA (Stuttgart) 1979;55.
- Schieroni MP, Revello MP. Medical, physical and rehabilitative treatment in a case load of 250 patients affected by scapulo-humeral periarthritis. Minerva Med 1985;76:941-3.
- Schomacher J. The effect of anterior versus posterior glide joint mobilization on external rotation range of motion in patients with shoulder adhesive capsulitis. J Orthop Sports Phys Ther 2007;37.
- Schultheis A, Reichwein F, Nebelung W. Frozen shoulder syndrome. MMW Fortschr Med 2009;151:36-7.
- Schultze J, Schlichting G, Galalae R, Koltze H, Kimmig B. Results of radiation therapy in periarthritis humeroscapularis. Rontgenpraxis 2004;55:160-4.
- Schulz J. Frozen shoulder. Med J Aust 1986;144.
- Schwitalle M, Betz U, Eckardt A, Rompe JD, Meurer A, Karbowski A. Rehabilitation success after shoulder manipulation under anaesthesia supported by continuous interscalene block. Eur J Phys Rehabil Med 1998;8:44-7.
- Scott NA, Guo B, Barton PM, Gerwin RD. Trigger point injections for chronic non-malignant musculoskeletal pain: a systematic review. Pain Med 2009;10:54-69.
- Segmuller HE, Taylor DE, Hogan CS, Saies AD, Hayes MG. Arthroscopic treatment of adhesive capsulitis. J Shoulder Elbow Surg 1995;4:403-8.
- Sharma RK, Bajekal RA, Bhan S. Frozen shoulder syndrome. A comparison of hydraulic distension and manipulation. Int Orthop 1993;17:275-8.
- Sharma SPS, Kvale AK, Baerheim AB. Outcome in shoulder capsulitis (frozen shoulder) treated with corticosteroid and corticosteroid with distension: a randomised pilot study. Int Musculoskelet Med 2009;31:75-80.
- Shchekotov GM. Humero-scapular periarthritis and its treatment with hydrocortisone. Vestn Khir Im I I Grek 1977;118:66-8.
- Shchepina TP. Physical therapy methods used in the rehabilitation of patients with periarthritis of the shoulder joint. Kurortol Fizioter 1989;26:33-5.
- Shehab D, Adham N. Comparative effectiveness of ultrasound and transcutaneous electrical stimulation in treatment of periarticular shoulder pain. Physiother Can 2000;52:208-10.
- Shiraishi T, Kubo T, Matsunaga T, Sugimoto K, Miyamoto H, Okumura SI, et al. Clinical study on tizanidine hydrochloride (Ternelin) therapy of tinnitus patients with shoulder stiffness. Practica Otologica 1991;84:1173-8.
- Sileghem A, Verstraeten A, Dequeker J. Double-blind, randomized, parallel-group study of the efficacy and safety of proglumetacin and naproxen in periarthritis of the shoulder or elbow. Curr Ther Res Clin Exp 1991;50:93-100.
- Simpson JK, Budge R. Treatment of frozen shoulder using distension arthrography (hydrodilatation): a case series. Australas Chiropr Osteopathy 2004;12:25-3.
- Singh OP, Goel MK, Goel SC, Singh GK. Periarthritis shoulder-treatment by manipulation and physiotherapy. Indian J Orthop 1980;14:201-3.
- Singh LN, Singh Ak J, Singh YN, Devi Sh P. Manipulation under anaesthesia for treatment of adhesive capsulitis with early physiotherapy. A pilot study. J Med Soc 2010;24:12-5.
- Skorogliadov AV, Sakalov DA, Seregin GI, Len’kova NA. Conduction blocks in the complex treatment of humeroscapular periarthritis. Ortop Travmatol Protez 1990:10-3.
- Skorogliadov AV, Gudkov VS, Okhotskaia OV, Sakalov DA. A method of treatment of humeroscapular periarthritis. Sov Med 1990:102-5.
- Skoroglyadov AV, Tektumanidze OV. Scapulohumeral periarthritis. Sov Med 1986:56-60.
- Slullitel D, Slullitel M, Blasco A, Periotti G, Yucra V, Cagliero G. Capsulitis adhesiva. Rev Argent Artrosc 2000;7:28-32.
- Smidt N, de Vet HCW, Bouter LM, Dekker J, Arendzen JH, de Bie RA, et al. Effectiveness of exercise therapy: a best-evidence summary of systematic reviews. Aust J Physiother 2005;51:71-85.
- Snow M, Boutros I, Funk L. Posterior arthroscopic capsular release in frozen shoulder. Arthroscopy 2009;25:19-23.
- Sokk J, Gapeyeva H, Ereline J, Kolts I, Paasuke M. Shoulder muscle strength and fatigability in patients with frozen shoulder syndrome: the effect of 4-week individualized rehabilitation. Electromyogr Clin Neurophysiol 2007;47:205-13.
- Spacca G, Cacchio A, Forgacs A, Monteforte P, Rovetta G, Rovati S. Analgesic efficacy of a lecithin-vehiculated diclofenac epolamine gel in shoulder periarthritis and lateral epicondylitis: a placebo-controlled, multicenter, randomized, double-blind clinical trial. Drugs Exp Clin Res 2005;31:147-54.
- Speed C, Hazleman B. Shoulder pain. Clin Evid 2004:1613-32.
- Speed C. Shoulder pain. Clin Evid 2006:1543-60.
- Spier W. Conservative therapy of shoulder stiffness. Hefte Unfallheilkd 1984;170:142-4.
- Spresser K. Extracorporeal shock wave therapy. J Am Chiropr Assoc 2002;39.
- Srivastava KP, Bhan BL, Bhatia IL. Scapulohumeral periarthritis. A clinical study and evaluation of end results of its treatment. J Indian Med Assoc 1972;59:275-8.
- Srour F. Use of cold for the management of acute retractile capsulitis of the shoulder joint. Kinesither Rev 2008;83:29-33.
- Stavnichii GV. Massage in humeroscapular periarthritis. Feldsher I Akusherka 1985;50:61-3.
- Stavnichii GV. Humeroscapular periarthritis. Its treatment by massage. Med Sestra 1986;45:11-3.
- Steinbrocker O, Argyros TG. Frozen shoulder: treatment by local injections of depot corticosteroids. Arch Phys Med Rehabil 1974;55:209-13.
- Stoddard A. Adhesive capsulitis of the shoulder and the sedimentation rate. Br J Phys Med 1955;18:9-10.
- Stodell MA, Sturrock RD. Frozen shoulder. Lancet 1981;2.
- Stoker DJ, Bruckner FE. Intra-articular injections in capsulitis. BMJ 1991;303.
- Stratz T, Stauber A, Muller W. Local treatment of periarthropathies with the 5-HT3-receptor-antagonist tropisetron. Z Orthop Ihre Grenzgeb 2002;140:394-8.
- Strobel G. Long-term therapeutic effect of different intra-articular injection treatments of the painful shoulder: effect on pain, mobility and work capacity. Rehabilitation 1996;35:176-8.
- Strunce JB, Walker MJ, Boyles RE, Young BA. The immediate effects of thoracic spine and rib manipulation on subjects with primary complaints of shoulder pain. J Man Manip Ther 2009;17:230-6.
- Sun SX. ECIWO Acupuncture Treatment of 151 Cases of Scapulohumeral Periarthritis n.d.:161-3.
- Sun KO, Chan KC, Lo SL, Fong DY. Acupuncture for frozen shoulder. Hong Kong Med J 2001;7:381-91.
- Sun G. Clinical experience in application of the acupoint futu. J Tradit Chin Med 2002;22:132-3.
- Surenkok O, Aytar A, Baltaci G. Acute effects of scapular mobilization in shoulder dysfunction: a double-blind randomized placebo-controlled trial. J Sport Rehabil 2009;18:493-501.
- Swirski J. Results of ultrasound treatment of humero-scapular periarthritis. Reumatologia 1971;9:159-63.
- Taller AJ. Treatment of periarthritis humeroscapularis with pirprofen. Double-blind study on 101 outpatients. Fortschr Med 1985;103:197-9.
- Talybov FI, Apenysheva NP. Thermography in patients with scapulohumeral periarthritis. Vopr Revm 1981:64-6.
- Talybov Yu F, Badokina GI, Sysoev VF, Agababova ER. Scintigraphy with the use of 99mTc-pyrophosphate in patients with scapulohumeral periarthritis. Ter Arkh 1981;53:25-8.
- Tam G. Effects of LLLT on the Periarthritis of the Shoulder: A Clinical Study on Different Treatments With Low Level Laser Therapy, Corticosteroid Injections or a Wait-and-See Policy 2010:S32-3.
- Tamai K, Yamato M, Hamada J, Mashitori H, Saotome K. Response of frozen shoulder to intraarticular corticosteroid and hyaluronate: a quantitative assessment with dynamic magnetic resonance imaging. Dokkyo J Med Sci 1999;26:235-41.
- Tamai K, Mashitori H, Ohno W, Hamada Ji, Sakai H, Saotome K. Synovial response to intraarticular injections of hyaluronate in frozen shoulder: a quantitative assessment with dynamic magnetic resonance imaging. J Orthop Sci 2004;9:230-4.
- Tan W. The recent status of physical medicine and rehabilitation in China. J Jpn Ass Phys Med Balneol Climatol 1995;59:10-4.
- Taskaynatan MA, Ozgul A, Yilmaz B, Kalyon TA. Suprascapular nerve block and steroid injection in nonspecific shoulder pain. J Rheumatol Med Rehabil 2004;15:9-15.
- Taskaynatan MA, Yilmaz B, Ozgul A, Yazicioglu K, Kalyon TA. Suprascapular nerve block versus steroid injection for non-specific shoulder pain. Tohoku J Exp Med 2005;205:19-25.
- Taverna E, Parrini M, Cabitza P. Lasertherapy vs placebo in the treatment of some bone and joints pathology. Minerva Ortop Traumatol 1990;41:631-6.
- Teys P, Bisset L, Vicenzino B. The initial effects of a Mulligan’s mobilization with movement technique on range of movement and pressure pain threshold in pain-limited shoulders. Man Therap 2008;13:37-42.
- Thakur S. Frozen shoulder. J Indian Med Assoc 1991;89:299-300.
- Thomas D, Williams RA, Smith DS. The frozen shoulder: a single blind controlled study of manipulation treatment compared with local hydrocortisone. Aust N Z J Med 1981;11:726-7.
- Thumb N, Kolarz G, Scherak O, Mayrhofer F. The efficacy and safety of fentiazac and diclofenac sodium in peri-arthritis of the shoulder: a multi-centre, double-blind comparison. J Int Med Res 1987;15:327-34.
- Todorov N. Treatment of humeroscapular periarthritis by an impulse ultrahigh-frequency field. Vopr Kurortol Fizioter Lech Fiz Kult 1972;37:410-11.
- Toker S, Kilincoglu V, Toker A, Gulcan E, Erturer E, Sahin L. Short-term outcome of combined corticosteriod and local anaestetic therapy with home-based exercise programme in painful shoulder conditions. Trop J Pharm Res 2008;7:1123-7.
- Toplicanec M. Use of cryotherapy in the treatment of a stiff and painful shoulder. Reumatizam 1986;33:22-8.
- Trehan RK, Patel S, Hill AM, Curtis MJ, Connell DA. Is it worthwhile to offer repeat hydrodilatation for frozen shoulder after 6 weeks?. Int J Clin Pract 2010;64:356-9.
- Tripathi RP, Verma AN, Sahu SC, Prasad BK. Treatment of periarthritis of the shoulder with intra-articular human placental extract: (a preliminary report). Clinician 1979;43:370-4.
- Tsarin VI. Manual therapy in the treatment of the humeroscapular periarthrosis syndrome. Med Sestra 1990;49:41-3.
- Tsukayama H, Yamashita H. Systematic review of clinical trials on acupuncture in the Japanese literature. Clin Acupunct Orient Med 2002;3:105-13.
- Tsun-Nin L. Injection of single acupuncture locus in treatment of posterior shoulder pain. A preliminary study. Orthop Rev 1977;6:63-6.
- Turner-Stokes L. MRI and arthroscopic surgery: a combined breakthrough in management of shoulder pain. Ann Rheum Dis 1996;55:405-6.
- Tuzlukov AP, Gorbatovskaia NS. Treatment of periarthritis of the shoulder joint. Ortop Travmatol Protez 1990:59-60.
- Ucuncu F, Capkin E, Karkucak M, Ozden G, Cakirbay H, Tosun M, et al. A comparison of the effectiveness of landmark-guided injections and ultrasonography guided injections for shoulder pain. Clin J Pain 2009;25:786-9.
- Ueno Y, Kuramoto K, Konno N, Koizumi T, Hoshiba T, Nunomura T, et al. Investigation on result of use after launch of ARTZ and ARTZ dispo: evaluation on the efficacy, safety and utility in the medication for osteoarthritis of the knee and periarthritis of the shoulder. Jpn Pharmacol Therapeut 1995;23:313-22.
- Uhlemann C, Callies R, Hippius M. Practical aspects of repeated iontophoresis per day. Z Physiother 1991;43:240-4.
- Uhthoff HK. Open surgical release for frozen shoulder. J Shoulder Elbow Surg 2002;11.
- Uitvlugt G, Detrisac DA, Johnson LL, Austin MD, Johnson C. Arthroscopic observations before and after manipulation of frozen shoulder. Arthroscopy 1993;9:181-5.
- Ulmer G. Shoulder mobilization under anesthesia in complete and partial shoulder stiffness. Helv Chir Acta 1982;48:611-14.
- Vad VB, Sakalkale D, Warren RF. The role of capsular distention in adhesive capsulitis. Arch Phys Med Rehabil 2003;84:1290-2.
- Valtonen EJ. Methylprednisolone acetate in the management of periarthritis of the shoulder and external humeral epicondylitis resistant to physical therapy. Ann Med Intern Fenn 1967;56:61-3.
- Valtonen EJ. Subacromial betamethasone therapy. II. Subacromial betamethasone and methylprednisolone injections in treatment of frozen shoulder and supra spinam tendinitis. A comparative trial. Ann Chir Gynaecol Fenn 1974;188:9-16.
- Valtonen EJ. Subacromial betamethasone therapy. I. The effect of subacromial injection of betamethasone in cases of painful shoulder resistant to physical therapy. Ann Chir Gynaecol Fenn 1974;188:5-8.
- van de Weg FB, van der Windt DAWM, Winters JC. Injection therapy in shoulder disorders, yes or no? A systematic review of the literature. Huisarts Wet 2004;47:178-83.
- van den Dolder PA, Roberts DL. A trial into the effectiveness of soft tissue massage in the treatment of shoulder pain. Aust J Physiother 2003;49:183-8.
- van der Heijden GJ, van der Windt DA, Kleijnen J, Koes BW, Bouter LM. Steroid injections for shoulder disorders: a systematic review of randomized clinical trials. Br J Gen Pract 1996;46:309-16.
- van der Heijden GJ, Leffers P, Wolters PJ, Verheijden JJ, van Mameren H, Houben JP, et al. No effect of bipolar interferential electrotherapy and pulsed ultrasound for soft tissue shoulder disorders: a randomised controlled trial. Ann Rheum Dis 1999;58:530-40.
- van der Windt D. Shoulder disorders in primary care. Amsterdam: Vrije Universiteit; 1997.
- van der Windt DA, Koes BW, Deville W, Boeke AJ, de Jong BA, Bouter LM. Effectiveness of corticosteroid injections versus physiotherapy for treatment of painful stiff shoulder in primary care: randomised trial. BMJ 1998;317:1292-6.
- van der Windt DA, van der Heijden GJ, van den Berg SG, ter Riet G, de Winter AF, Bouter LM. Ultrasound therapy for musculoskeletal disorders: a systematic review. Pain 1999;81:257-71.
- Arroll B. Corticosteroid injections were superior to physiotherapy for painful stiff shoulder. Evid Based Med 1999;4.
- van der Windt D, Koes B, Boeke J, Deville W, de Jong B, Bouter L. Corticosteroid injections or physiotherapy for painful, stiff shoulder? A randomized study in a general practice. Huisarts Wet 2000;43:420-5.
- van der Windt DAWM, Bouter LM. Physiotherapy or corticosteroid injection for shoulder pain?. Ann Rheum Dis 2003;62:385-7.
- van Laack W, Hennes A, Refisch A. Mobilization of the partially stiff shoulder under anesthesia (ankylosing humeroscapular periarthritis). Z Orthop Ihre Grenzgeb 1987;125:669-73.
- van Royen BJ, Pavlov PW. Treatment of frozen shoulder by distension and manipulation under local anaesthesia. Int Orthop 1996;20:207-10.
- van Royen B, Pavlov P. Frozen shoulder – manipulation after glenohumeral distension under local anesthesia. Acta Orthop Scand 1996;67.
- Vanni G, Galante M, Franchignoni FP. Orgotein in treatment of scapulohumeral periarthritis in hemiplegic patients. Riabilitazione 1985;18:251-6.
- Vas J, Ortega C, Olmo V, Perez-Fernandez F, Hernandez L, Medina I, et al. Single-point acupuncture and physiotherapy for the treatment of painful shoulder: a multicentre randomized controlled trial. Rheumatology 2008;47:887-93.
- Vaughn BF, Beam JW, Duschatko DM, Ingram-Rice B. Adhesive capsulitis: the ‘frozen shoulder’ syndrome. J Bodyw Mov Ther 2000;4:3-4.
- Venturi R. Medical treatment of acute periarthritis of the shoulder by drug injection directly into the scapulo-humeral joint and into the subachromial-deltoid bursa. Arthrographic and anatomical research. Chir Organi Mov 1979;65:157-76.
- Verkhozina EA. Treatment of traumatic synovitis arthritis and peri arthritis with ultrasound. Zdravookhr Beloruss 1972;18:58-9.
- Vermeulen HM, Verhoef J, Rozing PM, Obermann WR, Le Cessie S, van Houwelingen JC, et al. A comparison of end-range and mid-range mobilization techniques in the treatment of adhesive capsulitis: a randomized controlled trial. Ann Rheum Dis 2004;63.
- Vermeulen E. Physical therapy for shoulder disorders. Ned Tijdschr Fysiotherapie 2007;117:90-1.
- Verstraete A, Deckers Y, Dequeker J. Treatment of periarthritis: a single-blind evaluation of 2 injectable corticosteroids. Clin Trials J 1985;22:471-6.
- Vigano R, Guarino A, Nannei AM. Use of laser therapy associated with a lysine ketoprofen gel in various pathologies of orthopedic importance. Clin Ter 1986;118:13-7.
- Volhard J. The treatment of acute humeroscapuar periarthritis. Med Welt 1968;3.
- von Knorre VB, Keitel W. Comparative study of therapy: ultrasound, cryotherapy and intra-articulare cortisonoids to treat alterations of the shoulder joint due to inflammation. Z Physiother 1990;42:221-5.
- Vrettos BC, Neumann L, MacKie A, Damrel D, Wallace WA. One-stage arthroplasty of the ipsilateral shoulder and elbow. J Shoulder Elbow Surg 2005;14:425-8.
- Wagenhau FJ. Treatment of humeroscapular periarthritis. Dtsch Med Wochenschr 1969;94:1579-81.
- Walach H, Guthlin C, Konig M. Efficacy of massage therapy in chronic pain: a pragmatic randomized trial. J Altern Complement Med 2003;9:837-46.
- Waldburger M, Meier JL, Gobelet C. The frozen shoulder: diagnosis and treatment. Prospective study of 50 cases of adhesive capsulitis. Clin Rheumatol 1992;11:364-8.
- Wallny T, Melzer C, Wagner U, Wirth CJ, Schmitt O. [‘Primary’ shoulder stiffness: illness duration and therapeutic comparison. Z Orthop Ihre Grenzgeb 1997;135:222-7.
- Walsh DM, Howe TE, Johnson MI, Sluka KA. Transcutaneous electrical nerve stimulation for acute pain. Cochrane Database Syst Rev 2009.
- Wang W, Yin X, He Y, Wei J, Wang J, Di F. Treatment of periarthritis of the shoulder with acupuncture at the Zhongping (foot) extrapoint in 345 cases. J Tradit Chin Med 1990;10:209-12.
- Wang J, Wang W, Wang S. Treatment of periarthritis humeroscapularis with acupuncture and acupoint blocking. J Tradit Chin Med 1993;13:262-3.
- Wang W. 78 cases of periomarthritis treated with acupuncture. J Tradit Chin Med 1995;15:46-7.
- Wang LC. 50 cases of periarthritis of shoulder joint treated by electro-acupuncture and point injection therapy. Shanghai J Acupunct Moxibustion 1997;16.
- Wang W, Yu ZL, Xie J, Xu AS, Fang J. Relief of pain and improvement of dysfunction in shoulder periarthritis following Qi acupoint specific acupuncture versus conventional acupuncture. Chin J Clin Rehab 2005;9:48-50.
- Wang SS, Trudelle-Jackson EJ. Comparison of customized versus standard exercises in rehabilitation of shoulder disorders. Clin Rehabil 2006;20:675-85.
- Wang Z-L, Chen L-F, Zhu W-M. Observation on the transient analgesic effect of abdominal acupuncture TENS on pain of neck, shoulder, loin and legs. Zhongguo Zhenjiu 2007;27:657-9.
- Wang J-P, Huang T-F, Hung S-C, Ma H-L, Wu J-G, Chen T-H. Comparison of idiopathic, post-trauma and post-surgery frozen shoulder after manipulation under anesthesia. Int Orthop 2007;31:333-7.
- Wang Q-H, Du P-B, Gong H-T. Effects of adhesion releasing under nerve block and arthrolysis on adhesive scapulohumeral periarthritis: a comparative study. Chin Med J 2008;88:177-8.
- Warner JJ, Allen A, Marks PH, Wong P. Arthroscopic release for chronic, refractory adhesive capsulitis of the shoulder. J Bone Joint Surg Am 1996;78:1808-16.
- Wassef MR. Suprascapular nerve block. A new approach for the management of frozen shoulder. Anaesthesia 1992;47:120-4.
- Watson L, Dalziel R, Story I. Frozen shoulder: a 12-month clinical outcome trial. J Shoulder Elbow Surg 2000;9:16-22.
- Weber M, Prim J, Bugglin R, Michel BA, Gerber H. Long-term follow up of patients with frozen shoulder after mobilization under anesthesia, with special reference to the rotator cuff. Clin Rheumatol 1995;14:686-91.
- Weber KG. The 7-point shoulder treatment program. Krankengymnastik 2001;53:976-9.
- Weiser HI. Mobilization under local anesthesia for painful primary frozen shoulder. Harefuah 1976;90:215-19.
- Weiser HI. Painful primary frozen shoulder mobilization under local anesthesia. Arch Phys Med Rehabil 1977;58:406-8.
- Wen L. Analysis on effect of functional exercises to promote rehabilitation of patients wih periarthritis of shoulder. Chin Nurs Res 2009;23:1925-6.
- White AE, Tuite JD. The accuracy and efficacy of shoulder injections in restrictive capsulitis. J Bone Joint Surg Br 1995;77–B.
- White AET, Tuite JD. The accuracy and efficacy of shoulder injections in restrictive capsulitis. J Orthop Rheumatol 1996;9:37-40.
- White A. Chronic shoulder pain. Acupunct Med 2009;27.
- Widiastuti-Samekto M, Sianturi GP. Frozen shoulder syndrome: comparison of oral route corticosteroid and intra-articular corticosteroid injection. Med J Malaysia 2004;59:312-16.
- Wies JT, Niel Asher S, Latham M, Hazleman B, Speed CA. A pilot randomised placebo controlled trial of osteopathic and physiotherapy treatment for frozen shoulder. Br J Rheumatol 2003;42.
- Wiley AM. Frozen shoulder: surgical applications. Surg Shoulder 1995;1085:269-74.
- Wiley AM, Vastamaki M, Roth JH, Vastamaki M, Roth JH. Hand and upper extremity; arthroscopy of the upper extremity. New York: Churchill Livingstone; 1997.
- Winters JC, Sobel JS, Arendzen JH, Meyboom-de Jong B. The effect of corticosteroid injections in shoulder disorders. Huisarts Wet 1995;38:164-8.
- Winters JC, Sobel JS, Groenier KH, Arendzen HJ, Meyboom-de Jong B. Comparison of physiotherapy, manipulation, and corticosteroid injection for treating shoulder complaints in general practice: randomised, single blind study. BMJ 1997;314:1320-5.
- Winters JC, Jorritsma W, Groenier KH, Sobel JS, Meyboom-de Jong B, Arendzen HJ. Treatment of shoulder complaints in general practice: long term results of a randomised, single blind study comparing physiotherapy, manipulation, and corticosteroid injection. BMJ 1999;318:1395-6.
- Winters JC. The small art of the short term. Corticosteroid injections in the treatment of shoulder complaints. Pharm Weekbl 2000;135:690-3.
- Worsdorfer O, Wasmer G. Operative treatment of shoulder stiffness. Hefte Unfallheilkd 1984;170:150-8.
- Xiao JB, She SZ, Song WG, Zhang BG, Zeng ZW. Effects of super laser on the affected limb of acute and chronic pain 315 cases. Chin J Clin Rehab 2006;10:116-18.
- Xie KY, Zhao GF, Lu JM. Treatment of 103 cases of periarthritis of the shoulder by acupoint laser irradiation. J Tradit Chin Med 1988;8:265-6.
- Xu X, Fang JQ, Zhang Y, Liu KZ, Han CP. Treatment of early-stage adhesive shoulder periarthritis with transcutaneous electric stimulation on acupoints. J Acu Tuina Sci 2006;4:353-5.
- Xu B. Bone setting manipulation of Shangsiyuan chuobanchu for treatment of periarthritis of shouder. Zhongguo Gushang 2008;21.
- Xu K-S, Huang M-W, Yao L-Y, Chen J, Su J, Zheng J-Q. Clinically randomized controlled trials of moxibustion with salt in bamboo circle for treatment of periarthritis of shoulder. Zhongguo Zhenjiu 2009;29:77-80.
- Yamaguchi K, Sethi N, Bauer GS. Postoperative pain control following arthroscopic release of adhesive capsulitis: a short-term retrospective review study of the use of an intra-articular pain catheter. Arthroscopy 2002;18:359-65.
- Yamaguchi K, Buchbinder R, Green S, Forbes A, Hall S, Lawler G. Arthrographic distension with saline and steroid reduced pain and disability and improved range of motion in the short term in patients with painful stiff shoulder. J Bone Joint Surg Series A 2004;86.
- Yamaguchi K, Hollis R. Arthroscopic release of frozen shoulder. Semin Arthroplasty 2008;19:50-3.
- Yamamoto R, Namiki O, Iwata H, Shinmei M, Mikasa M, Ogawa N. Randomized comparative study of sodium hyaluronate (SPH) on periarthritis scapulohumeralis. Jpn J Clin Pharmacol Ther 1988;19:717-33.
- Yamamoto R, Tabata S, Mikasa M, Takagishi K, Ogawa N. Dose-range finding study of SLM-10 (sodium hyaluronate injection) in patients with periarthritis scapulohumeralis. Jpn Pharmacol Therapeut 1993;21:265-86.
- Yamamoto R, Tabata S, Mikasa M, Takagishi K, Ogawa N. Clinical trial of SLM-10 (sodium hyaluronate injection) compared with ARTZ in patients with periarthritis scapulohumeralis. Jpn Pharmacol Therapeut 1993;21:285-307.
- Yamshon LJ. Frozen shoulder: methods for bringing about early mobilization. Calif Med 1958;89:333-4.
- Yan XM, Li XY, Sun Y. Quantitative evaluation of the enhancement of therapeutic effect in pain therapy by combining nerve block and linear polarized near-infrared ray irradiation. Chin J Clin Rehab 2005;9:44-5.
- Yang J, Lu K. Sixty-four cases of scapulohumeral periarthritis treated by auricular plaster therapy. J Tradit Chin Med 2006;26:179-80.
- Yao HH, Chen YH. The effects of comprehensive rehabilitation therapy on scapulohumeral periarthritis. Chin J Clin Rehab 2002;6.
- Yaya Huaman R, Blasco Olcina R, Garcia Fernandez M, Vilches Padilla JJ. A study of 93 cases of painful shoulder and shoulder-band syndrome treated with hydergine. Rev Esp Otoneurooftalmol Neurocir 1975;33:131-5.
- Yegudin-Ash J, Afzal M, Cavaliere LF, Lee D, Zelicof S, Weinstein A. A comparative study of ultrasonography with magnetic resonance imaging in patients with painful shoulder using arthroscopy as a gold standard. Arthritis Rheum 1998;41.
- Yigiter K, Kerem M. A comparison of the effects of two different iontophoresis applications on pain in patients with adhesive capsulitis of the shoulder. Pain Clinic 2002;14:49-53.
- Yuan DW. Bioholographic acupuncture therapy for 44 cases of scapulohumeral periarthritis. Shanghai J Acupunct Moxibustion 1995;14.
- Zachepa AE. Surgical treatment of humero-scapular periarthroses. Ortop Travmatol Protez 1991:37-8.
- Zancan A, Gialanella B, Luisa A, Della Marta ME, D’Alessandro G, Casale R. Telethermographic effects and a comparative clinical assessment of the treatment of shoulder periarthritis using ultrasound. G Ital Med Lav 1993;15:55-8.
- Zeilig G, Guibert R. Terra Quant device for shoulder pain. Double blind trial. Positive Health 2005. www.positivehealth.com/article-view.php?articleid=1778 (accessed 3 June 2010).
- Zeng X-L, Lei L-M, Lin Z, Wang Z-T. Clinical observation on acupuncture at pain point combined with moxibustion at Zusanli (ST 36) for treatment of periarthritis of shoulder. Zhongguo Zhenjiu 2005;25:326-8.
- Zhang M. Treatment of peri-omarthritis with acupuncture at Yanglingquan (GB 34). J Tradit Chin Med 1991;11:9-10.
- Zhang W, Oetliker H, Schlapbach P, Gerber NJ. Rheumatology, Vol. 14. Physiotherapy: controlled trials and facts. Basel, Switzerland: S. Karger Ag; 1991.
- Zhang X, Wang Zd, Ou Ck. Needle scalpel versus acupuncture for scapulohumeral periarthritis in a staged treatment. Chin J Clin Rehab 2006;10:11-3.
- Zhang GX. Treatment of acute injury of soft tissue around shoulder joint by exercise needling and electroacupuncture as main. Zhongguo Zhen Jiu 2008;28:485-8.
- Zhang T-W, Cheng S-D, Wang H-F, Xu H-L. Shoulder-loosening techniques on severe frozen shoulder. Zhongguo Gushang 2009;22:718-19.
- Zhao L, Xia J, Fan J. The treatment of computer middle frequency on periorthritis of shoulder. Chin J Clin Rehab 2003;7.
- Zhao H, Zhao T, Liu B-Y, Liu Z-S. Observation on therapeutic effects of different needling methods at tiaokou (ST 38) on periarthritis of shoulder. Zhongguo Zhenjiu 2006;26:729-31.
- Zhou G, Yang Y. Clinical observations on the treatment of 76 shoulder periarthritis cases by meridional differentiation-based distant opposing acupuncture. Shanghai J Acupunct Moxibustion 2004;23.
- Zhu BW. 182 cases of periarthritis of shoulder joint treated by acupuncture and moxibustion and massage therapy. Shanghai J Acupunct Moxibustion 1997;16:23-4.
- Zhu ZZ, Liu CM, Feng WX. Analgesic effects of traditional Chinese medicine iontophoresis and rehabilitation training on periarthritis of shoulder: a randomized controlled study. Chin J Clin Rehab 2004;8:2698-9.
- Zivkovic M. Comparative Analysis of the Use of Kinesi Therapy and Other Methods of Physical Medicine in the Treatment of Scapulo Humeral Peri-Arthritis n.d.
- Zuecker W, Knuth C. Treatment of shoulder pain by appropriate methods of therapeutic swimming. Z Physiother 1977;29:407-9.
- Cyriax J, Russel G. Textbook of orthopaedic medicine. London: Ballière Tindall; 1977.
- Maitland GD. Treatment of the glenohumeral joint by passive movement. Physiotherapy 1983;69:3-7.
- Kaltenborn FM. Manual therapy for the extremity joints. Oslo: Olaf Norlis Bokhander; 1976.
- Vermeulen HM, Obermann WR, Burger BJ, Kok GJ, Rozing PM, van Den Ende CH. End-range mobilization techniques in adhesive capsulitis of the shoulder joint: a multiple-subject case report. Phys Ther 2000;80:1204-13.
- Mulligan BR. Mobilisations with movement. J Manual Manipulative Ther 1993;1:154-6.
Appendix 1 Search strategies for effectiveness review
BIOSIS Previews
Interface: Web of Science (ISI) on Web of Knowledge
Search date: 30 March 2010
Strategy: as below
Records identified: 800
| # 20 | 800 |
#19 OR #17 OR #15 OR #13 OR #11 OR #9 OR #5 OR #3 Databases=PREVIEWS Timespan=All Years |
| # 19 | 140 |
#18 AND #1 Databases=PREVIEWS Timespan=All Years |
| # 18 | 33,082 |
Topic=(“nerve block” or “neuromuscular blockade” or acupuncture or electroacupuncture or “electro acupuncture” or osteopuncture or “osteo puncture” or “perioste* stimulat*” or “perioste* therap*” or “perioste* needling” or massag* or acupressure or shiatsu or shiatzu or “zhi ya” or “chih ya” or “rehabilitat* program*” or “rehabilitat* protocol*” or “watch* wait*” or “conservative treat*” or “management decision*” or “management option*” or “management choice*”) Databases=PREVIEWS Timespan=All Years |
| # 17 | 454 |
#16 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 16 | > 100,000 |
Topic=(physiotherapy or physiotherapies or “physical therap*” or “manual therap*” or “passive motion” or “passive movement” or “muscle stretching” or “muscle stretches” or “muscle stretch” or mobilisation or mobilization or “exercis* program*” or “exercis* intervention*” or “exercis* train*” or “exercis* prescription*” or “exercis* prescrib*” or “exercis* therap*” or “therapeutic exercis*” or “home exercis*” or “supervis* exercis*” or “pendular exercis*” or “pendulum exercis*” or “isokinetic exercis*” or “resist* exercise*”) Databases=PREVIEWS Timespan=1969-2010 |
| # 15 | 38 |
#14 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 14 | > 100,000 |
Topic=(biofeedback) OR Topic=(thermograph* or thermotherap* or “thermo therap*” or hypertherm* or “hyper therm*” or diatherm* or cryotherap* or “cryo therap*”) OR Topic=(“cold pack*” or “cold therap*” or “cold treat*” or “ice pack*” or “ice therap*” or “ice treat*” or “heat pack*” or “heat therap*” or “heat treat*” or “hot pack*” or “heat therap*”) OR Topic=(“Laser Therapy” or “ultrasonic therapy” or “Interventional ultrasonography”) OR Topic=(“electrotherapeutic intervention*” or “electrotherapeutic treat*”) OR Topic=(“magnetic field therapy”) OR Topic=(“electromagnetic field therapy”) OR Topic=(“Biomagnetic therapy”) Databases=PREVIEWS Timespan=1969-2010 |
| # 13 | 31 |
#12 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 12 | 50,394 |
Topic=(TENS or ALTENS) OR Topic=(“electric* stimulat*” or “transcutaneous stimulat*”or “transdermal electrostimulat*”or “cutaneous electrostimulat*”or electroanalgesia or “electro analgesia”) OR Topic=(“muscle stimulat*”) OR Topic=(neuromodulation or “neuro modulation” or neurostimulation or “neuro stimulation”) OR Topic=(interferential) Databases=PREVIEWS Timespan=1969-2010 |
| # 11 | 30 |
#10 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 10 | 5938 |
Topic=(chiropractic*) OR Topic=(osteopath*) OR Topic=(manipulat* and (anesthesia or anaesthesia or anesthetic* or anaesthetic*)) OR Topic=(MUA) Databases=PREVIEWS Timespan=1969-2010 |
| # 9 | 112 |
#8 OR #7 Databases=PREVIEWS Timespan=1969-2010 |
| # 8 | 39 |
Topic=(“subacromial inject*” or “acromioclavicular inject*” or “glenohumeral inject*”) OR Topic=(“extra articular inject*” or “extraarticular inject*”) OR Topic=(“shoulder* inject*”) Databases=PREVIEWS Timespan=1969-2010 |
| # 7 | 81 |
#6 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 6 | 85,801 |
Topic=(injections) OR Topic=(“intraarticular injection*” or “intra-articular injection*” or “intra articular injection*”) OR Topic=(“bursa* inject*” or “intrabursa* inject*” or “intra bursa* inject*” or “periartic* inject*” or “peri artic* inject*”) Databases=PREVIEWS Timespan=1969-2010 |
| # 5 | 152 |
#4 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 4 | 5743 |
Topic=(Arthroscopy) OR Topic=(“arthroscop* releas*” or “arthroscop* decompress*” or “arthroscop* capsulotom*”) OR Topic=(“capsular releas*” or “interventional microadhesiolysis” or capsulotomy) Databases=PREVIEWS Timespan=1969-2010 |
| # 3 | 11 |
#2 AND #1 Databases=PREVIEWS Timespan=1969-2010 |
| # 2 | 25,371 |
Topic=(“arthrograph* distension*” or “arthograph* distention*”) OR Topic=(“arthrogram* distension*” or “arthogram* distention*”) OR Topic=(“glenohumeral distension*” or “glenohumeral distention*”) OR Topic=(dilatation or hydrodilat*) OR Topic=(arthography) Databases=PREVIEWS Timespan=1969-2010 |
| # 1 | 2354 |
Topic=(“frozen shoulder*”) OR Topic=(“stiff* shoulder*”) OR Topic=(“adhesive capsulitis”) OR Topic=(“shoulder bursitis”) OR Topic=(periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) OR Topic=(“shoulder pain*” or “pain* shoulder” or “shoulder complaint*”) OR Topic=(“shoulder* impinge*”) OR Topic=(“subacromial impingement syndrome”) Databases=PREVIEWS Timespan=1969-2010 |
Cumulative Index to Nursing and Allied Health Literature (CINAHL)
Interface: EBSCO
Search date: 26 March 2010
Strategy: as below
Records identified: 1757
| S73 | S18 or S22 or S27 or S33 or S40 or S51 or S56 or S62 or S66 or S70 or S72 |
| S72 | s14 and s71 |
| S71 | rehabilitat* N2 program* or rehabilitat* N2 protocol* or watch* N2 wait* or conservative N2 treat* or management N2 decision* or management N2 option* or management N2 choice* |
| S70 | S14 AND S69 |
| S69 | S67 or S68 |
| S68 | ( massag* or acupressure ) or ( shiatsu or shiatzu ) or zhi ya or chih ya |
| S67 | (MH “Massage”) |
| S66 | S14 and S65 |
| S65 | S63 or S64 |
| S64 | electroacupuncture* or osteopuncture* or perioste* N3 stimulat* or perioste* N3 therap* or perioste* N3 needling |
| S63 | (MH “Acupuncture+”) |
| S62 | S14 AND S61 |
| S61 | S57 or S58 or S59 or S60 |
| S60 | neuromuscular block* |
| S59 | (MH “Nerve Block”) |
| S58 | pulsed electromagnetic field therapy or electromagnetic* N3 field* or biomagnetic* or pulsed signal or PEMF |
| S57 | (MH “Magnetics+”) |
| S56 | S14 AND S55 |
| S55 | S52 or S53 or S54 |
| S54 | electrotherapeutic N2 intervention* or electrotherapeutic N2 treat* or laser N2 therap* or ultrasound N2 therap* |
| S53 | (MH “Ultrasonography+”) |
| S52 | (MH “Lasers+”) |
| S51 | S14 AND S50 |
| S50 | S43 or S44 or S45 or S46 or S47 or S48 or S49 |
| S49 | ( thermograph*or thermotherap* or hypertherm* or diatherm* or cryotherap* ) or thermo therap* or hyper therm* or cryo therap* or thermo graph* |
| S48 | hot N2 pack* or hot N2 therap* or hot N2 treat* |
| S47 | cold N2 pack* or cold N2 therap* or cold N2 treat* or ice N2 pack* or ice N2 therap* or ice N2 treat* or heat N2 pack* or heat N2 therap* or heat N2 treat* |
| S46 | (MH “Hyperthermia, Induced”) |
| S45 | (MH “Ice”) |
| S44 | (MH “Diathermy”) |
| S43 | (MH “Cryotherapy”) or (MH “Heat-Cold Application”) |
| S42 | S14 AND S41 |
| S41 | (MH “Biofeedback”) |
| S40 | S14 AND S39 |
| S39 | ( TENS or ALTENS ) or electric* N2 stimulat* or transcutaneous N2 stimulat* or transdermal N2 electrostimulat* or cutaneous N2 electrostimulat* or electroanalgesia or muscle N2 stimulat* or neuromodulation or neurostimulation or interferential |
| S38 | S14 and S37 |
| S37 | S34 or S35 or S36 |
| S36 | chiropractic* or osteopath* or manipulat* N3 anesthesia or manipulat* N3 anaesthesia or manipulat* N3 anesthetic* or manipulat* N3 anaesthetic* or MUA |
| S35 | (MH “Manual Therapy+”) |
| S34 | (MH “Manipulation, Chiropractic”) or (MH “Manipulation, Orthopedic”) or (MH “Manipulation, Osteopathic”) |
| S33 | S14 and S32 |
| S32 | S28 or S29 or S30 or S31 |
| S31 | exercise* N2 therap* or exercise* N2 therapeutic or home N2 exercis* or supervis*N2 exercis* or pendular exercis* or pendulum exercis* or isokinetic N2 exercise* or resist*N2 exercise* |
| S30 | passive motion or passive movement or CPM or ( stretching or stretches ) or ( mobilisation or mobilization ) or exercise* N2 program* or exercise* N2 strength* or exercise* N2 intervention* or exercise* N2 training or exercise* N2 prescription* or exercise* N2 prescrib* |
| S29 | physiotherapy or physiotherapies or physical therap* or manual therap* |
| S28 | (MH “Physical Therapy+”) |
| S27 | S24 or S25 or S26 |
| S26 | subacromial N3 inject* or acromioclavicular N3 inject* or glenohumeral N3 inject* or extraarticular N3 inject* or shoulder* N3 inject* |
| S25 | bursa*N3 inject* or intrabursa* N3 inject* or intra bursa* N3 inject* or periartic* N3 inject* or peri artic* N3 inject* or intraartic* N3 inject* or intra artic* N3 inject* |
| S24 | S14 and S23 |
| S23 | (MH “Injections+”) |
| S22 | S14 and S21 |
| S21 | S19 or S20 |
| S20 | arthroscop* N6 releas* or arthroscop* N6 decompress* or arthroscop* N6 capsulotom* or capsular N2 releas* or interventional microadhesiolysis or capsulotomy |
| S19 | (MH “Arthroscopy”) |
| S18 | S14 and S17 |
| S17 | S15 or S16 |
| S16 | arthrograph* N6 distension* or arthrograph* N6 distention* or arthrogram* N6 distension* or arthrogram* N6 distention* or glenohumeral N6 distension* or glenohumeral N6 distention* or ( dilatation or hydrodilat* ) |
| S15 | (MH “Arthrography”) |
| S14 | S1 or S2 or S3 or S4 or S5 or S6 or S7 or S8 or S9 or S10 or S11 or S12 or S13 |
| S13 | subacromial impingement syndrome |
| S12 | shoulder* N6 impinge* |
| S11 | (MH “Shoulder Impingement Syndrome”) |
| S10 | shoulder* N3 pain or shoulder* N3 pains or shoulder* N3 painful or shoulder* N3 complain* |
| S9 | (MH “Shoulder Pain”) |
| S8 | periarthritis N6 shoulder* or peri-arthritis N6 shoulder* or periarthritides N6 shoulder* or peri-arthritides N6 shoulder* or pericapsulitis N6 shoulder* or peri-capsulitis N6 shoulder* |
| S7 | capsulitis N6 shoulder* |
| S6 | bursitides N6 shoulder* |
| S5 | bursitis N6 shoulder* |
| S4 | adhesive N3 capsulitis |
| S3 | (MH “Adhesive Capsulitis”) |
| S2 | stiff* N3 shoulder* |
| S1 | frozen N6 shoulder* |
Cochrane Central Register of Controlled Trials (CENTRAL)
Interface: Cochrane Library Issue 3 2010
Search date: 23 March 2010
Strategy: as below
Records identified: 539
| ID | Search | Hits |
|---|---|---|
| #1 | (frozen NEAR/6 shoulder*):ti,ab,kw | 65 |
| #2 | (stiff* NEAR/3 shoulder*):ti,ab,kw | 63 |
| #3 | MeSH descriptor Bursitis explode all trees | 127 |
| #4 | (adhesive NEXT (capsulitis or capsulitides)):ti,ab,kw | 44 |
| #5 | (bursitis or bursitides) NEAR/6 shoulder*:ti,ab,kw | 11 |
| #6 | (capsulitis or capsulitides) NEAR/6 shoulder* | 39 |
| #7 | (periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) NEAR/6 shoulder*:ti,ab,kw | 77 |
| #8 | MeSH descriptor Shoulder Pain explode all trees | 240 |
| #9 | (shoulder* NEAR/3 (pain or pains or painful or complain*)):ti,ab,kw | 682 |
| #10 | MeSH descriptor Shoulder Impingement Syndrome explode all trees | 84 |
| #11 | (shoulder* NEAR/6 impinge*):ti,ab,kw | 107 |
| #12 | “subacromial impingement syndrome”:ti,ab,kw | 20 |
| #13 | (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12) | 964 |
| #14 | MeSH descriptor Arthrography, this term only | 148 |
| #15 | (arthrograph* NEAR/6 (distension* or distention*)):ti,ab,kw | 10 |
| #16 | (arthrogram* NEAR/6 (distension* or distention*)):ti,ab,kw or (glenohumeral NEAR/6 (distension* or distention*)):ti,ab,kw | 3 |
| #17 | MeSH descriptor Dilatation, this term only | 281 |
| #18 | (dilatation or hydrodilat*):ti,ab,kw | 2541 |
| #19 | (#14 OR #15 OR #16 OR #17 OR #18) | 2695 |
| #20 | (#13 AND #19) | 25 |
| #21 | MeSH descriptor Arthroscopy explode all trees | 835 |
| #22 | (arthroscop* NEAR/6 (releas* or decompress* or capsulotom*)):ti,ab,kw | 64 |
| #23 | (capsular NEAR/2 releas*):ti,ab,kw or “interventional microadhesiolysis”:ti,ab,kw or (capsulotomy):ti,ab,kw | 197 |
| #24 | (#21 OR #22 OR #23) | 1048 |
| #25 | (#13 AND #24) | 51 |
| #26 | MeSH descriptor Injections, Intra-Articular explode all trees | 628 |
| #27 | (#13 AND #26) | 62 |
| #28 | MeSH descriptor Injections explode all trees | 15,394 |
| #29 | (#13 AND #28) | 115 |
| #30 | (bursa* or intrabursa* or intra bursa* or periartic* or peri artic* or intraartic* or intra artic*) NEAR/3 inject*:ti,ab,kw | 813 |
| #31 | (#13 AND #30) | 79 |
| #32 | (subacromial or acromioclavicular or glenohumeral) NEAR/3 inject*:ti,ab,kw or (extra articular or extraarticular or shoulder*) NEAR/3 inject*:ti,ab,kw | 72 |
| #33 | (#27 OR #29 OR #31 OR #32) | 169 |
| #34 | MeSH descriptor Physical Therapy Modalities explode all trees | 9583 |
| #35 | (physiotherapy or physiotherapies or (physical therap*) or (manual therap*)):ti,ab,kw or (passive NEXT (motion or movement)):ti,ab,kw or (CPM):ti,ab,kw | 10,737 |
| #36 | MeSH descriptor Muscle Stretching Exercises explode all trees | 82 |
| #37 | (stretching or stretches):ti,ab,kw or (mobilisation or mobilization):ti,ab,kw or (exercise* NEAR/2 (program* or strength* or intervention* or training or prescription* or prescrib*)):ti,ab,kw or (exercise* NEAR/2 (therap* or therapeutic)):ti,ab,kw | 10,643 |
| #38 | (home or supervis*) NEAR/2 exercis*:ti,ab,kw or (pendular or pendulum) NEXT exercis*:ti,ab,kw or (isokinetic or resist*) NEAR/2 exercise*,:ti,ab,kw | 1658 |
| #39 | (#34 OR #35 OR #36 OR #37 OR #38) | 23,617 |
| #40 | (#13 AND #39) | 300 |
| #41 | MeSH descriptor Musculoskeletal Manipulations explode all trees | 4745 |
| #42 | (chiropractic*):ti,ab,kw or (osteopath*):ti,ab,kw or (manipulat* NEAR/3 (anesthesia or anaesthesia or anesthetic* or anaesthetic*)):ti,ab,kw or (MUA):ti,ab,kw | 728 |
| #43 | (#41 OR #42) | 5242 |
| #44 | (#13 AND #43) | 119 |
| #45 | (TENS or ALTENS):ti,ab,kw or (electric* NEAR/2 stimulat*) or (transcutaneous NEAR/2 stimulat*) or (transdermal NEAR/2 electrostimulat*) or (cutaneous NEAR/2 electrostimulat*) or electroanalgesia or (electro analgesia):ti,ab,kw or (muscle NEAR/2 stimulat*):ti,ab,kw or (neuromodulation or (neuro modulation) or neurostimulation or (neuro stimulation)):ti,ab,kw or (interferential):ti,ab,kw | 17,526 |
| #46 | (#13 AND #45) | 76 |
| #47 | MeSH descriptor Biofeedback (Psychology) explode all trees | 710 |
| #48 | (biofeedback):ti,ab,kw | 1348 |
| #49 | (#47 OR #48) | 1348 |
| #50 | (#13 AND #49) | 6 |
| #51 | MeSH descriptor Cryotherapy explode all trees | 866 |
| #52 | MeSH descriptor Ice explode all trees | 63 |
| #53 | MeSH descriptor Diathermy explode all trees | 620 |
| #54 | MeSH descriptor Hyperthermia, Induced explode all trees | 981 |
| #55 | MeSH descriptor Hot Temperature explode all trees | 1079 |
| #56 | (cold or ice or heat or hot) NEXT (pack* or therap* or treat*):ti,ab,kw or (thermograph* or thermotherap* or (thermo therap*) or hypertherm* or (hyper therm*)):ti,ab,kw or (diatherm* or cryotherap* or (cryo therap*)):ti,ab,kw | 2278 |
| #57 | (#51 OR #52 OR #53 OR #54 OR #55 OR #56) | 4273 |
| #58 | (#13 AND #57) | 51 |
| #59 | MeSH descriptor Laser Therapy explode all trees | 2372 |
| #60 | MeSH descriptor Ultrasonic Therapy explode all trees | 523 |
| #61 | MeSH descriptor Ultrasonography, Interventional explode all trees | 529 |
| #62 | (ultrasound):ti,ab,kw or (electrotherapeutic NEAXT (intervention* or treat*)):ti,ab,kw | 5557 |
| #63 | (#59 OR #60 OR #61 OR #62) | 8267 |
| #64 | (#13 AND #63) | 58 |
| #65 | MeSH descriptor Magnetic Field Therapy explode all trees | 369 |
| #66 | “pulsed electromagnetic field therapy”:ti,ab,kw or (electromagnetic* or magnetic*) NEAR/3 field*:ti,ab,kw or (biomagnetic* or (bio magnetic*) or (pulsed signal)):ti,ab,kw or (PEMF):ti,ab,kw | 793 |
| #67 | (#65 OR #66) | 1121 |
| #68 | (#13 AND #67) | 12 |
| #69 | MeSH descriptor Nerve Block explode all trees | 1994 |
| #70 | MeSH descriptor Neuromuscular Blockade explode all trees | 266 |
| #71 | (nerve NEAR/2 block*):ti,ab,kw | 2595 |
| #72 | (#69 OR #70 OR #71) | 2854 |
| #73 | (#13 AND #72) | 49 |
| #74 | MeSH descriptor Acupuncture Therapy explode all trees | 1830 |
| #75 | (acupuncture*):ti,ab,kw or (electroacupuncture* or (electro acupuncture*)):ti,ab,kw or (osteopuncture* or (osteo puncture*)):ti,ab,kw or (perioste* NEAR/3 (stimulat* or therap* or needling)):ti,ab,kw | 4213 |
| #76 | (#74 OR #75) | 4239 |
| #77 | (#13 AND #76) | 58 |
| #78 | MeSH descriptor Massage explode all trees | 595 |
| #79 | (massag* or acupressure or shiatsu or shiatzu or (zhi ya) or (chih ya)):ti,ab,kw | 1277 |
| #80 | (#78 OR #79) | 1277 |
| #81 | (#13 AND #80) | 20 |
| #82 | (rehabilitat* NEAR/2 (program* or protocol*)):ti,ab,kw or (watch* NEAR/2 wait*) or (conservative NEAR/2 treat*):ti,ab,kw or (management NEAR/2 (decision* or option* or choice*)):ti,ab,kw | 2974 |
| #83 | (#13 AND #82) | 34 |
| #84 | (#20 OR #25 OR #33 OR #40 OR #44 OR #46 OR #50 OR #58 OR #64 OR #68 OR #73 OR #77 OR #81 OR #83) | 588 |
Clinicaltrials.gov
Interface: http://clinicaltrials.gov/
Search date: 31 March 2010
Strategy: two separate searches carried out, one for ‘frozen shoulder’ and a second for ‘adhesive capsulitis’
Records identified: 29
Cochrane Database of Systematic Reviews (CDSR)
Interface: Cochrane Library Issue 3 2010
Search date: 23 March 2010
Strategy: as CENTRAL strategy above
Records identified: 17
Conference Proceedings Citation Index: Science
Interface: Web of Science (ISI) via Web of Knowledge
Search date: 30 March 2010
Search strategy: as below
Records identified: 82
| # 42 | 82 |
#41 OR #40 OR #39 OR #38 OR #37 OR #36 OR #35 OR #34 OR #33 OR #32 OR #31 OR #30 OR #29 OR #28 OR #27 OR #26 OR #25 OR #24 OR #23 OR #22 Databases=CPCI-S Timespan=1966–2010 |
| # 41 | 1 |
#21 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 40 | 7 |
#20 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 39 | 2 |
#19 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 38 | 11 |
#18 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 37 | 0 |
#17 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 36 | 0 |
#16 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 35 | 5 |
#15 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 34 | 0 |
#14 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 33 | 0 |
#13 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 32 | 0 |
#12 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 31 | 2 |
#11 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 30 | 0 |
#10 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 29 | 0 |
#9 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 28 | 12 |
#8 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 27 | 7 |
#7 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 26 | 35 |
#6 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 25 | 0 |
#5 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 24 | 13 |
#4 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 23 | 12 |
#3 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 22 | 1 |
#2 AND #1 Databases=CPCI-S Timespan=1966–2010 |
| # 21 | 2325 |
Topic=(“management decision*” or “management option*” or “management choice*”) Databases=CPCI-S Timespan=1966–2010 |
| # 20 | 1814 |
Topic=(“rehabilitat* program*” or “rehabilitat* protocol*” or “watch* wait*” or “conservative treat*”) Databases=CPCI-S Timespan=1966–2010 |
| # 19 | 427 |
Topic=(“perioste* stimulat*” or “perioste* therap*” or “perioste* needling” or massag* or acupressure or shiatsu or shiatzu or “zhi ya” or “chih ya”) Databases=CPCI-S Timespan=1966–2010 |
| # 18 | 1345 |
Topic=(“nerve block” or “neuromuscular blockade” or acupuncture or electroacupuncture or “electro acupuncture” or osteopuncture or “osteo puncture”) Databases=CPCI-S Timespan=1966–2010 |
| # 17 | 8 |
Topic=(“electromagnetic field therapy” or “Biomagnetic therapy”) Databases=CPCI-S Timespan=1966–2010 |
| # 16 | 18 |
Topic=(“Interventional ultrasonography” or “electrotherapeutic intervention*” or “electrotherapeutic treat*” or “magnetic field therapy”) Databases=CPCI-S Timespan=1966–2010 |
| # 15 | 16,084 |
Topic=(“heat pack*” or “heat therap*” or “heat treat*” or “hot pack*” or “heat therap*” or “Laser Therapy” or “ultrasonic therapy”) Databases=CPCI-S Timespan=1966–2010 |
| # 14 | 895 |
Topic=(diatherm* or cryotherap* or “cryo therap*” or “cold pack*” or “cold therap*” or “cold treat*” or “ice pack*” or “ice therap*” or “ice treat*”) Databases=CPCI-S Timespan=1966–2010 |
| # 13 | 895 |
Topic=(diatherm* or cryotherap* or “cryo therap*” or “cold pack*” or “cold therap*” or “cold treat*” or “ice pack*” or “ice therap*” or “ice treat*”) Databases=CPCI-S Timespan=1966–2010 |
| # 12 | 895 |
Topic=(diatherm* or cryotherap* or “cryo therap*” or “cold pack*” or “cold therap*” or “cold treat*” or “ice pack*” or “ice therap*” or “ice treat*”) Databases=CPCI-S Timespan=1966–2010 |
| # 11 | 6798 |
Topic=(biofeedback or thermograph* or thermotherap* or “thermo therap*” or hypertherm* or “hyper therm*”) Databases=CPCI-S Timespan=1966–2010 |
| # 10 | 360 |
Topic=(neurostimulation or “neuro stimulation” or interferential) Databases=CPCI-S Timespan=1966–2010 |
| # 9 | 574 |
Topic=(“cutaneous electrostimulat*”or electroanalgesia or “electro analgesia” or “muscle stimulat*” or neuromodulation or “neuro modulation”) Databases=CPCI-S Timespan=1966–2010 |
| # 8 | 11,210 |
Topic=(TENS or ALTENS or “electric* stimulat*” or “transcutaneous stimulat*”or “transdermal electrostimulat*”) Databases=CPCI-S Timespan=1966–2010 |
| # 7 | 18,081 |
Topic=(chiropractic* or osteopath* or manipulation or MUA) Databases=CPCI-S Timespan=1966–2010 |
| # 6 | 7995 |
Topic=(physiotherapy or physiotherapies or “physical therap*” or “manual therap*” or “passive motion” or “passive movement” or “muscle stretching” or “muscle stretches” or “muscle stretch” or mobilisation or mobilization or “exercis* program*” or “exercis* intervention*” or “exercis* train*” or “exercis* prescription*” or “exercis* prescrib*” or “exercis* therap*” or “therapeutic exercis*” or “home exercis*” or “supervis* exercis*” or “pendular exercis*” or “pendulum exercis*” or “isokinetic exercis*” or “resist* exercise*”) Databases=CPCI-S Timespan=1966–2010 |
| # 5 | 2 |
Topic=(“subacromial inject*” or “acromioclavicular inject*” or “glenohumeral inject*” or “extra articular inject*” or “extraarticular inject*” or “shoulder* inject*”) Databases=CPCI-S Timespan=1966–2010 |
| # 4 | 5583 |
Topic=(injections or “intraarticular injection*” or “intra-articular injection*” or “intra articular injection*” or “bursa* inject*” or “intrabursa* inject*” or “intra bursa* inject*” or “periartic* inject*” or “peri artic* inject*”) Databases=CPCI-S Timespan=1966–2010 |
| # 3 | 656 |
Topic=(Arthroscopy or “arthroscop* releas*” or “arthroscop* decompress*” or “arthroscop* capsulotom*” or “capsular releas*” or “interventional microadhesiolysis” or capsulotomy) Databases=CPCI-S Timespan=1966–2010 |
| # 2 | 2 |
Topic=(“arthrograph* distension*” or “arthograph* disten” or “arthrogram* distension*” or “arthogram* distention*” or “glenohumeral distension*” or “glenohumeral distention*” or arthography) Databases=CPCI-S Timespan=1966–2010 |
| # 1 | 275 |
Topic=(“frozen shoulder*” or “stiff* shoulder*” or “adhesive capsulitis” or “shoulder bursitis” or periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis or “shoulder pain*” or “pain* shoulder” or “shoulder complaint*” or “shoulder* impinge*” or “subacromial impingement syndrome”) Databases=CPCI-S Timespan=1966–2010 |
Database of Abstracts of Reviews of Effects (DARE)
Interface: Cochrane Library Issue 3 2010
Search date: 23 March 2010
Strategy: as CENTRAL strategy above
Records identified: 17
EMBASE
Interface: OvidSP
Search date: 22 March 2010
Strategy: as below
Records identified: 3785
An update search carried out on 5 January 2011 identified a further 521 records.
Database: EMBASE <1980 to 2010 Week 11>
-
humeroscapular periarthritis/(766)
-
(frozen adj6 shoulder$).ti. (215)
-
(stiff$adj3 shoulder$).ti. (73)
-
(adhesive adj (capsulitis or capsulitides)).ti. (161)
-
((bursitis or bursitides) adj6 shoulder$).ti. (17)
-
((capsulitis or capsulitides) adj6 shoulder$).ti. (86)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ti. (70)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 (1071)
-
(frozen adj6 shoulder$).ab. (440)
-
(stiff$adj3 shoulder$).ab. (313)
-
exp bursitis/(1723)
-
(adhesive adj (capsulitis or capsulitides)).ab. (279)
-
((bursitis or bursitides) adj6 shoulder$).ab. (51)
-
((capsulitis or capsulitides) adj6 shoulder$).ti,ab. (183)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ab. (98)
-
shoulder pain/(5140)
-
(shoulder$adj3 (pain or pains or painful or complain$)).ab. (3349)
-
Shoulder Impingement Syndrome/(680)
-
(shoulder$adj6 impinge$).ab. (364)
-
subacromial impingement syndrome.ab. (96)
-
9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 (9277)
-
Arthrography/(2891)
-
(arthrograph$adj6 (distension$or distention$)).ti,ab. (32)
-
(arthrogram$adj6 (distension$or distention$)).ti,ab. (1)
-
(glenohumeral adj6 (distension$or distention$)).ti,ab. (7)
-
Dilatation/(107)
-
(dilatation or hydrodilat$).ti,ab. (28,266)
-
or/22–27 (31,214)
-
21 and 28 (368)
-
Shoulder arthroscopy/(361)
-
(arthroscop$adj6 (releas$or decompress$or capsulotom$)).ti,ab. (535)
-
((capsular adj2 releas$) or capsulotomy or interventional microadhesiolysis).ti,ab. (1787)
-
or/30–32 (2581)
-
21 and 33 (280)
-
injection/(11,806)
-
35 and 21 (61)
-
((bursa$or intrabursa$or intra bursa$or periartic$or peri artic$or intraartic$or intra artic$) adj3 inject$).ti,ab. (2681)
-
37 and 21 (141)
-
((subacromial or acromioclavicular or glenohumeral) adj3 inject$).ti,ab. (137)
-
((extra articular or extraarticular or shoulder$) adj3 inject$).ti,ab. (136)
-
36 or 38 or 39 or 40 (400)
-
exp physiotherapy/(24,873)
-
(physiotherapies or physical therap$or manual therap$).ti,ab. (8968)
-
(passive adj (motion or movement)).ti,ab. (1271)
-
CPM.ti,ab. (3193)
-
muscle stretching/or stretching exercise/(3543)
-
(stretching or stretches).ti,ab. (12,491)
-
(mobilisation or mobilization).ti,ab. (28,856)
-
(exercise$adj2 (program$or strength$or intervention$or training or prescription$or prescrib$)).ti,ab. (14,622)
-
(exercise$adj2 (therap$or therapeutic)).ti,ab. (2185)
-
((home or supervis$) adj2 exercis$).ti,ab. (1554)
-
((pendular or pendulum) adj exercis$).ti,ab. (16)
-
((isokinetic or resist$) adj2 exercise$).ti,ab. (2223)
-
or/42–53 (91,822)
-
21 and 54 (1248)
-
orthopedic manipulation/(222)
-
chiropractic$.ti,ab. (1998)
-
osteopath$.ti,ab. (2213)
-
(manipulat$adj3 (anesthesia or anaesthesia or anesthetic$or anaesthetic$)).ti,ab. (284)
-
MUA.ti,ab. (3446)
-
56 or 57 or 58 or 59 or 60 (7965)
-
21 and 61 (109)
-
(TENS or ALTENS).ti,ab. (4441)
-
((electric$adj2 stimulat$) or (transcutaneous adj2 stimulat$) or (transdermal adj2 electrostimulat$) or (cutaneous adj2 electrostimulat$) or electroanalgesia or electro analgesia).ti,ab. (36,123)
-
(muscle adj2 stimulat$).ti,ab. (3878)
-
(neuromodulation or neuro modulation or neurostimulation or neuro stimulation).ti,ab. (2484)
-
interferential.ti,ab. (208)
-
or/63–67 (45,043)
-
21 and 68 (108)
-
biofeedback.ti,ab. (3532)
-
21 and 70 (7)
-
cryotherapy/(7404)
-
ice/(2468)
-
diathermy/(2178)
-
hyperthermia/(11,961)
-
thermograph$.ti,ab. (2186)
-
((cold or ice or heat or hot) adj (pack$or therap$or treat$)).ti,ab. (7854)
-
(thermotherap$or thermo therap$or hypertherm$or hyper therm$or diatherm$or cryotherap$or cryo therap$).ti,ab. (24,875)
-
or/72–78 (45,638)
-
21 and 79 (140)
-
low level laser Therapy/(4456)
-
ultrasound/(42,405)
-
echography/(111,055)
-
(electrotherapeutic adj (intervention$or treat$)).ti,ab. (4)
-
or/81–84 (154,738)
-
21 and 85 (717)
-
magnetotherapy/(317)
-
pulsed electromagnetic field therapy.ti,ab. (26)
-
((electromagnetic$or magnetic$) adj3 field$).ti,ab. (14,142)
-
(biomagnetic$or bio magnetic$or pulsed signal).ti,ab. (332)
-
PEMF.ti,ab. (223)
-
or/87–91 (14,579)
-
21 and 92 (23)
-
nerve block/(9791)
-
neuromuscular blocking/(6800)
-
nerve block$.ti,ab. (4505)
-
or/94–96 (17,905)
-
21 and 97 (141)
-
exp Acupuncture/(14,373)
-
acupuncture$.ti,ab. (8902)
-
(electroacupuncture$or electro acupuncture$).ti,ab. (1536)
-
(osteopuncture$or osteo puncture$).ti,ab. (2)
-
(perioste$adj3 (stimulat$or therap$or needling)).ti,ab. (114)
-
or/99–103 (15,019)
-
21 and 104 (166)
-
massage/(4230)
-
(massag$or acupressure or shiatsu or shiatzu or zhi ya or chih ya).ti,ab. (4120)
-
106 or 107 (6434)
-
21 and 108 (86)
-
(rehabilitat$adj2 (program$or protocol$)).ti,ab. (8634)
-
21 and 110 (143)
-
((watch$adj2 wait$) or (conservative$adj2 treat$)).ti,ab. (20,913)
-
21 and 112 (267)
-
(management adj2 (decision$or option$or choice$)).ti,ab. (6144)
-
21 and 114 (10)
-
111 or 113 or 115 (404)
-
8 or 29 or 34 or 41 or 55 or 62 or 69 or 71 or 80 or 86 or 93 or 98 or 105 or 109 or 116 (3785)
Health Management Information Consortium (HMIC)
Interface: OvidSP
Search date: 25 March 2010
Strategy: as below
Records identified: 8
Database: HMIC Health Management Information Consortium <January 2010>
-
(frozen adj6 shoulder$).ti,ab. (0)
-
(stiff$adj3 shoulder$).ti,ab. (4)
-
(adhesive adj (capsulitis or capsulitides)).ti,ab. (0)
-
((bursitis or bursitides) adj6 shoulder$).ti,ab. (0)
-
((capsulitis or capsulitides) adj6 shoulder$).ti,ab. (0)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ti,ab. (0)
-
(shoulder$adj3 (pain or pains or painful or complain$)).ti,ab. (34)
-
(shoulder$adj6 impinge$).ti,ab. (0)
-
subacromial impingement syndrome.ti,ab. (0)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 (35)
-
(arthrograph$adj6 (distension$or distention$)).ti,ab. (0)
-
(arthrogram$adj6 (distension$or distention$)).ti,ab. (0)
-
(glenohumeral adj6 (distension$or distention$)).ti,ab. (0)
-
(dilatation or hydrodilat$).ti,ab. (20)
-
(arthroscop$adj6 (releas$or decompress$or capsulotom$)).ti,ab. (0)
-
((capsular adj2 releas$) or interventional microadhesiolysis or capsulotomy).ti,ab. (0)
-
((bursa$or intrabursa$or intra bursa$or periartic$or peri artic$or intraartic$or intra artic$) adj3 inject$).ti,ab. (2)
-
((subacromial or acromioclavicular or glenohumeral) adj3 inject$).ti,ab. (0)
-
((extra articular or extraarticular or shoulder$) adj3 inject$).ti,ab. (0)
-
(physiotherapy or physiotherapies or physical therap$or manual therap$).ti,ab. (732)
-
(passive adj (motion or movement)).ti,ab. (0)
-
CPM.ti,ab. (6)
-
(stretching or stretches).ti,ab. (42)
-
(mobilisation or mobilization).ti,ab. (97)
-
(exercise$adj2 (program$or strength$or intervention$or training or prescription$or prescrib$)).ti,ab. (229)
-
(exercise$adj2 (therap$or therapeutic)).ti,ab. (27)
-
((home or supervis$) adj2 exercis$).ti,ab. (40)
-
((pendular or pendulum) adj exercis$).ti,ab. (0)
-
((isokinetic or resist$) adj2 exercise$).ti,ab. (11)
-
chiropractic$.ti,ab. (55)
-
osteopath$.ti,ab. (80)
-
(manipulat$adj3 (anesthesia or anaesthesia or anesthetic$or anaesthetic$)).ti,ab. (2)
-
MUA.ti,ab. (0)
-
(TENS or ALTENS).ti,ab. (27)
-
((electric$adj2 stimulat$) or (transcutaneous adj2 stimulat$) or (transdermal adj2 electrostimulat$) or (cutaneous adj2 electrostimulat$) or electroanalgesia or electro analgesia).ti,ab. (9)
-
(muscle adj2 stimulat$).ti,ab. (5)
-
(neuromodulation or neuro modulation or neurostimulation or neuro stimulation).ti,ab. (1)
-
interferential.ti,ab. (5)
-
biofeedback.ti,ab. (5)
-
((cold or ice or heat or hot) adj (pack$or therap$or treat$)).ti,ab. (18)
-
(thermograph$or thermotherap$or thermo therap$or hypertherm$or hyper therm$or diatherm$or cryotherap$or cryo therap$).ti,ab. (90)
-
((laser or ultrasonic) adj2 (therap$or treat$)).ti,ab. (25)
-
ultrasound.ti,ab. (260)
-
(electrotherapeutic adj (intervention$or treat$)).ti,ab. (0)
-
pulsed electromagnetic field therapy.ti,ab. (0)
-
((electromagnetic$or magnetic$) adj3 field$).ti,ab. (121)
-
(biomagnetic$or bio magnetic$or pulsed signal).ti,ab. (0)
-
PEMF.ti,ab. (0)
-
(nerve adj2 block$).ti,ab. (2)
-
neuromuscular block$.ti,ab. (3)
-
acupuncture$.ti,ab. (118)
-
(electroacupuncture$or electro acupuncture$).ti,ab. (1)
-
(osteopuncture$or osteo puncture$).ti,ab. (0)
-
(perioste$adj3 (stimulat$or therap$or needling)).ti,ab. (0)
-
(massag$or acupressure or shiatsu or shiatzu or zhi ya or chih ya).ti,ab. (67)
-
(rehabilitat$adj2 (program$or protocol$)).ti,ab. (182)
-
((watch$adj2 wait$) or (conservative adj2 treat$)).ti,ab. (30)
-
(management adj2 (decision$or option$or choice$)).ti,ab. (353)
-
or/11–58 (2475)
-
10 and 59 (8)
-
limit 60 to yr=“1966 -Current” (8)
-
from 61 keep 1–8 (8)
Health Technology Assessment (HTA) database
Interface: Cochrane Library Issue 3 2010
Search date: 23 March 2010
Strategy: as CENTRAL strategy above
Records identified: 3
Latin American and Caribbean Health Sciences Literature (LILACS)
Search date: 26 March 2010
Strategy: shoulder joint [Subject descriptor] or adhesive capsulitis [Words] or bursitis or periarthritis [Words]
Records identified: 298
Manual, Alternative and Natural Therapy (MANTIS)
Interface: Dialog Classic
Search date: 30 March 2010
Strategy: as below
Records identified: 189
? s frozen (6W) shoulder?/ti,ab,de
1317 FROZEN/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
S1 172 FROZEN (6W) SHOULDER?/TI,AB,DE
? s stiff? (3W) shoulder?/ti,ab,de
3859 STIFF?/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
S2 33 STIFF? (3W) SHOULDER?/TI,AB,DE
? s adhesive (W) (capsulitis or capsulitides)/ti,ab,de
437 ADHESIVE/TI,AB,DE
149 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
S3 115 ADHESIVE (W) (CAPSULITIS OR CAPSULITIDES)/TI,AB,DE
? s (bursitis or bursitides) (6W) shoulder?/ti,ab,de
373 BURSITIS/TI,AB,DE
0 BURSITIDES/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
S4 52 (BURSITIS OR BURSITIDES) (6W) SHOULDER?/TI,AB,DE
? s (capsulitis or capsulitides) (6W) shoulder?/ti,ab,de
149 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
S5 41 (CAPSULITIS OR CAPSULITIDES) (6W) SHOULDER?/TI,AB,DE
? s (periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) (6W) shoulder?/ti,ab,de
54 PERIARTHRITIS/TI,AB,DE
0 PERI-ARTHRITIS/TI,AB,DE
0 PERIARTHRITIDES/TI,AB,DE
0 PERI-ARTHRITIDES/TI,AB,DE
0 PERI-CAPSULITIS/TI,AB,DE
3 PERICAPSULITIS/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
S6 29 (PERIARTHRITIS OR PERI-ARTHRITIS OR PERIARTHRITIDES OR
PERI-ARTHRITIDES OR PERI-CAPSULITIS OR PERICAPSULITIS)
(6W) SHOULDER?/TI,AB,DE
? s shoulder? (3W) (pain or pains or painful or complain?)/ti,ab,de
6053 SHOULDER?/TI,AB,DE
40861 PAIN/TI,AB,DE
400 PAINS/TI,AB,DE
3210 PAINFUL/TI,AB,DE
4803 COMPLAIN?/TI,AB,DE
S7 1170 SHOULDER? (3W) (PAIN OR PAINS OR PAINFUL OR
COMPLAIN?)/TI,AB,DE
? s shoulder? (6W) impinge?/ti,ab,de
6053 SHOULDER?/TI,AB,DE
994 IMPINGE?/TI,AB,DE
S8 192 SHOULDER? (6W) IMPINGE?/TI,AB,DE
? s subacromial (W) impingement (W) syndrome/ti,ab,de
235 SUBACROMIAL/TI,AB,DE
934 IMPINGEMENT/TI,AB,DE
15975 SYNDROME/TI,AB,DE
S9 28 SUBACROMIAL (W) IMPINGEMENT (W) SYNDROME/TI,AB,DE
? s s1:s9
S10 1532 S1:S9
? s arthrograph? (6W) (distension? or distention?)/ti,ab,de
365 ARTHROGRAPH?/TI,AB,DE
204 DISTENSION?/TI,AB,DE
107 DISTENTION?/TI,AB,DE
S11 6 ARTHROGRAPH? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s arthrogram? (6W) (distension? or distention?)/ti,ab,de
126 ARTHROGRAM?/TI,AB,DE
204 DISTENSION?/TI,AB,DE
107 DISTENTION?/TI,AB,DE
S12 0 ARTHROGRAM? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s glenohumeral (6W) (distension? or distention?)/ti,ab,de
758 GLENOHUMERAL/TI,AB,DE
204 DISTENSION?/TI,AB,DE
107 DISTENTION?/TI,AB,DE
S13 1 GLENOHUMERAL (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s dilatation or hydrodilat?/ti,ab,de
301 DILATATION
4 HYDRODILAT?/TI,AB,DE
S14 304 DILATATION OR HYDRODILAT?/TI,AB,DE
? s s11:s14
S15 310 S11:S14
? s s10 and s15
1532 S10
310 S15
S16 11 S10 AND S15
? s arthroscop? (6W) (releas? or decompress? or capsulotom?)/ti,ab,de
2147 ARTHROSCOP?/TI,AB,DE
7144 RELEAS?/TI,AB,DE
2479 DECOMPRESS?/TI,AB,DE
52 CAPSULOTOM?/TI,AB,DE
S17 96 ARTHROSCOP? (6W) (RELEAS? OR DECOMPRESS? OR
CAPSULOTOM?)/TI,AB,DE
? s capsular (2W) releas? or interventional (W) microadhesiolysis or capsulotomy/ti,ab,de
540 CAPSULAR
7144 RELEAS?
34 CAPSULAR(2W)RELEAS?
352 INTERVENTIONAL
1 MICROADHESIOLYSIS
1 INTERVENTIONAL(W)MICROADHESIOLYSIS
42 CAPSULOTOMY/TI,AB,DE
S18 77 CAPSULAR (2W) RELEAS? OR INTERVENTIONAL (W)
MICROADHESIOLYSIS OR CAPSULOTOMY/TI,AB,DE
? s s17:s18
S19 164 S17:S18
? s s10 and s19
1532 S10
164 S19
S20 32 S10 AND S19
? s injection?/ti,ab,de
>>>Term “DE..” is not defined in file 91 and is ignored
S21 7403 INJECTION?/TI,AB,DE..
? s s10 and s21
1532 S10
7403 S21
S22 94 S10 AND S21
? s (bursa? or intrabursa? or intra (W) bursa? or periartic? or peri (W) artic? or intraartic? or intra (W) artic?) (3W) inject?/ti,ab,de
239 BURSA?/TI,AB,DE
2 INTRABURSA?/TI,AB,DE
3027 INTRA/TI,AB,DE
239 BURSA?/TI,AB,DE
1 INTRA/TI,AB,DE(W)BURSA?/TI,AB,DE
225 PERIARTIC?/TI,AB,DE
336 PERI/TI,AB,DE
23756 ARTIC?/TI,AB,DE
26 PERI/TI,AB,DE(W)ARTIC?/TI,AB,DE
592 INTRAARTIC?/TI,AB,DE
3027 INTRA/TI,AB,DE
23756 ARTIC?/TI,AB,DE
867 INTRA/TI,AB,DE(W)ARTIC?/TI,AB,DE
9111 INJECT?/TI,AB,DE
S23 335 (BURSA? OR INTRABURSA? OR INTRA (W) BURSA? OR PERIARTIC?
OR PERI (W) ARTIC? OR INTRAARTIC? OR INTRA (W) ARTIC?)
(3W) INJECT?/TI,AB,DE
? s s10 and s23
1532 S10
335 S23
S24 23 S10 AND S23
? s (subacromial or acromioclavicular or glenohumeral) (3W) inject?/ti,ab,de
235 SUBACROMIAL/TI,AB,DE
282 ACROMIOCLAVICULAR/TI,AB,DE
758 GLENOHUMERAL/TI,AB,DE
9111 INJECT?/TI,AB,DE
S25 24 (SUBACROMIAL OR ACROMIOCLAVICULAR OR GLENOHUMERAL) (3W)
INJECT?/TI,AB,DE
? s (extra (W) articular or extraarticular or shoulder?) (3W) inject?/ti,ab,de
1137 EXTRA/TI,AB,DE
6439 ARTICULAR/TI,AB,DE
227 EXTRA/TI,AB,DE(W)ARTICULAR/TI,AB,DE
143 EXTRAARTICULAR/TI,AB,DE
6053 SHOULDER?/TI,AB,DE
9111 INJECT?/TI,AB,DE
S26 17 (EXTRA (W) ARTICULAR OR EXTRAARTICULAR OR SHOULDER?) (3W)
INJECT?/TI,AB,DE
? s s22 or s24 or s25 or s26
94 S22
23 S24
24 S25
17 S26
S27 113 S22 OR S24 OR S25 OR S26
? s physiotherapy or physiotherapies or physical (W) therap? or manual(W) therap?/ti,ab,de
1824 PHYSIOTHERAPY
1 PHYSIOTHERAPIES
27577 PHYSICAL
58031 THERAP?
5273 PHYSICAL(W)THERAP?
3870 MANUAL/TI,AB,DE
58031 THERAP?/TI,AB,DE
662 MANUAL/TI,AB,DE(W)THERAP?/TI,AB,DE
S28 7178 PHYSIOTHERAPY OR PHYSIOTHERAPIES OR PHYSICAL (W) THERAP?
ORy MANUAL(W) THERAP?/TI,AB,DE
? s passive (W) (motion or movement)/ti,ab,de
3275 PASSIVE/TI,AB,DE
12870 MOTION/TI,AB,DE
8522 MOVEMENT/TI,AB,DE
S29 395 PASSIVE (W) (MOTION OR MOVEMENT)/TI,AB,DE
? s CPM/ti,ab,de
S30 117 CPM/TI,AB,DE
? s (stretching or stretches)/ti,ab,de
1453 STRETCHING/TI,AB,DE
200 STRETCHES/TI,AB,DE
S31 1555 (STRETCHING OR STRETCHES)/TI,AB,DE
? s (mobilisation or mobilization)/ti,ab,de
248 MOBILISATION/TI,AB,DE
1532 MOBILIZATION/TI,AB,DE
S32 1749 (MOBILISATION OR MOBILIZATION)/TI,AB,DE
? s exercise? (2W) (program? or strength? or intervention? or training or prescription? or prescrib?)/ti,ab,de
21844 EXERCISE?/TI,AB,DE
21676 PROGRAM?/TI,AB,DE
13678 STRENGTH?/TI,AB,DE
20015 INTERVENTION?/TI,AB,DE
15534 TRAINING/TI,AB,DE
2502 PRESCRIPTION?/TI,AB,DE
2943 PRESCRIB?/TI,AB,DE
S33 3545 EXERCISE? (2W) (PROGRAM? OR STRENGTH? OR INTERVENTION? OR
TRAINING OR PRESCRIPTION? OR PRESCRIB?)/TI,AB,DE
? s exercise? (2W) (therap? or therapeutic)/ti,ab,de
21844 EXERCISE?/TI,AB,DE
58031 THERAP?/TI,AB,DE
15559 THERAPEUTIC/TI,AB,DE
S34 1871 EXERCISE? (2W) (THERAP? OR THERAPEUTIC)/TI,AB,DE
? s (home or supervis?) (2W) exercis?/ti,ab,de
5568 HOME/TI,AB,DE
1564 SUPERVIS?/TI,AB,DE
21993 EXERCIS?/TI,AB,DE
S35 448 (HOME OR SUPERVIS?) (2W) EXERCIS?/TI,AB,DE
? s (pendular or pendulum) (W) exercis?/ti,ab,de
30 PENDULAR/TI,AB,DE
209 PENDULUM/TI,AB,DE
21993 EXERCIS?/TI,AB,DE
S36 5 (PENDULAR OR PENDULUM) (W) EXERCIS?/TI,AB,DE
? s (isokinetic or resist?) (2W) exercise?/ti,ab,de
1921 ISOKINETIC/TI,AB,DE
7695 RESIST?/TI,AB,DE
21844 EXERCISE?/TI,AB,DE
S37 720 (ISOKINETIC OR RESIST?) (2W) EXERCISE?/TI,AB,DE
? s s28:s37
S38 14795 S28:S37
? s s10 and s38
1532 S10
14795 S38
S39 313 S10 AND S38
? s chiropractic?/ti,ab,de
S40 18676 CHIROPRACTIC?/TI,AB,DE
? s osteopath?/ti,ab,de
S41 8326 OSTEOPATH?/TI,AB,DE
? s manipulat? (3W) (anesthesia or anaesthesia or anesthetic? or anaesthetic? or musculoskeletal)/ti,ab,de
8789 MANIPULAT?/TI,AB,DE
1764 ANESTHESIA/TI,AB,DE
384 ANAESTHESIA/TI,AB,DE
860 ANESTHETIC?/TI,AB,DE
293 ANAESTHETIC?/TI,AB,DE
6860 MUSCULOSKELETAL/TI,AB,DE
S42 237 MANIPULAT? (3W) (ANESTHESIA OR ANAESTHESIA OR ANESTHETIC?
OR ANAESTHETIC? OR MUSCULOSKELETAL)/TI,AB,DE
? s MUA/ti,ab,de
S43 41 MUA/TI,AB,DE
? s s40:s43
S44 26435 S40:S43
? s s10 and s44
1532 S10
26435 S44
S45 168 S10 AND S44
? s (TENS or ALTENS)/ti,ab,de
367 TENS/TI,AB,DE
4 ALTENS/TI,AB,DE
S46 369 (TENS OR ALTENS)/TI,AB,DE
? s electric? (2W) stimulat? or transcutaneous (2W) stimulat? or transdermal (2W) electrostimulat? or cutaneous (2W) electrostimulat? or electroanalgesia or electro(W)analgesia/ti,ab,de
5115 ELECTRIC?
13078 STIMULAT?
2781 ELECTRIC?(2W)STIMULAT?
716 TRANSCUTANEOUS
13078 STIMULAT?
592 TRANSCUTANEOUS(2W)STIMULAT?
184 TRANSDERMAL
116 ELECTROSTIMULAT?
0 TRANSDERMAL(2W)ELECTROSTIMULAT?
1545 CUTANEOUS
116 ELECTROSTIMULAT?
1 CUTANEOUS(2W)ELECTROSTIMULAT?
7 ELECTROANALGESIA
565 ELECTRO/TI,AB,DE
1688 ANALGESIA/TI,AB,DE
2 ELECTRO/TI,AB,DE(W)ANALGESIA/TI,AB,DE
S47 2821 ELECTRIC? (2W) STIMULAT? OR TRANSCUTANEOUS (2W) STIMULAT?
OR TRANSDERMAL (2W) ELECTROSTIMULAT? OR CUTANEOUS (2W)
ELECTROSTIMULAT? OR ELECTROANALGESIA OR
ELECTRO(W)ANALGESIA/TI,AB,DE
? s muscle (2W) stimulat?/ti,ab,de
25048 MUSCLE/TI,AB,DE
13078 STIMULAT?/TI,AB,DE
S48 317 MUSCLE (2W) STIMULAT?/TI,AB,DE
? s neuromodulation or neuro (W) modulation or neurostimulation or neuro (W) stimulation/ti,ab,de
60 NEUROMODULATION
492 NEURO
1698 MODULATION
1 NEURO(W)MODULATION
47 NEUROSTIMULATION
492 NEURO/TI,AB,DE
8205 STIMULATION/TI,AB,DE
0 NEURO/TI,AB,DE(W)STIMULATION/TI,AB,DE
S49 102 NEUROMODULATION OR NEURO (W) MODULATION OR
NEUROSTIMULATION OR NEURO (W) STIMULATION/TI,AB,DE
? s interferential/ti,ab,de
S50 76 INTERFERENTIAL/TI,AB,DE
? s s46:s50
S51 3178 S46:S50
? s s10 and s51
1532 S10
3178 S51
S52 44 S10 AND S51
? s biofeedback/ti,ab,de
S53 1148 BIOFEEDBACK/TI,AB,DE
? s s10 and s53
1532 S10
1148 S53
S54 2 S10 AND S53
? s (cold or ice or heat or hot) (W) (pack? or therap? or treat?)/ti,ab,de
2091 COLD/TI,AB,DE
805 ICE/TI,AB,DE
2522 HEAT/TI,AB,DE
1323 HOT/TI,AB,DE
1446 PACK?/TI,AB,DE
58031 THERAP?/TI,AB,DE
79900 TREAT?/TI,AB,DE
S55 306 (COLD OR ICE OR HEAT OR HOT) (W) (PACK? OR THERAP? OR
TREAT?)/TI,AB,DE
? s thermograph? or thermotherap? or thermo (W) therap? or hypertherm? or hyper (W) therm? or diatherm? or cryotherap? or cryo(W) therap?/ti,ab,de
413 THERMOGRAPH?
45 THERMOTHERAP?
32 THERMO
58031 THERAP?
1 THERMO(W)THERAP?
328 HYPERTHERM?
243 HYPER
2786 THERM?
1 HYPER(W)THERM?
113 DIATHERM?
272 CRYOTHERAP?
24 CRYO/TI,AB,DE
58031 THERAP?/TI,AB,DE
0 CRYO/TI,AB,DE(W)THERAP?/TI,AB,DE
S56 1124 THERMOGRAPH? OR THERMOTHERAP? OR THERMO (W) THERAP? OR
HYPERTHERM? OR HYPER (W) THERM? OR DIATHERM? OR
CRYOTHERAP? OR CRYO(W) THERAP?/TI,AB,DE
? s s55:s56
S57 1368 S55:S56
? s s10 and s57
1532 S10
1368 S57
S58 22 S10 AND S57
? s laser (W) therapy/ti,ab,de
1300 LASER/TI,AB,DE
43530 THERAPY/TI,AB,DE
S59 145 LASER (W) THERAPY/TI,AB,DE
? s ultrasonic (W) therapy/ti,ab,de
482 ULTRASONIC/TI,AB,DE
43530 THERAPY/TI,AB,DE
S60 115 ULTRASONIC (W) THERAPY/TI,AB,DE
? s ultrasound/ti,ab,de
S61 2326 ULTRASOUND/TI,AB,DE
? s ultrasonography/ti,ab,de
S62 1004 ULTRASONOGRAPHY/TI,AB,DE
? s electrotherapeutic (W) (intervention? or treat?)/ti,ab,de
12 ELECTROTHERAPEUTIC/TI,AB,DE
20015 INTERVENTION?/TI,AB,DE
79900 TREAT?/TI,AB,DE
S63 3 ELECTROTHERAPEUTIC (W) (INTERVENTION? OR TREAT?)/TI,AB,DE
? s s59:s63
S64 3169 S59:S63
? s s10 and s64
1532 S10
3169 S64
S65 61 S10 AND S64
? s pulsed (W) electromagnetic (W) field (W) therapy/ti,ab,de
533 PULSED/TI,AB,DE
754 ELECTROMAGNETIC/TI,AB,DE
6784 FIELD/TI,AB,DE
43530 THERAPY/TI,AB,DE
S66 7 PULSED (W) ELECTROMAGNETIC (W) FIELD (W) THERAPY/TI,AB,DE
? s (electromagnetic? or magnetic?) (3W) field?/ti,ab,de
787 ELECTROMAGNETIC?/TI,AB,DE
6966 MAGNETIC?/TI,AB,DE
8099 FIELD?/TI,AB,DE
S67 572 (ELECTROMAGNETIC? OR MAGNETIC?) (3W) FIELD?/TI,AB,DE
? s biomagnetic? or bio (W) magnetic? or pulsed signal/ti,ab,de
10 BIOMAGNETIC?
392 BIO
6966 MAGNETIC?
2 BIO(W)MAGNETIC?
0 PULSED SIGNAL/TI,AB,DE
S68 12 BIOMAGNETIC? OR BIO (W) MAGNETIC? OR PULSED
SIGNAL/TI,AB,DE
? s PEMF/ti,ab,de
S69 42 PEMF/TI,AB,DE
? s s66:s69
S70 578 S66:S69
? s s10 and s70
1532 S10
578 S70
S71 2 S10 AND S70
? s neuromuscular (W) blockade/ti,ab,de
3130 NEUROMUSCULAR/TI,AB,DE
802 BLOCKADE/TI,AB,DE
S72 81 NEUROMUSCULAR (W) BLOCKADE/TI,AB,DE
? s nerve (2W) block?/ti,ab,de
13003 NERVE/TI,AB,DE
6218 BLOCK?/TI,AB,DE
S73 453 NERVE (2W) BLOCK?/TI,AB,DE
? s s72:s73
S74 533 S72:S73
? s s10 and s74
1532 S10
533 S74
S75 9 S10 AND S74
? s acupuncture?/ti,ab,de
S76 7224 ACUPUNCTURE?/TI,AB,DE
? s electroacupuncture? or electro (W) acupuncture?/ti,ab,de
996 ELECTROACUPUNCTURE?
565 ELECTRO/TI,AB,DE
7224 ACUPUNCTURE?/TI,AB,DE
303 ELECTRO/TI,AB,DE(W)ACUPUNCTURE?/TI,AB,DE
S77 1223 ELECTROACUPUNCTURE? OR ELECTRO (W) ACUPUNCTURE?/TI,AB,DE
? s osteopuncture? or osteo (W) puncture?/ti,ab,de
0 OSTEOPUNCTURE?
86 OSTEO/TI,AB,DE
587 PUNCTURE?/TI,AB,DE
0 OSTEO/TI,AB,DE(W)PUNCTURE?/TI,AB,DE
S78 0 OSTEOPUNCTURE? OR OSTEO (W) PUNCTURE?/TI,AB,DE
? s perioste? (3W) (stimulat? or therap? or needling)/ti,ab,de
455 PERIOSTE?/TI,AB,DE
13078 STIMULAT?/TI,AB,DE
58031 THERAP?/TI,AB,DE
657 NEEDLING/TI,AB,DE
S79 7 PERIOSTE? (3W) (STIMULAT? OR THERAP? OR
NEEDLING)/TI,AB,DE
? s s76:s79
S80 7685 S76:S79
? s s10 and s80
1532 S10
7685 S80
S81 88 S10 AND S80
? s massag? or acupressure or shiatsu or shiatzu or zhi (W) ya or chih (W) ya/ti,ab,de
1535 MASSAG?
201 ACUPRESSURE
58 SHIATSU
0 SHIATZU
105 ZHI
21 YA
0 ZHI(W)YA
10 CHIH/TI,AB,DE
21 YA/TI,AB,DE
0 CHIH/TI,AB,DE(W)YA/TI,AB,DE
S82 1692 MASSAG? OR ACUPRESSURE OR SHIATSU OR SHIATZU OR ZHI (W)
YA OR CHIH (W) YA/TI,AB,DE
? s s10 and s82
1532 S10
1692 S82
S83 43 S10 AND S82
? s rehabilitat? (2W) (program? or protocol?)/ti,ab,de
12934 REHABILITAT?/TI,AB,DE
21676 PROGRAM?/TI,AB,DE
6815 PROTOCOL?/TI,AB,DE
S84 2040 REHABILITAT? (2W) (PROGRAM? OR PROTOCOL?)/TI,AB,DE
? s s10 and s84
1532 S10
2040 S84
S85 53 S10 AND S84
? s watch? (2W) wait? or conservative (2W) treat?/ti,ab,de
500 WATCH?
875 WAIT?
18 WATCH?(2W)WAIT?
3381 CONSERVATIVE/TI,AB,DE
79900 TREAT?/TI,AB,DE
1533 CONSERVATIVE/TI,AB,DE(2W)TREAT?/TI,AB,DE
S86 1551 WATCH? (2W) WAIT? OR CONSERVATIVE (2W) TREAT?/TI,AB,DE
? s s10 and s86
1532 S10
1551 S86
S87 51 S10 AND S86
? s management (2W) (decision? or option? or choice?)/ti,ab,de
18593 MANAGEMENT/TI,AB,DE
5371 DECISION?/TI,AB,DE
3950 OPTION?/TI,AB,DE
4823 CHOICE?/TI,AB,DE
S88 219 MANAGEMENT (2W) (DECISION? OR OPTION? OR
CHOICE?)/TI,AB,DE
? s s10 and s88
1532 S10
219 S88
S89 1 S10 AND S88
? s s85 or s87 or s89
53 S85
51 S87
1 S89
S90 101 S85 OR S87 OR S89
? s s16 or s20 or s27 or s39 or s45 or s52 or s54 or s58 or s65 or s71 or s75 or s81 or s83 or s90
11 S16
32 S20
113 S27
313 S39
168 S45
44 S52
2 S54
22 S58
61 S65
2 S71
9 S75
88 S81
43 S83
101 S90
S91 685 S16 OR S20 OR S27 OR S39 OR S45 OR S52 OR S54 OR S58 OR
S65 OR S71 OR S75 OR S81 OR S83 OR S90
? s py=1966:2010
S92 89245 PY=1966:2010
? s s91 and s92
685 S91
89245 S92
S93 189 S91 AND S92
NHS Economic Evaluation Database (NHS EED)
Interface: Cochrane Library Issue 3 2010
Search date: 23 March 2010
Strategy: as below
| ID | Search | Hits |
|---|---|---|
| #1 | (frozen NEAR/6 shoulder*):ti,ab,kw | 65 |
| #2 | (stiff* NEAR/3 shoulder*):ti,ab,kw | 63 |
| #3 | MeSH descriptor Bursitis explode all trees | 127 |
| #4 | (adhesive NEXT (capsulitis or capsulitides)):ti,ab,kw | 44 |
| #5 | (bursitis or bursitides) NEAR/6 shoulder*:ti,ab,kw | 11 |
| #6 | (capsulitis or capsulitides) NEAR/6 shoulder* | 39 |
| #7 | (periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) NEAR/6 shoulder*:ti,ab,kw | 77 |
| #8 | MeSH descriptor Shoulder Pain explode all trees | 240 |
| #9 | (shoulder* NEAR/3 (pain or pains or painful or complain*)):ti,ab,kw | 682 |
| #10 | MeSH descriptor Shoulder Impingement Syndrome explode all trees | 84 |
| #11 | (shoulder* NEAR/6 impinge*):ti,ab,kw | 107 |
| #12 | “subacromial impingement syndrome”:ti,ab,kw | 20 |
| #13 | (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12) | 964 |
| #14 | MeSH descriptor Arthrography, this term only | 148 |
| #15 | (arthrograph* NEAR/6 (distension* or distention*)):ti,ab,kw | 10 |
| #16 | (arthrogram* NEAR/6 (distension* or distention*)):ti,ab,kw or (glenohumeral NEAR/6 (distension* or distention*)):ti,ab,kw | 3 |
| #17 | MeSH descriptor Dilatation, this term only | 281 |
| #18 | (dilatation or hydrodilat*):ti,ab,kw | 2541 |
| #19 | (#14 OR #15 OR #16 OR #17 OR #18) | 2695 |
| #20 | (#13 AND #19) | 25 |
| #21 | MeSH descriptor Arthroscopy explode all trees | 835 |
| #22 | (arthroscop* NEAR/6 (releas* or decompress* or capsulotom*)):ti,ab,kw | 64 |
| #23 | (capsular NEAR/2 releas*):ti,ab,kw or “interventional microadhesiolysis”:ti,ab,kw or (capsulotomy):ti,ab,kw | 197 |
| #24 | (#21 OR #22 OR #23) | 1048 |
| #25 | (#13 AND #24) | 51 |
| #26 | MeSH descriptor Injections, Intra-Articular explode all trees | 628 |
| #27 | (#13 AND #26) | 62 |
| #28 | MeSH descriptor Injections explode all trees | 15,394 |
| #29 | (#13 AND #28) | 115 |
| #30 | (bursa* or intrabursa* or intra bursa* or periartic* or peri artic* or intraartic* or intra artic*) NEAR/3 inject*:ti,ab,kw | 813 |
| #31 | (#13 AND #30) | 79 |
| #32 | (subacromial or acromioclavicular or glenohumeral) NEAR/3 inject*:ti,ab,kw or (extra articular or extraarticular or shoulder*) NEAR/3 inject*:ti,ab,kw | 72 |
| #33 | (#27 OR #29 OR #31 OR #32) | 169 |
| #34 | MeSH descriptor Physical Therapy Modalities explode all trees | 9583 |
| #35 | (physiotherapy or physiotherapies or (physical therap*) or (manual therap*)):ti,ab,kw or (passive NEXT (motion or movement)):ti,ab,kw or (CPM):ti,ab,kw | 10,737 |
| #36 | MeSH descriptor Muscle Stretching Exercises explode all trees | 82 |
| #37 | (stretching or stretches):ti,ab,kw or (mobilisation or mobilization):ti,ab,kw or (exercise* NEAR/2 (program* or strength* or intervention* or training or prescription* or prescrib*)):ti,ab,kw or (exercise* NEAR/2 (therap* or therapeutic)):ti,ab,kw | 10,643 |
| #38 | (home or supervis*) NEAR/2 exercis*:ti,ab,kw or (pendular or pendulum) NEXT exercis*:ti,ab,kw or (isokinetic or resist*) NEAR/2 exercise*,:ti,ab,kw | 1658 |
| #39 | (#34 OR #35 OR #36 OR #37 OR #38) | 23,617 |
| #40 | (#13 AND #39) | 300 |
| #41 | MeSH descriptor Musculoskeletal Manipulations explode all trees | 4745 |
| #42 | (chiropractic*):ti,ab,kw or (osteopath*):ti,ab,kw or (manipulat* NEAR/3 (anesthesia or anaesthesia or anesthetic* or anaesthetic*)):ti,ab,kw or (MUA):ti,ab,kw | 728 |
| #43 | (#41 OR #42) | 5242 |
| #44 | (#13 AND #43) | 119 |
| #45 | (TENS or ALTENS):ti,ab,kw or (electric* NEAR/2 stimulat*) or (transcutaneous NEAR/2 stimulat*) or (transdermal NEAR/2 electrostimulat*) or (cutaneous NEAR/2 electrostimulat*) or electroanalgesia or (electro analgesia):ti,ab,kw or (muscle NEAR/2 stimulat*):ti,ab,kw or (neuromodulation or (neuro modulation) or neurostimulation or (neuro stimulation)):ti,ab,kw or (interferential):ti,ab,kw | 17,526 |
| #46 | (#13 AND #45) | 76 |
| #47 | MeSH descriptor Biofeedback (Psychology) explode all trees | 710 |
| #48 | (biofeedback):ti,ab,kw | 1348 |
| #49 | (#47 OR #48) | 1348 |
| #50 | (#13 AND #49) | 6 |
| #51 | MeSH descriptor Cryotherapy explode all trees | 866 |
| #52 | MeSH descriptor Ice explode all trees | 63 |
| #53 | MeSH descriptor Diathermy explode all trees | 620 |
| #54 | MeSH descriptor Hyperthermia, Induced explode all trees | 981 |
| #55 | MeSH descriptor Hot Temperature explode all trees | 1079 |
| #56 | (cold or ice or heat or hot) NEXT (pack* or therap* or treat*):ti,ab,kw or (thermograph* or thermotherap* or (thermo therap*) or hypertherm* or (hyper therm*)):ti,ab,kw or (diatherm* or cryotherap* or (cryo therap*)):ti,ab,kw | 2278 |
| #57 | (#51 OR #52 OR #53 OR #54 OR #55 OR #56) | 4273 |
| #58 | (#13 AND #57) | 51 |
| #59 | MeSH descriptor Laser Therapy explode all trees | 2372 |
| #60 | MeSH descriptor Ultrasonic Therapy explode all trees | 523 |
| #61 | MeSH descriptor Ultrasonography, Interventional explode all trees | 529 |
| #62 | (ultrasound):ti,ab,kw or (electrotherapeutic NEAXT (intervention* or treat*)):ti,ab,kw | 5557 |
| #63 | (#59 OR #60 OR #61 OR #62) | 8267 |
| #64 | (#13 AND #63) | 58 |
| #65 | MeSH descriptor Magnetic Field Therapy explode all trees | 369 |
| #66 | “pulsed electromagnetic field therapy”:ti,ab,kw or (electromagnetic* or magnetic*) NEAR/3 field*:ti,ab,kw or (biomagnetic* or (bio magnetic*) or (pulsed signal)):ti,ab,kw or (PEMF):ti,ab,kw | 793 |
| #67 | (#65 OR #66) | 1121 |
| #68 | (#13 AND #67) | 12 |
| #69 | MeSH descriptor Nerve Block explode all trees | 1994 |
| #70 | MeSH descriptor Neuromuscular Blockade explode all trees | 266 |
| #71 | (nerve NEAR/2 block*):ti,ab,kw | 2595 |
| #72 | (#69 OR #70 OR #71) | 2854 |
| #73 | (#13 AND #72) | 49 |
| #74 | MeSH descriptor Acupuncture Therapy explode all trees | 1830 |
| #75 | (acupuncture*):ti,ab,kw or (electroacupuncture* or (electro acupuncture*)):ti,ab,kw or (osteopuncture* or (osteo puncture*)):ti,ab,kw or (perioste* NEAR/3 (stimulat* or therap* or needling)):ti,ab,kw | 4213 |
| #76 | (#74 OR #75) | 4239 |
| #77 | (#13 AND #76) | 58 |
| #78 | MeSH descriptor Massage explode all trees | 595 |
| #79 | (massag* or acupressure or shiatsu or shiatzu or (zhi ya) or (chih ya)):ti,ab,kw | 1277 |
| #80 | (#78 OR #79) | 1277 |
| #81 | (#13 AND #80) | 20 |
| #82 | (rehabilitat* NEAR/2 (program* or protocol*)):ti,ab,kw or (watch* NEAR/2 wait*) or (conservative NEAR/2 treat*):ti,ab,kw or (management NEAR/2 (decision* or option* or choice*)):ti,ab,kw | 2974 |
| #83 | (#13 AND #82) | 34 |
| #84 | (#20 OR #25 OR #33 OR #40 OR #44 OR #46 OR #50 OR #58 OR #64 OR #68 OR #73 OR #77 OR #81 OR #83) | 588 |
National Technical Information Service (NTIS)
Interface: Dialog Classic
Search date: 30 March 2010
Strategy: as below
Records identified: 8
File 6:NTIS 1964–2010/Mar W4
(c) 2010 NTIS, Intl Cpyrght All Rights Res
? s frozen (6W) shoulder?/ti,ab,de
4683 FROZEN/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
S1 5 FROZEN (6W) SHOULDER?/TI,AB,DE
? s stiff? (3W) shoulder?/ti,ab,de
11654 STIFF?/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
S2 1 STIFF? (3W) SHOULDER?/TI,AB,DE
? s adhesive (W) (capsulitis or capsulitides)/ti,ab,de
4903 ADHESIVE/TI,AB,DE
1 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
S3 1 ADHESIVE (W) (CAPSULITIS OR CAPSULITIDES)/TI,AB,DE
? s (bursitis or bursitides) (6W) shoulder?/ti,ab,de
10 BURSITIS/TI,AB,DE
0 BURSITIDES/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
S4 0 (BURSITIS OR BURSITIDES) (6W) SHOULDER?/TI,AB,DE
? s (capsulitis or capsulitides) (6W) shoulder?/ti,ab,de
1 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
S5 1 (CAPSULITIS OR CAPSULITIDES) (6W) SHOULDER?/TI,AB,DE
? s (periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) (6W) shoulder?/ti,ab,de
3 PERIARTHRITIS/TI,AB,DE
0 PERI-ARTHRITIS/TI,AB,DE
0 PERIARTHRITIDES/TI,AB,DE
0 PERI-ARTHRITIDES/TI,AB,DE
0 PERI-CAPSULITIS/TI,AB,DE
0 PERICAPSULITIS/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
S6 0 (PERIARTHRITIS OR PERI-ARTHRITIS OR PERIARTHRITIDES OR PERI-ARTHRITIDES OR PERI-CAPSULITIS OR PERICAPSULITIS) (6W) SHOULDER?/TI,AB,DE
? s shoulder? (3W) (pain or pains or painful or complain?)/ti,ab,de
1917 SHOULDER?/TI,AB,DE
1751 PAIN/TI,AB,DE
121 PAINS/TI,AB,DE
245 PAINFUL/TI,AB,DE
2900 COMPLAIN?/TI,AB,DE
S7 19 SHOULDER? (3W) (PAIN OR PAINS OR PAINFUL OR COMPLAIN?)/TI,AB,DE
? s shoulder? (6W) impinge?/ti,ab,de
1917 SHOULDER?/TI,AB,DE
2858 IMPINGE?/TI,AB,DE
S8 1 SHOULDER? (6W) IMPINGE?/TI,AB,DE
? s subacromial (W) impingement (W) syndrome/ti,ab,de
0 SUBACROMIAL/TI,AB,DE
1922 IMPINGEMENT/TI,AB,DE
3547 SYNDROME/TI,AB,DE
S9 0 SUBACROMIAL (W) IMPINGEMENT (W) SYNDROME/TI,AB,DE
? s s1:s9
S10 26 S1:S9
? s arthrograph? (6W) (distension? or distention?)/ti,ab,de
4 ARTHROGRAPH?/TI,AB,DE
50 DISTENSION?/TI,AB,DE
39 DISTENTION?/TI,AB,DE
S11 0 ARTHROGRAPH? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s arthrogram? (6W) (distension? or distention?)/ti,ab,de
0 ARTHROGRAM?/TI,AB,DE
50 DISTENSION?/TI,AB,DE
39 DISTENTION?/TI,AB,DE
S12 0 ARTHROGRAM? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s glenohumeral (6W) (distension? or distention?)/ti,ab,de
5 GLENOHUMERAL/TI,AB,DE
50 DISTENSION?/TI,AB,DE
39 DISTENTION?/TI,AB,DE
S13 0 GLENOHUMERAL (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s dilatation or hydrodilat?/ti,ab,de
336 DILATATION
0 HYDRODILAT?/TI,AB,DE
S14 336 DILATATION OR HYDRODILAT?/TI,AB,DE
? s s11:s14
S15 336 S11:S14
? s s10 and s15
26 S10
336 S15
S16 0 S10 AND S15
? s arthroscop? (6W) (releas? or decompress? or capsulotom?)/ti,ab,de
11 ARTHROSCOP?/TI,AB,DE
45473 RELEAS?/TI,AB,DE
1628 DECOMPRESS?/TI,AB,DE
8 CAPSULOTOM?/TI,AB,DE
S17 0 ARTHROSCOP? (6W) (RELEAS? OR DECOMPRESS? OR CAPSULOTOM?)/TI,AB,DE
? s capsular (2W) releas? or interventional (W) microadhesiolysis or capsulotomy/ti,ab,de
121 CAPSULAR
46080 RELEAS?
0 CAPSULAR(2W)RELEAS?
72 INTERVENTIONAL
0 MICROADHESIOLYSIS
0 INTERVENTIONAL(W)MICROADHESIOLYSIS
5 CAPSULOTOMY/TI,AB,DE
S18 5 CAPSULAR (2W) RELEAS? OR INTERVENTIONAL (W) MICROADHESIOLYSIS OR CAPSULOTOMY/TI,AB,DE
? s s17:s18
S19 5 S17:S18
? s s10 and s19
26 S10
5 S19
S20 0 S10 AND S19
? s injection?/ti,ab,de
>>>Term “DE..” is not defined in file 6 and is ignored
S21 22468 INJECTION?/TI,AB,DE..
? s s10 and s21
26 S10
22468 S21
S22 1 S10 AND S21
? s (bursa? or intrabursa? or intra (W) bursa? or periartic? or peri (W) artic? or intraartic? or intra (W) artic?) (3W) inject?/ti,ab,de
54 BURSA?/TI,AB,DE
1 INTRABURSA?/TI,AB,DE
2614 INTRA/TI,AB,DE
54 BURSA?/TI,AB,DE
0 INTRA/TI,AB,DE(W)BURSA?/TI,AB,DE
2 PERIARTIC?/TI,AB,DE
84 PERI/TI,AB,DE
55035 ARTIC?/TI,AB,DE
1 PERI/TI,AB,DE(W)ARTIC?/TI,AB,DE
0 INTRAARTIC?/TI,AB,DE
2614 INTRA/TI,AB,DE
55035 ARTIC?/TI,AB,DE
16 INTRA/TI,AB,DE(W)ARTIC?/TI,AB,DE
37102 INJECT?/TI,AB,DE
S23 8 (BURSA? OR INTRABURSA? OR INTRA (W) BURSA? OR PERIARTIC? OR PERI (W) ARTIC? OR INTRAARTIC? OR INTRA (W) ARTIC?) (3W) INJECT?/TI,AB,DE
? s s10 and s23
26 S10
8 S23
S24 0 S10 AND S23
? s (subacromial or acromioclavicular or glenohumeral) (3W) inject?/ti,ab,de
0 SUBACROMIAL/TI,AB,DE
1 ACROMIOCLAVICULAR/TI,AB,DE
5 GLENOHUMERAL/TI,AB,DE
37102 INJECT?/TI,AB,DE
S25 0 (SUBACROMIAL OR ACROMIOCLAVICULAR OR GLENOHUMERAL) (3W) INJECT?/TI,AB,DE
? s (extra (W) articular or extraarticular or shoulder?) (3W) inject?/ti,ab,de
4370 EXTRA/TI,AB,DE
82 ARTICULAR/TI,AB,DE
0 EXTRA/TI,AB,DE(W)ARTICULAR/TI,AB,DE
0 EXTRAARTICULAR/TI,AB,DE
1917 SHOULDER?/TI,AB,DE
37102 INJECT?/TI,AB,DE
S26 2 (EXTRA (W) ARTICULAR OR EXTRAARTICULAR OR SHOULDER?) (3W) INJECT?/TI,AB,DE
? s s22 or s24 or s25 or s26
1 S22
0 S24
0 S25
2 S26
S27 3 S22 OR S24 OR S25 OR S26
? s physiotherapy or physiotherapies or physical (W) therap? or manual(W) therap?/ti,ab,de
38 PHYSIOTHERAPY
0 PHYSIOTHERAPIES
226243 PHYSICAL
19307 THERAP?
1223 PHYSICAL(W)THERAP?
37866 MANUAL/TI,AB,DE
18488 THERAP?/TI,AB,DE
1 MANUAL/TI,AB,DE(W)THERAP?/TI,AB,DE
S28 1253 PHYSIOTHERAPY OR PHYSIOTHERAPIES OR PHYSICAL (W) THERAP? OR MANUAL(W) THERAP?/TI,AB,DE
? s passive (W) (motion or movement)/ti,ab,de
13922 PASSIVE/TI,AB,DE
56426 MOTION/TI,AB,DE
19107 MOVEMENT/TI,AB,DE
S29 11 PASSIVE (W) (MOTION OR MOVEMENT)/TI,AB,DE
? s CPM/ti,ab,de
S30 356 CPM/TI,AB,DE
? s (stretching or stretches)/ti,ab,de
2022 STRETCHING/TI,AB,DE
319 STRETCHES/TI,AB,DE
S31 2331 (STRETCHING OR STRETCHES)/TI,AB,DE
? s (mobilisation or mobilization)/ti,ab,de
41 MOBILISATION/TI,AB,DE
2774 MOBILIZATION/TI,AB,DE
S32 2812 (MOBILISATION OR MOBILIZATION)/TI,AB,DE
? s exercise? (2W) (program? or strength? or intervention? or training or prescription? or prescrib?)/ti,ab,de
14367 EXERCISE?/TI,AB,DE
480068 PROGRAM?/TI,AB,DE
72882 STRENGTH?/TI,AB,DE
8460 INTERVENTION?/TI,AB,DE
71866 TRAINING/TI,AB,DE
2399 PRESCRIPTION?/TI,AB,DE
8170 PRESCRIB?/TI,AB,DE
S33 651 EXERCISE? (2W) (PROGRAM? OR STRENGTH? OR INTERVENTION? OR TRAINING OR PRESCRIPTION? OR PRESCRIB?)/TI,AB,DE
? s exercise? (2W) (therap? or therapeutic)/ti,ab,de
14367 EXERCISE?/TI,AB,DE
18488 THERAP?/TI,AB,DE
5073 THERAPEUTIC/TI,AB,DE
S34 37 EXERCISE? (2W) (THERAP? OR THERAPEUTIC)/TI,AB,DE
? s (home or supervis?) (2W) exercis?/ti,ab,de
14925 HOME/TI,AB,DE
10621 SUPERVIS?/TI,AB,DE
14861 EXERCIS?/TI,AB,DE
S35 17 (HOME OR SUPERVIS?) (2W) EXERCIS?/TI,AB,DE
? s (pendular or pendulum) (W) exercis?/ti,ab,de
39 PENDULAR/TI,AB,DE
952 PENDULUM/TI,AB,DE
14861 EXERCIS?/TI,AB,DE
S36 0 (PENDULAR OR PENDULUM) (W) EXERCIS?/TI,AB,DE
? s (isokinetic or resist?) (2W) exercise?/ti,ab,de
223 ISOKINETIC/TI,AB,DE
74785 RESIST?/TI,AB,DE
14367 EXERCISE?/TI,AB,DE
S37 68 (ISOKINETIC OR RESIST?) (2W) EXERCISE?/TI,AB,DE
? s s28:s37
S38 7465 S28:S37
? s s10 and s38
26 S10
7465 S38
S39 1 S10 AND S38
? s chiropractic?/ti,ab,de
S40 39 CHIROPRACTIC?/TI,AB,DE
? s osteopath?/ti,ab,de
S41 199 OSTEOPATH?/TI,AB,DE
? s manipulat? (3W) (anesthesia or anaesthesia or anesthetic? or anaesthetic? or musculoskeletal)/ti,ab,de
13446 MANIPULAT?/TI,AB,DE
1006 ANESTHESIA/TI,AB,DE
47 ANAESTHESIA/TI,AB,DE
723 ANESTHETIC?/TI,AB,DE
41 ANAESTHETIC?/TI,AB,DE
1646 MUSCULOSKELETAL/TI,AB,DE
S42 1 MANIPULAT? (3W) (ANESTHESIA OR ANAESTHESIA OR ANESTHETIC? OR ANAESTHETIC? OR MUSCULOSKELETAL)/TI,AB,DE
? s MUA/ti,ab,de
S43 27 MUA/TI,AB,DE
? s s40:s43
S44 257 S40:S43
? s s10 and s44
26 S10
257 S44
S45 0 S10 AND S44
? s (TENS or ALTENS)/ti,ab,de
2334 TENS/TI,AB,DE
0 ALTENS/TI,AB,DE
S46 2334 (TENS OR ALTENS)/TI,AB,DE
? s electric? (2W) stimulat? or transcutaneous (2W) stimulat? or transdermal (2W) electrostimulat? or cutaneous (2W) electrostimulat? or electroanalgesia or electro(W)analgesia/ti,ab,de
170950 ELECTRIC?
18749 STIMULAT?
518 ELECTRIC?(2W)STIMULAT?
84 TRANSCUTANEOUS
18749 STIMULAT?
13 TRANSCUTANEOUS(2W)STIMULAT?
93 TRANSDERMAL
17 ELECTROSTIMULAT?
0 TRANSDERMAL(2W)ELECTROSTIMULAT?
810 CUTANEOUS
17 ELECTROSTIMULAT?
1 CUTANEOUS(2W)ELECTROSTIMULAT?
3 ELECTROANALGESIA
6549 ELECTRO/TI,AB,DE
133 ANALGESIA/TI,AB,DE
1 ELECTRO/TI,AB,DE(W)ANALGESIA/TI,AB,DE
S47 524 ELECTRIC? (2W) STIMULAT? OR TRANSCUTANEOUS (2W) STIMULAT? OR TRANSDERMAL (2W) ELECTROSTIMULAT? OR CUTANEOUS (2W) ELECTROSTIMULAT? OR ELECTROANALGESIA OR ELECTRO(W)ANALGESIA/TI,AB,DE
? s muscle (2W) stimulat?/ti,ab,de
4028 MUSCLE/TI,AB,DE
18526 STIMULAT?/TI,AB,DE
S48 35 MUSCLE (2W) STIMULAT?/TI,AB,DE
? s neuromodulation or neuro (W) modulation or neurostimulation or neuro (W) stimulation/ti,ab,de
17 NEUROMODULATION
262 NEURO
15752 MODULATION
0 NEURO(W)MODULATION
2 NEUROSTIMULATION
246 NEURO/TI,AB,DE
7638 STIMULATION/TI,AB,DE
0 NEURO/TI,AB,DE(W)STIMULATION/TI,AB,DE
S49 19 NEUROMODULATION OR NEURO (W) MODULATION OR NEUROSTIMULATION OR NEURO (W) STIMULATION/TI,AB,DE
? s interferential/ti,ab,de
S50 29 INTERFERENTIAL/TI,AB,DE
? s s46:s50
S51 2929 S46:S50
? s s10 and s51
26 S10
2929 S51
S52 0 S10 AND S51
? s biofeedback/ti,ab,de
S53 218 BIOFEEDBACK/TI,AB,DE
? s s10 and s53
26 S10
218 S53
S54 0 S10 AND S53
? s (cold or ice or heat or hot) (W) (pack? or therap? or treat?)/ti,ab,de
26659 COLD/TI,AB,DE
15533 ICE/TI,AB,DE
118199 HEAT/TI,AB,DE
35308 HOT/TI,AB,DE
47432 PACK?/TI,AB,DE
18488 THERAP?/TI,AB,DE
132040 TREAT?/TI,AB,DE
S55 11411 (COLD OR ICE OR HEAT OR HOT) (W) (PACK? OR THERAP? OR TREAT?)/TI,AB,DE
? s thermograph? or thermotherap? or thermo (W) therap? or hypertherm? or hyper (W) therm? or diatherm? or cryotherap? or cryo(W) therap?/ti,ab,de
1380 THERMOGRAPH?
4 THERMOTHERAP?
2400 THERMO
19307 THERAP?
0 THERMO(W)THERAP?
735 HYPERTHERM?
654 HYPER
200235 THERM?
8 HYPER(W)THERM?
45 DIATHERM?
19 CRYOTHERAP?
241 CRYO/TI,AB,DE
18488 THERAP?/TI,AB,DE
0 CRYO/TI,AB,DE(W)THERAP?/TI,AB,DE
S56 2170 THERMOGRAPH? OR THERMOTHERAP? OR THERMO (W) THERAP? OR HYPERTHERM? OR HYPER (W) THERM? OR DIATHERM? OR CRYOTHERAP? OR CRYO(W) THERAP?/TI,AB,DE
? s s55:s56
S57 13561 S55:S56
? s s10 and s57
26 S10
13561 S57
S58 2 S10 AND S57
? s laser (W) therapy/ti,ab,de
61371 LASER/TI,AB,DE
14631 THERAPY/TI,AB,DE
S59 15 LASER (W) THERAPY/TI,AB,DE
? s ultrasonic (W) therapy/ti,ab,de
10590 ULTRASONIC/TI,AB,DE
14631 THERAPY/TI,AB,DE
S60 15 ULTRASONIC (W) THERAPY/TI,AB,DE
? s ultrasound/ti,ab,de
S61 1689 ULTRASOUND/TI,AB,DE
? s ultrasonography/ti,ab,de
S62 180 ULTRASONOGRAPHY/TI,AB,DE
? s electrotherapeutic (W) (intervention? or treat?)/ti,ab,de
6 ELECTROTHERAPEUTIC/TI,AB,DE
8460 INTERVENTION?/TI,AB,DE
132040 TREAT?/TI,AB,DE
S63 0 ELECTROTHERAPEUTIC (W) (INTERVENTION? OR TREAT?)/TI,AB,DE
? s s59:s63
S64 1841 S59:S63
? s s10 and s64
26 S10
1841 S64
S65 1 S10 AND S64
? s pulsed (W) electromagnetic (W) field (W) therapy/ti,ab,de
15810 PULSED/TI,AB,DE
38066 ELECTROMAGNETIC/TI,AB,DE
195700 FIELD/TI,AB,DE
14631 THERAPY/TI,AB,DE
S66 0 PULSED (W) ELECTROMAGNETIC (W) FIELD (W) THERAPY/TI,AB,DE
? s (electromagnetic? or magnetic?) (3W) field?/ti,ab,de
38583 ELECTROMAGNETIC?/TI,AB,DE
85629 MAGNETIC?/TI,AB,DE
243256 FIELD?/TI,AB,DE
S67 42083 (ELECTROMAGNETIC? OR MAGNETIC?) (3W) FIELD?/TI,AB,DE
? s biomagnetic? or bio (W) magnetic? or pulsed signal/ti,ab,de
96 BIOMAGNETIC?
2195 BIO
86544 MAGNETIC?
3 BIO(W)MAGNETIC?
0 PULSED SIGNAL/TI,AB,DE
S68 98 BIOMAGNETIC? OR BIO (W) MAGNETIC? OR PULSED SIGNAL/TI,AB,DE
? s PEMF/ti,ab,de
S69 5 PEMF/TI,AB,DE
? s s66:s69
S70 42137 S66:S69
? s s10 and s70
26 S10
42137 S70
S71 0 S10 AND S70
? s neuromuscular (W) blockade/ti,ab,de
839 NEUROMUSCULAR/TI,AB,DE
582 BLOCKADE/TI,AB,DE
S72 19 NEUROMUSCULAR (W) BLOCKADE/TI,AB,DE
? s nerve (2W) block?/ti,ab,de
6188 NERVE/TI,AB,DE
25659 BLOCK?/TI,AB,DE
S73 170 NERVE (2W) BLOCK?/TI,AB,DE
? s s72:s73
S74 180 S72:S73
? s s10 and s74
26 S10
180 S74
S75 0 S10 AND S74
? s acupuncture?/ti,ab,de
S76 87 ACUPUNCTURE?/TI,AB,DE
? s electroacupuncture? or electro (W) acupuncture?/ti,ab,de
6 ELECTROACUPUNCTURE?
6549 ELECTRO/TI,AB,DE
87 ACUPUNCTURE?/TI,AB,DE
4 ELECTRO/TI,AB,DE(W)ACUPUNCTURE?/TI,AB,DE
S77 8 ELECTROACUPUNCTURE? OR ELECTRO (W) ACUPUNCTURE?/TI,AB,DE
? s osteopuncture? or osteo (W) puncture?/ti,ab,de
0 OSTEOPUNCTURE?
6 OSTEO/TI,AB,DE
602 PUNCTURE?/TI,AB,DE
0 OSTEO/TI,AB,DE(W)PUNCTURE?/TI,AB,DE
S78 0 OSTEOPUNCTURE? OR OSTEO (W) PUNCTURE?/TI,AB,DE
? s perioste? (3W) (stimulat? or therap? or needling)/ti,ab,de
38 PERIOSTE?/TI,AB,DE
18526 STIMULAT?/TI,AB,DE
18488 THERAP?/TI,AB,DE
14 NEEDLING/TI,AB,DE
S79 1 PERIOSTE? (3W) (STIMULAT? OR THERAP? OR NEEDLING)/TI,AB,DE
? s s76:s79
S80 89 S76:S79
? s s10 and s80
26 S10
89 S80
S81 0 S10 AND S80
? s massag? or acupressure or shiatsu or shiatzu or zhi (W) ya or chih (W) ya/ti,ab,de
69 MASSAG?
2 ACUPRESSURE
0 SHIATSU
0 SHIATZU
2 ZHI
125 YA
0 ZHI(W)YA
12 CHIH/TI,AB,DE
122 YA/TI,AB,DE
0 CHIH/TI,AB,DE(W)YA/TI,AB,DE
S82 71 MASSAG? OR ACUPRESSURE OR SHIATSU OR SHIATZU OR ZHI (W) YA OR CHIH (W) YA/TI,AB,DE
? s s10 and s82
26 S10
71 S82
S83 0 S10 AND S82
? s rehabilitat? (2W) (program? or protocol?)/ti,ab,de
8675 REHABILITAT?/TI,AB,DE
480068 PROGRAM?/TI,AB,DE
14132 PROTOCOL?/TI,AB,DE
S84 864 REHABILITAT? (2W) (PROGRAM? OR PROTOCOL?)/TI,AB,DE
? s s10 and s84
26 S10
864 S84
S85 1 S10 AND S84
? s watch? (2W) wait? or conservative (2W) treat?/ti,ab,de
2243 WATCH?
2348 WAIT?
18 WATCH?(2W)WAIT?
5006 CONSERVATIVE/TI,AB,DE
132040 TREAT?/TI,AB,DE
28 CONSERVATIVE/TI,AB,DE(2W)TREAT?/TI,AB,DE
S86 46 WATCH? (2W) WAIT? OR CONSERVATIVE (2W) TREAT?/TI,AB,DE
? s s10 and s86
26 S10
46 S86
S87 0 S10 AND S86
? s management (2W) (decision? or option? or choice?)/ti,ab,de
218292 MANAGEMENT/TI,AB,DE
69503 DECISION?/TI,AB,DE
30662 OPTION?/TI,AB,DE
21235 CHOICE?/TI,AB,DE
S88 4609 MANAGEMENT (2W) (DECISION? OR OPTION? OR CHOICE?)/TI,AB,DE
? s s10 and s88
26 S10
4609 S88
S89 0 S10 AND S88
? s s85 or s87 or s89
1 S85
0 S87
0 S89
S90 1 S85 OR S87 OR S89
? s s16 or s20 or s27 or s39 or s45 or s52 or s54 or s58 or s65 or s71 or s75 or s81 or s83 or s90
0 S16
0 S20
3 S27
1 S39
0 S45
0 S52
0 S54
2 S58
1 S65
0 S71
0 S75
0 S81
0 S83
1 S90
S91 8 S16 OR S20 OR S27 OR S39 OR S45 OR S52 OR S54 OR S58 OR S65 OR S71 OR S75 OR S81 OR S83 OR S90
PASCAL
Interface: Dialog Classic
Search date: 30 March 2010
Strategy: as below
Records identified: 631
File 144:Pascal 1973–2010/Mar W1
(c) 2010 INIST/CNRS
? s frozen (6W) shoulder?/ti,ab,de
29523 FROZEN/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
S1 265 FROZEN (6W) SHOULDER?/TI,AB,DE
? s stiff? (3W) shoulder?/ti,ab,de
47795 STIFF?/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
S2 74 STIFF? (3W) SHOULDER?/TI,AB,DE
? s adhesive (W) (capsulitis or capsulitides)/ti,ab,de
31322 ADHESIVE/TI,AB,DE
165 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
S3 131 ADHESIVE (W) (CAPSULITIS OR CAPSULITIDES)/TI,AB,DE
? s (bursitis or bursitides) (6W) shoulder?/ti,ab,de
673 BURSITIS/TI,AB,DE
1 BURSITIDES/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
S4 38 (BURSITIS OR BURSITIDES) (6W) SHOULDER?/TI,AB,DE
? s (capsulitis or capsulitides) (6W) shoulder?/ti,ab,de
165 CAPSULITIS/TI,AB,DE
0 CAPSULITIDES/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
S5 66 (CAPSULITIS OR CAPSULITIDES) (6W) SHOULDER?/TI,AB,DE
? s (periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) (6W) shoulder?/ti,ab,de
109 PERIARTHRITIS/TI,AB,DE
0 PERI-ARTHRITIS/TI,AB,DE
1 PERIARTHRITIDES/TI,AB,DE
0 PERI-ARTHRITIDES/TI,AB,DE
0 PERI-CAPSULITIS/TI,AB,DE
0 PERICAPSULITIS/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
S6 25 (PERIARTHRITIS OR PERI-ARTHRITIS OR PERIARTHRITIDES OR PERI-ARTHRITIDES OR PERI-CAPSULITIS OR PERICAPSULITIS) (6W) SHOULDER?/TI,AB,DE
? s shoulder? (3W) (pain or pains or painful or complain?)/ti,ab,de
18440 SHOULDER?/TI,AB,DE
131547 PAIN/TI,AB,DE
1672 PAINS/TI,AB,DE
10410 PAINFUL/TI,AB,DE
19751 COMPLAIN?/TI,AB,DE
S7 1529 SHOULDER? (3W) (PAIN OR PAINS OR PAINFUL OR COMPLAIN?)/TI,AB,DE
? s shoulder? (6W) impinge?/ti,ab,de
18440 SHOULDER?/TI,AB,DE
5891 IMPINGE?/TI,AB,DE
S8 179 SHOULDER? (6W) IMPINGE?/TI,AB,DE
? s subacromial (W) impingement (W) syndrome/ti,ab,de
598 SUBACROMIAL/TI,AB,DE
3975 IMPINGEMENT/TI,AB,DE
394414 SYNDROME/TI,AB,DE
S9 147 SUBACROMIAL (W) IMPINGEMENT (W) SYNDROME/TI,AB,DE
? s s1:s9
S10 2177 S1:S9
? s arthrograph? (6W) (distension? or distention?)/ti,ab,de
2195 ARTHROGRAPH?/TI,AB,DE
5134 DISTENSION?/TI,AB,DE
1532 DISTENTION?/TI,AB,DE
S11 8 ARTHROGRAPH? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s arthrogram? (6W) (distension? or distention?)/ti,ab,de
228 ARTHROGRAM?/TI,AB,DE
5134 DISTENSION?/TI,AB,DE
1532 DISTENTION?/TI,AB,DE
S12 0 ARTHROGRAM? (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s glenohumeral (6W) (distension? or distention?)/ti,ab,de
1197 GLENOHUMERAL/TI,AB,DE
5134 DISTENSION?/TI,AB,DE
1532 DISTENTION?/TI,AB,DE
S13 1 GLENOHUMERAL (6W) (DISTENSION? OR DISTENTION?)/TI,AB,DE
? s dilatation or hydrodilat?/ti,ab,de
56041 DILATATION
7 HYDRODILAT?/TI,AB,DE
S14 56047 DILATATION OR HYDRODILAT?/TI,AB,DE
? s s11:s14
S15 56055 S11:S14
? s s10 and s15
2177 S10
56055 S15
S16 10 S10 AND S15
? s arthroscop? (6W) (releas? or decompress? or capsulotom?)/ti,ab,de
8274 ARTHROSCOP?/TI,AB,DE
261595 RELEAS?/TI,AB,DE
13807 DECOMPRESS?/TI,AB,DE
1046 CAPSULOTOM?/TI,AB,DE
S17 306 ARTHROSCOP? (6W) (RELEAS? OR DECOMPRESS? OR CAPSULOTOM?)/TI,AB,DE
? s capsular (2W) releas? or interventional (W) microadhesiolysis or capsulotomy/ti,ab,de
6434 CAPSULAR
261595 RELEAS?
92 CAPSULAR(2W)RELEAS?
9302 INTERVENTIONAL
0 MICROADHESIOLYSIS
0 INTERVENTIONAL(W)MICROADHESIOLYSIS
925 CAPSULOTOMY/TI,AB,DE
S18 1012 CAPSULAR (2W) RELEAS? OR INTERVENTIONAL (W) MICROADHESIOLYSIS OR CAPSULOTOMY/TI,AB,DE
? s s17:s18
S19 1264 S17:S18
? s s10 and s19
2177 S10
1264 S19
S20 99 S10 AND S19
? s injection?/ti,ab,de
>>>Term “DE..” is not defined in file 144 and is ignored
S21 225170 INJECTION?/TI,AB,DE..
? s s10 and s21
2177 S10
225170 S21
S22 161 S10 AND S21
? s (bursa? or intrabursa? or intra (W) bursa? or periartic? or peri (W) artic? or intraartic? or intra (W) artic?) (3W) inject?/ti,ab,de
3240 BURSA?/TI,AB,DE
54 INTRABURSA?/TI,AB,DE
66732 INTRA/TI,AB,DE
3240 BURSA?/TI,AB,DE
3 INTRA/TI,AB,DE(W)BURSA?/TI,AB,DE
1029 PERIARTIC?/TI,AB,DE
9053 PERI/TI,AB,DE
705828 ARTIC?/TI,AB,DE
162 PERI/TI,AB,DE(W)ARTIC?/TI,AB,DE
2544 INTRAARTIC?/TI,AB,DE
66732 INTRA/TI,AB,DE
705828 ARTIC?/TI,AB,DE
2857 INTRA/TI,AB,DE(W)ARTIC?/TI,AB,DE
309289 INJECT?/TI,AB,DE
S23 1060 (BURSA? OR INTRABURSA? OR INTRA (W) BURSA? OR PERIARTIC? OR PERI (W) ARTIC? OR INTRAARTIC? OR INTRA (W) ARTIC?) (3W) INJECT?/TI,AB,DE
? s s10 and s23
2177 S10
1060 S23
S24 34 S10 AND S23
? s (subacromial or acromioclavicular or glenohumeral) (3W) inject?/ti,ab,de
598 SUBACROMIAL/TI,AB,DE
667 ACROMIOCLAVICULAR/TI,AB,DE
1197 GLENOHUMERAL/TI,AB,DE
309289 INJECT?/TI,AB,DE
S25 62 (SUBACROMIAL OR ACROMIOCLAVICULAR OR GLENOHUMERAL) (3W) INJECT?/TI,AB,DE
? s (extra (W) articular or extraarticular or shoulder?) (3W) inject?/ti,ab,de
40008 EXTRA/TI,AB,DE
15525 ARTICULAR/TI,AB,DE
608 EXTRA/TI,AB,DE(W)ARTICULAR/TI,AB,DE
321 EXTRAARTICULAR/TI,AB,DE
18440 SHOULDER?/TI,AB,DE
309289 INJECT?/TI,AB,DE
S26 54 (EXTRA (W) ARTICULAR OR EXTRAARTICULAR OR SHOULDER?) (3W) INJECT?/TI,AB,DE
? s s22 or s24 or s25 or s26
161 S22
34 S24
62 S25
54 S26
S27 229 S22 OR S24 OR S25 OR S26
? s physiotherapy or physiotherapies or physical (W) therap? or manual(W) therap?/ti,ab,de
5062 PHYSIOTHERAPY
2 PHYSIOTHERAPIES
540408 PHYSICAL
661276 THERAP?
1884 PHYSICAL(W)THERAP?
35077 MANUAL/TI,AB,DE
661276 THERAP?/TI,AB,DE
120 MANUAL/TI,AB,DE(W)THERAP?/TI,AB,DE
S28 6839 PHYSIOTHERAPY OR PHYSIOTHERAPIES OR PHYSICAL (W) THERAP? OR MANUAL(W) THERAP?/TI,AB,DE
? s passive (W) (motion or movement)/ti,ab,de
62904 PASSIVE/TI,AB,DE
175972 MOTION/TI,AB,DE
109985 MOVEMENT/TI,AB,DE
S29 511 PASSIVE (W) (MOTION OR MOVEMENT)/TI,AB,DE
? s CPM/ti,ab,de
S30 2329 CPM/TI,AB,DE
? s (stretching or stretches)/ti,ab,de
18320 STRETCHING/TI,AB,DE
1979 STRETCHES/TI,AB,DE
S31 20093 (STRETCHING OR STRETCHES)/TI,AB,DE
? s (mobilisation or mobilization)/ti,ab,de
10059 MOBILISATION/TI,AB,DE
14519 MOBILIZATION/TI,AB,DE
S32 19880 (MOBILISATION OR MOBILIZATION)/TI,AB,DE
? s exercise? (2W) (program? or strength? or intervention? or training or prescription? or prescrib?)/ti,ab,de
83485 EXERCISE?/TI,AB,DE
496453 PROGRAM?/TI,AB,DE
289246 STRENGTH?/TI,AB,DE
201443 INTERVENTION?/TI,AB,DE
104948 TRAINING/TI,AB,DE
30046 PRESCRIPTION?/TI,AB,DE
30651 PRESCRIB?/TI,AB,DE
S33 6656 EXERCISE? (2W) (PROGRAM? OR STRENGTH? OR INTERVENTION? OR TRAINING OR PRESCRIPTION? OR PRESCRIB?)/TI,AB,DE
? s exercise? (2W) (therap? or therapeutic)/ti,ab,de
83485 EXERCISE?/TI,AB,DE
661276 THERAP?/TI,AB,DE
209433 THERAPEUTIC/TI,AB,DE
S34 704 EXERCISE? (2W) (THERAP? OR THERAPEUTIC)/TI,AB,DE
? s (home or supervis?) (2W) exercis?/ti,ab,de
46815 HOME/TI,AB,DE
33017 SUPERVIS?/TI,AB,DE
84078 EXERCIS?/TI,AB,DE
S35 629 (HOME OR SUPERVIS?) (2W) EXERCIS?/TI,AB,DE
? s (pendular or pendulum) (W) exercis?/ti,ab,de
498 PENDULAR/TI,AB,DE
3918 PENDULUM/TI,AB,DE
84078 EXERCIS?/TI,AB,DE
S36 3 (PENDULAR OR PENDULUM) (W) EXERCIS?/TI,AB,DE
? s (isokinetic or resist?) (2W) exercise?/ti,ab,de
2366 ISOKINETIC/TI,AB,DE
709778 RESIST?/TI,AB,DE
83485 EXERCISE?/TI,AB,DE
S37 1273 (ISOKINETIC OR RESIST?) (2W) EXERCISE?/TI,AB,DE
? s s28:s37
S38 57015 S28:S37
? s s10 and s38
2177 S10
57015 S38
S39 186 S10 AND S38
? s chiropractic?/ti,ab,de
S40 289 CHIROPRACTIC?/TI,AB,DE
? s osteopath?/ti,ab,de
S41 20440 OSTEOPATH?/TI,AB,DE
? s manipulat? (3W) (anesthesia or anaesthesia or anesthetic? or anaesthetic? or musculoskeletal)/ti,ab,de
73206 MANIPULAT?/TI,AB,DE
97112 ANESTHESIA/TI,AB,DE
15154 ANAESTHESIA/TI,AB,DE
37671 ANESTHETIC?/TI,AB,DE
7809 ANAESTHETIC?/TI,AB,DE
7339 MUSCULOSKELETAL/TI,AB,DE
S42 130 MANIPULAT? (3W) (ANESTHESIA OR ANAESTHESIA OR ANESTHETIC? OR ANAESTHETIC? OR MUSCULOSKELETAL)/TI,AB,DE
? s MUA/ti,ab,de
S43 258 MUA/TI,AB,DE
? s s40:s43
S44 21093 S40:S43
? s s10 and s44
2177 S10
21093 S44
S45 48 S10 AND S44
? s (TENS or ALTENS)/ti,ab,de
9868 TENS/TI,AB,DE
1 ALTENS/TI,AB,DE
S46 9869 (TENS OR ALTENS)/TI,AB,DE
? s electric? (2W) stimulat? or transcutaneous (2W) stimulat? or transdermal (2W) electrostimulat? or cutaneous (2W) electrostimulat? or electroanalgesia or electro(W)analgesia/ti,ab,de
640281 ELECTRIC?
347681 STIMULAT?
15569 ELECTRIC?(2W)STIMULAT?
3402 TRANSCUTANEOUS
347681 STIMULAT?
843 TRANSCUTANEOUS(2W)STIMULAT?
4939 TRANSDERMAL
1104 ELECTROSTIMULAT?
6 TRANSDERMAL(2W)ELECTROSTIMULAT?
40793 CUTANEOUS
1104 ELECTROSTIMULAT?
0 CUTANEOUS(2W)ELECTROSTIMULAT?
22 ELECTROANALGESIA
31673 ELECTRO/TI,AB,DE
20137 ANALGESIA/TI,AB,DE
4 ELECTRO/TI,AB,DE(W)ANALGESIA/TI,AB,DE
S47 15738 ELECTRIC? (2W) STIMULAT? OR TRANSCUTANEOUS (2W) STIMULAT? OR TRANSDERMAL (2W) ELECTROSTIMULAT? OR CUTANEOUS (2W) ELECTROSTIMULAT? OR ELECTROANALGESIA OR ELECTRO(W)ANALGESIA/TI,AB,DE
? s muscle (2W) stimulat?/ti,ab,de
248852 MUSCLE/TI,AB,DE
347681 STIMULAT?/TI,AB,DE
S48 1748 MUSCLE (2W) STIMULAT?/TI,AB,DE
? s neuromodulation or neuro (W) modulation or neurostimulation or neuro (W) stimulation/ti,ab,de
1804 NEUROMODULATION
11977 NEURO
134029 MODULATION
11 NEURO(W)MODULATION
428 NEUROSTIMULATION
11977 NEURO/TI,AB,DE
187063 STIMULATION/TI,AB,DE
17 NEURO/TI,AB,DE(W)STIMULATION/TI,AB,DE
S49 2211 NEUROMODULATION OR NEURO (W) MODULATION OR NEUROSTIMULATION OR NEURO (W) STIMULATION/TI,AB,DE
? s interferential/ti,ab,de
S50 264 INTERFERENTIAL/TI,AB,DE
? s s46:s50
S51 28886 S46:S50
? s s10 and s51
2177 S10
28886 S51
S52 26 S10 AND S51
? s biofeedback/ti,ab,de
S53 2966 BIOFEEDBACK/TI,AB,DE
? s s10 and s53
2177 S10
2966 S53
S54 0 S10 AND S53
? s (cold or ice or heat or hot) (W) (pack? or therap? or treat?)/ti,ab,de
95776 COLD/TI,AB,DE
45475 ICE/TI,AB,DE
392650 HEAT/TI,AB,DE
106420 HOT/TI,AB,DE
138939 PACK?/TI,AB,DE
661276 THERAP?/TI,AB,DE
1969126 TREAT?/TI,AB,DE
S55 87567 (COLD OR ICE OR HEAT OR HOT) (W) (PACK? OR THERAP? OR TREAT?)/TI,AB,DE
? s thermograph? or thermotherap? or thermo (W) therap? or hypertherm? or hyper (W) therm? or diatherm? or cryotherap? or cryo(W) therap?/ti,ab,de
7907 THERMOGRAPH?
1946 THERMOTHERAP?
16853 THERMO
661276 THERAP?
20 THERMO(W)THERAP?
15178 HYPERTHERM?
9780 HYPER
1038992 THERM?
48 HYPER(W)THERM?
1188 DIATHERM?
2472 CRYOTHERAP?
2537 CRYO/TI,AB,DE
661276 THERAP?/TI,AB,DE
5 CRYO/TI,AB,DE(W)THERAP?/TI,AB,DE
S56 27977 THERMOGRAPH? OR THERMOTHERAP? OR THERMO (W) THERAP? OR HYPERTHERM? OR HYPER (W) THERM? OR DIATHERM? OR CRYOTHERAP? OR CRYO(W) THERAP?/TI,AB,DE
? s s55:s56
S57 115050 S55:S56
? s s10 and s57
2177 S10
115050 S57
S58 6 S10 AND S57
? s laser (W) therapy/ti,ab,de
317814 LASER/TI,AB,DE
401563 THERAPY/TI,AB,DE
S59 1412 LASER (W) THERAPY/TI,AB,DE
? s ultrasonic (W) therapy/ti,ab,de
65537 ULTRASONIC/TI,AB,DE
401563 THERAPY/TI,AB,DE
S60 19 ULTRASONIC (W) THERAPY/TI,AB,DE
? s ultrasound/ti,ab,de
S61 87233 ULTRASOUND/TI,AB,DE
? s ultrasonography/ti,ab,de
S62 26006 ULTRASONOGRAPHY/TI,AB,DE
? s electrotherapeutic (W) (intervention? or treat?)/ti,ab,de
17 ELECTROTHERAPEUTIC/TI,AB,DE
201443 INTERVENTION?/TI,AB,DE
1969126 TREAT?/TI,AB,DE
S63 3 ELECTROTHERAPEUTIC (W) (INTERVENTION? OR TREAT?)/TI,AB,DE
? s s59:s63
S64 107999 S59:S63
? s s10 and s64
2177 S10
107999 S64
S65 92 S10 AND S64
? s pulsed (W) electromagnetic (W) field (W) therapy/ti,ab,de
67481 PULSED/TI,AB,DE
103327 ELECTROMAGNETIC/TI,AB,DE
810883 FIELD/TI,AB,DE
401563 THERAPY/TI,AB,DE
S66 7 PULSED (W) ELECTROMAGNETIC (W) FIELD (W) THERAPY/TI,AB,DE
? s (electromagnetic? or magnetic?) (3W) field?/ti,ab,de
105704 ELECTROMAGNETIC?/TI,AB,DE
508832 MAGNETIC?/TI,AB,DE
896260 FIELD?/TI,AB,DE
S67 167375 (ELECTROMAGNETIC? OR MAGNETIC?) (3W) FIELD?/TI,AB,DE
? s biomagnetic? or bio (W) magnetic? or pulsed signal/ti,ab,de
304 BIOMAGNETIC?
16712 BIO
508832 MAGNETIC?
10 BIO(W)MAGNETIC?
0 PULSED SIGNAL/TI,AB,DE
S68 314 BIOMAGNETIC? OR BIO (W) MAGNETIC? OR PULSED SIGNAL/TI,AB,DE
? s PEMF/ti,ab,de
S69 62 PEMF/TI,AB,DE
? s s66:s69
S70 167554 S66:S69
? s s10 and s70
2177 S10
167554 S70
S71 2 S10 AND S70
? s neuromuscular (W) blockade/ti,ab,de
26402 NEUROMUSCULAR/TI,AB,DE
30108 BLOCKADE/TI,AB,DE
S72 1096 NEUROMUSCULAR (W) BLOCKADE/TI,AB,DE
? s nerve (2W) block?/ti,ab,de
126557 NERVE/TI,AB,DE
266755 BLOCK?/TI,AB,DE
S73 2152 NERVE (2W) BLOCK?/TI,AB,DE
? s s72:s73
S74 3241 S72:S73
? s s10 and s74
2177 S10
3241 S74
S75 23 S10 AND S74
? s acupuncture?/ti,ab,de
S76 4547 ACUPUNCTURE?/TI,AB,DE
? s electroacupuncture? or electro (W) acupuncture?/ti,ab,de
699 ELECTROACUPUNCTURE?
31673 ELECTRO/TI,AB,DE
4547 ACUPUNCTURE?/TI,AB,DE
160 ELECTRO/TI,AB,DE(W)ACUPUNCTURE?/TI,AB,DE
S77 768 ELECTROACUPUNCTURE? OR ELECTRO (W) ACUPUNCTURE?/TI,AB,DE
? s osteopuncture? or osteo (W) puncture?/ti,ab,de
1 OSTEOPUNCTURE?
2078 OSTEO/TI,AB,DE
11990 PUNCTURE?/TI,AB,DE
0 OSTEO/TI,AB,DE(W)PUNCTURE?/TI,AB,DE
S78 1 OSTEOPUNCTURE? OR OSTEO (W) PUNCTURE?/TI,AB,DE
? s perioste? (3W) (stimulat? or therap? or needling)/ti,ab,de
3737 PERIOSTE?/TI,AB,DE
347681 STIMULAT?/TI,AB,DE
661276 THERAP?/TI,AB,DE
240 NEEDLING/TI,AB,DE
S79 19 PERIOSTE? (3W) (STIMULAT? OR THERAP? OR NEEDLING)/TI,AB,DE
? s s76:s79
S80 4792 S76:S79
? s s10 and s80
2177 S10
4792 S80
S81 23 S10 AND S80
? s massag? or acupressure or shiatsu or shiatzu or zhi (W) ya or chih (W) ya/ti,ab,de
2665 MASSAG?
92 ACUPRESSURE
18 SHIATSU
0 SHIATZU
97 ZHI
1802 YA
0 ZHI(W)YA
36 CHIH/TI,AB,DE
1802 YA/TI,AB,DE
0 CHIH/TI,AB,DE(W)YA/TI,AB,DE
S82 2754 MASSAG? OR ACUPRESSURE OR SHIATSU OR SHIATZU OR ZHI (W) YA OR CHIH (W) YA/TI,AB,DE
? s s10 and s82
2177 S10
2754 S82
S83 6 S10 AND S82
? s rehabilitat? (2W) (program? or protocol?)/ti,ab,de
42771 REHABILITAT?/TI,AB,DE
496453 PROGRAM?/TI,AB,DE
137423 PROTOCOL?/TI,AB,DE
S84 2831 REHABILITAT? (2W) (PROGRAM? OR PROTOCOL?)/TI,AB,DE
? s s10 and s84
2177 S10
2831 S84
S85 22 S10 AND S84
? s watch? (2W) wait? or conservative (2W) treat?/ti,ab,de
5496 WATCH?
15532 WAIT?
579 WATCH?(2W)WAIT?
29616 CONSERVATIVE/TI,AB,DE
1969126 TREAT?/TI,AB,DE
5597 CONSERVATIVE/TI,AB,DE(2W)TREAT?/TI,AB,DE
S86 6168 WATCH? (2W) WAIT? OR CONSERVATIVE (2W) TREAT?/TI,AB,DE
? s s10 and s86
2177 S10
6168 S86
S87 36 S10 AND S86
? s management (2W) (decision? or option? or choice?)/ti,ab,de
441599 MANAGEMENT/TI,AB,DE
154755 DECISION?/TI,AB,DE
66900 OPTION?/TI,AB,DE
104349 CHOICE?/TI,AB,DE
S88 7785 MANAGEMENT (2W) (DECISION? OR OPTION? OR CHOICE?)/TI,AB,DE
? s s10 and s88
2177 S10
7785 S88
S89 1 S10 AND S88
? s s85 or s87 or s89
22 S85
36 S87
1 S89
S90 58 S85 OR S87 OR S89
? s s16 or s20 or s27 or s39 or s45 or s52 or s54 or s58 or s65 or s71 or s75 or s81 or s83 or s90
10 S16
99 S20
229 S27
186 S39
48 S45
26 S52
0 S54
6 S58
92 S65
2 S71
23 S75
23 S81
6 S83
58 S90
S91 631 S16 OR S20 OR S27 OR S39 OR S45 OR S52 OR S54 OR S58 OR S65 OR S71 OR S75 OR S81 OR S83 OR S90
Physiotherapy Evidence Database (PEDro)
Interface: http://www.pedro.org.au/
Search date: 19 April 2010
Strategy: The database interface does not allow sophisticated searches to be undertaken so a broad search was carried out using the advanced search option. The body part ‘upper arm, shoulder or shoulder girdle’ was selected from the dropdown list provided and all the records identified from this search were downloaded.
Records identified: 718
PREMEDLINE
Interface: OvidSP
Search date: 25 March 2010
Strategy: as below
Records identified: 82
Database: Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations <March 24, 2010>
-
(frozen adj6 shoulder$).ti,ab. (22)
-
(stiff$adj3 shoulder$).ti,ab. (16)
-
(adhesive adj (capsulitis or capsulitides)).ti,ab. (19)
-
((bursitis or bursitides) adj6 shoulder$).ti,ab. (0)
-
((capsulitis or capsulitides) adj6 shoulder$).ti,ab. (9)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ti,ab. (7)
-
(shoulder$adj3 (pain or pains or painful or complain$)).ti,ab. (191)
-
(shoulder$adj6 impinge$).ti,ab. (23)
-
subacromial impingement syndrome.ti,ab. (11)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 (245)
-
(arthrograph$adj6 (distension$or distention$)).ti,ab. (1)
-
(arthrogram$adj6 (distension$or distention$)).ti,ab. (0)
-
(glenohumeral adj6 (distension$or distention$)).ti,ab. (0)
-
(dilatation or hydrodilat$).ti,ab. (780)
-
(arthroscop$adj6 (releas$or decompress$or capsulotom$)).ti,ab. (25)
-
((capsular adj2 releas$) or interventional microadhesiolysis or capsulotomy).ti,ab. (43)
-
((bursa$or intrabursa$or intra bursa$or periartic$or peri artic$or intraartic$or intra artic$) adj3 inject$).ti,ab. (92)
-
((subacromial or acromioclavicular or glenohumeral) adj3 inject$).ti,ab. (8)
-
((extra articular or extraarticular or shoulder$) adj3 inject$).ti,ab. (1)
-
(physiotherapy or physiotherapies or physical therap$or manual therap$).ti,ab. (929)
-
(passive adj (motion or movement)).ti,ab. (51)
-
CPM.ti,ab. (129)
-
(stretching or stretches).ti,ab. (2719)
-
(mobilisation or mobilization).ti,ab. (806)
-
(exercise$adj2 (program$or strength$or intervention$or training or prescription$or prescrib$)).ti,ab. (641)
-
(exercise$adj2 (therap$or therapeutic)).ti,ab. (112)
-
((home or supervis$) adj2 exercis$).ti,ab. (89)
-
((pendular or pendulum) adj exercis$).ti,ab. (1)
-
((isokinetic or resist$) adj2 exercise$).ti,ab. (126)
-
chiropractic$.ti,ab. (454)
-
osteopath$.ti,ab. (136)
-
(manipulat$adj3 (anesthesia or anaesthesia or anesthetic$or anaesthetic$)).ti,ab. (13)
-
MUA.ti,ab. (75)
-
(TENS or ALTENS).ti,ab. (1018)
-
((electric$adj2 stimulat$) or (transcutaneous adj2 stimulat$) or (transdermal adj2 electrostimulat$) or (cutaneous adj2 electrostimulat$) or electroanalgesia or electro analgesia).ti,ab. (759)
-
(muscle adj2 stimulat$).ti,ab. (93)
-
(neuromodulation or neuro modulation or neurostimulation or neuro stimulation).ti,ab. (93)
-
interferential.ti,ab. (41)
-
biofeedback.ti,ab. (119)
-
((cold or ice or heat or hot) adj (pack$or therap$or treat$)).ti,ab. (661)
-
(thermograph$or thermotherap$or thermo therap$or hypertherm$or hyper therm$or diatherm$or cryotherap$or cryo therap$).ti,ab. (821)
-
((laser or ultrasonic) adj2 (therap$or treat$)).ti,ab. (384)
-
ultrasound.ti,ab. (4964)
-
(electrotherapeutic adj (intervention$or treat$)).ti,ab. (2)
-
pulsed electromagnetic field therapy.ti,ab. (1)
-
((electromagnetic$or magnetic$) adj3 field$).ti,ab. (6171)
-
(biomagnetic$or bio magnetic$or pulsed signal).ti,ab. (29)
-
PEMF.ti,ab. (9)
-
(nerve adj2 block$).ti,ab. (233)
-
neuromuscular block$.ti,ab. (117)
-
acupuncture$.ti,ab. (676)
-
(electroacupuncture$or electro acupuncture$).ti,ab. (130)
-
(osteopuncture$or osteo puncture$).ti,ab. (0)
-
(perioste$adj3 (stimulat$or therap$or needling)).ti,ab. (4)
-
(massag$or acupressure or shiatsu or shiatzu or zhi ya or chih ya).ti,ab. (223)
-
(rehabilitat$adj2 (program$or protocol$)).ti,ab. (373)
-
((watch$adj2 wait$) or (conservative adj2 treat$)).ti,ab. (690)
-
(management adj2 (decision$or option$or choice$)).ti,ab. (328)
-
or/11–58 (23503)
-
10 and 59 (82)
-
limit 60 to yr=“1966 -Current” (82)
-
from 61 keep 1–82 (82)
-
from 62 keep 1–82 (82)
Science Citation Index
Interface: Web of Science (ISI) via Web of Knowledge
Search date: 30 March 2010
Strategy: as below
Records identified: 3140
| # 20 | 3140 |
#19 OR #17 OR #15 OR #13 OR #11 OR #9 OR #5 OR #3 Timespan=All Years |
| # 19 | 729 |
#18 AND #1 Timespan=All Years |
| # 18 | > 100,000 |
Topic=(“nerve block”) OR Topic=(“neuromuscular blockade”) OR Topic=(acupuncture) OR Topic=(electroacupuncture or “electro acupuncture”) OR Topic=(osteopuncture or “osteo puncture”) OR Topic=(“perioste* stimulat*” or “perioste* therap*” or “perioste* needling”) OR Topic=(massag* or acupressure or shiatsu or shiatzu or “zhi ya” or “chih ya”) OR Topic=(“rehabilitat* program*” or “rehabilitat* protocol*”) OR Topic=(“watch* wait*” or “conservative treat*”) OR Topic=(“management decision*” or “management option*” or “management choice*”) Timespan=All Years |
| # 17 | 221 |
#16 AND #1 Timespan=All Years |
| # 16 | > 100,000 |
Topic=(biofeedback) OR Topic=(thermograph* or thermotherap* or “thermo therap*” or hypertherm* or “hyper therm*” or diatherm* or cryotherap* or “cryo therap*”) OR Topic=(“cold pack*” or “cold therap*” or “cold treat*” or “ice pack*” or “ice therap*” or “ice treat*” or “heat pack*” or “heat therap*” or “heat treat*” or “hot pack*” or “heat therap*”) OR Topic=(“Laser Therapy” or “ultrasonic therapy” or “Interventional ultrasonography”) OR Topic=(“electrotherapeutic intervention*” or “electrotherapeutic treat*”) OR Topic=(“magnetic field therapy”) OR Topic=(“electromagnetic field therapy”) OR Topic=(“Biomagnetic therapy”) Timespan=All Years |
| # 15 | 162 |
#14 AND #1 Timespan=All Years |
| # 14 | > 100,000 |
Topic=(TENS or ALTENS) OR Topic=(“electric* stimulat*” or “transcutaneous stimulat*”or “transdermal electrostimulat*”or “cutaneous electrostimulat*”or electroanalgesia or “electro analgesia”) OR Topic=(“muscle stimulat*”) OR Topic=(neuromodulation or “neuro modulation” or neurostimulation or “neuro stimulation”) OR Topic=(interferential) Timespan=All Years |
| # 13 | 184 |
#12 AND #1 Timespan=All Years |
| # 12 | 19,962 |
Topic=(chiropractic*) OR Topic=(osteopath*) OR Topic=(manipulat* and (anesthesia or anaesthesia or anesthetic* or anaesthetic*)) OR Topic=(MUA) Timespan=All Years |
| # 11 | 1502 |
#10 AND #1 Timespan=All Years |
| # 10 | > 100,000 |
Topic=(physiotherapy or physiotherapies or “physical therap*” or “manual therap*”) OR Topic=(“passive motion” or “passive movement”) OR Topic=(“muscle stretching” or “muscle stretches” or “muscle stretch”) OR Topic=(mobilisation or mobilization) OR Topic=(“exercis* program*” or “exercis* intervention*” or “exercis* train*” or “exercis* prescription*” or “exercis* prescrib*”) OR Topic=(“exercis* therap*” or “therapeutic exercis*”) OR Topic=(“home exercis*” or “supervis* exercis*”) OR Topic=(“pendular exercis*” or “pendulum exercis*”) OR Topic=(“isokinetic exercis*” or “resist* exercise*”) Timespan=All Years |
| # 9 | 609 |
#8 OR #7 Timespan=All Years |
| # 8 | 121 |
Topic=(“subacromial inject*” or “acromioclavicular inject*” or “glenohumeral inject*”) OR Topic=(“extra articular inject*” or “extraarticular inject*”) OR Topic=(“shoulder* inject*”) Timespan=All Years |
| # 7 | 533 |
#6 AND #1 Timespan=All Years |
| # 6 | > 100,000 |
Topic=(injections) OR Topic=(“intraarticular injection*” or “intra-articular injection*” or “intra articular injection*”) OR Topic=(“bursa* inject*” or “intrabursa* inject*” or “intra bursa* inject*” or “periartic* inject*” or “peri artic* inject*”) Timespan=All Years |
| # 5 | 800 |
#4 AND #1 Timespan=All Years |
| # 4 | 20,598 |
Topic=(Arthroscopy) OR Topic=(“arthroscop* releas*” or “arthroscop* decompress*” or “arthroscop* capsulotom*”) OR Topic=(“capsular releas*” or “interventional microadhesiolysis” or capsulotomy) Timespan=All Years |
| # 3 | 12 |
#2 AND #1 Timespan=All Years |
| # 2 | 67 |
Topic=(“arthrograph* distension*” or “arthograph* disten”) OR Topic=(“arthrogram* distension*” or “arthogram* distention*”) OR Topic=(“glenohumeral distension*” or “glenohumeral distention*”) OR Topic=(arthography) Timespan=All Years |
| # 1 | 8973 |
Topic=(“frozen shoulder*”) OR Topic=(“stiff* shoulder*”) OR Topic=(“adhesive capsulitis”) OR Topic=(“shoulder bursitis”) OR Topic=(periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) OR Topic=(“shoulder pain*” or “pain* shoulder” or “shoulder complaint*”) OR Topic=(“shoulder* impinge*”) OR Topic=(“subacromial impingement syndrome”) Timespan=All Years |
Additional quality of life search strategy for information to inform the decision-analytic model
Database: Ovid MEDLINE(R) <1950 to October Week 4 2010>
-
(sf36 or sf 36).tw. (9054)
-
(eq5d or eq 5d or euroqol or euro qol).tw. (2024)
-
(short form 36 or shortform 36 or sf thirtysix or sf thirty six or shortform thirtysix or shortform thirty six or short form thirtysix or short form thirty six).tw. (4194)
-
(hrql or hrqol or h qol or hql or hqol).tw. (5810)
-
(hye or hyes or health$year$equivalent$or health utilit$).tw. (733)
-
health related quality of life.tw. (13228)
-
rosser.tw. (67)
-
(standard gamble$or time trade off or time tradeoff or tto or willingness to pay).tw. (2460)
-
(disutilities or disutility or daly or disability adjusted life).tw. (1022)
-
“Quality of Life”/(87323)
-
health status indicators/(15760)
-
quality adjusted life year/(4741)
-
(qaly$or quality adjusted life or quality of life or life quality).tw. (98027)
-
qwb$.tw. (157)
-
(quality of wellbeing or quality of well being).tw. (275)
-
factor analysis.tw. (15866)
-
preference based.tw. (418)
-
health status.tw. (27567)
-
(state adj2 (value or values or valuing or valued)).tw. (1507)
-
hspv.tw. (2)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 (185879)
-
“VAS 0–100mm”.mp. (4)
-
“VAS 0–10cm”.mp. (1)
-
(VAS and pain).mp. (7387)
-
10 point numeric scale.mp. (5)
-
“4 point category rating scale”.mp. (0)
-
Category rating scale.mp. (24)
-
Likert scale 0–10.mp. (2)
-
(likert scale and pain).mp. (295)
-
SPADI 5-item pain subscale.mp. (0)
-
SPADI.mp. (76)
-
22 or 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 (7739)
-
21 and 32 (1173)
-
American Shoulder & Elbow Surgeons score.mp. (2)
-
Constant score.mp. (869)
-
Flexilevel Scale of Shoulder Function.mp. (6)
-
Oxford shoulder scale.mp. (1)
-
Problem Elicitation Technique score.mp. (1)
-
Shoulder Disability score.mp. (1)
-
SPADI total score.mp. (3)
-
UCLA shoulder score.mp. (24)
-
Shoulder Disability Questionnaire.mp. (28)
-
Croft score.mp. (1)
-
DASH score.mp. (420)
-
(shoulder and Telephone questionnaire).mp. (4)
-
34 or 35 or 36 or 37 or 38 or 39 or 40 or 41 or 42 or 43 or 44 or 45 (1313)
-
21 and 46 (131)
-
33 or 47 (1282)
Appendix 2 Search strategy for review of views of people with frozen shoulder
Three databases (CINAHL, MEDLINE and PsycINFO) were searched to identify qualitative studies, with searches being restricted to English-language studies published from 1980 onwards. The search strategies used are given in full below.
Cumulative Index to Nursing and Allied Health Literature (CINAHL)
Interface: EBSCO
Search date: 3 June 2010, date coverage 1980–2010
Strategy: as below
Records identified: 311
| S24 | (S21 and S22 and S23) |
| S23 | (ZR “1980”) or (ZR “1981”) or (ZR “1982”) or (ZR “1983”) or (ZR “1984”) or (ZR “1985”) or (ZR “1986”) or (ZR “1987”) or (ZR “1988”) or (ZR “1989”) or (ZR “1990”) or (ZR “1991”) or (ZR “1992”) or (ZR “1993”) or (ZR “1994”) or (ZR “1995”) or (ZR “1996”) or (ZR “1997”) or (ZR “1998”) or (ZR “1999”) or (ZR “2000”) or (ZR “2001”) or (ZR “2002”) or (ZR “2003”) or (ZR “2004”) or (ZR “2005”) or (ZR “2006”) or (ZR “2007”) or (ZR “2008”) or (ZR “2009”) or (ZR “2010”) |
| S22 | (ZL “english”) |
| S21 | (S15 or S16 or S17 or S18 or S19) and (S14) |
| S20 | S15 or S16 or S17 or S18 or S19 |
| S19 | TX qualitative |
| S18 | (ZU “interviews”) |
| S17 | TX interview* |
| S16 | TX findings |
| S15 | TX findings |
| S14 | S1 or S2 or S3 or S4 or S5 or S6 or S7 or S8 or S9 or S10 or S11 or S12 or S13 |
| S13 | subacromial impingement syndrome |
| S12 | shoulder* N6 impinge* |
| S11 | (MH “Shoulder Impingement Syndrome”) |
| S10 | shoulder* N3 pain or shoulder* N3 pains or shoulder* N3 painful or shoulder* N3 complain* |
| S9 | (MH “Shoulder Pain”) |
| S8 | periarthritis N6 shoulder* or peri-arthritis N6 shoulder* or periarthritides N6 shoulder* or peri-arthritides N6 shoulder* or pericapsulitis N6 shoulder* or peri-capsulitis N6 shoulder* |
| S7 | capsulitis N6 shoulder* |
| S6 | bursitides N6 shoulder* |
| S5 | bursitis N6 shoulder* |
| S4 | adhesive N3 capsulitis |
| S3 | (MH “Adhesive Capsulitis”) |
| S2 | stiff* N3 shoulder* |
| S1 | frozen N6 shoulder* |
MEDLINE
Interface: OvidSP
Search date: 3 June 2010
Strategy: as below
Records identified: 915
Database: Ovid MEDLINE(R) <1950 to May Week 4 2010>
-
(frozen adj6 shoulder$).ti,ab. (599)
-
(stiff$adj3 shoulder$).ti,ab. (361)
-
exp bursitis/(3130)
-
(adhesive adj (capsulitis or capsulitides)).ti,ab. (336)
-
((bursitis or bursitides) adj6 shoulder$).ti,ab. (65)
-
((capsulitis or capsulitides) adj6 shoulder$).ti,ab. (195)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ti,ab. (176)
-
shoulder pain/(1861)
-
(shoulder$adj3 (pain or pains or painful or complain$)).ti,ab. (4029)
-
Shoulder Impingement Syndrome/(953)
-
(shoulder$adj6 impinge$).ti,ab. (505)
-
subacromial impingement syndrome.ti,ab. (117)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 (9213)
-
findings.af. (930557)
-
interview$.af. (174135)
-
Interview/(20706)
-
qualitative.af. (81067)
-
14 or 15 or 16 or 17 (1132990)
-
13 and 18 (1085)
-
limit 19 to english language (928)
-
limit 20 to yr=“1980 -Current” (915)
PsycINFO
Interface: OvidSP
Search date: 3 June 2011
Strategy: as below
Records identified: 79
Database: PsycINFO <1967 to May Week 4 2010>
-
(frozen adj6 shoulder$).ti,ab. (6)
-
(stiff$adj3 shoulder$).ti,ab. (22)
-
(adhesive adj (capsulitis or capsulitides)).ti,ab. (2)
-
((bursitis or bursitides) adj6 shoulder$).ti,ab. (2)
-
((capsulitis or capsulitides) adj6 shoulder$).ti,ab. (2)
-
((periarthritis or peri-arthritis or periarthritides or peri-arthritides or peri-capsulitis or pericapsulitis) adj6 shoulder$).ti,ab. (0)
-
(shoulder$adj3 (pain or pains or painful or complain$)).ti,ab. (234)
-
(shoulder$adj6 impinge$).ti,ab. (7)
-
subacromial impingement syndrome.ti,ab. (1)
-
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 (263)
-
findings.af. (407232)
-
interview$.af. (228353)
-
interviews/(4877)
-
qualitative.af. (117407)
-
11 or 12 or 13 or 14 (613112)
-
10 and 15 (81)
-
limit 16 to (english language and yr=“1980 -Current”) (79)
-
from 17 keep 1–79 (79)
Appendix 3 R code for standard deviation
Appendix 4 Quality assessment checklist
| Study quality assessment for RCTs and controlled trials | |
|---|---|
| 1. Was the number of participants randomised stated? | 1.1 Yes |
| 1.2 No | |
| 1.3 Unclear | |
| 1.4 Not applicable (N/A) | |
| 2. Was the method of randomisation adequate (e.g. use of random number table, computer random number generator, coin tossing, shuffling of cards or envelopes, throwing of dice)? | 2.1 Yes |
| 2.2 No | |
| 2.3 Unclear | |
| 2.4 Not applicable (N/A) | |
| 3. Was allocation concealment adequate (e.g. central allocation, sequentially numbered opaque sealed envelopes)? | 3.1 Yes |
| 3.2 No | |
| 3.3 Unclear | |
| 3.4 Not applicable (N/A) | |
| 4. Were the treatment groups comparable at baseline for important prognostic factors? | 4.1 Yes |
| 4.2 No | |
| 4.3 Unclear | |
| 4.4 Not applicable (N/A) | |
| 5. Was a suitable statistical method used to adjust for possible baseline imbalance? | 5.1 Yes |
| 5.2 No | |
| 5.3 Unclear | |
| 5.4 Not applicable (N/A) | |
| 6. Was the study reported as being at least double blind? | 6.1 Yes |
| 6.2 No | |
| 6.3 Unclear | |
| 6.4 Not applicable (N/A) | |
| 7. Were patients blinded? | 7.1 Yes |
| 7.2 No | |
| 7.3 Unclear | |
| 7.4 Not applicable (N/A) | |
| 8. Were outcome assessors blinded? | 8.1 Yes |
| 8.2 No | |
| 8.3 Unclear | |
| 8.4 Not applicable (N/A) | |
| 9. Were caregivers blinded? | 9.1 Yes |
| 9.2 No | |
| 9.3 Unclear | |
| 9.4 Not applicable (N/A) | |
| 10. Was ITT analysis used (in the analysis, participants were kept in the intervention groups to which they were randomised, regardless of the intervention they received)? | 10.1 Yes |
| 10.2 No | |
| 10.3 Unclear | |
| 10.4 Not applicable (N/A) | |
| 11. Were there any unexpected imbalances in dropouts between groups? | 11.1 Yes |
| 11.2 No | |
| 11.3 Unclear | |
| 11.4 Not applicable (N/A) | |
| 12. If there were any unexpected imbalances in dropouts were they explained or adjusted for? | 12.1 Yes |
| 12.2 No | |
| 12.3 Unclear | |
| 12.4 Not applicable (N/A) | |
| 13. Was the study powered for at least one outcome? | 13.1 Yes |
| 13.2 No | |
| 13.3 Unclear | |
| 13.4 Not applicable (N/A) | |
| Study quality assessment for case series | |
| 14. Were selection/eligibility criteria adequately reported? | 14.1 Yes |
| 14.2 No | |
| 14.3 Unclear | |
| 14.4 Not applicable (N/A) | |
| 15. Was the selected population representative of that seen in normal practice? | 15.1 Yes |
| 15.2 No | |
| 15.3 Unclear | |
| 15.4 Not applicable (N/A) | |
| 16. Was an appropriate measure of variability reported? | 16.1 Yes |
| 16.2 No | |
| 16.3 Unclear | |
| 16.4 Not applicable (N/A) | |
| 17. Was loss to follow-up reported or explained? | 17.1 Yes |
| 17.2 No | |
| 17.3 Unclear | |
| 17.4 Not applicable (N/A) | |
| 18. Were at least 90% of those included at baseline followed up? | 18.1 Yes |
| 18.2 No | |
| 18.3 Unclear | |
| 18.4 Not applicable (N/A) | |
| 19. Were patients recruited prospectively | 19.1 Yes |
| 19.2 No | |
| 19.3 Unclear | |
| 19.4 Not applicable (N/A) | |
| 20. Were patient recruited consecutively? | 20.1 Yes |
| 20.2 No | |
| 20.3 Unclear | |
| 20.4 Not applicable (N/A) | |
| 21. Did the study report relevant prognostic factors? | 21.1 Yes |
| 21.2 No | |
| 21.3 Unclear | |
| 21.4 Not applicable (N/A) | |
| 22. Any other additional limitations? | 22.1 Yes |
| 22.2 No | |
| 22.3 Unclear | |
| 22.4 Not applicable (N/A) | |
Appendix 5 List of excluded studies
| Study | Population | Intervention | Comparison | Outcome | Study design | Not a clinical study | Other |
|---|---|---|---|---|---|---|---|
| NCT00160784128 | ×a | ||||||
| NCT00840229129 | ×a | ||||||
| NCT00679887130 | × | ||||||
| NCT00261196131 | ×b | ||||||
| NCT00873158132 | ×a | ||||||
| NCT01087229133 | × | ||||||
| NCT00884065134 | × | ||||||
| NCT00415441135 | × | ||||||
| NCT00742846136 | ×a | ||||||
| NCT00694538137 | × | ||||||
| NCT00680472138 | × | ||||||
| NCT00377624139 | × | ||||||
| NCT00211718140 | × | ||||||
| NCT00929305141 | × | ||||||
| NCT00172601142 | ×a | ||||||
| NCT00587626143 | × | ||||||
| NCT00163124144 | × | ||||||
| NCT00992927145 | ×a | ||||||
| NCT00875862146 | × | ||||||
| NCT01029600147 | ×a | ||||||
| Anonymous 1999148 | × | ||||||
| Anonymous 2000149 | |||||||
| Anonymous 2001150 | |||||||
| Anonymous 2004151 | × | ||||||
| Anonymous 2005152 | × | ||||||
| Anonymous 2005153 | × | ||||||
| Anonymous 2009154 | × | ||||||
| Ahmad 2009155 | × | ||||||
| Ahn 2008156 | × | ||||||
| Ainsworth 2007157 | × | ||||||
| Alegre Marcet 1959158 | × | ||||||
| Alexander 2010159 | × | ||||||
| Allano 2005160 | × | ||||||
| Altman 2005161 | × | ||||||
| Alvado 200126 | ×c | ||||||
| Andersen 1998162 | × | ||||||
| Andersen 1996163 | × | ||||||
| Andren 1965164 | × | ||||||
| Ankermann 1986165 | × | ||||||
| Aoki 1982166 | × | ||||||
| Aoki 1991167 | × | ||||||
| Arias 1992168 | × | ||||||
| Arroll 200523 | × | ||||||
| Arslan 2001169 | × | ||||||
| Atra 1986170 | × | ||||||
| Avetisova 1980171 | ×d | ||||||
| Awad 1967172 | × | ||||||
| Azevedo 2008173 | × | ||||||
| Badalamente 2006174 | ×e | ||||||
| Badalamente 2009175 | ×b | ||||||
| Bancheri 1993176 | × | ||||||
| Baslund 1991177 | × | ||||||
| Batra 1985178 | × | ||||||
| Battisti 2007179 | × | ||||||
| Battisti 2009180 | × | ||||||
| Baumann 1981181 | × | ||||||
| Baumgartner 1981182 | × | ||||||
| Baums 2007183 | × | ||||||
| Beaufils 1996184 | × | ||||||
| Beaufils 1999185 | × | ||||||
| Beckerman 1993186 | × | ||||||
| Bell 2003187 | × | ||||||
| Bellmann 1969188 | × | ||||||
| Bennett 2000189 | × | ||||||
| Beres 1979190 | × | ||||||
| Berger 1980191 | × | ||||||
| Berghs 2004192 | × | ||||||
| Berglezov 1986193 | × | ||||||
| Bergman 2002194 | × | ||||||
| Bergman 2004195 | × | ||||||
| Bergman 2005196 | × | ||||||
| Bergman 2010197 | × | ||||||
| Bergman 2010121 | × | ||||||
| Bertoft 1999198 | × | ||||||
| Bettermann 1982199 | × | ||||||
| Bicer 2005200 | × | ||||||
| Bierner 1989201 | × | ||||||
| Bilgici 2002202 | ×f | ||||||
| Bingol 2005203 | × | ||||||
| Biswas 1979204 | × | ||||||
| Blaine 2008205 | × | ||||||
| Blanchard 1999206 | × | ||||||
| Blauth 1989207 | × | ||||||
| Booi 1986208 | × | ||||||
| Bosch Olives 1967209 | × | ||||||
| Boyer 1994210 | × | ||||||
| Boylan 2005211 | × | ||||||
| Boylan 2005212 | × | ||||||
| Boyles 2005213 | × | ||||||
| Bratanova 1978214 | × | ||||||
| Brigo 1981215 | × | ||||||
| Brox 2003216 | × | ||||||
| Buchbinder 2000217 | ×g | ||||||
| Buchbinder 2003218 | × | ||||||
| Buchbinder 2004219 | × | ||||||
| Buchbinder 2004220 | × | ||||||
| Buchbinder 2006221 | ×g | ||||||
| Buchbinder 2006222 | ×g | ||||||
| Buchbinder 2006223 | × | ||||||
| Buchbinder 2007224 | ×h | ||||||
| Buchbinder 2008225 | ×c | ||||||
| Bulgen 1984226 | ×h | ||||||
| Bumin 2001227 | ×g | ||||||
| Bunker 1998228 | × | ||||||
| Calabro 1982229 | × | ||||||
| Caldwell 1986230 | ×g | ||||||
| Callinan 2003231 | × | ||||||
| Camarinos 2009232 | ×e | ||||||
| Caniggia 1989233 | × | ||||||
| Capone 1994234 | × | ||||||
| Carette 2002235 | ×i | ||||||
| Carey 2008236 | × | ||||||
| Carter 2002237 | × | ||||||
| Casanova 1988238 | × | ||||||
| Castellarin 2004239 | × | ||||||
| Castelli 2006240 | × | ||||||
| Champion 2005241 | × | ||||||
| Chang 2009242 | × | ||||||
| Chavero Carrasco 2002243 | × | ||||||
| Chavez-Lopez 2009244 | × | ||||||
| Checchia 1991245 | ×g | ||||||
| Chen 1985246 | × | ||||||
| Chen 1988247 | × | ||||||
| Chen 2002248 | × | ||||||
| Chen 2005249 | × | ||||||
| Chen 2006250 | ×g | ||||||
| Chen 2006251 | × | ||||||
| Chen 2008252 | × | ||||||
| Chen 2009253 | × | ||||||
| Cheng 2008254 | × | ||||||
| Cherkashin 1969255 | ×d | ||||||
| Cho 2005256 | × | ||||||
| Ciapetti 2006257 | × | ||||||
| Cinar 2010258 | × | ||||||
| Cleland 200225 | ×c | ||||||
| Cloke 2008259 | × | ||||||
| Cohen 2000260 | ×h | ||||||
| Compernolle 1987261 | × | ||||||
| Connolly 1972262 | × | ||||||
| Connolly 1998263 | × | ||||||
| Coombes 1984264 | × | ||||||
| Corazza 1982265 | ×j | ||||||
| Corbeil 1992266 | × | ||||||
| Cossu 1993267 | × | ||||||
| Coudane 1988268 | × | ||||||
| Crawshaw 2009269 | × | ||||||
| Dacre 1987270 | ×j | ||||||
| Dahan 2000271 | × | ||||||
| Dal Conte 1990272 | × | ||||||
| Danneskiold-Samsoe 1996273 | × | ||||||
| De Bruijn 2007118 | × | ||||||
| de Jong 19914 | × | ||||||
| de Jong 1998274 | ×h | ||||||
| de la Serna 2004275 | × | ||||||
| de Macedo 2000276 | × | ||||||
| de Seze 1950277 | × | ||||||
| Debeyre 1971278 | × | ||||||
| Degen 1974279 | ×d | ||||||
| Denicolai 1986280 | × | ||||||
| Desproges-Gotteron 1980281 | × | ||||||
| Devitt 2002282 | × | ||||||
| Deyle 1999283 | × | ||||||
| Dias 20055 | × | ||||||
| DiMarcantonio 2006284 | × | ||||||
| Diwan 2005285 | × | ||||||
| Dodenhoff 2000286 | × | ||||||
| Dogru 2008287 | ×k | ||||||
| Dorian 1985288 | × | ||||||
| D’Orta 1985289 | ×g,j | ||||||
| Drakos 2008290 | × | ||||||
| Duke 1981291 | × | ||||||
| Duschatko 2000292 | × | ||||||
| Echternach 1966293 | × | ||||||
| Ekelund 1992294 | ×g | ||||||
| Elleuch 1994295 | × | ||||||
| Elleuch 2008296 | ×m | ||||||
| Erlendsson 1996297 | × | ||||||
| Ernst 2004298 | × | ||||||
| Ernst 2009299 | × | ||||||
| Escalante Triay 1981300 | × | ||||||
| Esposito 1993301 | × | ||||||
| Eulert 1981302 | × | ||||||
| Famaey 1984303 | × | ||||||
| Famaey 1982304 | × | ||||||
| Fan 2008305 | × | ||||||
| Fareed 1989306 | × | ||||||
| Farrell 2005307 | × | ||||||
| Feng 2003308 | × | ||||||
| Filshie 2005309 | × | ||||||
| Flannery 2007310 | × | ||||||
| Foster 2008311 | × | ||||||
| Fuhr 2005312 | × | ||||||
| Fujiwara 1993313 | ×n | ||||||
| Fukuhara 1999314 | × | ||||||
| Fura 1981315 | × | ||||||
| Gabrhelik 2009316 | × | ||||||
| Gado 1996317 | × | ||||||
| Galarraga 2002318 | × | ||||||
| Galgano 2005319 | × | ||||||
| Garrido 2009320 | × | ||||||
| Gaspar 2009321 | × | ||||||
| Gavant 1994322 | × | ||||||
| Geraets 2004323 | × | ||||||
| Geraets 2005324 | × | ||||||
| Geraets 2006120 | × | ||||||
| Gerber 2001325 | × | ||||||
| Gilula 1978326 | × | ||||||
| Ginn 1995327 | × | ||||||
| Ginn 1997328 | × | ||||||
| Ginn 1999329 | × | ||||||
| Ginn 2001330 | × | ||||||
| Ginn 2004331 | × | ||||||
| Ginn 2005332 | × | ||||||
| Ginn 2009333 | × | ||||||
| Ginsberg 1991334 | × | ||||||
| Gobezie 2007335 | × | ||||||
| Goh 1997336 | × | ||||||
| Gotte 1986337 | × | ||||||
| Gotter 1987338 | × | ||||||
| Graber 1997339 | × | ||||||
| Grabovoi 1986340 | ×b | ||||||
| Grammont 1982341 | × | ||||||
| Green 199829 | × | ||||||
| Green 2003342 | ×c | ||||||
| Green 2003343 | × | ||||||
| Green 2005344 | ×c | ||||||
| Green 2006345 | × | ||||||
| Green 2010346 | ×e | ||||||
| Grete 1981347 | × | ||||||
| Griggs 2000348 | × | ||||||
| Grossi 1986349 | × | ||||||
| Grubbs 1993350 | × | ||||||
| Gruehn 1965351 | × | ||||||
| Gu 1992352 | ×g,o | ||||||
| Gudushauri 1975353 | × | ||||||
| Guerra de Hoyos 2004354 | × | ||||||
| Guler-Uysal 2004355 | ×p | ||||||
| Guo 2006356 | × | ||||||
| Guo 2006357 | ×o | ||||||
| Guo 2007358 | × | ||||||
| Gusarova 1989359 | ×d | ||||||
| Gwilym 2007360 | × | ||||||
| Habib 2009361 | × | ||||||
| Haines 1982362 | ×g | ||||||
| Hall 1988363 | × | ||||||
| Hall 2005364 | × | ||||||
| Hamdan 2003365 | × | ||||||
| Hamer 1976366 | × | ||||||
| Han 2006367 | × | ||||||
| Hando 2010368 | × | ||||||
| Hannafin 2000369 | × | ||||||
| Harryman 1993370 | × | ||||||
| Harryman 1997371 | × | ||||||
| Hart 1976372 | × | ||||||
| Hauzeur 2004373 | × | ||||||
| Hay 2001374 | × | ||||||
| Hay 2003375 | × | ||||||
| He 2000376 | ×o | ||||||
| Heber 1983377 | × | ||||||
| Helbig 1983378 | × | ||||||
| Heller 2004379 | × | ||||||
| Hempel 1983380 | × | ||||||
| Herold 1982381 | × | ||||||
| Heuleu 1979382 | × | ||||||
| Hieber 1967383 | × | ||||||
| Ho 2009384 | × | ||||||
| Hoenle 1983385 | ×b | ||||||
| Hollingworth 1983386 | × | ||||||
| Hollis 2006387 | ×g | ||||||
| Hong 1982388 | × | ||||||
| Hormusjee 1980389 | × | ||||||
| Hossain 2008390 | × | ||||||
| Hosseini 2006391 | × | ||||||
| Hsu 1991392 | ×g | ||||||
| Hu 1993393 | × | ||||||
| Hu 2003394 | × | ||||||
| Hu 2004395 | × | ||||||
| Hu 2006396 | × | ||||||
| Huang 1996397 | × | ||||||
| Hulstyn 1993398 | × | ||||||
| Hummel-Berry 2001399 | × | ||||||
| Ibrahim 2006400 | × | ||||||
| Ide 2004401 | ×g | ||||||
| Imai 1983402 | × | ||||||
| Indeck 1990403 | × | ||||||
| Ingram-Rice 2000404 | × | ||||||
| Itel 1988405 | × | ||||||
| Itzkowitch 1996406 | × | ||||||
| Ivanov 1986407 | ×d | ||||||
| Jacchia 1968408 | × | ||||||
| Jacobs 1991409 | ×g | ||||||
| Jacobs 1992410 | ×g | ||||||
| Jacobs 2005411 | ×l | ||||||
| Jayson 1981412 | × | ||||||
| Jensen 1995413 | × | ||||||
| Ji 1988414 | × | ||||||
| Jia 1993415 | × | ||||||
| Jia 2008416 | ×o | ||||||
| Jiang 1982417 | × | ||||||
| Jiang 1991418 | × | ||||||
| Jin 2003419 | × | ||||||
| Johnson 2007420 | ×p | ||||||
| Jones 1997421 | × | ||||||
| Jones 1999422 | × | ||||||
| Joshi 1992423 | × | ||||||
| Judet 1985424 | × | ||||||
| Jurgel 2005425 | × | ||||||
| Kalke 1999426 | × | ||||||
| Kanai 2004427 | × | ||||||
| Kanai 2006428 | × | ||||||
| Kaptelin 1976429 | × | ||||||
| Karatas 2002430 | ×b | ||||||
| Karkan 1985431 | ×q | ||||||
| Katz 2000432 | × | ||||||
| Kay 1981433 | × | ||||||
| Kay 1990434 | × | ||||||
| Keilholz 1995435 | ×b | ||||||
| Kent 1961436 | × | ||||||
| Kent 1985437 | × | ||||||
| Keyl 1982438 | × | ||||||
| Khan 2005439 | × | ||||||
| Khan 2009440 | × | ||||||
| Khitrov 2007441 | ×b | ||||||
| Kirillova 1986442 | × | ||||||
| Kivimaki 1996443 | ×g | ||||||
| Kivimaki 2001444 | ×g | ||||||
| Klinger 2002445 | × | ||||||
| Knebl 2002446 | × | ||||||
| Kneer 1983447 | × | ||||||
| Kneer 1994448 | × | ||||||
| Knusel 1984449 | ×r | ||||||
| Koel 2008450 | × | ||||||
| Kong 2009451 | ×c | ||||||
| Kostadinov 1980452 | × | ||||||
| Koubaa 2006453 | × | ||||||
| Kovacs 1982454 | × | ||||||
| Kucukdeveci 2005455 | × | ||||||
| Kuijpers 2006456 | × | ||||||
| Kulenkampff 1989457 | × | ||||||
| Kuptniratsaikul 2002458 | × | ||||||
| Kurtais Gursel 2004459 | × | ||||||
| Laidley 2004460 | × | ||||||
| Lanfranchi 1968461 | × | ||||||
| Laroche 1998462 | × | ||||||
| Laskowski 1985463 | × | ||||||
| LaStayo 1994464 | × | ||||||
| Latham 1989465 | × | ||||||
| Laznicky 1989466 | × | ||||||
| Lech 1993467 | × | ||||||
| Leclaire 1991468 | × | ||||||
| Lee 1973469 | ×g | ||||||
| Lee 1974470 | ×g | ||||||
| Lee 1986471 | × | ||||||
| Lee 2006472 | ×f | ||||||
| Lee 2009473 | × | ||||||
| Lee 2010474 | ×c | ||||||
| Lehmann 1954475 | ×s | ||||||
| Levenets 1982476 | × | ||||||
| Li 1984477 | ×g | ||||||
| Li 2003478 | ×g | ||||||
| Liang 1973479 | ×s | ||||||
| Liao 2007480 | ×o | ||||||
| Liaw 2000481 | ×s | ||||||
| Lidstrom 1963482 | × | ||||||
| Liebolt 1970483 | × | ||||||
| Liem 2008484 | × | ||||||
| Liu 2003485 | × | ||||||
| Liu 2004486 | × | ||||||
| Lin 2005487 | ×s | ||||||
| Lin 2009488 | × | ||||||
| Livinya 1989489 | × | ||||||
| Long 1987490 | × | ||||||
| Lorbach 2010491 | × | ||||||
| Loyd 1983492 | × | ||||||
| Lu 1991493 | × | ||||||
| Lu 2008494 | ×o | ||||||
| Ludwig 1998495 | × | ||||||
| Lundberg 1965496 | × | ||||||
| Luo 2005497 | × | ||||||
| Luziatelli 1984498 | × | ||||||
| Ma 2004499 | ×o | ||||||
| Maiotti 2001500 | × | ||||||
| Maitland 1983501 | × | ||||||
| Mao 2003502 | ×o | ||||||
| Marcus 1994503 | × | ||||||
| Mardjuadi 1978504 | × | ||||||
| Marx 2007505 | × | ||||||
| Massoud 2002506 | × | ||||||
| Mattara 1994507 | × | ||||||
| Mavrikakis 1991508 | × | ||||||
| Mayerhofer 1981509 | × | ||||||
| McClatchie 2009510 | × | ||||||
| McHardy 2008511 | ×c | ||||||
| McKeever 1958512 | × | ||||||
| McQuay 1997513 | × | ||||||
| Meijer 2006514 | × | ||||||
| Melzer 1995515 | ×g | ||||||
| Mencke 1988516 | × | ||||||
| Menkes 1990517 | × | ||||||
| Mert 2009518 | × | ||||||
| Meyer 1952519 | ×t | ||||||
| Miccoli 1964520 | × | ||||||
| Michlovitz 2004521 | × | ||||||
| Miller 1991522 | × | ||||||
| Miller 2004523 | × | ||||||
| Miller 2009524 | × | ||||||
| Mior 2001525 | × | ||||||
| Miszczyk 2005526 | × | ||||||
| Mizuno 1976527 | × | ||||||
| Mohindra 1987528 | × | ||||||
| Molsberger 2010529 | × | ||||||
| Monreal González 2006530 | × | ||||||
| Montemagni 1989531 | ×j | ||||||
| Moore 1976532 | × | ||||||
| Morgan 1995533 | × | ||||||
| Morris 1994534 | × | ||||||
| Moutounet 1984535 | × | ||||||
| Mueller 1954536 | × | ||||||
| Mund-Hoym 1987537 | × | ||||||
| Munting 1978538 | × | ||||||
| Murnaghan 1955539 | ×s | ||||||
| Musil 2009540 | × | ||||||
| Nabeta 2000541 | ×n | ||||||
| Nabeta 2002542 | × | ||||||
| Naglic 2009543 | × | ||||||
| Naredo 2004544 | × | ||||||
| Narouze 2009545 | × | ||||||
| Nash 1989546 | ×c | ||||||
| Neviaser 1991547 | × | ||||||
| Ng 2009548 | ×g | ||||||
| Nicholson 1985549 | ×h | ||||||
| Nicholson 2003550 | ×h | ||||||
| Nicolova 1966551 | × | ||||||
| Nikolova 1970552 | × | ||||||
| Nobuhara 1990553 | ×h | ||||||
| Noel 1997554 | × | ||||||
| Noskov 2005555 | × | ||||||
| Nouijai 2006556 | × | ||||||
| Nykanen 1995557 | × | ||||||
| Ogilvie-Harris 1995558 | ×g | ||||||
| Ogilvie-Harris 1997559 | × | ||||||
| Ohshima 1987560 | ×n | ||||||
| Ohta 2005561 | × | ||||||
| Okamura 1995562 | × | ||||||
| Olejarova 2004563 | × | ||||||
| Omari 2001564 | × | ||||||
| Orr 1997565 | × | ||||||
| Ortolani 1985566 | × | ||||||
| Othman 2002567 | × | ||||||
| Ou 1989568 | × | ||||||
| Ozaki 1989569 | × | ||||||
| Ozaki 1995570 | × | ||||||
| Ozaki 1996571 | × | ||||||
| Pages 1982572 | × | ||||||
| Pap 1998573 | × | ||||||
| Parker 1989574 | × | ||||||
| Parker 1989575 | × | ||||||
| Parsons 1967576 | × | ||||||
| Patel 1989577 | × | ||||||
| Patte 1983578 | × | ||||||
| Pearsall 1999579 | × | ||||||
| Peng 1987580 | × | ||||||
| Peng 2006581 | ×o | ||||||
| Petri 1987582 | × | ||||||
| Petukhov 1979583 | ×d | ||||||
| Philadelphia Panel 2001584 | × | ||||||
| Piotte 2004585 | × | ||||||
| Pittler 2008586 | ×c | ||||||
| Placzek 1998587 | × | ||||||
| Polimeni 2003588 | × | ||||||
| Politano 1982589 | × | ||||||
| Pollock 1994590 | × | ||||||
| Poonam 1990591 | ×g | ||||||
| Qiu 2006592 | × | ||||||
| Quigley 1954593 | × | ||||||
| Quigley 1982594 | × | ||||||
| Quin 1965595 | ×s | ||||||
| Quraishi 2006596 | ×u | ||||||
| Quraishi 2008597 | ×g | ||||||
| Radaelli 1984598 | ×j | ||||||
| Rainbow 2008599 | × | ||||||
| Ramsey 2009600 | × | ||||||
| Refior 1984601 | × | ||||||
| Reichmister 1999602 | × | ||||||
| Ren 2006603 | × | ||||||
| Rendeiro 2006604 | × | ||||||
| Revel 1999605 | × | ||||||
| Rey 1991606 | × | ||||||
| Rhind 1982607 | × | ||||||
| Rich 1985608 | × | ||||||
| Richardson 1975609 | × | ||||||
| Rigato 2002610 | × | ||||||
| Ritchie 1996611 | × | ||||||
| Ritzmann 1999612 | × | ||||||
| Rizk 1982613 | ×s | ||||||
| Rizk 1983614 | ×s | ||||||
| Rizk 1994615 | ×h | ||||||
| Roberts 1965616 | × | ||||||
| Romoli 2000617 | × | ||||||
| Rompe 1981618 | × | ||||||
| Roques 1984619 | × | ||||||
| Roubal 1996620 | × | ||||||
| Rowlingson 1986621 | × | ||||||
| Saadat Niaki 2005622 | × | ||||||
| Saadeh 2005623 | × | ||||||
| Saeidian 2007624 | × | ||||||
| Saggini 1996625 | ×h | ||||||
| Sakeni 2007626 | × | ||||||
| Sanders 1990627 | × | ||||||
| Sandor 2000628 | × | ||||||
| Sandor 2000629 | × | ||||||
| Santalena 1998630 | × | ||||||
| Sauvain 1985631 | × | ||||||
| Scendoni 1987632 | × | ||||||
| Scheef 1979633 | × | ||||||
| Schieroni 1985634 | × | ||||||
| Schomacher 2007635 | × | ||||||
| Schultheis 2009636 | × | ||||||
| Schultze 2004637 | ×b | ||||||
| Schulz 1986638 | × | ||||||
| Schwitalle 1998639 | × | ||||||
| Scott 2009640 | × | ||||||
| Segmuller 1995641 | × | ||||||
| Shah 200722 | ×c | ||||||
| Sharma 1993642 | ×s | ||||||
| Sharma 2009643 | ×h | ||||||
| Shchekotov 1977644 | × | ||||||
| Shchepina 1989645 | ×d | ||||||
| Shehab 2000646 | × | ||||||
| Shiraishi 1991647 | × | ||||||
| Sileghem 1991648 | × | ||||||
| Simpson 2004649 | × | ||||||
| Singh 1980650 | × | ||||||
| Singh 2010651 | × | ||||||
| Skorogliadov 1990652 | × | ||||||
| Skorogliadov 1990653 | ×d | ||||||
| Skoroglyadov 1986654 | ×d | ||||||
| Slullitel 2000655 | ×e | ||||||
| Smidt 2005656 | × | ||||||
| Snow 2009657 | × | ||||||
| Sokk 2007658 | × | ||||||
| Spacca 2005659 | × | ||||||
| Speed 2004660 | × | ||||||
| Speed 2006661 | × | ||||||
| Spier 1984662 | × | ||||||
| Spresser 2002663 | × | ||||||
| Srivastava 1972664 | × | ||||||
| Srour 2008665 | ×g | ||||||
| Stavnichii 1985666 | ×d | ||||||
| Stavnichii 1986667 | ×d | ||||||
| Steinbrocker 1974668 | × | ||||||
| Stoddard 1955669 | × | ||||||
| Stodell 1981670 | × | ||||||
| Stoker 1991671 | × | ||||||
| Stratz 2002672 | ×b | ||||||
| Strobel 1996673 | × | ||||||
| Strunce 2009674 | × | ||||||
| Sun 1996675 | ×g | ||||||
| Sun 2001676 | ×g | ||||||
| Sun 2002677 | × | ||||||
| Surenkok 2009678 | × | ||||||
| Swirski 1971679 | × | ||||||
| Taller 1985680 | × | ||||||
| Talybov 1981681 | ×d | ||||||
| Talybov Yu 1981682 | ×b | ||||||
| Tam 2010683 | ×g | ||||||
| Tamai 1999684 | × | ||||||
| Tamai 2004685 | × | ||||||
| Tan 1995686 | × | ||||||
| Taskaynatan 2004687 | × | ||||||
| Taskaynatan 2005688 | × | ||||||
| Taverna 1990689 | ×g,j | ||||||
| Teys 2008690 | × | ||||||
| Thakur 1991691 | × | ||||||
| Thomas 1981692 | × | ||||||
| Thumb 1987693 | × | ||||||
| Todorov 1972694 | ×d | ||||||
| Toker 2008695 | × | ||||||
| Toplicanec 1986696 | ×f | ||||||
| Trehan 2010697 | × | ||||||
| Tripathi 1979698 | ×b | ||||||
| Tsarin 1990699 | × | ||||||
| Tsukayama 2002700 | ×c | ||||||
| Tsun-Nin 1977701 | × | ||||||
| Turner-Stokes 1996702 | × | ||||||
| Tuzlukov 1990703 | ×d | ||||||
| Ucuncu 2009704 | × | ||||||
| Ueno 1995705 | × | ||||||
| Uhlemann 1991706 | × | ||||||
| Uhthoff 2002707 | × | ||||||
| Uitvlugt 1993708 | × | ||||||
| Ulmer 1982709 | ×h | ||||||
| Vad 2003710 | × | ||||||
| Valtonen 1967711 | × | ||||||
| Valtonen 1974712 | × | ||||||
| Valtonen 1974713 | × | ||||||
| van de Weg 2004714 | ×c | ||||||
| van den Dolder 2003715 | × | ||||||
| van den Hout 200591 | × | ||||||
| van der Heijden 1996716 | ×c | ||||||
| van der Heijden 1999717 | × | ||||||
| van der Windt 1997718 | ×e | ||||||
| van der Windt 1998719 | × | ||||||
| van der Windt 1999720 | ×c | ||||||
| van der Windt 1999721 | × | ||||||
| van der Windt 2000722 | ×h,t | ||||||
| van der Windt 2003723 | × | ||||||
| van Laack 1987724 | × | ||||||
| van Royen 1996725 | × | ||||||
| van Royen 1996726 | × | ||||||
| Vanni 1985727 | × | ||||||
| Vas 2008728 | × | ||||||
| Vaughn 2000729 | × | ||||||
| Venturi 1979730 | × | ||||||
| Verkhozina 1972731 | ×d | ||||||
| Vermeulen 2004732 | ×g | ||||||
| Vermeulen 2007733 | × | ||||||
| Verstraete 1985734 | ×s | ||||||
| Vigano 1986735 | × | ||||||
| Volhard 1968736 | × | ||||||
| von Knorre 1990737 | × | ||||||
| Vrettos 2005738 | × | ||||||
| Wagenhau 1969739 | × | ||||||
| Walach 2003740 | × | ||||||
| Waldburger 1992741 | ×h | ||||||
| Wallny 1997742 | × | ||||||
| Walsh 2009743 | ×c | ||||||
| Wang 1990744 | × | ||||||
| Wang 1993745 | × | ||||||
| Wang 1995746 | × | ||||||
| Wang 1997747 | ×d | ||||||
| Wang 2005748 | ×o | ||||||
| Wang 2006749 | × | ||||||
| Wang 2007750 | × | ||||||
| Wang 2007751 | × | ||||||
| Wang 2008752 | × | ||||||
| Warner 1996753 | × | ||||||
| Wassef 1992754 | × | ||||||
| Watson 2000755 | ×g | ||||||
| Weber 1995756 | × | ||||||
| Weber 2001757 | × | ||||||
| Weiser 1976758 | ×f | ||||||
| Weiser 1977759 | × | ||||||
| Wen 2009760 | ×g,o | ||||||
| White 1995761 | × | ||||||
| White 1996762 | × | ||||||
| White 2009763 | × | ||||||
| Widiastuti-Samekto 2004764 | × | ||||||
| Wies 200371 | ×g | ||||||
| Wies 2003765 | ×g | ||||||
| Wiley 1995766 | × | ||||||
| Wiley 1997767 | × | ||||||
| Winters 1995768 | ×t | ||||||
| Winters 1997769 | × | ||||||
| Winters 1999770 | × | ||||||
| Winters 2000771 | ×t | ||||||
| Worsdorfer 1984772 | × | ||||||
| Xiao 2006773 | × | ||||||
| Xie 1988774 | × | ||||||
| Xu 2006775 | ×g | ||||||
| Xu 2008776 | ×o | ||||||
| Xu 2009777 | ×b | ||||||
| Yamaguchi 2002778 | × | ||||||
| Yamaguchi 2004779 | × | ||||||
| Yamaguchi 2008780 | × | ||||||
| Yamamoto 1988781 | ×h,n | ||||||
| Yamamoto 1993782 | ×n | ||||||
| Yamamoto 1993783 | × | ||||||
| Yamshon 1958784 | × | ||||||
| Yan 2005785 | × | ||||||
| Yang 2006786 | × | ||||||
| Yao 2002787 | ×h | ||||||
| Yaya Huaman 1975788 | × | ||||||
| Yegudin-Ash 1998789 | × | ||||||
| Yigiter 2002790 | × | ||||||
| Yuan 1995791 | ×o | ||||||
| Zachepa 1991792 | ×d | ||||||
| Zancan 1993793 | ×j | ||||||
| Zeilig 2005794 | × | ||||||
| Zeng 2005795 | ×o | ||||||
| Zhang 1991796 | × | ||||||
| Zhang 1991797 | × | ||||||
| Zhang 2006798 | ×o | ||||||
| Zhang 2008799 | ×o | ||||||
| Zhang 2009800 | ×o | ||||||
| Zhao 2003801 | × | ||||||
| Zhao 2006802 | ×o | ||||||
| Zhou 2004803 | ×o | ||||||
| Zhu 1997804 | ×o | ||||||
| Zhu 2004805 | ×o | ||||||
| Zivkovic 1969806 | × | ||||||
| Zuecker 1977807 | × |
Appendix 6 Study details
Appendix 6.1 Steroid injection
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|---|---|---|---|---|
|
Bal 2008 68 RCT Country, setting and treatment provider: Turkey; physical therapy and rehabilitation department of a hospital |
Inclusion criteria: Presence of shoulder pain with limitation of both active and passive movements of the glenohumeral joint of ≥ 25% in at least two directions; aged 18–70 years; symptom duration between 6 weeks and 6 months; and no treatment other than analgesics in the last 6 months Exclusion criteria: Uncontrolled diabetes mellitus, contraindications of injections and previous shoulder surgery Method of diagnosis: NR Condition terminology used: Adhesive capsulitis |
Age (years), mean (SD): Steroid injection: 56.9 (9.56); placebo injection: 56.3 (8.16) Female: 44% Participants with diabetes? Unclear/not reported |
Duration of FS at baseline: NR Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Steroid injection: intra-articular, posterior approach injection of methylprednisolone acetate, 40 mg, 1 ml. One injection only. 5-cm 21-gauge needle Home exercise |
Placebo injection: intra-articular, posterior approach injection of saline, 0.9% sodium chloride, 1 ml. One injection only. 5-cm 21-gauge needle Home exercise |
Oral paracetamol (1500 mg/day) was recommended to patients when needed Home exercise: Five sessions daily for 12 weeks (420 sessions in total) of home exercise consisting of pendulum circumduction and passive shoulder self-stretching in forward elevation, external rotation, horizontal adduction and internal rotation. When passive range of movement reached 90% of normal range the following exercises were added: isometric in all planes, theraband exercises (low, medium and high resistance), strengthening exercises for scapular stabilising muscles, and advanced muscle strengthening exercises with dumb-bells. Participants were advised to apply a heat pack before exercise and a cold pack after exercise |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Carette 2003 35 RCT Country, setting and treatment provider: Canada; outpatient rheumatology clinics at seven centres; all injections performed by trained radiologists and all physiotherapy supervised by physiotherapists with at least 3 years’ experience of musculoskeletal disorders |
Inclusion criteria: Adhesive capsulitis defined as the presence of shoulder pain with limitation of both active and passive movements of the glenohumeral joint of ≥ 25% in at least two directions (abduction, flexion, external rotation, internal rotation) compared with contralateral shoulder or with normal values. Patients eligible for inclusion if they met the definition for adhesive capsulitis and were aged ≥ 18 years, had been symptomatic for < 1 year and had a total SPADI score of ≥ 30. Partway through the trial patients with diabetes mellitus became eligible to be included Exclusion criteria: Adhesive capsulitis secondary to another cause, including inflammatory, degenerative, metabolic or infectious arthritis, cerebrovascular accident or fracture. Known blood coagulation disorder or allergy to radiological contrast material Method of diagnosis: Presence of pain and range of movement Condition terminology used: Adhesive capsulitis |
Age (years), mean (SD): Steroid injection + PT: 54.9 (10.5); steroid injection: 55.4 (10.0); PT: 54.2 (8.3); placebo injection: 56.5 (9.4) Female: 59% Any participants with diabetes? Yes. Steroid injection + PT: n = 2 (9.5%); steroid injection: n = 1 (4.3%); PT: n = 1 (3.9%); placebo injection: n = 2 (8.7%) |
Duration of FS at baseline (weeks), mean (SD): Steroid injection + PT: 22.1 (14.9); steroid injection: 21.2 (11.0); PT: 20.8 (11.2); placebo: 20.3 (7.3) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Steroid injection + PT: Injection of triamcinolone hexacetonide, 40 mg, 2 ml into shoulder joint space using fluoroscopic guidance (intra-articular) with patient in supine position with arm by their side and in internal rotation. One injection on day of randomisation only. 2.5- to 3-inch 21-gauge needle. PT: Patients with acute symptoms: 12 × 1-hour sessions, three times per week for 4 weeks consisting of TENS, mobilisation techniques, active range of movement exercises, ice application. Patients with chronic-like symptoms: 12 × 1-hour sessions, three times per week for 4 weeks consisting of ultrasound, mobilisation techniques, active and auto-assisted range of movement exercises, isometric strengthening exercises and ice application |
Steroid injection: Steroid injection as in combined intervention. Home exercise |
PT + placebo injection: Injection of saline, 2 ml into shoulder joint space using fluoroscopic guidance. One injection on day of randomisation only. PT only as in combined intervention Home exercise |
Placebo injection: Injection of saline, 2 ml into shoulder joint space using fluoroscopic guidance. One injection on day of randomisation only Home exercise |
Patients were given a supply of paracetamol tablets. All other medications for the treatment of adhesive capsulitis were stopped Home exercise: Home exercises of active and auto-assisted range of movement exercises in the planes of flexion, abduction, external rotation and internal rotation, 10 minutes twice daily for 12 weeks |
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Dacre 1989 67 RCT Country, setting and treatment provider: UK; physiotherapy was performed by a physiotherapist, the steroid was injected by a physician |
Inclusion criteria: Criteria of Bulgen et al. (1984): painful stiff shoulder for at least 4 weeks; inability to use arm with restriction of movement and loss of full function; pain at night causing sleep disturbance and inability to lie on affected side Exclusion criteria: Patients with predisposing conditions were excluded Method of diagnosis: Clinical diagnosis (examination, radiography and erythrocyte sedimentation rate) Terminology used: Periarthritis of the shoulder; painful stiff shoulder |
Age (years), mean: Steroid injection: 55.8; PT: 53.0; steroid injection + PT: 58.8 Female: 55% Any participants with diabetes? Yes. n = 3 |
Duration of FS at baseline: NR Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Steroid injection + PT: 20 mg triamcinolone with 1 ml 2% lidocaine was injected anteriorly. Mobilisation was the mainstay of physiotherapy (4–6 weeks’ duration). The specific method was chosen by the physiotherapist | Steroid injection: 20 mg triamcinolone with 1 ml 2% lidocaine was injected anteriorly | PT: Mobilisation was the mainstay of physiotherapy (4–6 weeks’ duration) | NR | |
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Rizk 1991 42 RCT Country, setting and treatment provider: USA; one physician performed all injections |
Inclusion criteria: Total range of movement < 50% of normal range (i.e. < 320°), shoulder pain for < 6 months, nocturnal accentuation of pain, no effusion in the glenohumeral joint, no history of recent trauma, no previous injections in the involved shoulder, no history of allergy to local anaesthetics or steroids, absence of polyarthritis or neurological disease that cause shoulder pain, no evidence of alternative causes of shoulder pain revealed in radiography carried out after onset of pain and within 2 months of study entry Method of diagnosis: Presence of symptoms Terminology used: FS; adhesive capsulitis |
Age (years), mean (range): Total: 55 (40–70); steroid (anterior) + lidocaine + PT: 55.9; steroid (lateral) + lidocaine + PT: 52.3; lidocaine (anterior) + PT: 57.7; lidocaine (lateral) + physiotherapy: 54.1 Female: 41.7% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (weeks), mean (range): Total: 13.2 (8 to 18) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Steroid (anterior approach) + PT: Once a week for 3 weeks intra-articular injection using the anterior approach (glenohumeral joint) of 1 ml repository aqueous suspension of methylprednisolone acetate (Depo-Medrol®), 40 mg/ml with 2 ml of 1% lidocaine. 1.5-inch, 21-gauge needle. Standardised weekly PT treatment for 11 weeks. Ultrasonic therapy of 1.5 W/cm2 for 7 minutes followed by therapeutic exercise (Codman and wall climbing) Home exercise |
Steroid (lateral approach) + PT: Same as for intra-articular steroid except using lateral approach (subacromial bursa) Home exercise |
Two arms for placebo injection: intra-articular lidocaine (anterior approach) + PT: Once a week for 3 weeks intra-articular injection using the anterior approach of 1% lidocaine. 1.5-inch, 21-gauge needle. PT as for steroid group; intrabursal lidocaine (lateral approach) + PT: Once a week for 3 weeks intrabursal injection using the lateral approach of 1% lidocaine. 1.5-inch, 21-gauge needle. PT as for steroid group. Home exercise |
Advised to continue NSAIDs prescribed by physician. All patients were receiving NSAIDs, with only minor difference in frequency distribution of various drugs between the treatment groups. Propoxyphene and other drugs were discontinued before entry into the study Home exercise: Instruction given in a home exercise programme (no further details provided) |
|
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Ryans 200541 RCT Country, setting and treatment provider: UK; direct access shoulder assessment clinic run by a GP with a special interest in rheumatology at a local district hospital. Injections provided by the same experienced clinician, physiotherapy provided by a single therapist or a nominated deputy if unavailable |
Inclusion criteria: Aged ≥ 18 years with a painful shoulder in the fifth cervical (C5) dermatone distribution of > 4 weeks’ and < 6 months’ duration; presence of restriction of active and passive range of movement in both external rotation and glenohumeral abduction of > 25% compared with other shoulder. Exclusion criteria: Previous intra-articular injection or previous physiotherapy for this episode of shoulder pain; limitation in only external rotation or glenohumeral abduction; evidence of glenohumeral osteoarthritis on radiography; clinical evidence of rotator cuff tear; history of significant trauma to the shoulder or history of inflammatory joint disease or of a cerebrovascular accident affecting the shoulder; bilateral adhesive capsulitis; patients with a contraindication to triamcinolone Method of diagnosis: Radiography Terminology used: Adhesive capsulitis |
Age (years), mean (SD): Steroid injection + PT: 56.3 (6.4); steroid injection: 52.3 (9.3); PT + placebo injection: 52.6 (7.7); placebo injection: 55.2 (9.4) Female: 59% Any participants with diabetes? Yes. Total: n = 5; steroid injection + PT: n = 1 (5%); steroid injection: n = 1 (5%); PT + placebo injection: n = 1 (5%); placebo injection: n = (11%) |
Duration of FS at baseline (weeks), mean (SD): Steroid injection + PT: 14.2 (4.4); steroid injection: 12.2 (5.3); PT + placebo injection: 14.4 (4.4); placebo injection: 14.9 (3.7) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: Unclear; 32% were reported to have experienced ‘minor trauma’. No further details were provided |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Steroid injection + PT: Injection of triamcinolone 20 mg, 1 ml and normal saline 2 ml using a combined non-guided approach to the shoulder: intra-articular, 1.5 ml injected by an anterior approach and 1.5 ml by a lateral approach. Physiotherapy consisted of eight sessions over 4 weeks: proprioceptive neuromuscular facilitation, Maitland mobilisations (which were progressed as the condition improved), standardised interferential modality and active exercise therapy with gym equipment Home exercise |
Steroid injection only: Injection of triamcinolone 20 mg, 1 ml and normal saline 2 ml using a combined non-guided approach to the shoulder: intra-articular, 1.5 ml injected by an anterior approach and 1.5 ml by a lateral approach Home exercise |
PT + placebo injection: Physiotherapy as for other group. Placebo injection of saline 3 ml using a combined non-guided approach to the shoulder: 1.5 ml injected by an anterior approach and 1.5 ml by a lateral approach Home exercise |
Placebo injection: Injection of saline 3 ml using a combined non-guided approach to the shoulder: 1.5 ml injected by an anterior approach and 1.5 ml by a lateral approach Home exercise |
Patients who were not already taking analgesics were issued with 50 × 500-mg paracetamol tablets for pain relief with suggestion to take one to two tablets 4- to 6-hourly as required for pain and taking no more than a maximum of 8 tablets daily. Patients recorded all analgesic and anti-inflammatory medication taken in a medication diary Home exercise: Patients were instructed by a physiotherapist in a home exercise programme using a video and home exercise instruction sheet |
Appendix 6.2 Sodium hyaluronate
| Study | Inclusion/exclusion criteria and diagnosis of frozen shoulder | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of frozen shoulder, previous treatments, secondary frozen shoulder) | |
|---|---|---|---|---|
|
Calis 2006 66 RCT Country, setting and treatment provider: Turkey; injection performed by a single physician |
Inclusion criteria: History of pain for at least 1 month; limited active and passive shoulder movement; decreased passive range of movement of ≥ 20%, in at least three movements, according to the American Medical Association guide for the evaluation of permanent impairment; no previous injection in the involved shoulder; no history of allergy to local anaesthetics, steroids or sodium hyaluronate; absence of cervical radiculopathy, fracture, dislocation and rotator cuff laceration; negative subacromial impingement test Method of diagnosis: Physical examination and laboratory tests. Subacromial impingement test (greatly improved range of movement after injection of 1% of 5 ml lidocaine into subacromial space of affected shoulder) to exclude subacromial impingement syndrome. All tests performed by one clinician Terminology used: Adhesive capsulitis |
Age (years), mean (SD): Sodium hyaluronate injection: 59.7 (9.81); steroid injection: 56.36 (11.3); PT: 52.33 (10.1); home exercise: 59.25 (6.8) Female: 63% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline: NR Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Sodium hyaluronate injection: Intra-articular, posterior approach injection of sodium hyaluronate (Orthovisc®), 30 mg, one injection once weekly for 2 weeks. 22-gauge needle used Home exercise |
Steroid injection: Intra-articular, posterior approach injection of triamcinolone acetonide (Kenakort-A®), 40 mg, one injection. 22-gauge needle used Home exercise |
PT: 10 daily sessions: a heat pack applied for 20 minutes; ultrasonic therapy for 5 minutes (1.5 W/cm2 intensity); TENS for 20 minutes at patient’s level of tolerance; and stretching exercises Home exercise |
No intervention: Home exercise only |
Paracetamol could be taken if necessary Home exercise: Stretching and Codman exercises |
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Rovetta 1998 69 RCT Country, setting and treatment provider: Italy; physiotherapy delivered by one physiotherapist |
Inclusion criteria: Clinical history of spontaneous shoulder pain; glenohumeral abduction and forward flexion < 90°, external rotation < 20°; clinical absence of signs of rotator cuff interruption; cervical examination excluding dysfunction in this area; plain radiographs in standard views; sonographic examination showing shrinking of the joint capsule with increased capsular echogenicity Exclusion criteria: Patients with a history of stroke, trauma, diabetes, ischaemic heart and generalised osteoarthritis disease; previous treatment with corticoid oral regimens Method of diagnosis: Clinical history and radiography or ultrasound Terminology used: Adhesive capsulitis |
Age (years), mean (?SD): Steroid + sodium hyaluronate injection + PT: 65.8 (9.1); steroid injection + PT: 62.3 (13) Female: 70% Any participants with diabetes? No |
Duration of FS at baseline, mean (?SD): Steroid + sodium hyaluronate injection + PT: 7.4 (? months) (4); steroid injection + PT: 9.0 (3.3) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Steroid + sodium hyaluronate injection + PT: Triamcinolone acetonide (20 mg) and sodium hyaluronate (20 mg) injection given intra-articularly (posterior approach, Cyriax and Russell) at 15-day intervals in the first month, then monthly for 6 months. The most appropriate PT was decided by physiotherapist: passive mobilisation, active exercises and facilitation exercises performed for 4–12 weeks | Steroid injection + PT: Triamcinolone acetonide (Bristol-Myers Squibb) (20 mg) injection given intra-articularly (posterior approach, Cyriax and Russell808) at 15-day intervals in the first month, then monthly for 6 months. PT as for other group | NR | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Takagishi 1996 70 RCT Country, setting and treatment provider: Japan; university hospital medical department |
Inclusion criteria (including definition of FS): Patients with FS who have < 120° of shoulder joint flexion and those who suffer from pain on exercise of > 4 on the VAS and suffer from pain during night-time and daytime Exclusion criteria: Previous shoulder injury Method of diagnosis: Clinical examination Terminology used: Gojukata (Japanese; English translation ‘Fifties shoulder’) |
Age (years), mean: Sodium hyaluronate: 56; steroid: 48 Female: 75% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (months), mean: Sodium hyaluronate: 4.4; steroid: 3.5 Stage of FS at baseline: NR |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Sodium hyaluronate: one intra-articular injection of 2 mg sodium hyaluronate once a week for 5 weeks | Steroid injection: one intra-articular injection of 2 mg dexamethasone once a week for 5 weeks | Topical NSAIDs | ||
Appendix 6.3 Acupuncture
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|---|---|---|---|---|
|
Cheing 2008 81 RCT Country, setting and treatment provider: Hong Kong; treatment delivered by physiotherapist accredited to deliver acupuncture. The same individual delivered all of the treatments |
Inclusion criteria: Localised pain over one shoulder, night pain and restricted active and passive shoulder motion Exclusion criteria: History of trauma, fractures, previous shoulder surgery, cervical or thoracic pain syndrome, complex regional pain syndrome, malignancies or anticoagulant therapy or acupuncture treatment to the painful shoulder in the previous 6 months Method of diagnosis: Diagnosed by orthopaedic surgeon Terminology used: FS; adhesive capsulitis |
Age (years), range: 33 to 90 Female: NR Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (months), mean (SD): Electroacupuncture: 6.71 (6.50); interferential electrotherapy: 6.70 (6.05); control: 8.26 (7.94) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Electroacupuncture: 10 sessions, two to three times per week, over a 4-week period. Three points needled (one trigger point, LI15 and ST38) with a needle of 0.30 × 40 mm diameter, at a depth of 15–25 mm. ‘De qi’ sensation was felt by the patient and 2–100 Hz electrical stimulation was used at a pulse duration of 100–400 µs (just below pain threshold) for 20 minutes Home exercise |
Interferential electrotherapy: 10 × 20-minute sessions over 4 weeks at 80–120 Hz. Four suction-type electrodes were placed around the shoulder region in a coplanar arrangement Home exercise |
No intervention: Waiting list controls |
NR Home exercise: Participants followed a chart and performed a standard set of mobilisation exercises five times per day over 6 months that included four directions (forward flexion, external rotation, horizontal adduction, internal rotation) |
|
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Fang 2006 82 RCT Country, setting and treatment provider: China; Hospital outpatient department |
Inclusion criteria: Used recognised criteria Exclusion criteria: Aged > 65 years, too sensitive to electrotherapy, fracture of shoulder, receiving long-term concomitant medication Method of diagnosis: Clinical examination Terminology used: Periarthritis of the shoulder |
Age (years), mean (SD): TENS: 51.5 (3.1); electroacupuncture: 52.9 (3.8) Female: 55% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (weeks/months??), mean (SD): TENS: 6.1 (3.5); electroacupuncture: 6.8 (3.2) Stage of FS at baseline: Stage 1 (pre-adhesive): Patient feels pain around the shoulder, with increased pain at night (which even affects quality of sleep). The ranges of motion (or functional activities of the shoulder) are normal or slightly affected. Stage 2 (adhesive): Patient feels some reduction in pain but feels uncomfortable because of the pain. The functional activities of the shoulder are seriously affected; the ranges of motion are considerably reduced, which even affects activities of normal life Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Electroacupuncture: 10 × 40-minute sessions every other day. Four acupuncture points [Waiguan (SJ5) and Hegu (LI4) and, alternating each session, Jianliao (SJ14) and Jian qian (Ex-UE), or Jianyu (LI15) and Naoshu (SI10)]. 0.30 mm × 40 mm needles inserted to depth of 20–25 mm. Electricity was administered after De qi was felt (not reported whether De qi was felt by acupuncturist or by patient) | TENS: 10 × 40-minute sessions every other day. Electrodes placed at four acupuncture points [Waiguan (SJ5) and Hegu (LI4) and, alternating each session, Jianliao (SJ14) and Jian qian (Ex-UE), or Jianyu (LI15) and Naoshu (SI10)]. High frequency (100 Hz) for 10 minutes and low frequency (2 Hz) for 30 minutes. Intensity of current 10 ± 2 mA | NR | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Ma 2006 83 RCT Country, setting and treatment provider: Taiwan; medical centre |
Inclusion criteria: Shoulder pain for at least 3 months, could not lift arms more than 135° Exclusion criteria: Non-spontaneous FS caused by nervous system diseases, acute inflammation and broken bones; acupuncture syncope and skin infection surrounding acupuncture points Method of diagnosis: Clinical history Terminology used: FS |
Age (years), mean: PT: 54.1; acupuncture: 56.4; acupuncture + PT: 52.8 Female: 52% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (weeks), mean: 25.8 Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Acupuncture: 15-minute session twice a week for 4 weeks. Each session consisted of therapeutic principles in promoting flow of qi and blood, driving out the wind and cold, removing dampness and activating meridians; therapeutic methods on three yang meridians of the hand; and prescriptions with jianliao, jianlu (LI15), fengchi (GB20), hegu (L14) and yanglingquan (GB34) | Acupuncture + PT: Acupuncture as for other group. Five sessions of physiotherapy per week for 4 weeks. Each session consisted of heat pack for 15 minutes, joint mobilisation for 5–10 minutes and active shoulder exercises for 5–10 minutes | PT: Five sessions of physiotherapy per week for 4 weeks. Each session consisted of heat pack for 15 minutes, joint mobilisation for 5–10 minutes and active shoulder exercises for 5–10 minutes | NR | |
Appendix 6.4 Physical therapy
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|---|---|---|---|---|
|
Diercks 2004 73 Controlled trial Country, setting and treatment provider: Netherlands; NR |
Inclusion criteria: Idiopathic FS diagnosed between January 1997 and January 2001; the Lundberg criteria were used (> 50% motion restriction of the glenohumeral joint in all directions for at least 3 months) Exclusion criteria: Patients with significant injury to the ipsilateral shoulder or arm, with surgical procedures on the shoulder, arm, cervical spine, thorax or breast within the previous 2 years or with intra-articular deformities, degenerative arthritis, inflammatory arthritis or diabetes mellitus were excluded Method of diagnosis: Glenohumeral joint movement was measured using a Cybex inclinometer Terminology used: FS |
Age (years), mean (SD): Supervised neglect: 50 (6); PT: 51 (7) Female: 61% Any participants with diabetes? No |
Duration of FS at baseline (months), ?mean (range): Supervised neglect: 5 (3 to 12); PT: 5 (3 to 10) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
PT: Patients were prescribed a standardised treatment protocol, carried out by a physical therapist, of active exercises up to and beyond the pain threshold, passive stretching and manipulation of the glenohumeral joint and home exercises aimed at stretching and maximal reaching. Duration was not reported Home exercise |
Supervised neglect: Patients were provided with an explanation of the natural course of the disease and were instructed not to exercise in excess of their pain threshold and to carry out pendulum exercises and active exercises within this painless range and resume all activities that were tolerated. Duration was not reported Home exercise |
NSAIDs or analgesics were prescribed to both groups where necessary | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Dogru 2008 51 RCT Country, setting and treatment provider: Turkey; outpatient clinic |
Inclusion criteria: Shoulder pain of at least 3 months’ duration and no major trauma; at least 25% loss of shoulder motion in all planes; pain on motion of at least 40 mm on VAS; normal findings on radiograph of the glenohumeral joint; absence of arthritis, malignancy and conditions such as cardiac disease, infection and coagulation disorders Exclusion criteria: Secondary adhesive capsulitis due to rotator cuff tears, fractures, dislocations and reflex sympathetic dystrophy Method of diagnosis: Routine systemic and neurological examination and measurement of active and passive range of movement. Passive range of movement was measured in all planes with a long-arm goniometer while patient supine. Complete blood count, erythrocyte sedimentation rate and routine biochemical analysis for exclusion of secondary factors. Shoulder radiographs were taken Terminology used: Adhesive capsulitis |
Age (years), mean (?SD): Total: 55.4 (7.6); ultrasound + PT: 53.9 (7.8); sham ultrasound + PT: 56.8 (7.3) Female: 57% Any participants with diabetes? Yes. Total: n = 18 (37%); ultrasound + PT: n = 8 (32%); sham ultrasound + PT: n = 10 (42%) |
Duration of FS at baseline (months), mean (?SD, range): Total: 5.7 (3.3, 3 to 12); ultrasound + PT: 6.3 (3.5); sham ultrasound + PT: 5.2 (2.9) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Ultrasound + PT: 10 daily sessions of 50 minutes over 2 weeks (no treatment at weekends). Each session consisted of 20 minutes of superficial heat (heat packs at 60°C), 10 minutes of ultrasound (3 MHz frequency and 1.5 W/cm2 intensity) and a 20-minute exercise programme (Codman’s exercises and wall climbing followed by glenohumeral joint stretching exercises to the patient’s tolerance) Home exercise |
Sham ultrasound + PT: Same as ultrasound intervention except the ultrasound machine was not switched to ‘on’ Home exercise |
Paracetamol, maximum of 1000 mg/day Home exercise: A daily exercise programme consisting of Codman’s exercises, active ROM and stretching exercises |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Dundar 2009 74 RCT Country, setting and treatment provider: Turkey; ?hospital |
Inclusion criteria: FS patients with gradually increasing shoulder pain and stiffness (painful phase and stiff phase) Exclusion criteria: Patients with rotator cuff pathology or those with secondary FS or stiff shoulder associated with a fracture, arthritis, abnormal shoulder radiographs or any significant trauma were excluded Method of diagnosis: Diagnosis was made on the basis of history, physical examination, radiography findings and, occasionally, magnetic resonance imaging, by specialists in physical medicine and rehabilitation. All patients had radiography of the shoulder joint Terminology used: FS |
Age (years), mean (SD): Continuous passive motion: 56.3 (7.8); conventional PT: 57.1 (8.3) Female: 68% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (months), mean (SD): Continuous passive motion: 6.3 (4.2); conventional PT: 5.9 (4.0) Stage of FS at baseline: Patients with phase 1 and/or phase 2 FS were included Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Continuous passive motion: Treatments involved a gradual increase in motion for 1 hour per day for 20 days over 4 weeks (5 days per week) using an external motorised device Home exercise |
Conventional PT: Daily physiotherapy by a physiotherapist involving active stretching and pendulum exercises for 1 hour per day for 20 days over 4 weeks (5 days per week) Home exercise |
No NSAIDs or other analgesics allowed. A 1-week washout period was required before therapy Home exercise: A standardised home exercise programme of passive range of movement and pendulum exercise every day until week 12. This was demonstrated by a physiotherapist on one occasion and written advice was given |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Leung 2008 75 RCT Country, setting and treatment provider: Hong Kong; NR |
Inclusion criteria: Patients with idiopathic FS who experienced shoulder pain and limited shoulder movement for at least 8 weeks Exclusion criteria: Patients with history of trauma to the shoulder, acute signs of shoulder inflammation and intrinsic shoulder pathology; patients with impaired sensation of hot and cold; pregnant patients; patients taking analgesic or anti-inflammatory drugs, with metal implants or with a cardiac pacemaker Method of diagnosis: Diagnosis made by orthopaedic surgeon Terminology used: FS |
Age (years), mean (SD): SWD + stretching: 59.8 (12.9); heat pack +stretching: 62.5 (12.1); stretching only: 57.3 (13.1) Female: 70% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline: NR Stage of FS at baseline: Stiff phase Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
SWD + stretching: Treatment was 3 × 20-minute sessions per week for 4 weeks. SWD machine with an operating frequency of 27.12 MHz was used. Patients sat on a wooden chair with back and affected arm supported and a pair of disc electrodes were placed on the affected glenohumeral joint. The intensity of the current was adjusted according to the patient’s subjective feeling of warmth and to maintain the feeling of comfortable warmth throughout. Immediately after heat treatment four stretching exercises were performed in a fixed sequence (stretching in external rotation and in flexion followed by stretching hand behind back and cross-body adduction). Each stretch was sustained for 30 seconds with a 10-second rest between stretches Home exercise |
Heat pack + stretching: Superficial heat was delivered using an electrical heat pack (35.5 × 68.5 cm). The temperature was set to 63°C but patients were informed that heating was to produce a feeling of comfortable warmth and if the heat was felt to be excessive this was adjusted. Stretching exercises as for SWD Home exercise |
No intervention: Home exercise only | Home exercise: Patients were asked to perform stretches at home every day | |
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Maricar 1999 76 RCT Country, setting and treatment provider: Singapore; NR |
Inclusion criteria: Major complaint of limitation in shoulder range of movement with secondary complaint of pain; able to place arms behind head and back reaching vertebral column; 90-degree abduction Exclusion criteria: History of previous shoulder trauma or previous episode of FS; existing or previous diagnosis of several other conditions (detailed in paper) including uncontrolled diabetes Method of diagnosis: Diagnosis (late stage 2 to stage 3) determined by the research physiotherapist. Radiographs were also reviewed Terminology used: FS; shoulder capsulitis |
Age (years), mean (SD): Manual therapy + exercise: 57.9 (9.5); exercise only: 54.9 (5.4) Female: 41% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline: Average duration for both groups: 3 months (two patients in each group had onset between 6 months and < 2 years) Stage of FS at baseline: Late stage 2 or stage 3 Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Manual therapy + exercise: Manual therapy (mobilisation of the shoulder quadrant, shoulder capsular stretch, shoulder flexion, shoulder abduction, shoulder external and internal rotation using Maitland grade III+ and IV) for eight weekly sessions. A 15-minute exercise circuit of nine exercises was also performed (including a strengthening regime for the rotator cuff muscles from week 5 onwards) Home exercise |
Exercise only: A 15-minute exercise circuit of nine exercises (including a strengthening regime for the rotator cuff muscles from week 5 onwards) Home exercise |
Home exercise: Home exercises were taught by a research physiotherapist and exercise sheets issued | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Pajareya 2004 77 RCT Country, setting and treatment provider: Thailand; hospital based; therapy delivered by three physiotherapists whose performance had been standardised |
Inclusion criteria: Shoulder pain and limitation of a passive range of shoulder motion in all directions that interfered with activities of daily living; attendance at the orthopaedic and rehabilitation clinic at Siriraj Hospital Exclusion criteria: Secondary adhesive capsulitis; intrinsic or extrinsic causes of shoulder problems; generalised arthritis; bilateral involvement; contraindications for NSAIDs; bleeding tendencies Method of diagnosis: NR Terminology used: Adhesive capsulitis |
Age (years), mean (SD): PT: 56.3 (10.6); standard care: 57.7 (10) Female: 68% Any participants with diabetes? Yes. Total: n = 20; PT: n = 10 (16.7%); standard care: n = 10 (16.7%) |
Duration of FS at baseline: PT: < 6 weeks: n = 13; 6 to < 12 weeks: n = 20; at least 12 weeks: n = 27; standard care: < 6 weeks: n = 6; 6 to < 12 weeks: n = 20; at least 12 weeks: n = 33 Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: Unclear/NR; 47% reported history of minor trauma before onset |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| PT: Three times per week for 3 weeks consisting of SWD (20 minutes), mobilisation and passive glenohumeral joint stretching exercises to the patient’s tolerance. On non-PT days patients were advised to perform pulley exercises (actively assisted exercises for 5 minutes) and active non-assisted exercises using a towel and wall (5 minutes after applying a heat pack for 20 minutes). The exercise guideline was based on Cyriax. Exercise was contraindicated if the patient felt pain with the passive movements before the end of the range | No intervention: Information only |
Ibuprofen (400 mg, three times daily, for 3 weeks) Patients were asked not to have any adjuvant therapy except oral paracetamol (up to 6 g/day) An information sheet advising on protection of the shoulder from vigorous activities was also given. Patients were encouraged to use their arms in a normal fashion for other activities of daily living |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Stergioulas 2008 16 RCT Country, setting and treatment provider: Greece; Peania Physical Therapy Centre; physiotherapists performed treatment |
Inclusion criteria: Painful and limited passive glenohumeral mobility; more restricted lateral rotation (< 8%) relative to abduction and medial rotation; no clear signs that shoulder pain was caused by another condition Exclusion criteria: Insulin-dependent diabetes; bilateral symptoms; systemic inflammatory joint disease; corticosteroid or physiotherapy treatment in preceding 6 months; surgery, dislocation or fracture/s of shoulder; calcification of the shoulder joint; complete rotator cuff tear Method of diagnosis: History of limited movement of glenohumeral joint, with pain at the available range of motion. Clinical history Terminology used: FS |
Age (years), mean (SD): Laser: 55.5 (5.8); sham laser: 56.8 (6.8) Female: 37% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (weeks/months??), mean (?SD): Laser: 26.5 (12.8); placebo: 27.1 (13.6) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: Unclear/NR |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Laser therapy: 3B Laser M1 1000 (a Ga-Al-As laser) applied around shoulder joint (810 nm, continuous mode, 60 mW, spot size 0.5 cm2, duty cycle 50%, 3.6 J/cm2) for 30 seconds over 8 weeks (12 sessions; two per week in the first 4 weeks, then one per week). Dose per point was 1.8 J; total dose/session 14.4 J. Home exercise |
Placebo laser: Placebo laser that appeared identical to active laser was used for the same regimen as active laser (12 sessions over 8 weeks; two per week in the first 4 weeks, then one per week) Home exercise |
Not reported Home exercise: Patients were instructed to perform pendulum and pain-free active exercises at home |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Vermeulen 2006 40 RCT Country, setting and treatment provider: Netherlands; outpatient clinic of Department of Physical Therapy at Leiden University Medical Centre; physical therapists performed treatment |
Inclusion criteria: ≥ 50% loss of passive movement of shoulder joint relative to non-affected side, in one or more of three movement directions (abduction in frontal plane, forward flexion or external rotation); duration of complaints of ≥ 3 months; ability to complete questionnaires in Dutch Exclusion criteria: Previous MUA of the affected shoulder; other conditions involving the shoulder (e.g. rheumatoid arthritis, osteoarthritis, damage of the glenohumeral cartilage, Hill–Sachs lesion, osteoporosis, malignancies in the shoulder region); neurological deficits affecting shoulder function in normal daily activities; pain or disorders of the cervical spine, elbow, wrist or hand; and injection with corticosteroid in the affected shoulder in the preceding 4 weeks Method of diagnosis: NR Terminology used: Adhesive capsulitis |
Age (years), mean (SD): HGMT: 51.6 (7.6); LGMT: 51.7 (8.6) Female: 66% Any participants with diabetes? Yes. Total: n = 16: HGMT: n = 8 (16%); LGMT: n = 8 (16%) |
Duration of FS at baseline (months), mean (range): HGMT: 8 (5 to 14.5); LGMT: 8 (6 to 14) Stage of FS at baseline: NR Previous treatments for FS: Previous PT: HGMT: n = 39 (79%), LGMT: n = 42 (82%); previous steroid injections: HGMT: n = 32 (65%), LGMT: n = 29 (57%); previous surgery: HGMT: n = 3 (6%), LGMT: n = 3 (6%) Participants with secondary FS: No |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| HGMT: One 30-minute session twice weekly for a maximum of 12 weeks consisting of 5-minute assessment of range of movement by performing all three physiological movements of the glenohumeral joint passively. At each position of the shoulder, the end-feel movement was assessed in order to apply the mobilisation technique into the stiffness zone. Mobilisation techniques were applied with intensities according to Maitland grades III and IV. The duration of prolonged stress on the shoulder capsule in the end-range position varied according to the participant’s tolerance. Participants were instructed to inform the therapist about the degree and nature of the pain during and after treatment. If the pain influenced the execution of mobilisation techniques by increasing the reflex muscle activity, then the therapist altered the direction or degree of mobilisation. If pain worsened or continued for more than 4 hours the intensity of the mobilisation technique was decreased in the next session | LGMT: One 30-minute session twice weekly for a maximum of 12 weeks consisting of 5-minute assessment of range of movement by performing all three physiological movements of the glenohumeral joint passively. At each position of the shoulder, the end-feel movement was assessed in order to apply the mobilisation technique within the pain-free zone. Participants were informed that all techniques should be performed without causing pain in the shoulder. Mobilisation techniques were performed according to Maitland grades I and II. Reflex muscle activity was monitored for indications of joint pain. If joint mobility increased, then mobilisation techniques were adjusted, and the amplitudes of movements were increased without reaching the limits of range of movement. For the last 3 minutes of each treatment passive proprioceptive neuromuscular facilitation patterns within the pain-free zone in the supine position were applied. Codman pendular exercises were performed for 2 minutes in a prone position |
Pain medication, prescribed and non-prescribed, was allowed. No other concomitant treatments for FS, including intra-articular corticosteroid injections into any joint, were allowed in the first 3 months Home exercise: Neither treatment group received a home exercise programme but both were advised to use the affected shoulder in daily activities whenever possible |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Yang 2007 78 RCT Country, setting and treatment provider: Taiwan; therapy delivered by a single physical therapist with 8 years’ experience in manual therapy Note: Because of limitations in the study design and data reported, only data comparing ERM + MRM with MWM + MRM at 6 weeks were extracted |
Inclusion criteria: Painful stiff shoulder for at least 3 months; range of movement losses of at least 25% compared with the non-involved shoulder in at least two of glenohumeral flexion, abduction or medial or lateral rotation Exclusion criteria: Diabetes mellitus, history of surgery on relevant shoulder, rheumatoid arthritis, painful stiff shoulder after severe trauma, fracture of shoulder complex, rotator cuff rupture, tendon calcification Method of diagnosis: NR Terminology used: FS |
Age (years), mean (SD): ERM + MRM: 53.3 (6.5); MWM + MRM: 58 (10.1) Female: 86% Any participants with diabetes? No |
Duration of FS at baseline (weeks), mean (SD): ERM + MRM: 18 (8); MWM + MRM: 22 (10) Stage of FS at baseline: Stiff phase Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| ERM + MRM: Mobilisation techniques were given in the following order: MRM (Maitland809 and Kaltenborn810) 10–15 repetitions, ERM (Vermeulen et al.811 and Maitland809) 10–15 repetitions, MRM (Maitland809 and Kaltenborn810), MWM (Mulligan812) 3 sets of 10 repetitions. There were 3 weeks in each phase. Mobilisation techniques were performed twice per week for 30 minutes. A simple exercise programme comprising pendular exercises and scapular setting (isometric scapular retraction) was also given | MWM + MRM: Mobilisation techniques were given in the following order: MRM (Maitland809 and Kaltenborn810), MWM (Mulligan812), MRM (Maitland809 and Kaltenborn810), ERM (Vermeulen et al.811 and Maitland809). Technique as for other group |
No other interventions allowed (including other physical therapies) Home exercise: Home exercise was discouraged |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Yan 2005 72 RCT Country, setting and treatment provider: China; primary care |
Inclusion criteria: ‘Standard shoulder periarthritis diagnostic criteria in “Clinical pain therapy”’ Exclusion criteria: Patients with functional disability due to neural-, muscle- or bone-related diseases Method of diagnosis: Clinical examination Terminology used: FS |
Age (years), mean (SD): Dumb-bell gymnastics: 56.6 (12.4); bare-handed exercise: 54.2 (11.6) Female: 20% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (years), mean: Dumb-bell gymnastics: 6.8; barehanded exercises: 5.8 Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None stated |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Dumb-bell gymnastics: Exercises performed 5–10 minutes, two to three times a day for 3 months using dumb-bells weighing 2–5 kg | Barehanded exercises: Bare-handed exercises performed for 3 months | NR | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Wies 2003 71 RCT Country, setting and treatment provider: UK; NR |
Inclusion criteria: NR (all patients had been diagnosed with primary FS; confirmed through correspondence with author) Exclusion criteria: NR Method of diagnosis: NR Terminology used: FS |
Age: NR Female: NR Any participants with diabetes? Unclear/NR |
Duration of FS at baseline: NR Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: No |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| PT: Physiotherapy consisting of manual therapy and therapeutic exercise for 9 weeks. No further details reported | Osteopathy technique: The Niel-Asher technique, consisting of a progression of deep-tissue manipulation, for 9 weeks | Control: Breathing exercises, effleurage massage and pain-free range of movement exercises for 9 weeks | NR | |
Appendix 6.5 Manipulation under anaesthesia
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|---|---|---|---|---|
|
Amir-us-Saqlain 2007 84 RCT Country, setting and treatment provider: Pakistan; orthopaedic outpatients department |
Inclusion criteria: History of pain and stiffness of the shoulder joint for which no cause could be identified, restriction of glenohumeral motion of < 50% of abduction and < 50% of external rotation compared with the motion of the contralateral shoulder joint and normal radiography finding in the shoulder joint. All symptoms had to be of 3 weeks’ duration Exclusion criteria: Intrinsic problems with the shoulder, such as recent surgical repair of soft tissue of the shoulder, history of fracture around shoulder joint, instability and recurrence following previous manipulation, significant osteopenia, tear of the rotator cuff, arthritis involving the glenohumeral or acromioclavicular joint, sympathetic dystrophy and abnormality of plain radiography of the shoulder joint. Extrinsic problems such as a neuromuscular disorder (Parkinsonism) or referred pain from an associated condition, such as extrusion of cervical disc with radiculopathy Method of diagnosis: Standardised physical examination in upright position; active and passive range of movement of both shoulders was measured using a standard goniometer. Shoulder radiographs were taken Terminology used: FS; adhesive capsulitis |
Age (years), mean (range): Total: 54 (38 to 65) Female: 61% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (weeks), mean (range): Total: 10 (3 weeks to 10 months) Stage of FS at baseline: 67% pain and stiffness, 12% pain, 21% stiffness Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT: Local injection of 80 mg Kenacort combined with 2% Xylocaine in shoulder joint 1 cm distal and 1 cm lateral to coracoid process. Manipulated extremity was kept in 160° of abduction, with 90° of external rotation, using a cotton bandage secured to the wrist and tied to the back of the bed for 24 hours. Patients were allowed to untie their extremity for toileting only. All patients were discharged after 24 hours with daily physiotherapy sessions of 30 minutes for 3 weeks | MUA + steroid injection + PT: Local injection of 80 mg Kenacort combined with 2% Xylocaine in shoulder joint 1 cm distal and 1 cm lateral to coracoid process. All patients were discharged after 24 hours with daily physiotherapy sessions of 30 minutes for 3 weeks | NR | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Jacobs 2009 85 RCT Country, setting and treatment provider: UK; MUA performed by orthopaedic surgeon. MUA patients shown exercises by physiotherapist. Distension with steroids performed in hospital orthopaedic outpatient department |
Inclusion criteria (including definition of FS): Consecutive patients who presented to the upper limb service at the Royal Oldham Hospital with primary FS Exclusion criteria: Type 1 or 2 diabetes and other medical conditions known to be associated with FS; steroid injection in the affected shoulder before referral Method of diagnosis: Medical history; clinical examination including radiography Terminology used: FS; adhesive capsulitis |
Age (years), median: MUA: 56.5; steroid injection + distension: 57 Female: 66% Any participants with diabetes? No |
Duration of FS at baseline (weeks), median: MUA: 19; steroid injection + distension: 16 Stage of FS at baseline: Most patients were in ‘freezing phase’ of FS (based on duration of symptoms) Previous treatments for FS: NR Participants with secondary FS: No |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
MUA: Anaesthetised patients were positioned on the opposite side to that being manipulated. The assistant placed the heel of the hand on the lateral border of the ipsilateral scapula to stabilise it. Using a short lever arm, the patient’s arm was manipulated into full adduction and forward flexion, full external rotation, full internal rotation and, finally, full abduction. All patients were treated as day cases Home exercise |
Steroid injection + distension: Three steroid and distension treatments at 6-week intervals. The injection consisted of 1 ml of triamcinolone (40 mg), 5 ml of 2% lidocaine, 10 ml of 0.25% bupivacaine and 5 ml of air and was given by the posterior route. The needle was inserted 1–2 cm below the corner of the acromion into the ‘soft spot’ and directed towards the index finger, entering the glenohumeral joint. The air provides a palpable and occasionally audible ‘squelch’ confirming that the injection is in the glenohumeral joint and that the joint capsule has not been ruptured by the injection Home exercise |
NR Home exercise: For MUA as shown by the physiotherapist; the steroid injection group was given a sheet detailing the same exercises |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Kivimaki 2007 39 RCT Country, setting and treatment provider: Finland; three regional hospitals in southern Finland; a physician performed MUA and physiotherapists gave physiotherapy advice |
Inclusion criteria: Adult patients with gradually increasing shoulder pain and stiffness; shoulder mobility of no more than 140° in elevation and 30° in external rotation Exclusion criteria: Arthritis, osteoarthritis or traumatic bone or tendon changes in the affected shoulder; in patients with weak external rotation or abduction ultrasound examination was performed and patients with verified rotator cuff rupture were excluded Method of diagnosis: Specialists in physical medicine and rehabilitation diagnosed FS by radiography and measuring range of movement Terminology used: Frozen shoulder |
Age (years), mean (SD): MUA: 53 (8.4); control: 53 (8.6) Female: 68% Any participants with diabetes? Yes. 18 patients (reported as equal in both groups) |
Duration of FS at baseline: Mean duration of shoulder pain was 7 months in both groups (range 3 to 22 months) Stage of FS at baseline: Stiff and painful Previous treatments for FS: In the 3 months before randomisation no differences were reported between groups regarding reported physiotherapy, massage or chiropractic manipulations, and patients had used analgesics for 36 days equally in both groups Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| MUA + PT: Manipulations were performed < 2 weeks after randomisation under short general anaesthesia. Patients were supine and, after confirming the capsular contracture, the physician lifted the affected extremity and pushed the upper arm into flexion and abduction while supporting the scapula against the thoracic cage. After the shoulder was stretched into flexion the elbow was raised to a right angle and the upper arm was gently rotated into internal and external rotation. Any cracking sound in the shoulder joint during manipulation was recorded. Normal or near-normal mobility was achieved. The patients received physiotherapy advice in two sessions and written instructions for a daily training programme (home exercise) | Home exercise: The patients received physiotherapy advice in two sessions and written instructions for a daily training programme (home exercise) |
The most frequently used treatment was a prescription for analgesics. Home exercise: Daily training programme that included pendulum exercises for the arm and stretching exercises for the shoulder joint |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Quraishi 2007 38 RCT Country, setting and treatment provider: UK; MUA performed by consultant orthopaedic surgeon; arthrographic distension performed by radiologist |
Inclusion criteria: Aged > 18 years, stage II adhesive capsulitis, global loss of active and passive shoulder movement, restriction of rotation < 50% of normal and normal anteroposterior and axillary lateral radiography of the glenohumeral joint Exclusion criteria: Post-traumatic or other extrinsic cause, suspected osteoporosis, unfit for general anaesthesia Method of diagnosis: Clinical assessment by consultant surgeon Terminology used: Adhesive capsulitis |
Age (years), mean (range): MUA: 54.5 (36 to 69); arthrographic distension: 55.2 (44 to 70) Female: 58% Any participants with diabetes? Yes. Total: n = 6; MUA: n = 3 (18%, all type 1); arthrographic distension: n = 3 (16%, all type 1) |
Duration of FS at baseline (weeks), mean (range): MUA: 39.8 (16 to 102); arthrographic distension: 37.4 (12 to 76) Stage of FS at baseline: Stage II primary adhesive capsulitis Previous treatments for FS: Physiotherapy: n = 16; steroid injection: n = 22; physiotherapy + steroid injection: n = 11 Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
MUA + steroid injection: Restoration of shoulder movement following a specific protocol to ensure safe breakage of adhesions by using a short lever arm. 2 ml of 2% lidocaine and 0.75 ml (30 mg) of triamcinolone acetonide was injected anteriorly into the glenohumeral joint without guidance Home exercise |
Arthrographic distension: Needle inserted into glenohumeral joint using anterior approach. Position of needle was checked by image intensifiers before and after injection of radio-opaque contrast material. Normal saline (10–55 ml) was then injected to progressively distend the capsule until it ruptured, which was usually through the subscapularis bursa, but occasionally down the biceps sheath Home exercise |
NR Home exercise: Self-exercise programme of pendular exercises and wall-climbing movements |
||
Appendix 6.6 Distension
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|---|---|---|---|---|
|
Buchbinder 2004 43 RCT Country, setting and treatment provider: Australia; single radiologist; community-based rheumatology practice |
Inclusion criteria: Aged > 18 years, pain and stiffness in predominantly one shoulder for ≥ 3 months, restriction of passive range of movement of > 30° in two or more planes of movement, measured to onset of pain Exclusion criteria: Severe pain at rest (> 7 out of 10 on VAS), previous arthrographic distension, systemic inflammatory joint disease (including rheumatoid arthritis, polymyalgia rheumatica); radiological evidence of osteoarthritis of the shoulder or fracture, calcification about the shoulder joint, reason to suspect a complete rotator cuff tear Method of diagnosis: Passive range of movement was measured to onset of pain using a gravity inclinometer; radiography Terminology used: Painful stiff shoulder |
Age (years), mean (SD): Arthrographic distension: 57.2 (8.6); sham distension: 57.5 (8.1) Female: 80% Any participants with diabetes? Yes. Arthrographic distension: n = 8 (32%); sham distension: n = 5 (24%) |
Duration of FS at baseline (days), median (range): Arthrographic distension: 118 (102 to 194); sham distension: 114 (96 to 402) Stage of FS at baseline: Not reported Previous treatments for FS: Previous corticosteroid injection: arthrographic distension: n = 7 (28%); sham distension: n = 6 (28.6%) Participants with secondary FS: Yes. Total: n = 1 (2.2%); arthrographic distension: n = 0; sham distension: n = 1 (postoperative capsulitis) |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
|
Arthrographic distension + steroid: Participants were in a supine position with the affected arm externally rotated and a sandbag on the hand. The image intensifier was centred on the glenohumeral joint, cone open to include scapula and upper third of the humerus, and the image intensifier to table top distance set at 50 cm. The skin was marked for arthrogram needle site and infiltrated with local anaesthetic. The arthrogram needle was positioned, connected to the connector tap and tube and 0.5–1 ml of contrast injected and a radiographic image taken; 40 mg of methylprednisolone acetate (Depo-Medrol®, 1 ml) and up to 82 ml of normal saline was then injected (total volume 30–90 ml). The end point of the procedure was filling of the subscapular bursa, capsular rupture, injection of the total volume of liquid or the participant requesting termination of the procedure Home exercise |
Placebo: Arthrogram only. Same as for intervention except that there was no injection of steroid and saline Home exercise |
Paracetamol; codeine preparations allowed. Participants were asked to stop taking any NSAIDs No manual treatment (e.g. physiotherapy, massage, chiropractic) or other medical interventions (e.g. intra-articular steroid injection) were allowed Home exercise: Participants received a simple exercise programme comprising pendular exercises and scapular setting (isometric scapular retraction) |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Gam 1998 86 RCT Country, setting and treatment provider: Denmark; hospital |
Inclusion criteria: FS of > 6 weeks’ duration; aged between 18 and 70 years; nocturnal accentuation of pain; passive range of shoulder external rotation < 50% of opposite shoulder; no effusion in glenohumeral joint; normal radiography of affected shoulder; normal erythrocyte sedimentation rate, haemoglobin, leucocytes and alkaline phosphatase and negative immunoglobulin M rheumatoid factor Exclusion criteria: Trauma to shoulder in previous 6 months that caused pain or restricted movement of shoulder within 1 week (trivial minor injuries accepted); diabetes; treatment for FS (except analgesics) during study period Method of diagnosis: Diagnosis by authors. Diagnosis was defined after clinical examination, blood samples and radiography or diagnostic ultrasound Terminology used: FS; adhesive capsulitis; scapulohumeral periarthritis |
Age (years), median (25–75 percentiles): Steroid injection: 47 (43–54); steroid injection + distension: 53.5 (50–63) Female: 59% Any participants with diabetes? No |
Duration of FS at baseline (months), median (25–75 percentiles): Steroid injection: 4.5 (3.3–5.8); steroid injection + distension: 5 (4.3 –6.0) Stage of FS at baseline: NR Previous treatments for FS: NR Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Distension + steroid: Intra-articular injection, confirmed by ultrasound, of 20 mg of triamcinolone hexacetonide into the affected glenohumeral joint using the posterior approach with a 1.5-inch needle perpendicular to the scapular spine. Once per week for a maximum of 6 weeks or until no symptoms. An additional 19 ml of lidocaine 0.5% was injected for distension | Steroid injection: As for combined intervention except no injection of lidocaine for distension | Analgesic use (details not specified) | ||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) | |
|
Tveita 2008 36 RCT Country, setting and treatment provider: Norway; hospital |
Inclusion criteria: Limitation of passive movement in the glenohumeral joint compared with the unaffected side, > 30° for at least two of forward flexion, abduction or external rotation; patients with previous FS in opposite shoulder accepted even if difference between sides was < 30°; pain in predominantly one shoulder lasting > 3 months and < 2 years; age > 18 years or > 70 years Exclusion criteria: Patients who could not comply with range of movement measurement procedures; diabetes mellitus; trauma to shoulder in the previous 6 months that required hospital care; serious mental illness; various contraindications to injections; patients currently taking corticosteroid tablets; reduction of glenohumeral range for reasons other than ‘classic’ adhesive capsulitis Method of diagnosis: Clinical history and radiography Terminology used: Adhesive capsulitis |
Age (years), mean (SD): Steroid injection: 52 (7); hydrodilatation: 51 (6) Female: 59% Any participants with diabetes? No |
Duration of FS at baseline (months), mean (SD): Steroid injection: 7 (4); hydrodilatation: 7 (4) Stage of FS at baseline: NR Previous treatments for FS: At baseline: patients undergoing physiotherapy n = 13 (8 in steroid injection group, 5 in hydrodilatation group); patients taking analgesics daily n = 20 (9 in steroid injection group, 11 in hydrodilatation group) Participants with secondary FS: None reported |
|
| Intervention 1 | Intervention 2 | Intervention 3 | Control | Concomitant treatment and details of home exercise |
| Arthrographic distension + steroid: Dilatation with a corticosteroid, a contrast agent, local anaesthetic and saline necessary for dilatation and rupturing of the joint capsule. Injections were performed as for the corticosteroid group, except that the 4 ml of contrast medium, 2 ml of triamcinolone acetonide, 4 ml of local anaesthetic and 10 ml of saline were injected slowly into the joint. When resistance was met the injection was halted for a while and then continued. The capsule would usually rupture in the wall of the subscapular recess or sometimes in the wall of the bicipital or axillary recess, which was recorded as a loss of resistance, and contrast leakage was identified by fluoroscopy. If rupture had not occurred, more Ulravist/Marcain was injected until rupture. Three injections with 2-week intervals were given. The steps of the dilatation procedure were documented with repeat radiography examinations | Steroid injection: The Kaye–Schneider technique was used to perform arthrograms. Patients were supine and a mark was made with a pen on the glenohumeral joint space at about the junction of its middle and lower third using image-intensified fluoroscopy. The joint was punctured by a needle (22-gauge intramuscular needle); 3–4 ml of contrast medium (iopromide, Ultravist 300, Schering AG), 2 ml of triamcinolone acetonide (Kenacort, 10 mg/ml) and 3–4 ml of bupivacaine hydrochloride (Marcain, 5 mg/ml) were injected slowly. The position of the needle was checked frequently using fluoroscopy. Three injections with 2-week intervals were given. The steps of the injection were documented with repeat radiography examinations |
Pain medication organised by the patients’ primary care physicians Patients were allowed to proceed with their current physiotherapy programme; no patients were prescribed new physiotherapy programmes during the study |
||
Appendix 6.7 Capsular release
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) |
|---|---|---|---|
|
Austgulen 2007 87 Case series Country, setting and treatment provider: Norway; Bergen Surgical Hospital; one surgeon performed surgery |
Inclusion criteria: Patients with primary FS and stiffness typical of FS. Physiotherapy must have been tried previously without a satisfactory result Exclusion criteria: Secondary causes of FS (e.g. trauma) excluded Method of diagnosis: Diagnosis of FS was confirmed during anaesthesia; limited outward rotation of < 20° and < 45° in abduction with a fixed scapula Terminology used: FS |
Age (years), average (range): 53 (34 to 71) Female: 67% Any participants with diabetes? Yes. n = 11 with diabetes |
Duration of FS at baseline (months), average (range): 13 (3 to 60 months) Stage of FS at baseline: NR Previous treatments for FS: Physiotherapy Participants with secondary FS: None reported |
| Intervention | Concomitant treatment and details of home exercise | ||
|
Arthroscopic capsular and ligament release and PT: Surgery was performed using intravenous anaesthesia with addition of local infiltration. Patients were operated on in a beach chair position. Shoulder arthroscopy was performed using normal technique, with access to the shoulder joint from behind and surgical instruments entered in front in the rotator interval. The rotator interval was cleaned and the entire frontal capsule and glenohumeral ligament and coracohumeral ligament were split from the bicep tendon to 6 o’clock. Both capsules and ligaments were split with Acufex Upbiter Scissor punch. The subacromial space was inspected and adherences were loosened. The space and displacement were evaluated. Where spaces were narrow subacromial decompression was performed until it was possible to outwardly rotate the shoulder to maximum and abduct to 180°. All patients received aggressive rehabilitation with a physiotherapist from the first day after surgery Home exercise |
NSAIDs and other pain relief given as needed. Oxycodone was given sometimes postoperatively. Patients received 40-mg bupivacaine injections into shoulder joints and cold packs pre- and postoperatively Home exercise: All did home exercises every day and performed stretches at home every day |
||
| Study | Inclusion/exclusion criteria and diagnosis of FS | Participant characteristics (age, sex, diabetes) | Condition-related characteristics (duration and stage of FS, previous treatments, secondary FS) |
|
Chen 2002 88 Case series Country, setting and treatment provider: Taiwan; Kaohisiung Medical University Hospital |
Inclusion criteria: Basic criteria for definition of idiopathic FS Exclusion criteria: NR Method of diagnosis: Idiopathic FS diagnosed using history, physical examination, X-ray and arthrography Terminology used: FS |
Age (years), range: 32 to 79 Female: 75% Any participants with diabetes? Unclear/NR |
Duration of FS at baseline (months), average (range): 8 (3 months to 4 years) Stage of FS at baseline: NR Previous treatments for FS: Participants had not responded to conservative treatment of at least 12 weeks’ duration Participants with secondary FS: No |
| Intervention | Concomitant treatment and details of home exercise | ||
| Arthroscopic brisement (distension, debride, release) followed by gentle manipulation and PT: Distension was first undertaken to allow insertion of the arthroscope. The synovium was removed by arthroscopic shaver or vaporisation. The authors state that an attempt should be made to resect the areas of synovitis in the axillary pouch. In stage 3 disease residual synovial thickening or fibrotic changes are seen but the hypervascular appearance has resolved. The sheet of capsular tissue was debrided carefully. On removal of the arthroscopic instruments, a gentle manipulation was performed. The arm was elevated in the scapular plane (which was usually associated with audible popping of the contracted capsule) and then externally rotated and then internally rotated at varying degrees of abduction. This was done with gradual pressure and stopped if unyielding resistance was met. Repetition of these steps led to tearing of the capsular structures. The arm was then kept in the abduction–external rotation position for 2 days during which the patient was confined to bed. Passive and active exercise of the shoulder was then allowed, with a rehabilitation programme at the hospital rehabilitation facility (further details of the procedure are provided in the paper) | NR | ||
Appendix 7 Data extraction tables
Appendix 7.1 Steroid injection
| Study: Bal 200868 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain at night) measured using VAS 0–100 mm | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 77.5 | 20.0 | |
| Placebo injection | 40 (40 shoulders) | 70.0 | 40.0 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | –36.5 | 25.1 | 0.07 |
| Placebo injection | 24 shoulders | –26.5 | 25.1 | ||
| Steroid injection | 12 weeks | 42 shoulders | –53.1 | 27.8 | 0.552 |
| Placebo injection | 24 shoulders | –51.7 | 28.1 | ||
| Outcome: Pain (pain on activity) measured using SPADI 5-item pain subscale | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 71 | 39.7 | |
| Placebo injection | 40 (40 shoulders) | 66 | 25.0 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | –30.1 | 22.1 | 0.041 |
| Placebo injection | 24 shoulders | –19 | 17.6 | ||
| Steroid injection | 12 weeks | 42 shoulders | –42.4 | 25.5 | 0.684 |
| Placebo injection | 24 shoulders | –44.8 | 19.4 | ||
| Study: Bal 200868 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using UCLA Shoulder scale | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | Baseline data not reported | ||
| Placebo injection | 40 (40 shoulders) | ||||
| Intervention a | Time point | No. analysed | Median | IQR | |
| Steroid injection | 2 weeks | 42 shoulders | 26.5 | 4.5 | |
| Placebo injection | 24 shoulders | 23 | 6.5 | ||
| Steroid injection | 12 weeks | 42 shoulders | 32.5 | 6.2 | |
| Placebo injection | 24 shoulders | 31.5 | 7.7 | ||
| Outcome: Function and disability measured using SPADI total score | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 69.4 | 40.5 | |
| Placebo injection | 40 (40 shoulders) | 70.5 | 25.6 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | –30.9 | 19.9 | 0.047 |
| Placebo injection | 24 shoulders | –20.2 | 15 | ||
| Steroid injection | 12 weeks | 42 shoulders | –44.4 | 24 | 0.407 |
| Placebo injection | 24 shoulders | –48.2 | 16.3 | ||
| Outcome: Function and disability measured using SPADI 8-item disability subscale | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 63.4 | 38.1 | |
| Placebo injection | 40 (40 shoulders) | 70.5 | 24.8 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | –28.8 | 21.2 | 0.301 |
| Placebo injection | 24 shoulders | –23.1 | 17.8 | ||
| Steroid injection | 12 weeks | 42 shoulders | –42.2 | 26.3 | 0.156 |
| Placebo injection | 24 shoulders | –49.8 | 18.8 | ||
| Study: Bal 200868 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement (°) – passive abduction | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 107.5 | 41.2 | |
| Placebo injection | 40 (40 shoulders) | 90 | 27.5 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | 36.5 | 27.1 | 0.033 |
| Placebo injection | 24 shoulders | 18.7 | 26.8 | ||
| Steroid injection | 12 weeks | 42 shoulders | 57.8 | 27.9 | 0.639 |
| Placebo injection | 24 shoulders | 54.2 | 29.1 | ||
| Outcome: Range of movement (°) – passive external rotation | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 50 | 31.2 | |
| Placebo injection | 40 (40 shoulders) | 40 | 17.5 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | 18.4 | 16.3 | 0.173 |
| Placebo injection | 24 shoulders | 12.9 | 13.4 | ||
| Steroid injection | 12 weeks | 42 shoulders | 27.4 | 19.5 | 0.421 |
| Placebo injection | 24 shoulders | 31.2 | 20.1 | ||
| Outcome: Range of movement (°) – passive internal rotation | |||||
| Intervention a | Time point | No. randomised | Median | IQR | |
| Steroid injection | Baseline | 40 (42 shoulders) | 55 | 25.0 | |
| Placebo injection | 40 (40 shoulders) | 47.5 | 10.0 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SD | p-value b |
| Steroid injection | 2 weeks | 42 shoulders | 16.5 | 19.1 | 0.088 |
| Placebo injection | 24 shoulders | 9.8 | 14.9 | ||
| Steroid injection | 12 weeks | 42 shoulders | 25.7 | 19.1 | 0.693 |
| Placebo injection | 24 shoulders | 54.2 | 29.1 | ||
| Study: Bal 200868 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Bal 200868 |
|---|
| Outcome: Other |
| Not reported |
| Study: Bal 200868 |
|---|
| Outcome: Adverse events |
| No side effects were noted during the drug or exercise therapy sessions |
| Study: Carette 200335 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using SPADI 5-item pain subscale | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection + PT | Baseline | 21 | 69.0 | 17.1 |
| Steroid injection | 23 | 70.2 | 16.4 | |
| PT + placebo injection | 26 | 65.4 | 19.7 | |
| Placebo injection | 23 | 69.1 | 18.3 | |
| Intervention a | Time point | No. analysed | Mean change from baseline | SE |
| Steroid injection + PT | 6 weeks | 21 | –48.7 | 5.9 |
| Steroid injection | 23 | –39.1 | 5.6 | |
| PT + placebo injection | 26 | –21.8 | 5.3 | |
| Placebo injection | 23 | –17.3 | 5.6 | |
| Steroid injection + PT | 3 months | 21 | –52.1 | 5.9 |
| Steroid injection | 23 | –48.1 | 5.6 | |
| PT + placebo injection | 26 | –38.1 | 5.3 | |
| Placebo injection | 23 | –30.1 | 5.6 | |
| Steroid injection + PT | 6 months | 21 | –52.8 | 5.9 |
| Steroid injection | 23 | –54.9 | 5.6 | |
| PT + placebo injection | 26 | –43.8 | 5.3 | |
| Placebo injection | 23 | –36.4 | 5.6 | |
| Steroid injection + PT | 12 months | 21 | –48.4 | 5.9 |
| Steroid injection | 23 | –52.6 | 5.6 | |
| PT + placebo injection | 26 | –46.1 | 5.3 | |
| Placebo injection | 23 | –46.0 | 5.6 | |
| Outcome: Pain (pain overall) measured using SPADI 5-item pain subscale – calculated valuesb | ||||
| Intervention a | Time point | No. analysed | Mean | SD c |
| Steroid injection + PT | 3 months | 21 | –52.1 | 13.1 |
| Steroid injection | 23 | –48.1 | 13.1 | |
| PT + placebo injection | 26 | –38.1 | 13.1 | |
| Placebo injection | 23 | –30.1 | 13.1 | |
| Study: Carette 200335 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using SPADI total score | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 66.4 | 15.5 | |
| Steroid injection | 23 | 66.6 | 17.6 | ||
| PT + placebo injection | 26 | 61.5 | 16.5 | ||
| Placebo injection | 23 | 67.3 | 17.5 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | –46.5 | 5.3 | 24.3 |
| Steroid injection | 23 | –36.7 | 5.1 | 24.5 | |
| PT + placebo injection | 26 | –22.2 | 4.8 | 24.5 | |
| Placebo injection | 23 | –18.9 | 5.1 | 24.5 | |
| Steroid injection + PT | 3 months | 21 | –50.4 | 5.3 | 24.3 |
| Steroid injection | 23 | –45.5 | 5.1 | 24.5 | |
| PT + placebo injection | 26 | –37.9 | 4.8 | 24.5 | |
| Placebo injection | 23 | –29.4 | 5.1 | 24.5 | |
| Steroid injection + PT | 6 months | 21 | –52.5 | 5.3 | 24.3 |
| Steroid injection | 23 | –51.3 | 5.1 | 24.5 | |
| PT + placebo injection | 26 | –43.1 | 4.8 | 24.5 | |
| Placebo injection | 23 | –38.4 | 5.1 | 24.5 | |
| Steroid injection + PT | 12 months | 21 | –48.3 | 5.3 | 24.3 |
| Steroid injection | 23 | –50.1 | 5.1 | 24.5 | |
| PT + placebo injection | 26 | –45.5 | 4.8 | 24.5 | |
| Placebo injection | 23 | –47.2 | 5.1 | 24.5 | |
| Outcome: Function and disability measured using SPADI 8-item disability subscale | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 63.8 | 16.9 | |
| Steroid injection | 23 | 63 | 20.6 | ||
| PT + placebo injection | 26 | 57.6 | 17.3 | ||
| Placebo injection | 23 | 65.5 | 19.3 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | –44.3 | 5.2 | 23.8 |
| Steroid injection | 23 | –34.2 | 5 | 24.0 | |
| PT + placebo injection | 26 | –22.7 | 4.7 | 24.0 | |
| Placebo injection | 23 | –20.4 | 4.9 | 23.5 | |
| Steroid injection + PT | 3 months | 21 | –48.7 | 5.2 | 23.8 |
| Steroid injection | 23 | –43 | 5 | 24.0 | |
| PT + placebo injection | 26 | –37.7 | 4.7 | 24.0 | |
| Placebo injection | 23 | –28.7 | 4.9 | 23.5 | |
| Steroid injection + PT | 6 months | 21 | –52.1 | 5.2 | 23.8 |
| Steroid injection | 23 | –47.6 | 5 | 24.0 | |
| PT + placebo injection | 26 | –42.3 | 4.7 | 24.0 | |
| Placebo injection | 23 | –40.5 | 4.9 | 23.5 | |
| Steroid injection + PT | 12 months | 21 | –48.1 | 5.2 | 23.8 |
| Steroid injection | 23 | –47.6 | 5 | 24.0 | |
| PT + placebo injection | 26 | –45 | 4.7 | 24.0 | |
| Placebo injection | 23 | –48.4 | 4.9 | 23.5 | |
| Study: Carette 200335 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 57.5 | 11.8 | |
| Steroid injection | 23 | 58.2 | 10.4 | ||
| PT + placebo injection | 26 | 57.1 | 9.8 | ||
| Placebo injection | 23 | 56.7 | 14.4 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | 21 | 3 | 13.7 |
| Steroid injection | 23 | 12.9 | 2.9 | 13.9 | |
| PT + placebo injection | 26 | 9.7 | 2.7 | 13.8 | |
| Placebo injection | 23 | 5.7 | 2.9 | 13.9 | |
| Steroid injection + PT | 3 months | 21 | 24.4 | 3 | 13.7 |
| Steroid injection | 23 | 20.4 | 2.9 | 13.9 | |
| PT + placebo injection | 26 | 14 | 2.7 | 13.8 | |
| Placebo injection | 23 | 8.1 | 2.9 | 13.9 | |
| Steroid injection + PT | 6 months | 21 | 27.3 | 3 | 13.7 |
| Steroid injection | 23 | 22.1 | 2.9 | 13.9 | |
| PT + placebo injection | 26 | 19.4 | 2.7 | 13.8 | |
| Placebo injection | 23 | 14.3 | 2.9 | 13.9 | |
| Steroid injection + PT | 12 months | 21 | 29.7 | 3 | 13.7 |
| Steroid injection | 23 | 22.8 | 2.9 | 13.9 | |
| PT + placebo injection | 26 | 24.7 | 2.7 | 13.8 | |
| Placebo injection | 23 | 24 | 2.9 | 13.9 | |
| Outcome: Range of movement – passive external rotation (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 7.9 | 11.6 | |
| Steroid injection | 23 | 15.1 | 14 | ||
| PT + placebo injection | 26 | 14.9 | 14.1 | ||
| Placebo injection | 23 | 17.9 | 18.3 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | 26.5 | 3.6 | 16.5 |
| Steroid injection | 23 | 18.3 | 3.4 | 16.3 | |
| PT + placebo injection | 26 | 9.6 | 3.2 | 16.3 | |
| Placebo injection | 23 | 7.1 | 3.4 | 16.3 | |
| Steroid injection + PT | 3 months | 21 | 31 | 3.6 | 16.5 |
| Steroid injection | 23 | 25.5 | 3.4 | 16.3 | |
| PT + placebo injection | 26 | 18 | 3.2 | 16.3 | |
| Placebo injection | 23 | 13.4 | 3.4 | 16.3 | |
| Steroid injection + PT | 6 months | 21 | 34.1 | 3.6 | 16.5 |
| Steroid injection | 23 | 29.5 | 3.4 | 16.3 | |
| PT + placebo injection | 26 | 23 | 3.2 | 16.3 | |
| Placebo injection | 23 | 20.8 | 3.4 | 16.3 | |
| Steroid injection + PT | 12 months | 21 | 38 | 3.6 | 16.5 |
| Steroid injection | 23 | 30.8 | 3.4 | 16.3 | |
| PT + placebo injection | 26 | 31.6 | 3.2 | 16.3 | |
| Placebo injection | 23 | 30.4 | 3.4 | 16.3 | |
| Outcome: Range of movement – hand behind back (cm) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 34.1 | 10.4 | |
| Steroid injection | 23 | 34.7 | 13.1 | ||
| PT + placebo injection | 26 | 37.2 | 10.9 | ||
| Placebo injection | 23 | 34 | 11 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | –19.8 | 2.5 | 11.5 |
| Steroid injection | 23 | –11.3 | 2.4 | 11.5 | |
| PT + placebo injection | 26 | –10.5 | 2.3 | 11.7 | |
| Placebo injection | 23 | –10.8 | 2.4 | 11.5 | |
| Steroid injection + PT | 3 months | 21 | –21.8 | 2.5 | 11.5 |
| Steroid injection | 23 | –15.8 | 2.4 | 11.5 | |
| PT + placebo injection | 26 | –15.9 | 2.3 | 11.7 | |
| Placebo injection | 23 | –14.7 | 2.4 | 11.5 | |
| Steroid injection + PT | 6 months | 21 | –22.8 | 2.5 | 11.5 |
| Steroid injection | 23 | –17.4 | 2.4 | 11.5 | |
| PT + placebo injection | 26 | –18.1 | 2.3 | 11.7 | |
| Placebo injection | 23 | –16.9 | 2.4 | 11.5 | |
| Steroid injection + PT | 12 months | 21 | –22.7 | 2.5 | 11.5 |
| Steroid injection | 23 | –18.4 | 2.4 | 11.5 | |
| PT + placebo injection | 26 | –21.3 | 2.3 | 11.7 | |
| Placebo injection | 23 | –21.2 | 2.4 | 11.5 | |
| Study: Carette 200335 | |||||
|---|---|---|---|---|---|
| Outcome: Quality of life – SF-36 PCS | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 35.2 | 7.8 | |
| Steroid injection | 23 | 37.5 | 9.1 | ||
| PT + placebo injection | 26 | 37.6 | 7.7 | ||
| Placebo injection | 23 | 36.8 | 7.7 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | 6.4 | 1.9 | 8.7 |
| Steroid injection | 23 | 4.4 | 1.9 | 9.1 | |
| PT + placebo injection | 26 | 1.1 | 1.9 | 9.7 | |
| Placebo injection | 23 | 2.5 | 1.9 | 9.1 | |
| Steroid injection + PT | 3 months | 21 | 8.6 | 1.9 | 8.7 |
| Steroid injection | 23 | 10.1 | 1.9 | 9.1 | |
| PT + placebo injection | 26 | 6.4 | 1.9 | 9.7 | |
| Placebo injection | 23 | 5 | 1.9 | 9.1 | |
| Steroid injection + PT | 6 months | 21 | 8.8 | 1.9 | 8.7 |
| Steroid injection | 23 | 10.1 | 1.9 | 9.1 | |
| PT + placebo injection | 26 | 9.1 | 1.9 | 9.7 | |
| Placebo injection | 23 | 6.8 | 1.9 | 9.1 | |
| Steroid injection + PT | 12 months | 21 | 11.5 | 1.9 | 8.7 |
| Steroid injection | 23 | 11.1 | 1.9 | 9.1 | |
| PT + placebo injection | 26 | 9.4 | 1.9 | 9.7 | |
| Placebo injection | 23 | 10.1 | 1.9 | 9.1 | |
| Outcome: Quality of life – SF-36 MCS | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection + PT | Baseline | 21 | 43.1 | 12.9 | |
| Steroid injection | 23 | 49.4 | 10.2 | ||
| PT + placebo injection | 26 | 49.8 | 11.8 | ||
| Placebo injection | 23 | 49 | 12.5 | ||
| Intervention a | Time point | No. analysed | Mean change from baseline | SE | SD b |
| Steroid injection + PT | 6 weeks | 21 | 5.7 | 2.3 | 10.5 |
| Steroid injection | 23 | 1.5 | 2.2 | 10.6 | |
| PT + placebo injection | 26 | 2 | 2.2 | 11.2 | |
| Placebo injection | 23 | 2.6 | 2.2 | 10.6 | |
| Steroid injection + PT | 3 months | 21 | 6.6 | 2.3 | 10.5 |
| Steroid injection | 23 | 2.2 | 2.2 | 10.6 | |
| PT + placebo injection | 26 | 3.8 | 2.2 | 11.2 | |
| Placebo injection | 23 | 1.2 | 2.2 | 10.6 | |
| Steroid injection + PT | 6 months | 21 | 9.2 | 2.3 | 10.5 |
| Steroid injection | 23 | 3.8 | 2.2 | 10.6 | |
| PT + placebo injection | 26 | 1 | 2.2 | 11.2 | |
| Placebo injection | 23 | 2.1 | 2.2 | 10.6 | |
| Steroid injection + PT | 12 months | 21 | 9.3 | 2.3 | 10.5 |
| Steroid injection | 23 | 3.5 | 2.2 | 10.6 | |
| PT + placebo injection | 26 | 2.4 | 2.2 | 11.2 | |
| Placebo injection | 23 | 3.2 | 2.2 | 10.6 | |
| Study: Carette 200335 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Carette 200335 |
|---|
| Outcome: Other |
| Not reported |
| Study: Dacre 198967 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain at night, pain with active and passive movement, day pain) measured using 10-cm VAS scalea | ||||
| Intervention | Time point | No. randomised | Mean | p-value |
| Steroid injection + PT | Baseline | Unclearb | Baseline data not reported | |
| Steroid injection | Unclearb | |||
| PT | Unclearb | |||
| Intervention | Time point | No. analysed | Mean | p-value |
| Steroid injection + PT | 6 weeks | 20 |
Mean measures of pain improved by 57–86% at 6 months Pain on active movement improved by means of between 5% and 58% after 6 weeks |
All groups showed significant reduction in pain after 6 weeks (p < 0.001) |
| Steroid injection | 22 | |||
| PT | 20 | |||
| Steroid injection + PT | 6 months | 20 |
Mean measures of pain improved by 57–86% at 6 months Pain on active movement improved by means of between 62% and 78% at 6 months in the three groups (not significant) |
|
| Steroid injection | 22 | |||
| PT | 20 | |||
| Study: Dacre 198967 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Dacre 198967 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – complete shoulder abduction, glenohumeral abduction and external rotation | ||||
| Intervention | Time point | No. randomised | Mean | p-value |
| Steroid injection + PT | Baseline | (Unclear)b | Baseline data not reported | |
| Steroid injection | (Unclear)b | |||
| PT | (Unclear)b | |||
| Intervention | Time point | No. analysed | Mean | p-value |
| Steroid injection + PT | 6 weeks | 20 | Mean measures of movement improved by between 11% and 31% at 6 weeks | |
| Steroid injection | 22 | Total abduction improved by means of between 10% and 11% after 6 weeks in the three groups (not significant) | p > 0.05 | |
| PT | 20 | |||
| Steroid injection + PT | 6 months | 20 | Mean measures of movement improved by between 10% and 34% at 6 months | |
| Steroid injection | 22 | Total abduction improved by means of between 15% and 23% at 6 months in the three groups | p > 0.05 | |
| PT | 20 | |||
| Study: Dacre 198967 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Dacre 198967 |
|---|
| Outcome: Other |
| Not reported |
| Study: Dacre 198967 |
|---|
| Outcome: Adverse events |
| Stated that no patients had adverse reactions |
| Study: Rizk 199142 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using 0–5 Likert scale (0 = no pain, 5 = extreme pain) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Steroid injection (anterior approach) + PT | Baseline | 16 | 3.88 | NR | |
| Steroid injection (lateral approach) + PT | 16 | 3.7 | NR | ||
| Two arms for placebo injection (anterior approach and lateral) + PTb | 16 | 4.07 | NR | ||
| Intervention a | Time point | No. analysed | Mean | Combined mean c | SD d |
| Steroid injection (anterior approach) + PT | 4 weeks | 15 | 3.87 | NA | NA |
| Steroid injection (lateral approach) + PT | 14 | 3.71 | |||
| Two arms for placebo injection (anterior approach and lateral) + PTb | 15 | 3.93 | NA | NA | |
| Steroid injection (anterior approach) + PT | 11 weeks | 15 | 3.47 | 3.417 | 1.557 |
| Steroid injection (lateral approach) + PT | 14 | 3.36 | |||
| Two arms for placebo injection (anterior approach and lateral) + PTb | 15 | 3.13 | 3.13 | 1.653 | |
| Steroid injection (anterior approach) + PT | 24 weeks | 15 | 3.2 | 3.1034 | 1.742 |
| Steroid injection (lateral approach) + PT | 14 | 3 | |||
| Two arms for placebo injection (anterior approach and lateral) + PTb | 15 | 3 | 3 | 1.673 | |
| Study: Rizk 199142 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Rizk 199142 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection (anterior approach) + PT | Baseline | 16 | 72.7 | NR |
| Steroid injection (lateral approach) + PT | 16 | 76.1 | NR | |
| Placebo injection (anterior approach) + PT | 8 | 71.9 | NR | |
| Placebo injection (lateral approach) + PT | 8 | 72.5 | NR | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Steroid injection (anterior approach) + PT | 1 week | Unclear | 72.8 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 76.4 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 71.9 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 72.5 | NR | |
| Steroid injection (anterior approach) + PT | 2 weeks | Unclear | 72.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 75.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 72.1 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 72.5 | NR | |
| Steroid injection (anterior approach) + PT | 3 weeks | Unclear | 72.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 75.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 72.3 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 72.8 | NR | |
| Steroid injection (anterior approach) + PT | 4 weeks | Unclear | 73 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 75.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 72.6 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 73.1 | NR | |
| Steroid injection (anterior approach) + PT | 5 weeks | Unclear | 73.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 75.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 72.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 73.3 | NR | |
| Steroid injection (anterior approach) + PT | 6 weeks | Unclear | 73.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 76.3 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 73.1 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 73.3 | NR | |
| Steroid injection (anterior approach) + PT | 7 weeks | Unclear | 74.4 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 76.8 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 73.3 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 73.7 | NR | |
| Steroid injection (anterior approach) + PT | 8 weeks | Unclear | 75.3 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 77.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 74.2 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 74.2 | NR | |
| Steroid injection (anterior approach) + PT | 9 weeks | Unclear | 75.8 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 78.3 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 74.2 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 74.2 | NR | |
| Steroid injection (anterior approach) + PT | 10 weeks | Unclear | 75.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 79.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 74.4 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 74.5 | NR | |
| Steroid injection (anterior approach) + PT | 11 weeks | Unclear | 76.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 79.8 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 74.5 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 74.7 | NR | |
| Steroid injection (anterior approach) + PT | 15 weeks | Unclear | 77.5 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 79.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 74.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 75.4 | NR | |
| Steroid injection (anterior approach) + PT | 6 months | Unclear | 78.4 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 81.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 76.6 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 80.1 | NR | |
| Outcome: Range of movement – passive external rotation (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection (anterior approach) + PT | Baseline | 16 | 33.6 | NR |
| Steroid injection (lateral approach) + PT | 16 | 30.1 | NR | |
| Placebo injection (anterior approach) + PT | 8 | 30.4 | NR | |
| Placebo injection (lateral approach) + PT | 8 | 32.3 | NR | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Steroid injection (anterior approach) + PT | 1 week | Unclear | 33.8 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 30.5 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.2 | NR | |
| Steroid injection (anterior approach) + PT | 2 weeks | Unclear | 33.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.2 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 30.5 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.3 | NR | |
| Steroid injection (anterior approach) + PT | 3 weeks | Unclear | 34 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 30.9 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.3 | NR | |
| Steroid injection (anterior approach) + PT | 4 weeks | Unclear | 34.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 31.2 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.3 | NR | |
| Steroid injection (anterior approach) + PT | 5 weeks | Unclear | 34.2 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 31.4 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.8 | NR | |
| Steroid injection (anterior approach) + PT | 6 weeks | Unclear | 34.4 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 31.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 31.5 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 32.8 | NR | |
| Steroid injection (anterior approach) + PT | 7 weeks | Unclear | 34.8 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 31.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 31.9 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 33 | NR | |
| Steroid injection (anterior approach) + PT | 8 weeks | Unclear | 34.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 33.2 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 32.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 33.4 | NR | |
| Steroid injection (anterior approach) + PT | 9 weeks | Unclear | 34.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 33.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 32.9 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 33.3 | NR | |
| Steroid injection (anterior approach) + PT | 10 weeks | Unclear | 34.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 34.8 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 33.3 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 33.3 | NR | |
| Steroid injection (anterior approach) + PT | 11 weeks | Unclear | 34.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 36.7 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 33.4 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 33.3 | NR | |
| Steroid injection (anterior approach) + PT | 15 weeks | Unclear | 36.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 37.3 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 34.9 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 34.2 | NR | |
| Steroid injection (anterior approach) + PT | 6 months | Unclear | 40.2 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 41 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 40.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 39.9 | NR | |
| Outcome: Range of movement – passive internal rotation (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection (anterior approach) + PT | Baseline | 16 | 24.8 | NR |
| Steroid injection (lateral approach) + PT | 16 | 25.2 | NR | |
| Placebo injection (anterior approach) + PT | 8 | 24.9 | NR | |
| Placebo injection (lateral approach) + PT | 8 | 24.3 | NR | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Steroid injection (anterior approach) + PT | 1 week | Unclear | 24.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 25.3 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 24.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 24.4 | NR | |
| Steroid injection (anterior approach) + PT | 2 weeks | Unclear | 24.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 25.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 24.4 | NR | |
| Steroid injection (anterior approach) + PT | 3 weeks | Unclear | 24.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 24.4 | NR | |
| Steroid injection (anterior approach) + PT | 4 weeks | Unclear | 24.9 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.1 | NR | |
| Steroid injection (anterior approach) + PT | 5 weeks | Unclear | 25.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.1 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.3 | NR | |
| Steroid injection (anterior approach) + PT | 6 weeks | Unclear | 25.3 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.7 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.1 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.7 | NR | |
| Steroid injection (anterior approach) + PT | 7 weeks | Unclear | 25.8 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.2 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.7 | NR | |
| Steroid injection (anterior approach) + PT | 8 weeks | Unclear | 26.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.6 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.3 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.8 | NR | |
| Steroid injection (anterior approach) + PT | 9 weeks | Unclear | 26.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 24.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.6 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.7 | NR | |
| Steroid injection (anterior approach) + PT | 10 weeks | Unclear | 26.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 25.5 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 25.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.7 | NR | |
| Steroid injection (anterior approach) + PT | 11 weeks | Unclear | 26.1 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 25.9 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 26.1 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 25.8 | NR | |
| Steroid injection (anterior approach) + PT | 15 weeks | Unclear | 28.7 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 26.1 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 26.7 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 26.4 | NR | |
| Steroid injection (anterior approach) + PT | 6 months | Unclear | 32.4 | NR |
| Steroid injection (lateral approach) + PT | Unclear | 30.8 | NR | |
| Placebo injection (anterior approach) + PT | Unclear | 31.8 | NR | |
| Placebo injection (lateral approach) + PT | Unclear | 30.6 | NR | |
| Study: Rizk 199142 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Rizk 199142 |
|---|
| Outcome: Other |
| Not reported |
| Study: Rizk 199142 |
|---|
| Outcome: Adverse events |
| Study authors stated: ‘No withdrawals due to adverse effects’ |
| Study: Ryans 200541 | ||||
|---|---|---|---|---|
| Outcome: Pain (daytime pain at rest) measured using 100-mm VAS | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection + PT | Baseline | 20 | 31.2 | 21 |
| Steroid injection | 20 | 37.8 | 19.8 | |
| PT + placebo injection | 20 | 45.8 | 24.7 | |
| Placebo injection | 20 | 44.1 | 33.7 | |
| Intervention a | Time point | No. analysed | Mean change from baseline | SD |
| Steroid injection + PT | 6 weeks | 17 | –16.3 | 26.4 |
| Steroid injection | 17 | –9.7 | 18.5 | |
| PT + placebo injection | 20 | –17.6 | 39.1 | |
| Placebo injection | 17 | –5.4 | 27.8 | |
| Steroid injection + PT | 16 weeks | 17 | –16.1 | 24.4 |
| Steroid injection | 13 | –9.8 | 24.7 | |
| PT + placebo injection | 16 | –29.3 | 33.7 | |
| Placebo injection | 12 | –24.5 | 34.2 | |
| Outcome: Pain (daytime pain at rest) measured using 100-mm VAS – calculated values | ||||
| Intervention a | Time point | No. analysed | Mean | SD b |
| Steroid injection + PT | 6 weeks | 17 | 14.9 | 10.39 |
| Steroid injection | 17 | 28.1 | 10.39 | |
| PT + placebo injection | 20 | 28.2 | 10.39 | |
| Placebo injection | 17 | 38.7 | 10.39 | |
| Study: Ryans 200541 | ||||
|---|---|---|---|---|
| Outcome: Function and disability, measured using UK SDQ/Croft score | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection + PT | Baseline | 20 | 13.6 | 4 |
| Steroid injection | 20 | 15.8 | 4.5 | |
| PT + placebo injection | 20 | 14.9 | 4.8 | |
| Placebo injection | 20 | 14.1 | 4.6 | |
| Intervention a | Time point | No. analysed | Mean change from baseline | SD |
| Steroid injection + PT | 6 weeks | 17 | –7.8 | 5.7 |
| Steroid injection | 17 | –6.1 | 6.4 | |
| PT + placebo injection | 20 | –3.5 | 4.9 | |
| Placebo injection | 17 | –3.1 | 3.4 | |
| Steroid injection + PT | 16 weeks | 17 | –7.6 | 5.8 |
| Steroid injection | 13 | –7.8 | 5.9 | |
| PT + placebo injection | 16 | –5.6 | 5.8 | |
| Placebo injection | 12 | –6.6 | 5.4 | |
| Outcome: Function and disability, self-assessed global function measured using 100-mm VAS | ||||
| Intervention a | Time point | Number randomised | Mean | SD |
| Steroid injection + PT | Baseline | 20 | 57.2 | 16.2 |
| Steroid injection | 20 | 65 | 17.2 | |
| PT + placebo injection | 20 | 63.9 | 21.4 | |
| Placebo injection | 20 | 62.7 | 26.9 | |
| Intervention a | Time point | No. analysed | Mean change from baseline | SD |
| Steroid injection + PT | 6 weeks | 17 | –37.8 | 18.1 |
| Steroid injection | 17 | –28.4 | 24.1 | |
| PT + placebo injection | 20 | –26.1 | 26.7 | |
| Placebo injection | 17 | –16.8 | 22 | |
| Steroid injection + PT | 16 weeks | 17 | –39.2 | 25.8 |
| Steroid injection | 13 | –35.6 | 26.9 | |
| PT + placebo injection | 16 | –42.7 | 29.4 | |
| Placebo injection | 12 | –40.7 | 26.7 | |
| Study: Ryans 200541 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – passive external rotation (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Steroid injection + PT | Baseline | 20 | 31.6 | 13.3 |
| Steroid injection | 20 | 31.7 | 14.1 | |
| PT + placebo injection | 20 | 28.1 | 15 | |
| Placebo injection | 20 | 26.7 | 10.3 | |
| Intervention a | Time point | No. analysed | Mean change from baseline | SD |
| Steroid injection + PT | 6 weeks | 17 | 21 | 16.5 |
| Steroid injection | 17 | 14.3 | 15.2 | |
| PT + placebo injection | 20 | 16.7 | 13.2 | |
| Placebo injection | 17 | 6.6 | 13.2 | |
| Steroid injection + PT | 16 weeks | 17 | 19.7 | 19.7 |
| Steroid injection | 13 | 19.1 | 19.2 | |
| PT + placebo injection | 16 | 18 | 14 | |
| Placebo injection | 12 | 22.2 | 18.2 | |
| Study: Ryans 200541 |
|---|
| Outcome: Quality of life – SF-36 domain score and HADS anxiety and HADS depression score |
| Baseline values reported only, therefore data not extracted |
| Study: Ryans 200541 |
|---|
| Outcome: Other |
| Not reported |
| Study: Ryans 200541 |
|---|
| Outcome: Adverse events |
| Not reported |
Appendix 7.2 Sodium hyaluronate
| Study: Calis 200666 |
|---|
| Outcome: Pain (pain overall) measured using VAS |
| Data provided in graphical format only. Text states that there was significant improvement in pain severity at 3 months within all treatment groups including no intervention group (p < 0.001). There was significantly greater improvement in the steroid group than in the placebo group (p = 0.02) |
| Study: Calis 200666 | |||||
| Outcome: Function and disability measured using the Constant score | |||||
|---|---|---|---|---|---|
| Intervention a | Time point | No. randomised | Mean | SD | |
| Sodium hyaluronate injection | Baseline | 24 (27 shoulders) | 50.1 | 8.9 | |
| Steroid injection | 25 (26 shoulders) | 54.6 | 9.7 | ||
| PT | 21 (22 shoulders) | 55.3 | 9.4 | ||
| No intervention | 20 (20 shoulders) | 51.2 | 12.1 | ||
| Intervention a | Time point | No. analysed | Final value mean | SD | p-value |
| Sodium hyaluronate injection | 15 days | 24 (27 shoulders) | 58.4 | 11 | |
| Steroid injection | 25 (26 shoulders) | 66.5 | 11.6 | ||
| PT | 21 (22 shoulders) | 70.2 | 11.6 | < 0.05b | |
| No intervention | 20 (20 shoulders) | 57.9 | 11.5 | ||
| Sodium hyaluronate injection | 3 months | 24 (27 shoulders) | 70.1 | 10.3 | < 0.05c |
| Steroid injection | 25 (26 shoulders) | 70.3 | 9.9 | < 0.05d | |
| PT | 21 (22 shoulders) | 76.1 | 10.7 | < 0.05e | |
| No intervention | 20 (20 shoulders) | 61.2 | 10.8 | ||
| Study: Calis 200666 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Sodium hyaluronate injection | Baseline | 24 (27 shoulders) | 109.6 | 17.8 | |
| Steroid injection | 25 (26 shoulders) | 117.8 | 23.6 | ||
| PT | 21 (22 shoulders) | 116.8 | 16.4 | ||
| No intervention | 20 (20 shoulders) | 114.2 | 22.1 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value |
| Sodium hyaluronate injection | 15 days | 24 (27 shoulders) | 127.2 | 19 | |
| Steroid injection | 25 (26 shoulders) | 135.1 | 23.4 | ||
| PT | 21 (22 shoulders) | 145.4 | 19.2 | < 0.05b | |
| No intervention | 20 (20 shoulders) | 125 | 20.1 | ||
| Sodium hyaluronate injection | 3 months | 24 (27 shoulders) | 145.9 | 21 | |
| Steroid injection | 25 (26 shoulders) | 150.3 | 19.6 | < 0.05c | |
| PT | 21 (22 shoulders) | 158.4 | 18.3 | < 0.05d | |
| No intervention | 20 (20 shoulders) | 133.5 | 15.3 | ||
| Outcome: Range of movement – passive external rotation (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Sodium hyaluronate injection | Baseline | 24 (27 shoulders) | 43.7 | 10.5 | |
| Steroid injection | 25 (26 shoulders) | 47.3 | 12.5 | ||
| PT | 21 (22 shoulders) | 50 | 10.6 | ||
| No intervention | 20 (20 shoulders) | 45.7 | 9 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value |
| Sodium hyaluronate injection | 15 days | 24 (27 shoulders) | 52.9 | 10.7 | |
| Steroid injection | 25 (26 shoulders) | 54.8 | 10.5 | ||
| PT | 21 (22 shoulders) | 63.8 | 11.7 | < 0.05e | |
| No intervention | 20 (20 shoulders) | 52.7 | 9.3 | ||
| Sodium hyaluronate injection | 3 months | 24 (27 shoulders) | 63.3 | 11.4 | |
| Steroid injection | 25 (26 shoulders) | 63 | 10.8 | ||
| PT | 21 (22 shoulders) | 73.8 | 10.4 | < 0.05e | |
| No intervention | 20 (20 shoulders) | 55 | 8.1 | ||
| Study: Calis 200666 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Calis 200666 |
|---|
| Outcome: Other |
| Not reported |
| Study: Calis 200666 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Rovetta 200868 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain on passive joint motion), measured using VAS 0–10 cm | |||||
| Intervention | Time point | No. randomised | Mean a | SD b | |
| Sodium hyaluronate + steroid injection + PT | Baseline | 14 | 7.7 | 1 | |
| Steroid injection + PT | 16 | 7.6 | 1 | ||
| Intervention | Time point | No. analysed | Mean a | SD b | p-value c |
| Sodium hyaluronate + steroid injection + PT | 6 months | 14 | 6 | 1 | < 0.0001 |
| Steroid injection + PT | 16 | 5.2 | 1 | < 0.0001 | |
| Study: Rovetta 200868 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Rovetta 200868 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean a | SD b | |
| Sodium hyaluronate + steroid injection + PT | Baseline | 14 | 77.8 | 6 | |
| Steroid injection + PT | 16 | 76.5 | 56 | ||
| Intervention | Time point | No. analysed | Mean a | SD b | p-value c |
| Sodium hyaluronate + steroid injection + PT | 6 months | 14 | 95.5 | 31 | < 0.01 |
| Steroid injection + PT | 16 | 98.7 | 30 | < 0.05 | |
| Outcome: External rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean a | SD b | |
| Sodium hyaluronate + steroid injection + PT | Baseline | 14 | 16.4 | 5 | |
| Steroid injection + PT | 16 | 12.2 | 3 | ||
| Intervention | Time point | No. analysed | Mean a | SD b | p-value c |
| Sodium hyaluronate + steroid injection + PT | 6 months | 14 | 38.5 | 21 | < 0.001 |
| Steroid injection + PT | 16 | 37.8 | 19 | < 0.001 | |
| Outcome: Range of movement – internal rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean a | SD b | |
| Sodium hyaluronate + steroid injection + PT | Baseline | 14 | 25.7 | 7 | |
| Steroid injection + PT | 16 | 25.3 | 7 | ||
| Intervention | Time point | No. analysed | Mean a | SD b | p-value c |
| Sodium hyaluronate + steroid injection + PT | 6 months | 14 | 27.5 | 10 | <0.001 |
| Steroid injection + PT | 16 | 51.2 | 18 | <0.01 | |
| Study: Rovetta 200868 |
|---|
| Outcome: Quality of Life |
| Not reported |
| Study: Rovetta 200868 |
|---|
| Outcome: Other |
| Joint capsule thickness was reported but data were not extracted as this was not an outcome of interest |
| Study: Rovetta 200868 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Takagishi 199670 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain on activity), measured using VAS 0–10 | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Sodium hyaluronate injection | Baseline | 10 | 8 | 1.8 |
| Steroid injection | 10 | 7.2 | 2.1 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Sodium hyaluronate injection | 5 weeksa | 10 | 3.7 | 1.7 |
| Steroid injection | 10 | 4.2 | 2.7 | |
| Outcome: Pain (no. of patients experiencing pain at night) | ||||
| Intervention | Time point | No. randomised | No. of patients | |
| Sodium hyaluronate injection | Baseline | 10 | 8 | |
| Steroid injection | 10 | 9 | ||
| Intervention | Time point | No. analysed | No. of patients | |
| Sodium hyaluronate injection | 5 weeksa | 10 | 4 | |
| Steroid injection | 10 | 0 | ||
| Outcome: Pain (no. of patients experiencing daytime pain) | ||||
| Intervention | Time point | No. randomised | No. of patients | |
| Sodium hyaluronate + steroid injection + PT | Baseline | 10 | 8 | |
| Steroid injection + PT | 10 | 8 | ||
| Intervention | Time point | No. analysed | No. of patients | |
| Sodium hyaluronate + steroid injection + PT | 5 weeksa | 10 | 4 | |
| Steroid injection + PT | 10 | 2 | ||
| Study: Takagishi 199670 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using activities of daily living (ADL) questionnairea | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| Sodium hyaluronate injection | Baseline | 10 | 5.35 | 1.83 | |
| Steroid injection | 10 | 4.95 | 2.5 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-value b |
| Sodium hyaluronate injection | 5 weeksc | 10 | NR | NR | p < 0.08 |
| Steroid injection | 10 | NR | NR | p < 0.05 | |
| Study: Takagishi 199670 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – external rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Sodium hyaluronate injection | Baseline | 10 | 18 | 13.6 |
| Steroid injection | 10 | 22.5 | 24.4 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Sodium hyaluronate injection | 5 weeksa | 10 | 24 | 22 |
| Steroid injection | 10 | 26 | 22 | |
| Study: Takagishi 199670 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Takagishi 199670 |
|---|
| Outcome: Other |
| Not reported |
| Study: Takagishi 199670 |
|---|
| Outcome: Adverse events |
| Not reported |
Appendix 7.3 Physical therapy
| Study: Diercks 200473 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Diercks 200473 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using the Constant score | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 32 | 29.97 | 8.46 | |
| Supervised neglect | 45 | 28.6 | 8.64 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| PT | 3 months | 32 | 39.5 | 8.45 | 0.000 |
| Supervised neglect | 45 | 55.87 | 14.26 | ||
| PT | 6 months | 32 | 47.91 | 7.51 | 0.000 |
| Supervised neglect | 45 | 63.31 | 15 | ||
| PT | 9 months | 32 | 54.59 | 7.89 | 0.000 |
| Supervised neglect | 45 | 69.96 | 15.44 | ||
| PT | 12 months | 32 | 58.97 | 8.79 | 0.000 |
| Supervised neglect | 45 | 76.71 | 13.6 | ||
| PT | 15 months | 32 | 65.06 | 11.12 | 0.000 |
| Supervised neglect | 45 | 81.2 | 13.45 | ||
| PT | 18 months | 32 | 70.69 | 12.47 | 0.000 |
| Supervised neglect | 45 | 86.82 | 14.41 | ||
| PT | 21 months | 32 | 76.75 | 14.41 | 0.001 |
| Supervised neglect | 45 | 87.8 | 12.8 | ||
| PT | 24 months | 32 | 79.56 | 16.09 | 0.004 |
| Supervised neglect | 45 | 88.78 | 11.26 | ||
| Study: Diercks 200473 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – Constant score for external rotation | |||||
| Intervention a | Time point | No. randomised | Median | Range | p-value |
| PT | Baseline | 32 | 2 | NR | NR |
| Supervised neglect | 45 | 2 | NR | ||
| Intervention a | Time point | No. analysed | Median | Range | p-value |
| PT | 24 months | 32 | 10 | NR | NR |
| Supervised neglect | 45 | 8 | NR | ||
| Study: Diercks 200473 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Diercks 200473 |
|---|
| Outcome: Other |
| Not reported |
| Study: Diercks 200473 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Dogru 200851 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using SPADI 5-item pain subscale | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 66.9 | 13.8 | |
| Sham ultrasound + PT | 25 (24b) | 57.7 | 18 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 40.1 | 18.6 | 0.37 |
| Sham ultrasound + PT | 24 | 35.6 | 13.7 | ||
| Ultrasound + PT | 3 months | 25 | 31 | 20 | 0.39 |
| Sham ultrasound + PT | 24 | 25.5 | 18.3 | ||
| Outcome: Pain (pain on activity) measured using 0–100 mm VAS | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 80.8 | 18.2 | |
| Sham ultrasound + PT | 25 (24b) | 78 | 18.4 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| Ultrasound + PT | 2 weeks | 25 | 39.6 | 25.3 | 0.56 |
| Sham ultrasound + PT | 24 | 40.7 | 20.3 | ||
| Ultrasound + PT | 3 months | 25 | 24.8 | 29.9 | 0.83 |
| Sham ultrasound + PT | 24 | 23.6 | 25.5 | ||
| Study: Dogru 200851 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using SPADI total score | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 66.6 | 14.6 | |
| Sham ultrasound + PT | 25 (24b) | 62.1 | 17.3 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 37 | 18.6 | 0.72 |
| Sham ultrasound + PT | 24 | 38.2 | 17.8 | ||
| Ultrasound + PT | 3 months | 25 | 29.5 | 21.6 | 0.5 |
| Sham ultrasound + PT | 24 | 26.4 | 19.6 | ||
| Outcome: Function and disability measured by SPADI 8-item disability subscale | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 66.5 | 13.7 | |
| Sham ultrasound + PT | 25 (24b) | 63.1 | 13.8 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 38.6 | 17.4 | 1 |
| Sham ultrasound + PT | 24 | 38.1 | 15.9 | ||
| Ultrasound + PT | 3 months | 25 | 30 | 20.9 | 0.45 |
| Sham ultrasound + PT | 24 | 25.5 | 17.8 | ||
| Study: Dogru 200851 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 101.4 | 20.9 | |
| Sham ultrasound + PT | 25 (24b) | 113.5 | 14.1 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 142.8 | 25.9 | 0.72 |
| Sham ultrasound + PT | 24 | 146 | 26.2 | ||
| Ultrasound + PT | 3 months | 25 | 147.8 | 30.1 | 0.98 |
| Sham ultrasound + PT | 24 | 148 | 26.5 | ||
| Outcome: Range of movement – external rotation (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 34.8 | 14.7 | |
| Sham ultrasound + PT | 25 (24b) | 55.8 | 17.2 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 25 | 58 | 0.004 |
| Sham ultrasound + PT | 24 | 24 | 71.3 | ||
| Ultrasound + PT | 3 months | 25 | 25 | 65.7 | 0.05 |
| Sham ultrasound + PT | 24 | 24 | 75.4 | ||
| Outcome: Range of movement – internal rotation (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 29.2 | 15.7 | |
| Sham ultrasound + PT | 25 (24b) | 47.3 | 18.8 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 2 weeks | 25 | 52.2 | 15.7 | 0.12 |
| Sham ultrasound + PT | 24 | 58.3 | 15.5 | ||
| Ultrasound + PT | 3 months | 25 | 57.4 | 13.8 | 0.21 |
| Sham ultrasound + PT | 24 | 60.9 | 15.3 | ||
| Study: Dogru 200851 | |||||
|---|---|---|---|---|---|
| Outcome: Quality of life – SF-36 PCS | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 38.9 | 7.9 | |
| Sham ultrasound + PT | 25 (24b) | 36.6 | 9.8 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 3 months | 25 | 44.2 | 8.4 | 0.83 |
| Sham ultrasound + PT | 24 | 44.6 | 8.8 | ||
| Outcome: Quality of life – SF-36 MCS | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| Ultrasound + PT | Baseline | 25 | 43.5 | 10.2 | |
| Sham ultrasound + PT | 25 (24b) | 42 | 7.7 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value c |
| Ultrasound + PT | 3 months | 25 | 44.8 | 11.5 | 0.81 |
| Sham ultrasound + PT | 24 | 43.8 | 10.6 | ||
| Study: Dogru 200851 |
|---|
| Outcome: Other outcome |
| Not reported |
| Study: Dogru 200851 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Dundar 200974 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain at rest) measured using VAS 0–10 cm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 5.44 | 1.51 |
| Conventional PT | 28 | 5.54 | 1.64 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 2.86 | 1.96 |
| Conventional PT | 28 | 4.11 | 2.03 | |
| Continuous passive motion | 12 weeks | 29 | 2.41 | 1.72 |
| Conventional PT | 28 | 3.76 | 1.91 | |
| Outcome: Pain (pain on movement) measured using VAS 0–10 cm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 6.34 | 1.99 |
| Conventional PT | 28 | 6.31 | 1.86 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 4.06 | 2.13 |
| Conventional PT | 28 | 4.93 | 1.87 | |
| Continuous passive motion | 12 weeks | 29 | 3.75 | 1.92 |
| Conventional PT | 28 | 4.65 | 1.65 | |
| Outcome: Pain (pain at night) measured using VAS 0–10 cm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 6.1 | 1.75 |
| Conventional PT | 28 | 6 | 1.69 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 2 weeks | 29 | 3.91 | 2.61 |
| Conventional PT | 28 | 4.84 | 1.66 | |
| Continuous passive motion | 12 weeks | 29 | 3.74 | 2.14 |
| Conventional PT | 28 | 4.64 | 1.77 | |
| Outcome: Pain (pain overall) measured using VAS 0–10 cm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 5.96 | 2 |
| Conventional PT | 28 | 5.92 | 1.8 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 2 weeks | 29 | 4.01 | 2.1 |
| Conventional PT | 28 | 4.58 | 1.28 | |
| Continuous passive motion | 12 weeks | 29 | 3.79 | 2.01 |
| Conventional PT | 28 | 4.39 | 1.82 | |
| Study: Dundar 200974 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured by SPADI 8-item disability subscale | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 5.78 | 1.7 |
| PT | 28 | 5.69 | 1.84 | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 4.03 | 1.58 |
| PT | 28 | 4.29 | 1.91 | |
| Continuous passive motion | 12 weeks | 29 | 3.82 | 1.61 |
| PT | 28 | 3.99 | 1.84 | |
| Outcome: Function and disability measured by Constant score | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 58.86 | 9.54 |
| PT | 28 | 57.59 | 9.32 | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 74.86 | 9.64 |
| PT | 28 | 70.54 | 9.38 | |
| Continuous passive motion | 12 weeks | 29 | 79.63 | 9.45 |
| PT | 28 | 76.26 | 9.45 | |
| Study: Dundar 200974 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 106.86 | 24.5 |
| PT | 28 | 103.45 | 23.6 | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 137.96 | 16.26 |
| PT | 28 | 127.67 | 26.66 | |
| Continuous passive motion | 12 weeks | 29 | 141.75 | 13.11 |
| PT | 28 | 137.33 | 15.31 | |
| Outcome: Range of movement – passive external rotation (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 48.8 | 21.3 |
| PT | 28 | 49.7 | 21.2 | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 65.82 | 17.54 |
| PT | 28 | 64.9 | 21.52 | |
| Continuous passive motion | 12 weeks | 29 | 68.22 | 17.11 |
| PT | 28 | 68.98 | 14.22 | |
| Outcome: Range of movement – passive internal rotation (°) | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| Continuous passive motion | Baseline | 29 | 44.19 | 19.06 |
| PT | 28 | 45.02 | 19.1 | |
| Intervention a | Time point | No. analysed | Mean | SD |
| Continuous passive motion | 4 weeks | 29 | 62.89 | 19.96 |
| PT | 28 | 64.45 | 17.8 | |
| Continuous passive motion | 12 weeks | 29 | 66.27 | 17.14 |
| PT | 28 | 67.19 | 18.47 | |
| Study: Dundar 200974 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Dundar 200974 |
|---|
| Outcome: Other outcome |
| Not reported |
| Study: Dundar 200974 |
|---|
| Outcome: Adverse events |
| No side effects were observed during the study |
| Study: Leung 200775 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Leung 200775 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured by SPADI 8-item disability subscale | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| SWD + stretching | Baseline | 10 | 41.5 | 12.1 | |
| HP + stretching | 10 | 38.9 | 11.8 | ||
| No intervention | 10 | 33.3 | 12.5 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| SWD + stretching | 2 weeks | 10 | 56.3 | 15 | 0.046 |
| HP + stretching | 10 | 54.2 | 15.4 | ||
| No intervention | 10 | 45.3 | 11.2 | ||
| SWD + stretching | 4 weeks | 10 | 67.8 | 15.1 | NR |
| HP + stretching | 10 | 56.5 | 14.1 | ||
| No intervention | 10 | 46.1 | 12.7 | ||
| SWD + stretching | 8 weeks | 10 | 71.3 | 19.3 | NR |
| HP + stretching | 10 | 57.8 | 16.3 | ||
| No intervention | 10 | 53.8 | 16.5 | ||
| Study: Leung 200775 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – external rotation (arm by side) (°) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| SWD + stretching | Baseline | 10 | 50.4 | 14.1 | |
| HP + stretching | 10 | 28.2 | 23.4 | ||
| No intervention | 10 | 39.5 | 21.7 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| SWD + stretching | 2 weeks | 10 | 59.3 | 19.8 | 0.09 |
| HP + stretching | 10 | 27.6 | 18.7 | ||
| No intervention | 10 | 39.5 | 20.6 | ||
| SWD + stretching | 4 weeks | 10 | 60.9 | 14.5 | NR |
| HP + stretching | 10 | 32.6 | 21.1 | ||
| No intervention | 10 | 43.3 | 22.6 | ||
| SWD + stretching | 8 weeks | 10 | 62.1 | 11.5 | NR |
| HP + stretching | 10 | 32.6 | 21.7 | ||
| No intervention | 10 | 41.1 | 23.2 | ||
| Outcome: Range of movement – external rotation (arm at 90° abduction) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| SWD + stretching | Baseline | 10 | 51.6 | 18.2 | |
| HP + stretching | 10 | 26.7 | 26 | ||
| No intervention | 10 | 42.5 | 18.7 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| SWD + stretching | 2 weeks | 10 | 57.8 | 22.7 | 0.021 |
| HP + stretching | 10 | 27 | 26.5 | ||
| No intervention | 10 | 43.4 | 20.8 | ||
| SWD + stretching | 4 weeks | 10 | 59.6 | 19.3 | NR |
| HP+ stretching | 10 | 30.1 | 26.8 | ||
| No intervention | 10 | 45.7 | 23.3 | ||
| SWD + stretching | 8 weeks | 10 | 60.6 | 11 | NR |
| HP + stretching | 10 | 30.5 | 24.4 | ||
| No intervention | 10 | 49 | 27.2 | ||
| Outcome: Range of movement – hand behind back (cm) | |||||
| Intervention a | Time point | No. randomised | Mean | SD | |
| SWD + stretching | Baseline | 10 | 12.3 | 4.8 | NR |
| HP + stretching | 10 | 24.9 | 11.5 | ||
| No intervention | 10 | 16 | 9.6 | ||
| Intervention a | Time point | No. analysed | Mean | SD | p-value b |
| SWD + stretching | 2 weeks | 10 | 7.2 | 6.1 | 0.004 |
| HP + stretching | 10 | 22.2 | 11.5 | ||
| No intervention | 10 | 14.7 | 8.1 | ||
| SWD + stretching | 4 weeks | 10 | 7.6 | 5.7 | NR |
| HP + stretching | 10 | 18.5 | 8.9 | ||
| No intervention | 10 | 14.7 | 8 | ||
| SWD + stretching | 8 weeks | 10 | 6 | 7.3 | NR |
| HP + stretching | 10 | 18.3 | 7.5 | ||
| No intervention | 10 | 13 | 6.7 | ||
| Study: Leung 200775 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Leung 200775 |
|---|
| Outcome: Other |
| Not reported |
| Study: Leung 200775 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Maricar 199976 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Maricar 199976 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Maricar 199976 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – external rotation, internal rotation and hand behind back | ||||
| Intervention a | Time point | No. randomised | Mean | SD |
| PT | Baseline | 54 | NR | NR |
| Exercises | NR | NR | ||
| Intervention a | Time point | No. analysed | Mean change from baseline, p-values | |
| PT | 3, 5, 7 and 8 weeks | 16 | Both groups showed significant improvement in all shoulder range of movements (p < 0.001). There was no significant difference between groups at weeks 3, 5, 7 or 8 | |
| Exercises | 16 | |||
| Study: Maricar 199976 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Maricar 199976 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Maricar 199976 |
|---|
| Outcome: Other outcome |
| Not reported |
| Study: Pajareya 200477 | ||||
|---|---|---|---|---|
| Outcome: Pain (no. of analgesics tablets taken) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| PT | Baseline | 61 | Baseline data not reported | |
| No intervention (information only) | 61 | |||
| Intervention | Time point | No. analysed | Mean | SD |
| PT | 3 weeks | 60 | 61.38 | |
| No intervention (information only) | 59 | 58.59 | ||
| Study: Pajareya 200477 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured by SPADI 8-item disability subscale | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| PT | Baseline | 61 (60a) | 54.93 | 21.3 |
| No intervention (information only) | 61 (59a) | 50.6 | 16.6 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SD |
| PT | 3 weeks | 60 | 20.5 | 15.4 |
| No intervention (information only) | 59 | 11.9 | 14.2 | |
| Outcome: Function and disability measured by global rating of pain and disability | ||||
| Intervention | Time point | No. randomised | No shoulder complaint, n (%) | |
| PT | Baseline | 61 (60a) | 0 | |
| No intervention (information only) | 61 (59a) | 0 | ||
| Intervention | Time point | No. randomised | Some pain or limitation but does not interfere with everyday life, n (%) | |
| PT | Baseline | 61 (60a) | 0 | |
| No intervention (information only) | 61 (59a) | 0 | ||
| Intervention | Time point | No. randomised | Minimal inconvenience, n (%) | |
| PT | Baseline | 61 (60a) | 9 (15%) | |
| No intervention (information only) | 61 (59a) | 12 (20.3%) | ||
| Intervention | Time point | No. randomised | Moderate inconvenience, n (%) | |
| PT | Baseline | 61 (60a) | 35 (58.3%) | |
| No intervention (information only) | 61 (59a) | 34 (57.6%) | ||
| Intervention | Time point | No. randomised | Marked inconvenience, n (%) | |
| PT | Baseline | 61 (60a) | 16 (26.7%) | |
| No intervention (information only) | 61 (59a) | 13 (22.3%) | ||
| Study: Pajareya 200477 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – abduction (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| PT | Baseline | 61 (60a) | 121.9 | 27.8 |
| No intervention (information only) | 61 (59a) | 121.3 | 27.8 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SD |
| PT | 3 weeks | 60 | 21.9 | 21 |
| No intervention (information only) | 59 | 14.7 | 18.1 | |
| Outcome: Range of movement – external rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| PT | Baseline | 61 (60a) | 74.8 | 22.1 |
| No intervention (information only) | 61 (59a) | 75.3 | 16 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SD |
| PT | 3 weeks | 60 | 21.3 | 15.3 |
| No intervention (information only) | 59 | 18.3 | 15.4 | |
| Outcome: Range of movement – internal rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| PT | Baseline | 61 (60a) | 41.2 | 10.6 |
| No intervention (information only) | 61 (59a) | 41.1 | 10.3 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SD |
| PT | 3 weeks | 60 | 6.3 | 7.7 |
| No intervention (information only) | 59 | 3 | 7 | |
| Study: Pajareya 200477 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Pajareya 200477 | |
|---|---|
| Outcome: Adverse events Patients were asked, ‘Have the trial drugs and/or treatment programme upset you in any way?’, and were examined for signs of echymosis or burn during range of movement evaluation |
|
| PT | 10 episodes of pain (in 4 patients) that persisted for > 2 hours after treatment |
| No intervention (information only) | 15 patients had gastrointestinal side effects (6 had severe dyspepsia and discontinued NSAIDs; 2 had severe oedema; 1 had severe headache that subsided with discontinuation of NSAIDs) |
| Study: Pajareya 200477 | |||
|---|---|---|---|
| Outcome: Other – treatment success | |||
| Intervention | Time point | No. randomised | n (%) patients who had successful treatment |
| PT | Baseline | 61 (60a) | NA |
| No intervention (information only) | 61 (59a) | NA | |
| Intervention | Time point | No. analysed | n (%) patients who had successful treatment |
| PT | 3 weeks | 60 | 21 (35) |
| No intervention (information only) | 59 | 11 (18.6) | |
| PT | 6 weeks | 60 | 35 (61.4) |
| No intervention (information only) | 59 | 21 (60.8) | |
| PT | 12 weeks | 60 | 43 (76.8) |
| No intervention (information only) | 59 | 31 (60.8) | |
| PT | 24 weeks | 60 | 45 (80.4) |
| No intervention (information only) | 59 | 42 (82.4) | |
| Outcome: Other – satisfactionb | |||
| Intervention | Time point | No. analysed | No. patients ‘very satisfied’ |
| PT | 3 weeks | 60 | 5 |
| No intervention (information only) | 59 | 1 | |
| Intervention | Time point | No. analysed | No. patients ‘moderately satisfied’ |
| PT | 3 weeks | 60 | 7 |
| No intervention (information only) | 59 | 1 | |
| Intervention | Time point | No. analysed | No. patients ‘unsatisfied’ |
| PT | 3 weeks | 60 | 24 |
| No intervention (information only) | 59 | 13 | |
| Intervention | Time point | No. analysed | No. patients ‘very unsatisfied’ |
| PT | 3 weeks | 60 | 23 |
| No intervention (information only) | 59 | 45 | |
| Study: Stergioulas 200816 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Laser therapy | Baseline | 37 | 70.90 | 8.51 |
| Placebo laser | 37 | 67.03 | 8.12 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Laser therapy | 4 weeks | 31 | 32.34 | 7.44 |
| Placebo laser | 32 | 51.15 | 8.22 | |
| Laser therapy | 8 weeks | 31 | 27.41 | 6.72 |
| Placebo laser | 32 | 40.18 | 7.99 | |
| Laser therapy | 16 weeks | 31 | 23.92 | 6.11 |
| Placebo laser | 32 | 36.6 | 7.09 | |
| Outcome: Pain (pain at night) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Laser therapy | Baseline | 37 | 77.91 | 9.23 |
| Placebo laser | 37 | 72.39 | 8.86 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Laser therapy | 4 weeks | 31 | 41.42 | 7.69 |
| Placebo laser | 32 | 55.67 | 8.49 | |
| Laser therapy | 8 weeks | 31 | 24.18 | 6.56 |
| Placebo laser | 32 | 49.33 | 8.05 | |
| Laser therapy | 16 weeks | 31 | 19.38 | 5.77 |
| Placebo laser | 32 | 42.35 | 7.57 | |
| Outcome: Pain (pain on activity) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Laser therapy | Baseline | 37 | 80.55 | 8.82 |
| Placebo laser | 37 | 73.66 | 8.74 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Laser therapy | 4 weeks | 31 | 45.57 | 8.27 |
| Placebo laser | 32 | 67.75 | 8.03 | |
| Laser therapy | 8 weeks | 31 | 30.82 | 6.88 |
| Placebo laser | 32 | 51.39 | 8.58 | |
| Laser therapy | 16 weeks | 31 | 22.54 | 6.02 |
| Placebo laser | 32 | 39.78 | 7.65 | |
| Study: Stergioulas 200816 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured by SPADI total score | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Laser | Baseline | 37 | 65.78 | 13.23 | |
| Placebo laser | 37 | 61.67 | 14.22 | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| Laser | 4 weeks | 31 | 36.57 | 11.31 | < 0.05 |
| Placebo laser | 32 | 48.35 | 13.61 | ||
| Laser | 8 weeks | 31 | 25.73 | 10.72 | < 0.01 |
| Placebo laser | 32 | 39.84 | 11.11 | ||
| Laser | 16 weeks | 31 | 19.92 | 10.04 | < 0.01 |
| Placebo laser | 32 | 33.75 | 10.43 | ||
| Outcome: Function and disability measured by Croft score | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Laser | Baseline | 37 | 13.85 | 4.44 | |
| Placebo laser | 37 | 15.63 | 4.79 | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| Laser | 4 weeks | 31 | 7.69 | 4.03 | < 0.05 |
| Placebo laser | 32 | 14.52 | 4.37 | ||
| Laser | 8 weeks | 31 | 6.93 | 3.87 | < 0.05 |
| Placebo laser | 32 | 11.27 | 4.23 | ||
| Laser | 16 weeks | 31 | 5.52 | 4.00 | < 0.005 |
| Placebo laser | 32 | 12.65 | 4.31 | ||
| Outcome: Function and disability measured by DASH score | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Laser | Baseline | 37 | 48.56 | 14.19 | |
| Placebo laser | 37 | 43.09 | 13.78 | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| Laser | 4 weeks | 31 | 26.67 | 10.44 | NR |
| Placebo laser | 32 | 27.35 | 11.39 | ||
| Laser | 8 weeks | 31 | 20.64 | 9.89 | < 0.05 |
| Placebo laser | 32 | 29.88 | 10.78 | ||
| Laser | 16 weeks | 31 | 15.23 | 7.98 | < 0.005 |
| Placebo laser | 32 | 25.74 | 11.45 | ||
| Outcome: Function and disability measured by HAQ score | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Laser | Baseline | 37 | 2.03 | 0.81 | |
| Placebo laser | 37 | 2.24 | 0.92 | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| Laser | 4 weeks | 31 | 1.43 | 0.72 | < 0.001 |
| Placebo laser | 32 | 2.14 | 0.78 | ||
| Laser | 8 weeks | 31 | 1.27 | 0.56 | < 0.005 |
| Placebo laser | 32 | 2.02 | 0.8 | ||
| Laser | 16 weeks | 31 | 1.23 | 0.54 | NR |
| Placebo laser | 32 | 1.54 | 0.77 | ||
| Study: Stergioulas 200816 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Laser | Baseline | 37 | 65.56 | 12.05 |
| Placebo laser | 37 | 59.37 | 10.89 | |
| Interventiona | Time point | No. analysed | Mean | SD |
| Laser | 4 weeks | 31 | 78.67 | 13.76 |
| Placebo laser | 32 | 69.68 | 12.87 | |
| Laser | 8 weeks | 31 | 81.94 | 13.71 |
| Placebo laser | 32 | 76.47 | 9.65 | |
| Laser | 16 weeks | 31 | 85.63 | 13.95 |
| Placebo laser | 32 | 80.43 | 13.58 | |
| Outcome: Range of movement – external rotation (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Laser | Baseline | 37 | 31.52 | 9.53 |
| Placebo laser | 37 | 28.63 | 8.79 | |
| Interventiona | Time point | No. analysed | Mean | SD |
| Laser | 4 weeks | 31 | 35.33 | 9.91 |
| Placebo laser | 32 | 33.56 | 9.12 | |
| Laser | 8 weeks | 31 | 37.13 | 9.97 |
| Placebo laser | 32 | 35.08 | 9.44 | |
| Laser | 16 weeks | 31 | 42.72 | 10.05 |
| Placebo laser | 32 | 38.53 | 9.9 | |
| Study: Stergioulas 200816 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Stergioulas 200816 |
|---|
| Outcome: Other |
| Not reported |
| Study: Stergioulas 200816 |
|---|
| Outcome: Adverse events |
| Authors stated no complications were reported |
| Study: Vermeulen 200640 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain at rest) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 28 | 15.0 to 65.0 |
| LGMT | 51 | 36 | 20.0 to 64.5 | |
| Intervention | Time point | No. analysed | Mean change from baseline | 95% CI |
| HGMT | 3 months | 49 | –15 | –5.9 to –24.0 |
| LGMT | 51 | –22.1 | –15.6 to –28.7 | |
| HGMT | 6 months | 49 | –22.3 | –32.1 to –12.4 |
| LGMT | 51 | –24.3 | –31.3 to –17.4 | |
| HGMT | 12 months | 49 | –23.9 | –31.7 to –16.0 |
| LGMT | 51 | –23 | –30.8 to –15.2 | |
| Pain (pain during movement) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 59 | 46.0 to 78.0 |
| LGMT | 51 | 62 | 37.5 to 77.0 | |
| Intervention | Time point | No. analysed | Mean change from baseline | 95% CI |
| HGMT | 3 months | 49 | –26.9 | –19.0 to –34.7 |
| LGMT | 51 | –24.3 | –16.1 to –32.5 | |
| HGMT | 6 months | 49 | –31.4 | –39.5 to –23.2 |
| LGMT | 51 | –31.9 | –39.2 to –24.5 | |
| HGMT | 12 months | 49 | –39.2 | –47.2 to –31.2 |
| LGMT | 51 | –32.6 | –41.4 to –23.8 | |
| Pain (pain at night) measured using VAS 0–100 mm | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 72 | 47.0 to 84.0 |
| LGMT | 51 | 63 | 31.0 to 78.5 | |
| Intervention | Time point | No. analysed | Mean change from baseline | 95% CI |
| HGMT | 3 months | 49 | –31.3 | –20.3 to –42.4 |
| LGMT | 51 | –27.5 | –19.0 to –35.9 | |
| HGMT | 6 months | 49 | –38.8 | –50.8 to –26.9 |
| LGMT | 51 | –31.7 | –40.1 to –23.4 | |
| HGMT | 12 months | 49 | –43.7 | –53.6 to –33.8 |
| LGMT | 51 | –35.9 | –44.4 to –27.4 | |
| Study: Vermeulen 200640 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured using the shoulder rating questionnaire | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 37.5 | 28.7 to 47.0 |
| LGMT | 51 | 39.5 | 31.0 to 49.6 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SDa |
| HGMT | 3 months | 49 | 25.8 | 17.4 |
| LGMT | 51 | 23.4 | 15.1 | |
| HGMT | 6 months | 49 | 32.3 | 19.3 |
| LGMT | 51 | 27.8 | 15.6 | |
| HGMT | 12 months | 49 | 38.3 | 19.2 |
| LGMT | 51 | 31.7 | 17.6 | |
| Outcome: Function and disability measured using the shoulder disability questionnaire (Dutch version) | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 81.2 | 75.0 to 87.5 |
| LGMT | 51 | 81.2 | 71.9 to 93.7 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SDa |
| HGMT | 3 months | 49 | –29.9 | 25.2 |
| LGMT | 51 | –24.7 | 24.5 | |
| HGMT | 6 months | 49 | –38.9 | 32.0 |
| LGMT | 51 | –33.2 | 27.5 | |
| HGMT | 12 months | 49 | –50.0 | 30.5 |
| LGMT | 51 | –38.8 | 27.5 | |
| Study: Vermeulen 200640 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | |||||
| Intervention | Time point | No. randomised | Median | IQR | |
| HGMT | Baseline | 49 | 75 | 60.0 to 90.0 | |
| LGMT | 51 | 75 | 67.5 to 85.0 | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SDa | |
| HGMT | 3 months | 49 | 46.3 | 33.0 | |
| LGMT | 51 | 36.3 | 28.8 | ||
| HGMT | 6 months | 49 | 55.8 | 37.1 | |
| LGMT | 51 | 46.9 | 31.8 | ||
| HGMT | 12 months | 49 | 72.9 | 31.5 | |
| LGMT | 51 | 60.3 | 32.5 | ||
| Outcome: Range of movement – active external rotation (°) | |||||
| Intervention | Time point | No. randomised | Median | IQR | |
| HGMT | Baseline | 49 | 20 | 10.0 to 25.0 | |
| LGMT | 51 | 20 | 7.5 to 30.0 | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SDa | |
| HGMT | 3 months | 49 | 11.6 | 11.3 | |
| LGMT | 51 | 9.3 | 14.6 | ||
| HGMT | 6 months | 49 | 15.9 | 12.7 | |
| LGMT | 51 | 13.2 | 13.1 | ||
| HGMT | 12 months | 49 | 20.8 | 12.0 | |
| LGMT | 51 | 15.9 | 16.2 | ||
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention | Time point | No. randomised | Median | IQR | |
| HGMT | Baseline | 49 | 85 | 70.0 to 95.0 | |
| LGMT | 51 | 85 | 80.0 to 95.0 | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SDa | p-valueb |
| HGMT | 3 months | 49 | 47.9 | 31.7 | < 0.05 |
| LGMT | 51 | 34.8 | 26.5 | ||
| HGMT | 6 months | 49 | 57.1 | 34.5 | |
| LGMT | 51 | 46.1 | 29.4 | ||
| HGMT | 12 months | 49 | 72.4 | 29.4 | < 0.05 |
| LGMT | 51 | 59.9 | 29.1 | ||
| Outcome: Range of movement – passive external rotation (°) | |||||
| Intervention | Time point | No. randomised | Median | IQR | |
| HGMT | Baseline | 49 | 20 | 10.0 to 30.0 | |
| LGMT | 51 | 20 | 12.5 to 35.0 | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SDa | p-valueb |
| HGMT | 3 months | 49 | 13.1 | 11.8 | |
| LGMT | 51 | 11.7 | 13.1 | ||
| HGMT | 6 months | 49 | 16.8 | 13.4 | |
| LGMT | 51 | 12.7 | 12.4 | ||
| HGMT | 12 months | 49 | 21.9 | 14.3 | < 0.05 |
| LGMT | 51 | 15.4 | 17.4 | ||
| Study: Vermeulen 200640 | ||||
|---|---|---|---|---|
| Outcome: Quality of life – SF-36 PCS | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 43.8 | 31.9 to 54.2 |
| LGMT | 51 | 45.1 | 36.3 to 57.5 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SDa |
| HGMT | 3 months | 49 | 14.2 | 16.0 |
| LGMT | 51 | 13.6 | 17.8 | |
| HGMT | 6 months | 49 | 19.2 | 18.8 |
| LGMT | 51 | 17.1 | 17.8 | |
| HGMT | 12 months | 49 | 23.2 | 21.9 |
| LGMT | 51 | 22.8 | 19.7 | |
| Outcome: Quality of life – SF-36 MCS | ||||
| Intervention | Time point | No. randomised | Median | IQR |
| HGMT | Baseline | 49 | 73.4 | 51.9 to 87.0 |
| LGMT | 51 | 73.2 | 51.9 to 83.5 | |
| Intervention | Time point | No. analysed | Mean change from baseline | SDa |
| HGMT | 3 months | 49 | 8.6 | 17.1 |
| LGMT | 51 | 4.5 | 23.4 | |
| HGMT | 6 months | 49 | 8.2 | 20.4 |
| LGMT | 51 | 7.9 | 21.7 | |
| HGMT | 12 months | 49 | 7.7 | 20.4 |
| LGMT | 51 | 10.2 | 4.8 | |
| Study: Vermeulen 200640 |
|---|
| Outcome: Other outcome |
| Not reported |
| Study: Vermeulen 200640 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Yang 200778 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Yang 200778 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured using the Flexilevel Scale of Shoulder Function | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| ERM + MRM | Baseline | 14 | NR | NR |
| MWM + MRM | 14 | NR | NR | |
| Intervention | Time point | No. analysed | Mean % of change | SD |
| ERM + MRM | 6 weeks | 14 | 19.9 | 8.1 |
| MWM + MRM | 13 | 17.25 | 12.2 | |
| Study: Yang 200778 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – external rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| ERM + MRM | Baseline | 14 | NR | NR |
| MWM + MRM | 14 | NR | NR | |
| Intervention | Time point | No. analysed | Mean % of change | SD |
| ERM + MRM | 6 weeks | 14 | 36.4 | 24.3 |
| MWM + MRM | 13 | 34.2 | 14.3 | |
| Outcome: Range of movement – internal rotation (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| ERM + MRM | Baseline | 14 | NR | NR |
| MWM + MRM | 14 | NR | NR | |
| Intervention | Time point | No. analysed | Mean % of change | SD |
| ERM + MRM | 6 weeks | 14 | 20.5 | 24.4 |
| MWM + MRM | 13 | 45.6 | 38.5 | |
| Study: Yang 200778 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Yang 200778 |
|---|
| Outcome: Other |
| Not reported |
| Study: Yang 200778 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Yan 200572 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Yan 200572 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Yan 200572 |
|---|
| Outcome: Range of movement |
| Measures of interest not reported |
| Study: Yan 200572 | |||
|---|---|---|---|
| Outcome: Other – rate of improvementa | |||
| Intervention | Time point | No. analysed | No. of patients with bad outcome |
| Dumb-bell gymnastics | 3 months | 26 | 0 |
| Barehanded exercises | 28 | 7 | |
| Intervention | Time point | No. analysed | No. of patients with average outcome |
| Dumb-bell gymnastics | 3 months | 26 | 0 |
| Barehanded exercises | 28 | 16 | |
| Intervention | Time point | No. analysed | No. of patients with good outcome |
| Dumb-bell gymnastics | 3 months | 26 | 2 |
| Barehanded exercises | 28 | 5 | |
| Intervention | Time point | No. analysed | No. of patients with excellent outcome |
| Dumb-bell gymnastics | 3 months | 26 | 24 |
| Barehanded exercises | 28 | 0 | |
| Study: Yan 200572 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Yan 200572 |
|---|
| Outcome: Other |
| Not reported |
| Study: Yan 200572 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Wies 200371 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Wies 200371 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured by SPADI total score | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 10 | NR | NR | |
| Osteopathy technique | 10 | NR | NR | ||
| Control | 10 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SD | p-valuea |
| PT | 9 weeks | 10 | 18.8 | 23.6 | 0.059 |
| Osteopathy technique | 10 | 38.7 | 22.5 | ||
| Control | 10 | 22.8 | 18.2 | ||
| Study: Wies 200371 | |||||
|---|---|---|---|---|---|
| Outcome – Range of movement – active abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 10 | NR | NR | |
| Osteopathy technique | 10 | NR | NR | ||
| Control | 10 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean change from baseline | SD | p-valuea |
| PT | 9 weeks | 10 | 39.6 | 35.8 | NR |
| Osteopathy technique | 10 | 46 | 23 | ||
| Control | 10 | 0.8 | 39.5 | ||
| Study: Wies 200371 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Wies 200371 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Wies 200371 |
|---|
| Outcome: Other outcome |
| Not reported |
Appendix 7.4 Acupuncture (with or without physical therapy)
| Study: Cheing 200881 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain at moment) measured using VAS 0–10 cm | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Electroacupuncture | Baseline | 25 | 6.5 | 2.1 |
| Interferential electrotherapy | 24 | 6.5 | 2 | |
| Interventiona | Time point | No. analysed | Mean | SD |
| Electroacupuncture | 4 weeks | 24 | 3.5 | 1.9 |
| Interferential electrotherapy | 23 | 3.4 | 1.9 | |
| Electroacupuncture | Approx. 2 months | 24 | 3.1 | 2.2 |
| Interferential electrotherapy | 23 | 2.4 | 1.7 | |
| Electroacupuncture | Approx. 4 months | 24 | 2.4 | 2.2 |
| Interferential electrotherapy | 23 | 2 | 1.5 | |
| Electroacupuncture | Approx. 7 months | 24 | 1.7 | 2.3 |
| Interferential electrotherapy | 23 | 1.3 | 1.4 | |
| Study: Cheing 200881 | ||||
|---|---|---|---|---|
| Outcome: Function and disability – Constant Murley Assessment Score | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Electroacupuncture | Baseline | 25 | 65.5 | 16.7 |
| Interferential electrotherapy | 24 | 59.6 | 15.4 | |
| Interventiona | Time point | No. analysed | Mean | SD |
| Electroacupuncture | 4 weeks | 24 | 86 | 8.2 |
| Interferential electrotherapy | 23 | 84.9 | 8.4 | |
| Electroacupuncture | Approx. 2 months | 24 | 89.3 | 4.8 |
| Interferential electrotherapy | 23 | 92.1 | 5.9 | |
| Electroacupuncture | Approx. 4 months | 24 | 93.3 | 6.0 |
| Interferential electrotherapy | 23 | 90.2 | 9.7 | |
| Electroacupuncture | Approx. 7 months | 24 | 93.8 | 6.4 |
| Interferential electrotherapy | 23 | 95.5 | 4.1 | |
| Study: Cheing 200881 |
|---|
| Outcome: Range of movement |
| Not reported |
| Study: Cheing 200881 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Cheing 200881 |
|---|
| Outcome: Other |
| Not reported |
| Study: Cheing 200881 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Fang 200682 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using VAS (range and units of measurement unspecified) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Electroacupunctureb | Baseline | 77b | 7.03 | 1.33 |
| Electroacupuncturec | 97c | 5.51 | 2.54 | |
| TENSb | 88b | 7.26 | 1.15 | |
| TENSc | 98c | 6.01 | 2.59 | |
| Interventiona | Time point | No. analysed | Mean | SD |
| Electroacupunctureb | Post treatment | 77b | 1.65 | 2.12 |
| Electroacupuncturec | 97c | 2.31 | 2.11 | |
| TENSb | 88b | 2.28 | 2.4 | |
| TENSc | 98c | 2.65 | 2.06 | |
| Study: Fang 200682 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Fang 200682 |
|---|
| Outcome: Range of movement |
| Not reported |
| Study: Fang 200682 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Fang 200682 | ||||
|---|---|---|---|---|
| Outcome: Other – efficacy rate | ||||
| Interventiona | Time point | No. randomised | Meanb | SD |
| Interventiona | Time point | No. analysed | Cured (mean %) | SD |
| Electroacupunctureb | Post treatment | 77 | 32 | 41.6 |
| Electroacupuncturec | 97 | 11 | 11.3 | |
| TENSb | 88 | 29 | 33 | |
| TENSc | 98 | 9 | 9.2 | |
| Interventiona | Time point | No. analysed | Large improvement (mean %) | SD |
| Electroacupunctureb | Post treatment | 77 | 27 | 35 |
| Electroacupuncturec | 97 | 49 | 50.5 | |
| TENSb | 88 | 33 | 37.5 | |
| TENSc | 98 | 40 | 40.8 | |
| Interventiona | Time point | No. analysed | Improvement (mean %) | SD |
| Electroacupunctureb | Post treatment | 77 | 13 | 16.9 |
| Electroacupuncturec | 97 | 35 | 36.1 | |
| TENSb | 88 | 23 | 26.1 | |
| TENSc | 98 | 46 | 46.9 | |
| Interventiona | Time point | No. analysed | No improvement (mean %) | SD |
| Electroacupunctureb | Post treatment | 77 | 5 | 6.5 |
| Electroacupuncturec | 97 | 2 | 2.1 | |
| TENSb | 88 | 3 | 3.4 | |
| TENSc | 98 | 3 | 3.1 | |
| Interventiona | Time point | No. analysed | Total efficacy rate (%) | SD |
| Electroacupunctureb | Post treatment | 77 | 93.5 | NR |
| Electroacupuncturec | 97 | 97.9 | NR | |
| TENSb | 88 | 96.6 | NR | |
| TENSc | 98 | 96.9 | NR | |
| Study: Fang 200682 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Ma 200683 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain at rest) measured using 0–10 numeric scale | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 1.8 | NR | |
| Acupuncture | 30 | 2.3 | NR | ||
| Acupuncture + PT | 15 | 2.6 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 0.9 | NR | 0.0448 |
| Acupuncture | 30 | 0.8 | NR | ||
| Acupuncture + PT | 15 | 0.9 | NR | ||
| PT | 4 weeks | 30 | 0.4 | NR | 0.0238 |
| Acupuncture | 30 | 0.7 | NR | ||
| Acupuncture + PT | 15 | 0.7 | NR | ||
| Outcome: Pain (pain on activity) measured using 0–10 numeric scale | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 6.4 | NR | |
| Acupuncture | 30 | 7.3 | NR | ||
| Acupuncture + PT | 15 | 7.4 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 5.4 | NR | 0.0398 |
| Acupuncture | 30 | 5.7 | NR | ||
| Acupuncture + PT | 15 | 4.1 | NR | ||
| PT | 4 weeks | 30 | 3.7 | NR | 0.0137 |
| Acupuncture | 30 | 4.3 | NR | ||
| Acupuncture + PT | 15 | 3.3 | NR | ||
| Study: Ma 200683 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 70.6 | NR | |
| Acupuncture | 30 | 88.8 | NR | ||
| Acupuncture + PT | 15 | 79.5 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 84.9 | NR | 0.2645 |
| Acupuncture | 30 | 90.7 | NR | ||
| Acupuncture + PT | 15 | 90.3 | NR | ||
| PT | 4 weeks | 30 | 98.3 | NR | 0.1634 |
| Acupuncture | 30 | 115.7 | NR | ||
| Acupuncture + PT | 15 | 110.1 | NR | ||
| Outcome: Range of movement – active external rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 15.9 | NR | |
| Acupuncture | 30 | 32.1 | NR | ||
| Acupuncture + PT | 15 | 20.7 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 27.3 | NR | 0.5453 |
| Acupuncture | 30 | 42.7 | NR | ||
| Acupuncture + PT | 15 | 30.1 | NR | ||
| PT | 4 weeks | 30 | 28.3 | NR | 0.0367 |
| Acupuncture | 30 | 43.2 | NR | ||
| Acupuncture + PT | 15 | 36.1 | NR | ||
| Outcome: Range of movement – active internal rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 30 | NR | |
| Acupuncture | 30 | 41.3 | NR | ||
| Acupuncture + PT | 15 | 33.6 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 37.5 | NR | 0.0656 |
| Acupuncture | 30 | 48.5 | NR | ||
| Acupuncture + PT | 15 | 47.6 | NR | ||
| PT | 4 weeks | 30 | 43.9 | NR | 0.0656 |
| Acupuncture | 30 | 51.7 | NR | ||
| Acupuncture + PT | 5 | 54.4 | NR | ||
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 78 | NR | |
| Acupuncture | 30 | 94.9 | NR | ||
| Acupuncture + PT | 15 | 84.8 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 91.8 | NR | 0.0735 |
| Acupuncture | 30 | 99.2 | NR | ||
| Acupuncture + PT | 15 | 100.5 | NR | ||
| PT | 4 weeks | 30 | 103.3 | NR | 0.0675 |
| Acupuncture | 30 | 120 | NR | ||
| Acupuncture + PT | 15 | 110.7 | NR | ||
| Outcome: Range of movement – passive external rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 23.4 | NR | |
| Acupuncture | 30 | 43.2 | NR | ||
| Acupuncture + PT | 15 | 29.6 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 33.9 | NR | 0.6952 |
| Acupuncture | 30 | 55.8 | NR | ||
| Acupuncture + PT | 15 | 34.2 | NR | ||
| PT | 4 weeks | 30 | 33.8 | NR | 0.8952 |
| Acupuncture | 30 | 52.3 | NR | ||
| Acupuncture + PT | 15 | 38.5 | NR | ||
| Outcome: Range of movement – passive internal rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 37.6 | NR | |
| Acupuncture | 30 | 52.8 | NR | ||
| Acupuncture + PT | 15 | 45.7 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 44.1 | NR | 0.7345 |
| Acupuncture | 30 | 59.3 | NR | ||
| Acupuncture + PT | 15 | 50.1 | NR | ||
| PT | 4 weeks | 30 | 51.3 | NR | 0.4345 |
| Acupuncture | 30 | 56.3 | NR | ||
| Acupuncture + PT | 15 | 59.2 | NR | ||
| Study: Ma 200683 | |||||
|---|---|---|---|---|---|
| Outcome: Quality of life – SF-36 physical function | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 74.8 | NR | |
| Acupuncture | 30 | 71.3 | NR | ||
| Acupuncture + PT | 15 | 75.3 | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valuea |
| PT | 2 weeks | 30 | 79 | NR | 0.3743 |
| Acupuncture | 30 | 78.8 | NR | ||
| Acupuncture + PT | 15 | 86.9 | NR | ||
| PT | 4 weeks | 30 | 86.1 | NR | 0.8746 |
| Acupuncture | 30 | 81.3 | NR | ||
| Acupuncture + PT | 15 | 86.9 | NR | ||
| Outcome: Quality of life – SF-36 role physical | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 20.2 | NR | |
| Acupuncture | 30 | 43.8 | NR | ||
| Acupuncture + PT | 15 | 32.4 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 41.7 | NR | 0.7445 |
| Acupuncture | 30 | 56.7 | NR | ||
| Acupuncture + PT | 15 | 45.7 | NR | ||
| PT | 4 weeks | 30 | 49.4 | NR | 0.0745 |
| Acupuncture | 30 | 45 | NR | ||
| Acupuncture + PT | 15 | 45.1 | NR | ||
| Outcome: Quality of life – SF-36 bodily pain | |||||
| Intervention | Time point | No randomised | Mean | SD | |
| PT | Baseline | 30 | 46.3 | NR | |
| Acupuncture | 30 | 50.9 | NR | ||
| Acupuncture + PT | 15 | 42.6 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 58.9 | NR | 0.4033 |
| Acupuncture | 30 | 54.7 | NR | ||
| Acupuncture + PT | 15 | 50.4 | NR | ||
| PT | 4 weeks | 30 | 58.7 | NR | 0.6783 |
| Acupuncture | 30 | 66.3 | NR | ||
| Acupuncture + PT | 15 | 53.4 | NR | ||
| Outcome: Quality of life – SF-36 general health | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 58.9 | NR | |
| Acupuncture | 30 | 50.5 | NR | ||
| Acupuncture + PT | 15 | 54.3 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 63.3 | NR | 0.6004 |
| Acupuncture | 30 | 58.3 | NR | ||
| Acupuncture + PT | 15 | 60.1 | NR | ||
| PT | 4 weeks | 30 | 73.3 | NR | 0.0604 |
| Acupuncture | 30 | 61.3 | NR | ||
| Acupuncture + PT | 15 | 57.3 | NR | ||
| Outcome: Quality of life – SF-36 vitality | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 58.1 | NR | |
| Acupuncture | 30 | 53.4 | NR | ||
| Acupuncture + PT | 15 | 52.4 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 69.3 | NR | 0.7894 |
| Acupuncture | 30 | 62.5 | NR | ||
| Acupuncture + PT | 15 | 65.3 | NR | ||
| PT | 4 weeks | 30 | 73.3 | NR | 0.8894 |
| Acupuncture | 30 | 65 | NR | ||
| Acupuncture + PT | 15 | 67.1 | NR | ||
| Outcome: Quality of life – SF-36 social functioning | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 83.9 | NR | |
| Acupuncture | 30 | 60.2 | NR | ||
| Acupuncture + PT | 15 | 77.3 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 93.3 | NR | 0.0567 |
| Acupuncture | 30 | 68.8 | NR | ||
| Acupuncture + PT | 15 | 89.5 | NR | ||
| PT | 4 weeks | 30 | 93.1 | NR | 0.0677 |
| Acupuncture | 30 | 75 | NR | ||
| Acupuncture + PT | 15 | 83.5 | NR | ||
| Outcome: Quality of life – SF-36 role emotional | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 57 | NR | |
| Acupuncture | 30 | 75 | NR | ||
| Acupuncture + PT | 15 | 70.4 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 61.1 | NR | 0.3458 |
| Acupuncture | 30 | 77.8 | NR | ||
| Acupuncture + PT | 15 | 75.8 | NR | ||
| PT | 4 weeks | 30 | 66.3 | NR | 0.0578 |
| Acupuncture | 30 | 78.6 | NR | ||
| Acupuncture + PT | 15 | 79.3 | NR | ||
| Outcome: Quality of life – SF-36 mental health | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| PT | Baseline | 30 | 62.6 | NR | |
| Acupuncture | 30 | 69 | NR | ||
| Acupuncture + PT | 15 | 65 | NR | ||
| Intervention | Time point | No. analysed | Final value mean | SD | p-valuea |
| PT | 2 weeks | 30 | 65.9 | NR | 0.061 |
| Acupuncture | 30 | 72.7 | NR | ||
| Acupuncture + PT | 15 | 68.6 | NR | ||
| PT | 4 weeks | 30 | 66.9 | NR | 0.091 |
| Acupuncture | 30 | 73 | NR | ||
| Acupuncture + PT | 15 | 69.1 | NR | ||
| Study: Ma 200683 |
|---|
| Outcome: Other |
| Not reported |
| Study: Ma 200683 |
|---|
| Outcome: Adverse events |
| Not reported |
Appendix 7.5 Manipulation under anaesthesia
| Study: Amir-us-Saqlain 200784 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Amir-us-Saqlain 200784 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Amir-us-Saqlain 200784 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 33.75 | 11.03 | |
| MUA + steroid injection + PT | 20 (17)a | 29.71 | 13.05 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 151.81 | 13.19 | 0.00 |
| MUA + steroid injection + PT | 17 | 122.82 | 21.08 | ||
| Outcome: Range of movement – active external rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 28.56 | 24.45 | |
| MUA + steroid injection + PT | 20 (17)a | 33.53 | 22.96 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 83.38 | 6.61 | 0.213 |
| MUA + steroid injection + PT | 17 | 76.76 | 19.76 | ||
| Outcome: Range of movement – active internal rotationc | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 1.00 | 0.00 | |
| MUA + steroid injection + PT | 20 (17)a | 1.12 | 0.33 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 3.56 | 0.51 | 0.00 |
| MUA + steroid injection + PT | 17 | 2.65 | 0.7 | ||
| Outcome: Range of movement – passive abduction (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 45.88 | 14.91 | |
| MUA + steroid injection + PT | 20 (17)a | 40.18 | 17.01 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 160.25 | 12.1 | 0.00 |
| MUA + steroid injection + PT | 17 | 137.76 | 15.21 | ||
| Outcome: Range of movement – passive external rotation (°) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 37 | 23.22 | |
| MUA + steroid injection + PT | 20 (17)a | 41.47 | 23.77 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 91.25 | 6.45 | 0.200 |
| MUA + steroid injection + PT | 17 | 86.41 | 13.38 | ||
| Outcome: Range of movement – passive internal rotationc | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | Baseline | 23 (16)a | 1 | 0.00 | |
| MUA + steroid injection + PT | 20 (17)a | 1.18 | 0.39 | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection + manipulated extremity kept in abduction and external rotation + PT | 12 weeks | 16 | 3.88 | 0.34 | 0.00 |
| MUA + steroid injection + PT | 17 | 2.88 | 0.49 | ||
| Study: Amir-us-Saqlain 200784 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Amir-us-Saqlain 200784 |
|---|
| Outcome: Other |
| Not reported |
| Study: Amir-us-Saqlain 200784 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Jacobs 200985 | |||||
|---|---|---|---|---|---|
| Outcome: Pain measured using 100-point VAS | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA | Baseline | 28 | Baseline data not reported | ||
| Steroid + distension | 25 | ||||
| Intervention | Time point | No. analysed | Mean regression coefficient | SE | p-value, 95% CI |
| MUA | 16 weeks | 19 | –2.77 | 0.33 | Not significant,b –1.11 to 1.15 |
| Steroid + distension | 24 | –2.75 | 0.42 | ||
| Study: Jacobs 200985 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured by the Constant scorea | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA | Baseline | 28 | Baseline data not reported | ||
| Steroid + distension | 25 | ||||
| Intervention | Time point | No. analysed | Mean regression coefficient | SE | p-value,b 95% CI |
| MUA | 16 weeks | 19 | 3.13 | 0.24 | Not significant, –0.90 to 1.11 |
| Steroid + distension | 24 | 3.23 | 0.42 | ||
| Study: Jacobs 200985 |
|---|
| Outcome: Range of movement |
| Not reported |
| Study: Jacobs 200985 | |||||
|---|---|---|---|---|---|
| Outcome: Quality of life, SF-36 components (general health, bodily pain, physical role, emotional role, social functioning, bodily pain, vitality, mental health)a | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| MUA | Baseline | 28 | Baseline data not reported | ||
| Steroid + distension | 25 | ||||
| Intervention | Time point | No. analysed | Results reported | p-valueb | |
| MUA | 24 months | 19 | All components of the SF-36 improved for all patients during the course of treatment. The physical role and bodily pain components showed the greatest improvement | Not significant | |
| Steroid + distension | 24 | ||||
| Study: Jacobs 200985 |
|---|
| Outcome: Other |
| Not reported |
| Study: Jacobs 200985 |
|---|
| Outcome: Adverse events |
| Not reported |
| Study: Kivimaki 200739 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain intensity) measured using Likert 0–10 scale | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT | Baseline | 65 | 6.6 | 0.3 |
| Home exercise | 60 | 6.4 | 0.3 | |
| Interventiona | Time point | No. analysed | Mean | SDb |
| MUA + PT | 6 weeks | 55 | 4.9 | NR |
| Home exercise | 55 | 4.7 | NR | |
| MUA + PT | 3 months | 51 | 4.9 | 2.735 |
| Home exercise | 50 | 4.7 | 2.735 | |
| MUA + PT | 6 months | 38 | 2.0 | NR |
| Home exercise | 45 | 2.8 | NR | |
| MUA + PT | 12 months | 37 | 1.5 | NR |
| Home exercise | 42 | 2.2 | NR | |
| Outcome: Pain (pain intensity) measured using Likert 0–10 scale | ||||
| Interventiona | Time point | No. analysed | Mean | SDb |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 0.2 | –0.64 to 1.02 |
| MUA + PT vs home exercise | 3 months | 101 | 0.2 | –1.06 to 1.10 |
| MUA + physical therapy vs home exercise | 6 months | 83 | –0.8 | –1.80 to 0.20 |
| MUA + PT vs home exercise | 12 months | 79 | –0.7 | –1.80 to 0.40 |
| Study: Kivimaki 200739 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured with a modified version of the SDQ | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 4 | –3.8 to 11.8 |
| MUA + PT vs home exercise | 3 months | 101 | 0.3 | –2.69 to 2.75 |
| MUA + PT vs home exercise | 6 months | 83 | –1.7 | –5.3 to 1.9 |
| MUA + PT vs home exercise | 12 months | 79 | 0 | –3.2 to 3.2 |
| Study: Kivimaki 200739 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – passive abduction (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 10 | –3.2 to 23.2 |
| MUA + PT vs home exercise | 3 months | 101 | 9 | –6 to 24 |
| MUA + PT vs home exercise | 6 months | 83 | 9 | –4 to 22 |
| MUA + PT vs home exercise | 12 months | 79 | 7 | –5 to 19 |
| Outcome: Range of movement – passive internal rotation (cm) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 4 | –1 to 9 |
| MUA + PT vs home exercise | 3 months | 101 | –3 | –7.4 to 2.4 |
| MUA + PT vs home exercise | 6 months | 83 | –2 | –7.4 to 3.4 |
| MUA + PT vs home exercise | 12 months | 79 | –1 | –4.1 to 6.1 |
| Outcome: Range of movement – external rotation (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 5 | –2 to 12 |
| MUA + PT vs home exercise | 3 months | 101 | 6 | –3to 15 |
| MUA + PT vs home exercise | 6 months | 83 | 6 | –2 to 14 |
| MUA + PT vs home exercise | 12 months | 79 | 4 | –4.2 to 12.2 |
| Study: Kivimaki 200739 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Kivimaki 200739 | ||||
|---|---|---|---|---|
| Outcome: Other – working ability | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| MUA + PT vs home exercise | Baseline | 125 | NA | NA |
| Interventiona | Time point | No. analysed | Mean difference | 95% CI |
| MUA + PT vs home exercise | 6 weeks | 110 | 0.4 | –4.2 to 1.8 |
| MUA + PT vs home exercise | 3 months | 101 | 0 | –0.8 to 0.8 |
| MUA + PT vs home exercise | 6 months | 83 | 0.5 | –0.6 to 1.6 |
| MUA + PT vs home exercise | 12 months | 79 | 0.1 | –0.8 to1.0 |
| Study: Kivimaki 200739 |
|---|
| Outcome: Adverse events |
| There were no major complications during manipulation |
| Study: Quraishi 200738 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain overall), unspecified VAS | |||||
| Interventiona | Time point | No. randomised | Mean | Range | SD |
| MUA + steroid injection | Baseline | 17 (18 shoulders) | 5.7 | 3.0–8.5 | NR |
| Arthrographic distension | 19 (20 shoulders) | 6.1 | 4.0–10 | NR | |
| Interventiona | Time point | No. analysed | Mean | Range | SD |
| MUA + steroid injection | 2 months | 15 (16 shoulders) | 4.7 | 0–8.5 | NR |
| Arthrographic distension | 18 (18 shoulders) | 2.4 | 0–8.0 | NR | |
| MUA + steroid injection | 6 months | 15 (16 shoulders) | 2.7 | 0–9.0 | 0.64 |
| Arthrographic distension | 18 (18 shoulders) | 1.7 | 0–7.0 | 0.64 | |
| Study: Quraishi 200738 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using the Constant score | |||||
| Interventiona | Time point | No. randomised | Mean | Range | |
| MUA + steroid injection | Baseline | 17 (18 shoulders) | 36 | 26–66 | |
| Arthrographic distension | 19 (20 shoulders) | 28.8 | 18–55 | ||
| Interventiona | Time point | No. analysed | Mean | SDb | p-valuec |
| MUA + steroid injection | 2 months | 15 (16 shoulders) | 58.5 | NA | |
| Arthrographic distension | 18 (18 shoulders) | 57.4 | NA | ||
| MUA + steroid injection | 6 months | 15 (16 shoulders) | 59.5 | 0.24 | 0.02 |
| Arthrographic distension | 18 (18 shoulders) | 65.9 | 0.24 | ||
| Study: Quraishi 200738 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – external rotation (°) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| MUA + steroid injection | Baseline | 17 (18 shoulders) | NR | NR | |
| Arthrographic distension | 19 (20 shoulders) | NR | NR | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection | 6 months | 15 (16 shoulders) | NR | NR | 0.13 |
| Arthrographic distension | 18 (18 shoulders) | NR | NR | ||
| Outcome: Range of movement – internal rotation (°) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | p-value |
| MUA + steroid injection | Baseline | 17 (18 shoulders) | NR | NR | NR |
| Arthrographic distension | 19 (20 shoulders) | NR | NR | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection | 6 months | 15 (16 shoulders) | NR | NR | 0.48 |
| Arthrographic distension | 18 (18 shoulders) | NR | NR | ||
| Outcome: Range of movement – abduction (°) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | p-value |
| MUA + steroid injection | Baseline | 17 (18 shoulders) | NR | NR | NR |
| Arthrographic distension | 19 (20 shoulders) | NR | NR | ||
| Interventiona | Time point | No. analysed | Mean | SD | p-valueb |
| MUA + steroid injection | 6 months | 15 (16 shoulders) | NR | NR | 0.62 |
| Arthrographic distension | 18 (18 shoulders) | NR | NR | ||
| Study: Quraishi 200738 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Quraishi 200738 |
|---|
| Outcome: Other – satisfaction |
| 94% of patients were satisfied or very satisfied after arthrographic distension compared with 81% after MUA |
| Study: Quraishi 200738 |
|---|
| Outcome: Adverse events |
| Not reported |
Appendix 7.6 Distension
| Study: Buchbinder 200443 | ||||
|---|---|---|---|---|
| Outcome: Pain (pain overall) measured using Likert 0–10 scale | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 6.0 | 2.0 |
| Placebo (arthrogram only) | 21 | 5.7 | 2.1 | |
| Interventiona | Time point | No. analysed | Mean change from baselineb | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 2.5 | 2.5 |
| Placebo (arthrogram only) | 21 | 0.2 | 1.7 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 2.4 | 2.7 |
| Placebo (arthrogram only) | 21 | 1.0 | 2.4 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 3.1 | 2.4 |
| Placebo (arthrogram only) | 21 | 2.4 | 2.8 | |
| Outcome: Pain (pain overall) measured using Likert 0–10 scale – calculated valuesc | ||||
| Interventiona | Time point | No. analysed | Mean | SDd |
| Arthrographic distension + steroid | 6 weeks | 25 | 3.6 | 2.69 |
| Placebo (arthrogram only) | 6 weeks | 21 | 4.7 | 2.73 |
| Arthrographic distension + steroid | 12 weeks | 25 | 2.9 | 2.69 |
| Placebo (arthrogram only) | 12 weeks | 21 | 3.3 | 2.73 |
| Study: Buchbinder 200443 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured using SPADI total score | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 53.8 | 18.5 |
| Placebo (arthrogram only) | 21 | 55.7 | 22.6 | |
| Interventiona | Time point | No. analysed | Mean change from baseline | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 21.8 | 19.3 |
| Placebo (arthrogram only) | 21 | 4.7 | 19.8 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 21.6 | 23.0 |
| Placebo (arthrogram only) | 21 | 16.6 | 21.4 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 28.7 | 23.9 |
| Placebo (arthrogram only) | 21 | 24.3 | 25.8 | |
| Outcome: Function and disability measured using the problem elicitation technique score | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 172.6 | 48.2 |
| Placebo (arthrogram only) | 21 | 169.9 | 76.7 | |
| Interventiona | Time point | No. analysed | Mean change from baseline | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 62.5 | 75.5 |
| Placebo (arthrogram only) | 21 | 8.7 | 29.0 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 71.3 | 81 |
| Placebo (arthrogram only) | 21 | 25.3 | 34.4 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 107.7 | 74.2 |
| Placebo (arthrogram only) | 21 | 53.3 | 70.3 | |
| Study: Buchbinder 200443 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 79.8 | 27.5 |
| Placebo (arthrogram only) | 21 | 72.9 | 21.2 | |
| Interventiona | Time point | No. analysed | Mean change from baseline | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 20.2 | 30.2 |
| Placebo (arthrogram only) | 21 | –0.2 | 22.5 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 24.4 | 34.2 |
| Placebo (arthrogram only) | 21 | 15.5 | 26.1 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 32.8 | 38.1 |
| Placebo (arthrogram only) | 21 | 28.3 | 29.9 | |
| Outcome: Range of movement – external rotation (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 41.7 | 20.2 |
| Placebo (arthrogram only) | 21 | 35.8 | 27.0 | |
| Interventiona | Time point | No. analysed | Mean change from baseline | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 9.0 | 18.5 |
| Placebo (arthrogram only) | 21 | 3.2 | 19.9 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 8.8 | 28.0 |
| Placebo (arthrogram only) | 21 | 15.3 | 33.7 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 19.9 | 27.9 |
| Placebo (arthrogram only) | 21 | 17.8 | 29.2 | |
| Outcome: Range of movement – hand behind back (°) | ||||
| Interventiona | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 25 | 16.0 | 3.9 |
| Placebo (arthrogram only) | 21 | 16.7 | 4.2 | |
| Interventiona | Time point | No. analysed | Mean change from baseline | SD |
| Arthrographic distension + steroid | 3 weeks | 25 | 3.2 | 3.7 |
| Placebo (arthrogram only) | 21 | –0.2 | 3.8 | |
| Arthrographic distension + steroid | 6 weeks | 25 | 3.4 | 4.3 |
| Placebo (arthrogram only) | 21 | 1.5 | 4.8 | |
| Arthrographic distension + steroid | 12 weeks | 25 | 4.2 | 4.4 |
| Placebo (arthrogram only) | 21 | 3.4 | 5.0 | |
| Study: Buchbinder 200443 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Buchbinder 200443 |
|---|
| Outcome: Other |
| Not reported |
| Study: Buchbinder 200443 |
|---|
| Outcome: Adverse events |
| Total reported adverse events: arthrographic distension (n = 25): 9 (36%), placebo (n = 21): 1 (4%) |
| Pain associated with procedure: arthrographic distension (n = 25): 4 (16%), placebo (n = 21): 1 (4%) |
| Increased pain for up to 48 hours after procedure: arthrographic distension (n = 25): 3 (12%), placebo (n = 21): 1 (4%) |
| Claustrophobia at time of procedure: arthrographic distension (n = 25): 1 (4%), placebo (n = 21): 1 (4%) |
| Unsettled, anxious and hot: arthrographic distension (n = 25): 0 (0%), placebo (n = 21): 1 (4%) |
| Shoulder noisy (i.e. fluid noises): arthrographic distension (n = 25): 1 (4%), placebo (n = 21): 0 (0%) |
| Study: Gam 199886 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (pain at rest, pain on activity) measured using VAS 0–10 cm | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| Distension + steroid | Baseline | 12 | NR | NR | |
| Steroid injection | 8 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-value |
| Distension + steroid | Unclear | 11 | NR | NR | No difference between groups (p = 0.1) |
| Steroid injection | 7 | ||||
| Outcome: Pain (analgesic consumption) | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| Distension + steroid | Baseline | 12 | NR | NR | |
| Steroid injection | 8 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-value |
| Distension + steroid | Week 11 | 11 | NR | NR | Analgesic consumption significantly lower in the distension group than in the steroid group (p = 0.008) |
| Steroid injection | 7 | ||||
| Study: Gam 199886 |
|---|
| Outcome: Function and disability |
| Not reported |
| Study: Gam 199886 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – passive abductiona | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| Distension + steroid | Baseline | 12 | NR | NR | |
| Steroid injection | 8 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| Distension + steroid | Unclear | 11 | There was no significant difference between groups (reported in the text) | NR | |
| Steroid injection | 7 | ||||
| Outcome: Range of movement – passive external rotationa | |||||
| Intervention | Time point | No. randomised | Mean | SD | |
| Distension + steroid | Baseline | 12 | NR | NR | |
| Steroid injection | 8 | NR | NR | ||
| Intervention | Time point | No. analysed | Mean | SD | p-valueb |
| Distension + steroid | Unclear | 11 | There was a significant improvement with distension compared with steroid (reported in the text) | 0.0007 | |
| Steroid injection | 7 | ||||
| Study: Gam 199886 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Gam 199886 |
|---|
| Outcome: Other |
| Not reported |
| Study: Gam 199886 |
|---|
| Outcome: Adverse events |
| Two cases of unacceptable pain (dropouts) |
| Study: Tveita 200836 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Tveita 200836 | ||||
|---|---|---|---|---|
| Outcome: Function and disability measured using SPADI total scorea | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 63 | 20 |
| Steroid injection | 39 | 59 | 20 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 26 | 19 |
| Steroid injection | 39 | 20 | 17 | |
| Study: Tveita 200836 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – active abduction (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 57 | 21 |
| Steroid injection | 39 | 55 | 20 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 83 | 37 |
| Steroid injection | 39 | 86 | 34 | |
| Outcome: Range of movement – active external rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 23 | 15 |
| Steroid injection | 39 | 22 | 16 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 37 | 17 |
| Steroid injection | 39 | 39 | 20 | |
| Outcome: Range of movement – active internal rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 46 | 15 |
| Steroid injection | 39 | 45 | 16 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 66 | 18 |
| Steroid injection | 39 | 68 | 17 | |
| Outcome: Range of movement – passive abduction (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 31 | 11 |
| Steroid injection | 39 | 31 | 11 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 46 | 13 |
| Steroid injection | 39 | 12 | 17 | |
| Outcome: Range of movement – passive external rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 19 | 13 |
| Steroid injection | 39 | 16 | 14 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 29 | 16 |
| Steroid injection | 39 | 27 | 17 | |
| Outcome: Range of movement – passive internal rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthrographic distension + steroid | Baseline | 37 | 34 | 14 |
| Steroid injection | 39 | 32 | 13 | |
| Intervention | Time point | No. analysed | Mean | SD |
| Arthrographic distension + steroid | 6 weeks after last injection | 37 | 48 | 15 |
| Steroid injection | 39 | 45 | 12 | |
| Study: Tveita 200836 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Tveita 200836 |
|---|
| Outcome: Other |
| Not reported |
| Study: Tveita 200836 |
|---|
| Outcome: Adverse events |
| Injections reported as very painful: distension: n = 5, steroid: n = 6 |
| Other possible side effects reported: distension: n = 14, steroid: n = 20 |
| Complaints of flushing or disturbances in heat regulation: distension: n = 9, steroid: n = 13 |
| Minor loss of sensation and motor control loss in affected arm: distension: n = 2; steroid: n = 2 |
| Loss of sleep, nausea or dizziness: number and treatment group unspecified |
| Glenohumeral joint infection: treatment group unspecified: n = 1 |
Appendix 7.7 Capsular release
| Study: Austgulen 200787 |
|---|
| Outcome: Pain |
| Not reported |
| Study: Austgulen 200787 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using the Oxford Shoulder score | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 41 | 75 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 18.4 | 7.3 | < 0.001 |
| Outcome: Function and disability measured using a telephone questionnaire (ability to work) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 2.4 | 2.6 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 7.4 | 2.5 | < 0.001 |
| Outcome: Function and disability measured using a telephone questionnaire (physical activity) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 2.3 | 2.5 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 7.4 | 2.4 | < 0.001 |
| Outcome: Function and disability measured using a telephone questionnaire (sleep at night) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 1.7 | 2.5 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 7.2 | 2.6 | < 0.001 |
| Study: Austgulen 200787 | |||||
|---|---|---|---|---|---|
| Outcome: Range of movement – abduction (°) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 34 | 8 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 154 | 37 | < 0.001 |
| Outcome: Range of movement – external rotation (°) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | 3 | 5 | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 39 | 23 | < 0.001 |
| Study: Austgulen 200787 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Austgulen 200787 |
|---|
| Outcome: Adverse events |
| Two patients had FS again and had repeat surgery. No deep infections, nerve damage or other complications reported |
| Study: Austgulen 200787 | |||||
|---|---|---|---|---|---|
| Outcome: Other (satisfaction, 10-point scale) | |||||
| Interventiona | Time point | No. randomised | Mean | SD | |
| Arthroscopic capsular release and PT | Baseline | 66 (70 shoulders) | NR | NR | |
| Interventiona | Time point | No. analysed | Mean | SD | p-value |
| Arthroscopic capsular release and PT | Postoperativelyb | Unclear | 8.6 | 1.6 | NR |
| Study: Chen 200288 | |||||
|---|---|---|---|---|---|
| Outcome: Pain (reduction of pain) | |||||
| Intervention | Time point | No. randomised | Mean | SDa | |
| Arthroscopic capsular release + manipulation + PT | Baseline | 183 (186 shoulders) | NR | NR | |
| Intervention | Time point | No. analysed | Description of results provided in paper | ||
| Arthroscopic capsular release + PT | Within 1 months | Unclear | Half of patients had pain relief within 1 month whether in motion or not | ||
| Arthroscopic capsular release + PT | After 3 months | All except eight shoulders were pain free in any direction of shoulder movement | |||
| Study: Chen 200288 | |||||
|---|---|---|---|---|---|
| Outcome: Function and disability measured using a modified version of the ASES score | |||||
| Intervention | Time point | No. randomised | Mean | SDa | |
| Arthroscopic capsular release + manipulation + PT | Baseline | 183 (186 shoulders) | 41 | 13 | |
| Intervention | Time point | No. analysed | Mean improvement | SDa | p-value |
| Arthroscopic capsular release + PT | Postoperativelyb | Unclear | 87 | 11 | < 0.005 |
| Study: Chen 200288 | ||||
|---|---|---|---|---|
| Outcome: Range of movement – external rotation(°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthroscopic capsular release + manipulation + PT | Baseline | 183 (186 shoulders) | NR | NR |
| Intervention | Time point | No. analysed | Average gain in external rotation | SD |
| Arthroscopic capsular release + PT | Postoperativelya | Unclear | 35° | NR |
| Outcome: Range of movement – internal rotation (°) | ||||
| Intervention | Time point | No. randomised | Mean | SD |
| Arthroscopic capsular release + manipulation + PT | Baseline | 183 (186 shoulders) | NR | NR |
| Intervention | Time point | No. analysed | Average gain in external rotation | SD |
| Arthroscopic capsular release + PT | Postoperativelya | Unclear | 30° | NR |
| Study: Chen 200288 |
|---|
| Outcome: Quality of life |
| Not reported |
| Study: Chen 200288 |
|---|
| Outcome: Other |
| Not reported |
| Study: Chen 200288 |
|---|
| Outcome: Adverse events |
| Complications occurred in only one patient (superficial wound infection) |
Appendix 8 Study quality
Controlled trials
| Study | No. of participants randomised stated | Method of randomisation | Allocation concealment | Comparability at baseline | Adjustment for baseline imbalance | Double blinded | Patients blinded | Outcome assessors blinded | Caregivers blinded | ITT analysis | Imbalances in dropouts | Imbalances in dropouts explained/adjusted for | Study powered |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amir-us-Saqlain 200784 | Y | U | U | Ya | N | U | U | U | N | U | N | NA | U/NR |
| Bal 200868 | Y | U | U | U | N | Y | Y | Y | N | N | Y | Y | U/NR |
| Buchbinder 200443 | Y | Y | Y | Y | NA | Y | Y | Y | N | Y | Y | Y | N |
| Calis 200666 | Y | U | U | Y | NA | N | U | Y | N | Y | N | NA | U/NR |
| Carette 200335 | Y | Y | Y | Yb | Y | U | U | Y | U | Y | Y | Y | N |
| Cheing 200881 | Y | U | U | Y | NA | Y | U | Y | U | N | N | NA | U/NR |
| Dacre 198967 | Y | U | U | U | N | N | N | Y | N | N | U | N | Y |
| Diercks 200473 | N | N | N | Y | NA | N | N | N | N | NA | N | NA | NA |
| Dogru 200851 | Y | Uc | U | N | N | N | N | Y | U | N | N | NA | Nd |
| Dundar 200974 | Y | U | U | Y | NA | N | U | U | U | U | N | NA | U |
| Fang 200682 | Y | U | U | U | N | N | Y | U | N | U | U | U | U/NR |
| Gam 199886 | Y | Y | U | U | NA | U | U | Y | U | N | N | NA | U/NR |
| Jacobs 200985 | Y | U | U | Ye | Y | N | N | U | N | N | U | U | N |
| Kivimaki 200739 | Y | Y | Y | Y | NA | N | N | Y | N | U | Y | N | Yf |
| Leung 200875 | Y | Y | U | Y | NA | N | N | Y | N | Y | N | NA | U/NR |
| Ma 200683 | Y | Ug | U | Y | NA | N | N | U | N | U | U | NA | U/NR |
| Maricar 199976 | Uh | U | U | Y | N | N | U | U | U | N | U | NA | U/NR |
| Pajareya 200477 | Y | Ni | Y | Y | NA | N | U | Uj | N | N | Y | N | Nd |
| Quraishi 200738 | Y | Y | U | Y | NA | N | N | U | N | U | N | NA | U/NR |
| Rizk 199142 | Y | U | U | Y | NA | N | N | Y | N | N | N | NA | N |
| Rovetta 199869 | Y | U | U | Y | NA | N | U | U | U | U | Uk | NA | U/NR |
| Ryans 200541 | Y | Y | U | Y | NA | N | Y | Y | N | N | Y | Y | N |
| Stergioulas 200816 | Y | Y | Ul | Y | NA | Y | Y | Y | U | N | N | NA | U/NR |
| Takagishi 199670 | Y | U | U | U | U | U | U | U | U | U | U | NA | U/NR |
| Tveita 200836 | Y | Y | U | Um | Yn | N | N | N | N | Y | N | NA | No |
| Vermeulen 200640 | Y | Y | U | Y | NA | N | N | Y | N | Y | N | NA | Y |
| Wies 200371 | N | U | U | U | NA | N | N | Y | N | U | U | U | U/NR |
| Yan 200572 | Y | U | U | U | NA | U | U | U | U | Y | N | NA | U/NR |
| Yang 200778 | Y | Y | Y | Y | NA | N | N | Y | U | Y | N | NA | Nd |
Observational studies
| Austgulen 200787 | Chen 200288 | |
|---|---|---|
| Selection/eligibility criteria reported | Y | Y |
| Representativeness of population | P | U |
| Measure of variability | Y | N |
| Loss to follow-up reported/explained | U | U |
| ≥ 90% follow-up | U | U |
| Patients recruited prospectively | Y | U |
| Patients recruited consecutively | U | U |
| Relevant prognostic factors reported | U | N |
Appendix 9 WinBUGS code
Appendix 10 Mixed-treatment comparison
Network 1: studies of any intervention (i.e. conservative and invasive) and any quality
| Prior: Uniform (0, 0.8) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 4 | –1.71 | –2.75 to –0.67a |
| Steroid + physiotherapy | 6 | –1.53 | –2.41 to –0.75a |
| Electroacupuncture | 7 | 1.36 | –2.84 to 0.11 |
| Physiotherapy + placebo | 2 | –1.22 | –1.98 to –0.42a |
| Steroid | 5 | –1.15 | –2.03 to –0.26a |
| Physiotherapy | 3 | –0.94 | –2.20 to 0.38 |
| MUA + physiotherapy | 8 | –0.87 | –2.44 to 0.78 |
| Sodium hyaluronate | 10 | –0.21 | –1.13 to 0.69 |
| Arthrographic distension + steroid | 9 | –0.15 | –1.19 to 0.90 |
| Prior: Uniform (0, 2) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 4 | –1.71 | –3.27 to –0.15a |
| Steroid + physiotherapy | 6 | –1.57 | –2.80 to –0.53a |
| Electroacupuncture | 7 | –1.36 | –3.55 to 0.84 |
| Physiotherapy + placebo | 2 | –1.21 | –2.27 to –0.06a |
| Steroid | 5 | –1.14 | –2.31 to 0.05 |
| Physiotherapy | 3 | –0.93 | –2.78 to 1.02 |
| MUA + physiotherapy | 8 | –0.85 | –3.24 to 1.63 |
| Sodium hyaluronate | 10 | –0.21 | –1.71 to 1.27 |
| Arthrographic distension + steroid | 9 | –0.15 | –1.70 to 1.41 |
| Prior: Uniform (0, 10) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 4 | –1.70 | –3.46 to 0.06 |
| Steroid + physiotherapy | 6 | –1.57 | –2.93 to –0.42a |
| Electroacupuncture | 7 | –1.35 | –3.84 to 1.11 |
| Physiotherapy + placebo | 2 | –1.21 | –2.40 to 0.08 |
| Steroid | 5 | –1.14 | –2.44 to 0.19 |
| Physiotherapy | 3 | –0.94 | –3.03 to 1.26 |
| MUA + physiotherapy | 8 | –0.86 | –3.57 to 1.95 |
| Sodium hyaluronate | 10 | –0.21 | –1.90 to 1.46 |
| Arthrographic distension + steroid | 9 | –0.14 | –1.89 to 1.62 |
| Prior: Uniform (0, 15) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 4 | –1.71 | –3.42 to 0.003 |
| Steroid + physiotherapy | 6 | –1.57 | –2.93 to –0.44a |
| Electroacupuncture | 7 | –1.36 | –3.77 to 1.04 |
| Physiotherapy + placebo | 2 | –1.22 | –2.38 to 0.056 |
| Steroid | 5 | –1.143 | –2.45 to 0.17 |
| Physiotherapy | 3 | –0.94 | –2.98 to 1.24 |
| MUA + physiotherapy | 8 | –0.86 | –3.49 to 1.88 |
| Sodium hyaluronate | 10 | –0.21 | –1.87 to 1.45 |
| Arthrographic distension + steroid | 9 | –0.15 | –1.89 to 1.58 |
Network 2: studies of any intervention that were of good or satisfactory quality (i.e. method of randomisation was adequate and outcome assessment was blinded)
There were five trials with data that could be used in the analysis of pain at or close to 3 months. Five interventions formed part of a connected network with placebo and the evidence was informed by four trials. The network is presented in Figure 27.
FIGURE 27.
Network 2. PT, physical therapy.
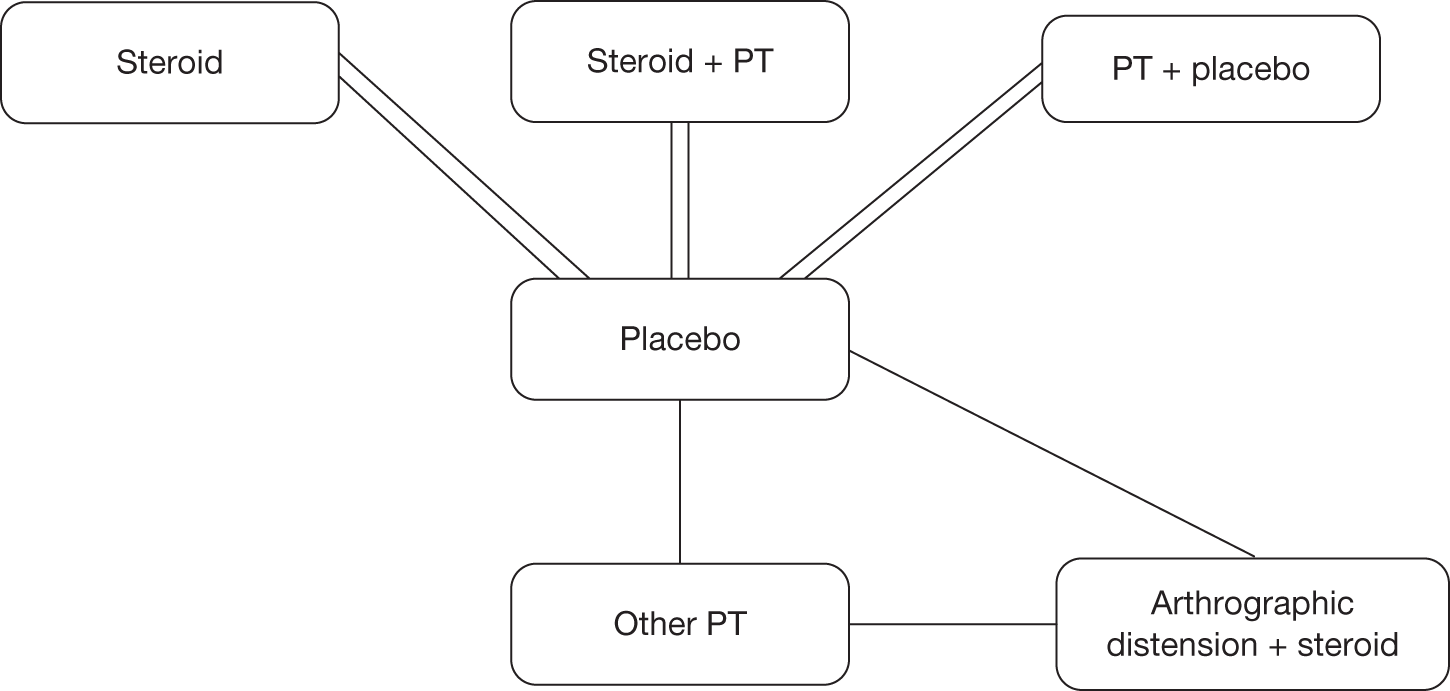
Each line represents one comparison, for example there were two studies available for the comparison of steroid with placebo, and one study for the comparison of other physical therapy with placebo.
| Prior: Uniform (0, 0.8) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.90 | –2.79 to –1.02a |
| Physical therapy without mobilisation | 5 | –1.70 | –2.66 to –0.75a |
| Steroid | 3 | –1.14 | –1.98 to –0.31a |
| Physiotherapy + placebo | 4 | –0.93 | –1.75 to –0.12a |
| Arthrographic distension + steroid | 6 | –0.15 | –1.09 to 0.80 |
| Prior: Uniform (0, 2) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.90 | –3.06 to –0.78a |
| Physical therapy without mobilisation | 5 | –1.71 | –3.15 to –0.29a |
| Steroid | 3 | –1.14 | –2.24 to –0.03a |
| Physiotherapy + placebo | 4 | –0.92 | –2.03 to 0.16 |
| Arthrographic distension + steroid | 6 | –0.14 | –1.59 to 1.29 |
| Prior: Uniform (0, 5) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.90 | –3.24 to –0.61a |
| Physical therapy without mobilisation | 5 | –1.71 | –3.43 to 0.002 |
| Steroid | 3 | –1.14 | –2.42 to 0.14 |
| Physiotherapy + placebo | 4 | –0.93 | –2.20 to 0.34 |
| Arthrographic distension + steroid | 6 | –0.15 | –1.87 to 1.55 |
| Prior: Uniform (0, 10) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.909 | –3.31 to –0.54a |
| Physical therapy without mobilisation | 5 | –1.705 | –3.54 to 0.12 |
| Steroid | 3 | –1.15 | –2.51 to 0.22 |
| Physiotherapy + placebo | 4 | –0.9271 | –2.9 to 0.43 |
| Arthrographic distension + steroid | 6 | –0.1412 | –1.97 to 1.70 |
Network 3: studies of conservative treatments of any quality
There were six trials with data that could be used in the analysis of pain at or close to 3 months. Six interventions formed part of a connected network with placebo and the evidence was informed by six trials. The network is presented in Figure 28.
FIGURE 28.
Network 3. PT, physical therapy.
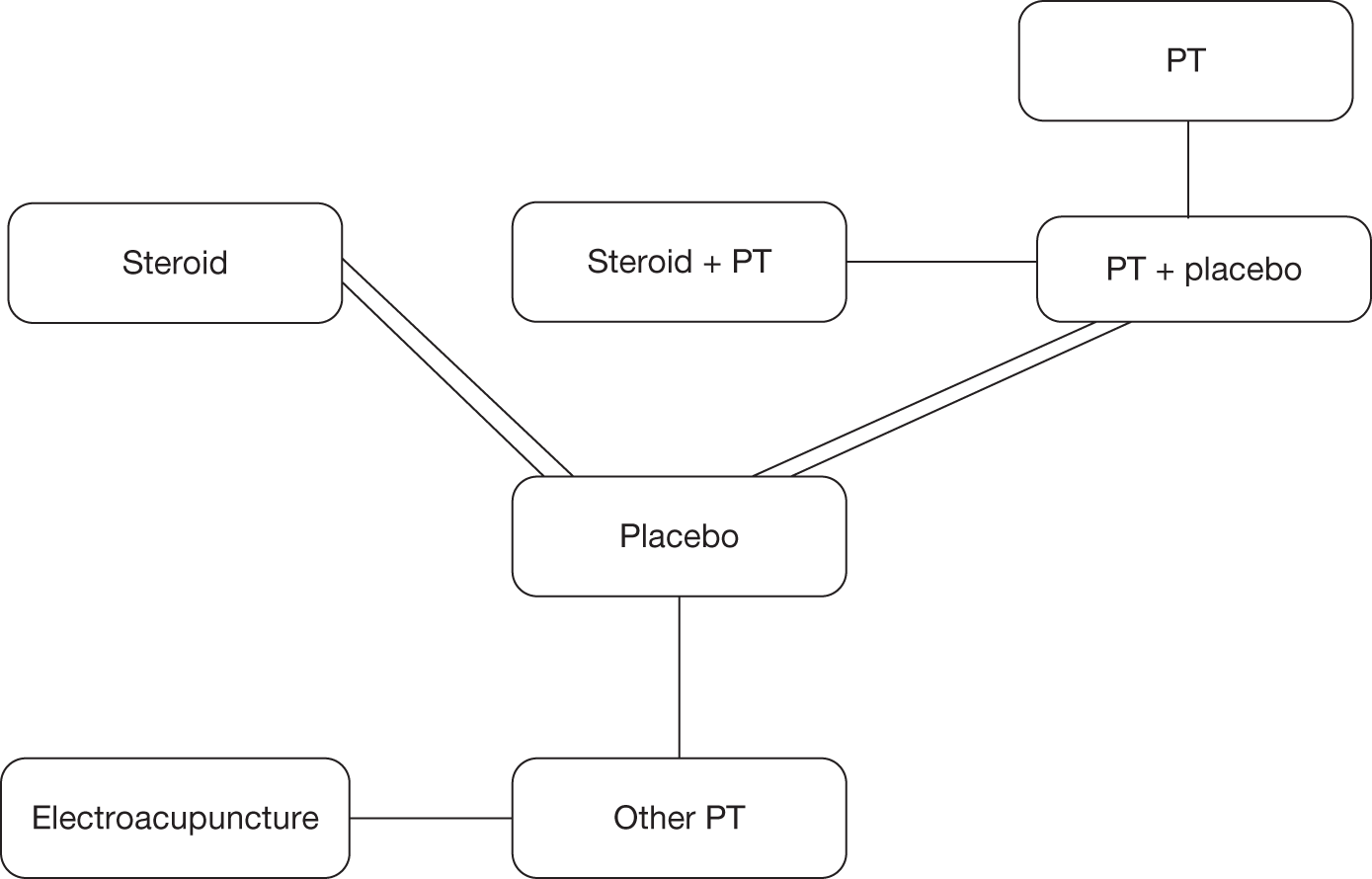
Each line represents one comparison, for example there were two studies available for the comparison of steroid with placebo, and one study for the comparison of other physical therapy with placebo.
| Prior: Uniform (0, 0.8) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 6 | –1.71 | –2.73 to –0.68a |
| Steroid + physiotherapy | 5 | –1.53 | –2.40 to –0.75a |
| Electroacupuncture | 7 | –1.36 | –2.81 to 0.092 |
| Physiotherapy + placebo | 2 | –1.22 | –1.98 to –0.42a |
| Steroid | 4 | –1.15 | –2.02 to –0.27a |
| Physiotherapy | 3 | –0.94 | –2.20 to 0.38 |
| Prior: Uniform (0, 2) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 6 | –1.71 | –3.24 to –0.19a |
| Steroid + physiotherapy | 5 | –1.56 | –2.77 to –0.54a |
| Electroacupuncture | 7 | –1.36 | –3.52 to 0.79 |
| Physiotherapy + placebo | 2 | –1.21 | –2.25 to –0.067a |
| Steroid | 4 | –1.14 | –2.31 to 0.034 |
| Physiotherapy | 3 | –0.92 | –2.73 to 1.00 |
| Prior: Uniform (0, 5) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 6 | –1.70 | –3.43 to –0.002a |
| Steroid + physiotherapy | 5 | –1.58 | –2.92 to –0.46a |
| Electroacupuncture | 7 | –1.35 | –3.80 to 1.05 |
| Physiotherapy + placebo | 2 | –1.21 | –2.38 to 0.037 |
| Steroid | 4 | –1.15 | –2.43 to 0.14 |
| Physiotherapy | 3 | –0.93 | –2.98 to 1.21 |
| Prior: Uniform (0, 10) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Physical therapy without mobilisation | 6 | –1.70 | –3.43 to 0.059 |
| Steroid + physiotherapy | 5 | –1.57 | –2.92 to –0.43a |
| Electroacupuncture | 7 | –1.35 | –3.81 to 1.13 |
| Physiotherapy + placebo | 2 | –1.21 | –2.39 to 0.060 |
| Steroid | 4 | –1.15 | –2.45 to 0.15 |
| Physiotherapy | 3 | –0.92 | –2.99 to 1.25 |
Network 4: studies of conservative treatments that were of good or satisfactory quality (i.e. method of randomisation was adequate and outcome assessment was blinded)
There were three trials with data that could be used in the analysis of pain at or close to 3 months. Four interventions formed part of a connected network with placebo and the evidence was informed by four trials. The network is presented in Figure 29.
FIGURE 29.
Network 4. PT, physical therapy.
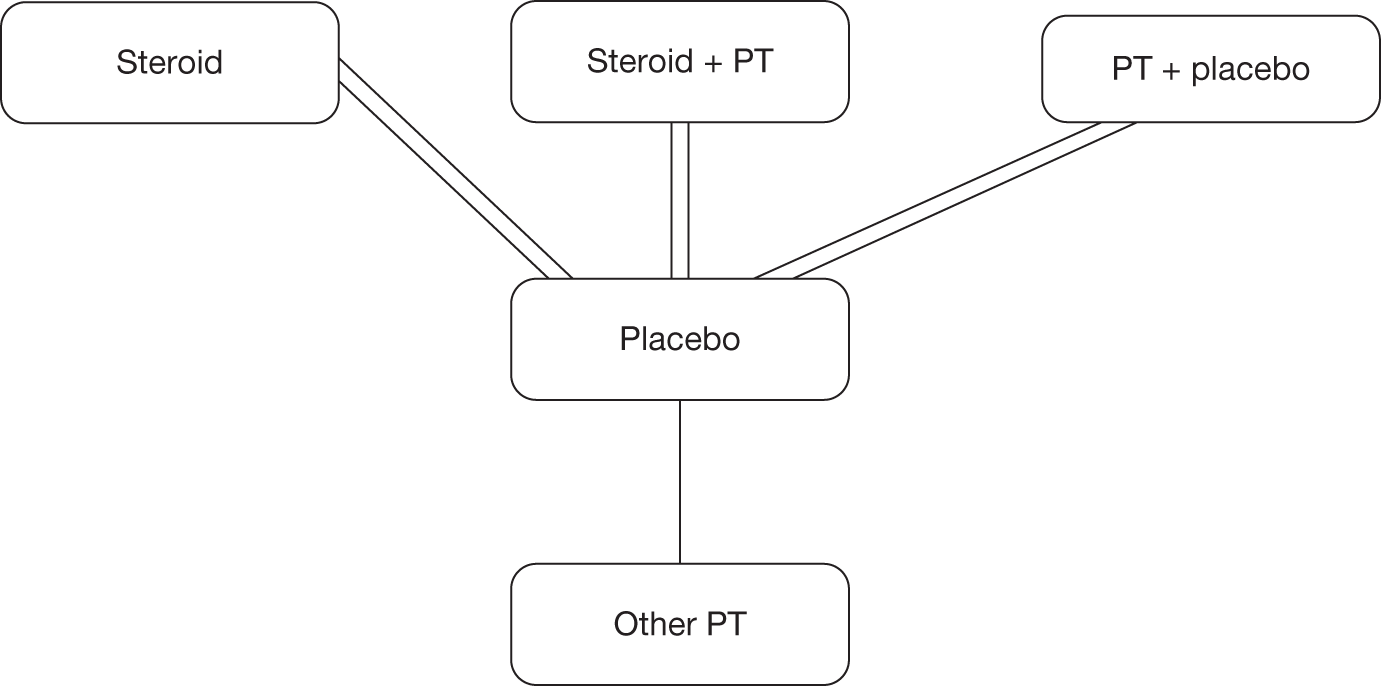
Each line represents one comparison, for example there were two studies available for the comparison of steroid with placebo, and one study for the comparison of other physical therapy with placebo.
| Prior: Uniform (0, 0.8) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.9 | –2.79 to –1.02a |
| Physical therapy without mobilisation | 5 | –1.7 | –2.66 to –0.76a |
| Steroid | 3 | –1.14 | –1.97 to –0.31a |
| Physiotherapy + placebo | 4 | –0.94 | –2.66 to –0.76a |
| Prior: Uniform (0, 2) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.911 | –3.06 to –0.81a |
| Physical therapy without mobilisation | 5 | –1.706 | –3.12 to –0.30a |
| Steroid | 3 | –1.146 | –2.23 to –0.06a |
| Physiotherapy + placebo | 4 | –0.9251 | –2.01 to 0.14 |
| Prior: Uniform (0, 5) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.912 | –3.24 to –0.62a |
| Physical therapy without mobilisation | 5 | –1.71 | –3.43 to 0.008 |
| Steroid | 3 | –1.14 | –2.43 to 0.16 |
| Physiotherapy + placebo | 4 | –0.93 | –2.22 to 0.34 |
| Prior: Uniform (0, 10) | |||
|---|---|---|---|
| Comparator: placebo | |||
| Treatment | Node no. | SMD | |
| Mean | 95% CrI | ||
| Steroid + physiotherapy | 2 | –1.91 | –3.35 to –0.54a |
| Physical therapy without mobilisation | 5 | –1.71 | –3.55 to 0.14 |
| Steroid | 3 | –1.15 | –2.51 to 0.25 |
| Physiotherapy + placebo | 4 | –0.93 | –2.32 to 0.43 |
Appendix 11 Economic evaluation study quality checklist
Appendix 12 Economic evaluation data extraction/summary
Study assessed: van den Hout 200593
Type of economic evaluation: Cost–utility analysis.
Study objective: To compare high-grade and low-grade mobilisation techniques in patients with frozen shoulder (FS).
Interventions: The interventions compared were high-grade mobilisation comprising mobilisation techniques that are performed in the end ranges of the limited joint mobility of the shoulder. The duration of prolonged stress in the end-range position was varied according to individual patient tolerance. Low-grade mobilisation was performed under the explicit instruction that the technique should be performed without causing any pain to the patient. Patients were treated for 30 minutes twice weekly over a period of 12 weeks. From 6 weeks onwards treatments could be reduced in frequency, or stopped if the therapist felt that the shoulder had returned to a normal range of motion. The only concomitant treatment allowed was pain relief. The history of treatment was variable although a high percentage of patients in each group (64% HGMT and 58% LGMT) had received multiple steroid injections prior to enrolling in the trial.
Location/setting: The study was conducted in a tertiary care setting in the Netherlands.
Analytical approach: The economic evaluation was conducted concurrently with a RCT. 40 The trial enrolled 100 patients who had suffered from unilateral adhesive capsulitis for at least 3 months and who had experienced at least a 50% decrease in passive joint mobility. An ITT analysis was conducted. Missing data were imputed by carrying forward the last available measurement. Differences were tested using double-sided non-parametric bootstrapping. The time horizon appears to have been 12 months and the analysis was conducted from a societal perspective.
Effectiveness/utility data: No clinical efficacy data were presented in the paper. Data on utilities were estimated using the SF-6D utility index, which was calculated from SF-36 response data collected alongside the trial at baseline and at 3, 6 and 12 months’ follow-up. Only SF-6D data were presented.
Resource use/cost data: Resource-use information was collected using quarterly cost questionnaires. Dutch standard prices, which were designed to reflect social costs and standardise economic evaluations, were used. Where standard prices were not available, charges were used. Costs included treatment sessions, alternative medicine sessions, hospitalisations (MUA, acromioplasty), home nursing care, medication, travel costs and non-health-care costs (labour and domestic help).
Results: The average annual health-care costs per patient were €2552 for HGMT and €2293 for LGMT, a difference of €259 in favour of LGMT (p = 0.58, 95% CI –€644 to €1162). The average annual societal costs per patient were €8809 for HGMT and €6911 for LGMT, a difference of €1898 in favour of LGMT (p = 0.37, 95% CI –€2551 to €5711).
The average QALYs were 0.695 for HGMT and 0.702 for LGMT, a difference of 0.007 (p = 0.71, 95% CI –0.32 to 0.049).
Although results did not reach statistical significance, both cost and utility results favoured LGMT over HGMT.
Appendix 13 Resource-use table
Resource use for the interventions identified from the primary studies included in the review
| Intervention | Resource use | ||
|---|---|---|---|
| Health professionals involved (physiotherapist, GP, other) | Setting (community, hospital) | Quantities (no. of sessions, duration, etc.) | |
| Steroid therapy alone | Guided injection (hospital doctor, either trainee or consultant, orthopaedic surgeon, rheumatologist, radiologist, anaesthetist) | Hospital | Three sessions: first session (initial assessment), 30 minutes; two follow-up sessions each 1 hour with injection |
| Unguided injection delivered by physiotherapist or GP | Community |
Physiotherapist: three sessions: first session (initial assessment) 20 minutes; two follow-up sessions with injection each 10–20 minutes GP: three sessions: first session (initial assessment) 10–12.5 minutes; two follow-up sessions each 10–12.5 minutes |
|
| Active PT | Physiotherapist | Community/hospital |
Six sessions of 30 minutes’ duration delivered in either setting Following guided injection, referral to physiotherapist for six sessions of 30 minutes’ duration Following injection from physiotherapist, six sessions of 30 minutes’ duration Following injection from GP, referral to physiotherapist for six sessions of 30 minutes’ duration OR Active PT is delivered at the same time as injection in all settings = no extra sessions/time required |
| Sodium hyaluronate | Hospital consultant | Hospital | One injection, 30–60 minutes; one review visit |
| Electroacupuncturea + other PT | Physiotherapist | Either | Delivered in six sessions plus additional component of PT by a physiotherapist, adding maybe 15 minutes to a 20- to 30-minute session |
| ‘Normal’ acupuncture + active PT | As above | Either | As above – delivered in six sessions plus additional component of PT by a physiotherapist, adding maybe 15 minutes to a 20- to 30-minute session |
| MUA | Orthopaedic surgeon and anaesthetist; operating theatre staff, usually one anaesthetic nurse and a surgical scrub nurse | Secondary care hospital |
General anaesthesia and MUA likely to take on average 15 minutes. Recovery from anaesthesia variable, typically may take a further 15 minutes. Plus final/follow-up review with orthopaedic surgeon Base case 30 minutes, general surgical procedure |
| Arthrographic distension | Radiologist or orthopaedic surgeon | Secondary care hospital | The procedure was assumed to require around 15 minutes and be delivered as a single injection over one visit |
| Capsular release | Physiotherapist, anaesthetist, orthopaedic surgeon, anaesthetic nurse, scrub nurse and assistant, recovery nurse | Secondary care hospital | 30–45 minutes for the anaesthesia and release procedure. Recovery from anaesthesia further 15 minutes |
Appendix 14 Exploratory mapping analysis
Mapping from SF-36 PCS and MCS onto EQ-5D
Pattern of EQ-5D scores and SF-36 (PCS, MCS) scores within the SAPPHIRE data set (for complete measurements at 1 month)
| EQ-5D grouping | n | EQ-5D (mean) | SF-36 PCS (mean) | SF-36 MCS (mean) |
|---|---|---|---|---|
| < 0 | 11 | –0.07 | 28.11 | 27.46 |
| 0–0.249 | 23 | 0.11 | 33.47 | 40.18 |
| 0.25–0.499 | 4 | 0.29 | 39.86 | 52.83 |
| 0.5–0.699 | 60 | 0.62 | 36.96 | 43.82 |
| 0.7–0.799 | 35 | 0.76 | 43.78 | 53.18 |
| 0.8–0.899 | 20 | 0.81 | 50.08 | 51.43 |
| 0.9–1.0 | 0 | NA | NA | NA |
| Full index | 18 | 1 | 52.48 | 55.05 |
| Overall | 171 | 0.59 | 40.49 | 46.19 |
Pattern of EQ-5D scores and SF-36 (PCS, MCS) scores within the SAPPHIRE data set (for complete measurements at 3 months)
| EQ-5D grouping | n | EQ-5D (mean) | SF-36 PCS (mean) | SF-36 MCS (mean) |
|---|---|---|---|---|
| < 0 | 10 | –0.07 | 35.96 | 45.70 |
| 0–0.249 | 18 | 0.11 | 39.18 | 47.35 |
| 0.25–0.499 | 1 | 0.26 | 36.64 | 41.14 |
| 0.5–0.699 | 44 | 0.63 | 39.68 | 42.01 |
| 0.7–0.799 | 21 | 0.75 | 44.07 | 50.23 |
| 0.8–0.899 | 28 | 0.82 | 42.91 | 53.2 |
| 0.9–1.0 | 0 | NA | NA | NA |
| Full index | 11 | 1.0 | 40.68 | 48.95 |
| Overall | 133 | 0.59 | 40.79 | 47.21 |
Pattern of EQ-5D scores and SF-36 (PCS, MCS) scores within the SAPPHIRE data set (for complete measurements at 12 months)
| EQ-5D grouping | n | EQ-5D (mean) | SF-36 PCS (mean) | SF-36 MCS (mean) |
|---|---|---|---|---|
| < 0 | 11 | –0.12 | 27.81 | 29.83 |
| 0–0.249 | 11 | 0.09 | 32.00 | 38.27 |
| 0.25–0.499 | 1 | 0.26 | 21.51 | 47.41 |
| 0.5–0.699 | 49 | 0.61 | 34.52 | 43.54 |
| 0.7–0.799 | 30 | 0.75 | 45.54 | 48.71 |
| 0.8–0.899 | 32 | 0.82 | 50.13 | 53.35 |
| 0.9–1.0 | 0 | NA | NA | NA |
| Full index | 29 | 1 | 54.87 | 55.79 |
| Overall | 163 | 0.66 | 42.53 | 47.34 |
Pattern of EQ-5D scores and SF-36 PCS and MCS scores within the SAPPHIRE data set (for complete responses) at different time points
FIGURE 30.
One-month follow-up. (a) EQ-5D and SF-36 PCS, (b) EQ-5D and SF-36 MCS.
FIGURE 31.
Three-month follow-up. (a) EQ-5D and SF-36 PCS, (b) EQ-5D and SF-36 MCS.
FIGURE 32.
Twelve-month follow-up. (a) EQ-5D and SF-36 PCS, (b) EQ-5D and SF-36 MCS.
Regression models using main effects with and without squared terms and interaction term (using individual-level data at 1, 3 and 12 months)
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLSa | OLSa | OLSa | TOBIT | CLAD | |||||
|
Independent variables |
PCS MCS | PCS MCS, squared terms | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | |||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | –0.2645051 | 0.004122 | –0.947965 | 0.1748065 | –1.378458 | 0.0449899 | –1.171123 | 0.3070996 | –1.839797 | 0.4490999 |
| PCS | 0.0139756 | 0.0015533 | –0.0201276 | 0.0113677 | 0.0335792 | 0.0080814 | 0.023285 | 0.0108202 | 0.0568059 | 0.0155343 |
| MCS | 0.0065544 | 0.2287304 | 0.0366059 | 0.0081761 | 0.04406 | 0.0117933 | 0.0415846 | 0.0095459 | 0.0446596 | 0.0092294 |
| PCS*PCS | –0.000091 | 0.000089 | –0.0001066 | 0.0000965 | –0.0000106 | 0.0001109 | –0.000332 | 0.0001687 | ||
| MCS*MCS | –0.003444 | 0.0001092 | –0.000322 | 0.0001171 | –0.0003238 | 0.0001014 | –0.0002661 | 0.0000738 | ||
| PCS*MCS | –0.0002528 | 0.0000856 | –0.0001693 | 0.0001162 | –0.0003647 | 0.0001247 | ||||
| Adjusted R2 | 0.3840 | 0.4162 | 0.4284 | – | – | |||||
| ME | –0.0025 | –0.0039 | –0.0020 | –0.0235 | –0.0265 | |||||
| MAE | 0.1889 | 0.1871 | 0.1861 | 0.1877 | 0.1815 | |||||
| RMSE | 0.254899 | 0.259112 | 0.25906 | 0.26508 | 0.266589 | |||||
| |Difference|b | ||||||||||
| |Δ|≤ 0.10 | 154 | 82% | 151 | 81% | 154 | 82% | 156 | 83% | 154 | 82% |
| |Δ|≤ 0.05 | 136 | 73% | 130 | 70% | 137 | 70% | 139 | 74% | 145 | 78% |
| |Δ|≤ 0.01 | 85 | 45% | 76 | 41% | 88 | 47% | 91 | 49% | 102 | 55% |
| Actual | Predict | Predict | Predict | Predict | Predict | |||||
| Mean | 0.6219 | 0.6244 | 0.6258 | 0.6239 | 0.6454 | 0.6484 | ||||
| SD | 0.29191 | 0.19633 | 0.20683 | 0.20939 | 0.22937 | 0.21983 | ||||
| Min. | –0.18 | 0.20 | –0.10 | –0.09 | –0.10 | –0.07 | ||||
| Max. | 1.00 | 0.92 | 0.90 | 0.92 | 1.01 | 0.86 | ||||
| Rangec | 1.18 | 0.72 | 1.01 | 1.01 | 1.11 | 0.93 | ||||
Regression models using the main effects with and without squared terms and interaction term (using models estimated from 3-month data and used to predict EQ-5D scores at 12 months)
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLSa | OLSa | OLSa | TOBIT | CLAD | |||||
|
Independent variables |
PCS MCS | PCS MCS, squared terms | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | PCS MCS, squared terms and interaction term | |||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | 0.2842682 | 0.1811317 | –0.6233533 | 0.5770837 | –1.552434 | 0.749881 | –1.814838 | 0.7887539 | –0.6770489 | 1.035821 |
| PCS | 0.004559 | 0.003425 | 0.0018081 | 0.0201721 | 0.0249701 | 0.0232969 | 0.0286508 | 0.0244527 | 0.0157259 | 0.0273862 |
| MCS | 0.0026323 | 0.0030736 | 0.0516204 | 0.0221907 | 0.073418 | 0.0246642 | 0.0831682 | 0.0259928 | 0.0399689 | 0.0316185 |
| PCS*PCS | 0.0000186 | 0.000258 | 0.0000464 | 0.0002541 | 0.000041 | 0.0002683 | 0.0000152 | 0.0002815 | ||
| MCS*MCS | –0.0005561 | 0.0002495 | –0.0005877 | 0.0002459 | –0.0006711 | 0.0002584 | –0.0002875 | 0.0002772 | ||
| PCS*MCS | –0.00052 | 0.0002744 | –0.0005816 | 0.0002883 | –0.0002641 | 0.0003168 | ||||
| Adjusted R2 | 0.0147 | 0.0513 | 0.0830 | – | – | |||||
| ME | 0.0662 | 0.0703 | 0.0761 | 0.0654 | –0.0100 | |||||
| MAE | 0.1969 | 0.2045 | 0.2039 | 0.2672 | 0.1531 | |||||
| RMSE | 0.247936 | 0.266884 | 0.260014 | 0.351159 | 0.221678 | |||||
| |Difference|b | ||||||||||
| |Δ|≤ 0.10 | 51 | 31% | 55 | 34% | 58 | 36% | 58 | 36% | 94 | 58% |
| |Δ|≤ 0.05 | 23 | 14% | 34 | 21% | 39 | 24% | 36 | 22% | 56 | 34% |
| |Δ|≤ 0.01 | 7 | 4% | 15 | 9% | 8 | 8% | 17 | 6% | 16 | 10% |
| Actual | Predict | Predict | Predict | Predict | Predict | |||||
| Mean | 0.6690 | 0.6028 | 0.5987 | 0.5929 | 0.6037 | 0.6790 | ||||
| SD | 0.29254 | 0.07250 | 0.1277 | 0.15807 | 0.19692 | 0.12368 | ||||
| Min. | –0.24 | 0.46 | –0.20 | –0.24 | –0.50 | 0.10 | ||||
| Max. | 1.00 | 0.71 | 0.76 | 0.86 | 0.91 | 0.84 | ||||
| Range c | 1.02 | 0.256 | 0.607 | 0.755 | 0.846 | 0.541 | ||||
Actual versus predicted EQ-5D: mapping SF-36 PCS and MCS to EQ-5D
FIGURE 33.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, OLS1 model.
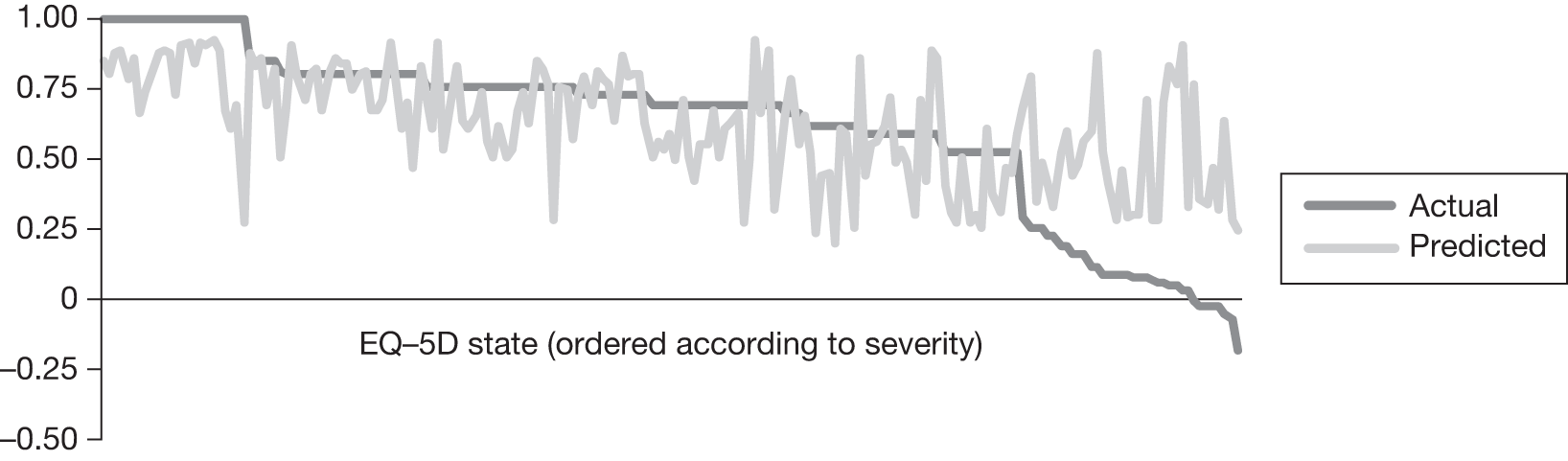
FIGURE 34.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, OLS2 model.
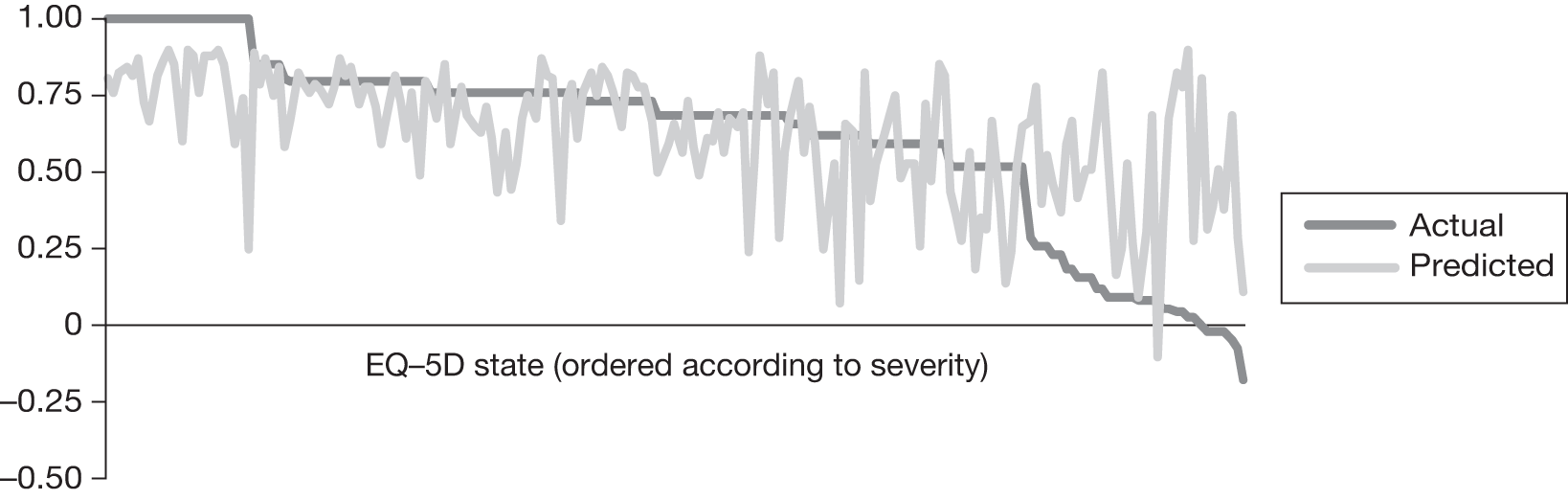
FIGURE 35.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, OLS3 model.
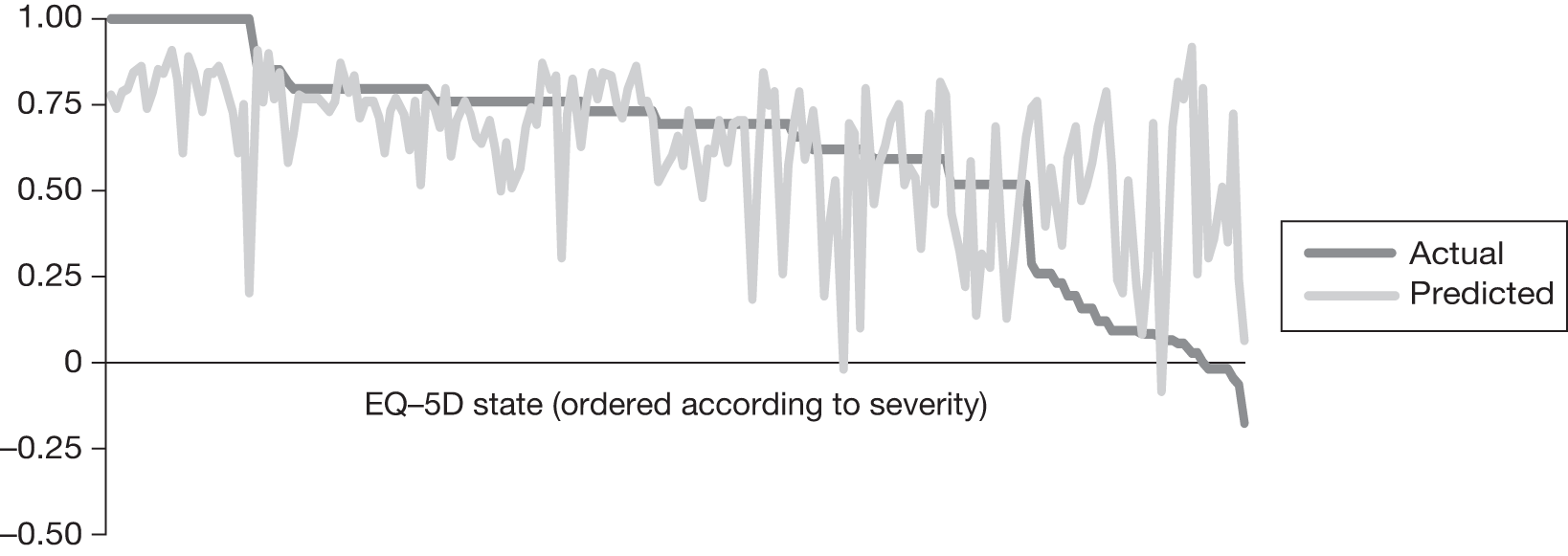
FIGURE 36.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, TOBIT model.
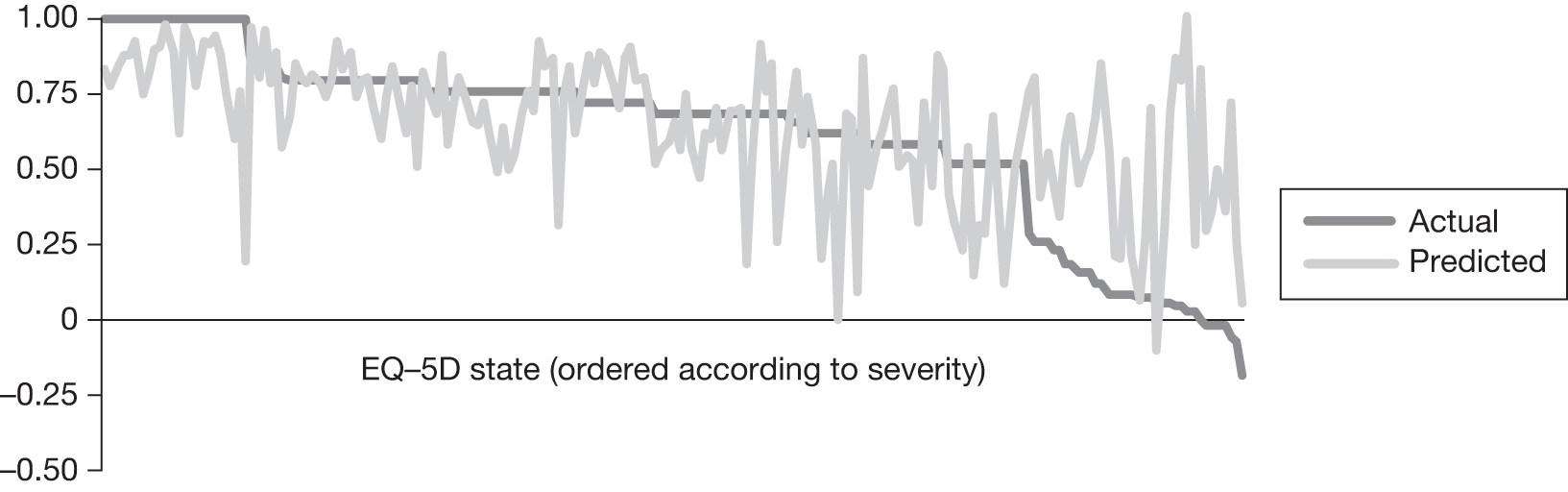
FIGURE 37.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, CLAD model.
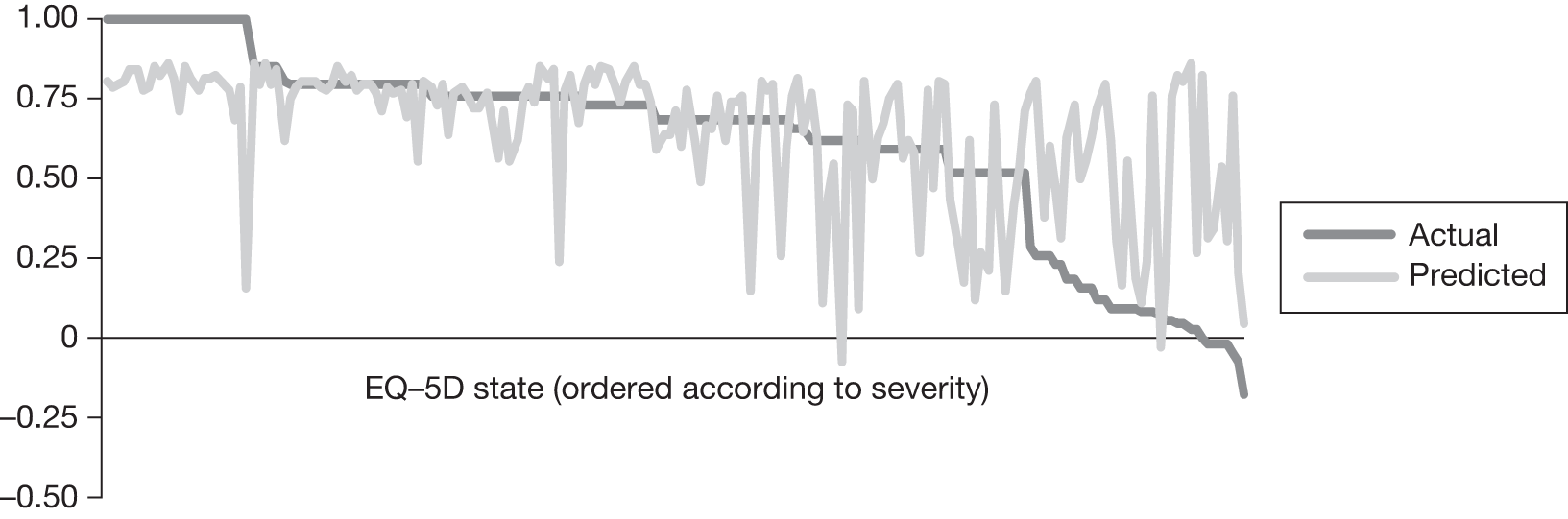
Mapping from pain visual analogue scale onto EQ-5D
Pattern of EQ-5D scores and pain visual analogue scale scores within the SAPPHIRE data set (for complete measurements at 1 month)
| EQ-5D grouping | n | EQ-5D (mean) | Pain VAS (mean) |
|---|---|---|---|
| < 0 | 11 | –0.07 | 80.91 |
| 0–0.249 | 23 | 0.11 | 58.26 |
| 0.25–0.499 | 4 | 0.29 | 26.25 |
| 0.5–0.699 | 68 | 0.63 | 51.00 |
| 0.7–0.799 | 34 | 0.76 | 33.91 |
| 0.8–0.899 | 20 | 0.81 | 29.4 |
| 0.9–1.0 | 19 | 1.0 | 19.74 |
| Full index | 19 | 1.0 | 19.74 |
| Overall | 179 | 0.59 | 44.13 |
Pattern of EQ-5D scores and pain visual analogue scale scores within the SAPPHIRE data set (for complete measurements at 3 months)
| EQ-5D grouping | n | EQ-5D (mean) | Pain VAS (mean) |
|---|---|---|---|
| < 0 | 8 | –0.05 | 18.13 |
| 0–0.249 | 15 | 0.1 | 29.27 |
| 0.25–0.499 | 1 | 0.26 | 50 |
| 0.5–0.699 | 52 | 0.63 | 41.87 |
| 0.7–0.799 | 24 | 0.75 | 40.17 |
| 0.8–0.899 | 26 | 0.83 | 38.88 |
| 0.9–1.0 | 15 | 1.0 | 34.67 |
| Full index | 15 | 1.0 | 34.67 |
| Overall | 141 | 0.63 | 37.63 |
Pattern of EQ-5D scores and pain visual analogue scale scores within the SAPPHIRE data set (for complete measurements at 12 months)
| EQ-5D grouping | n | EQ-5D (mean) | Pain VAS (mean) |
|---|---|---|---|
| < 0 | 12 | –0.05 | 57.83 |
| 0–0.249 | 13 | 0.1 | 55.77 |
| 0.25–0.499 | 1 | 0.26 | 5 |
| 0.5–0.699 | 50 | 0.63 | 39.16 |
| 0.7–0.799 | 30 | 0.75 | 34.63 |
| 0.8–0.899 | 31 | 0.83 | 17.00 |
| 0.9–1.0 | 34 | 1.0 | 5.29 |
| Full index | 34 | 1.0 | 5.29 |
| Overall | 171 | 0.63 | 29.99 |
Pattern of EQ-5D scores and pain visual analogue scale scores within the SAPPHIRE data set (for complete responses) at different time points
FIGURE 38.
One-month follow-up.
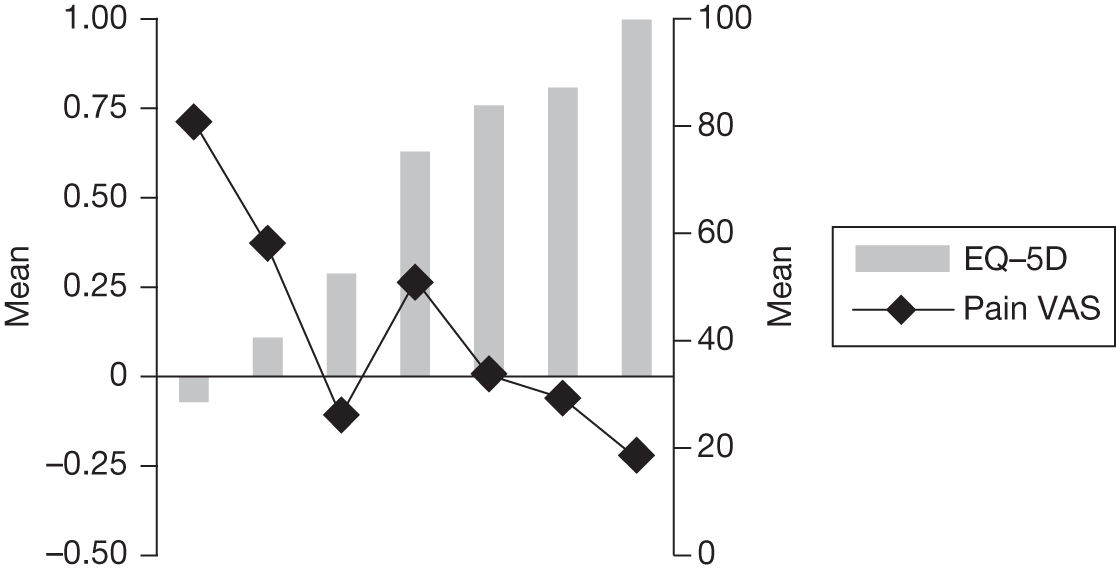
FIGURE 39.
Three-month follow-up.
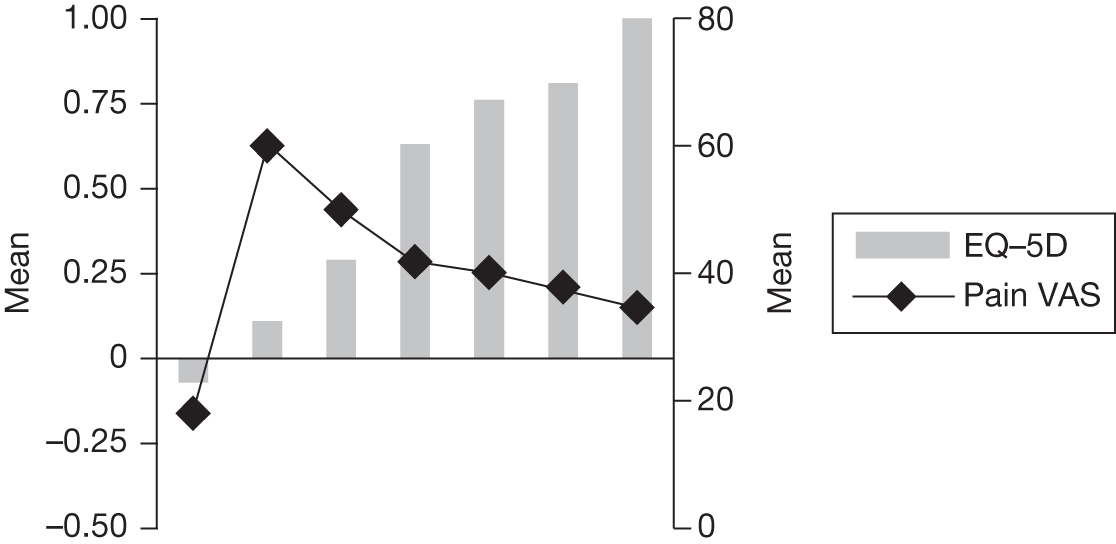
FIGURE 40.
Twelve-month follow-up.
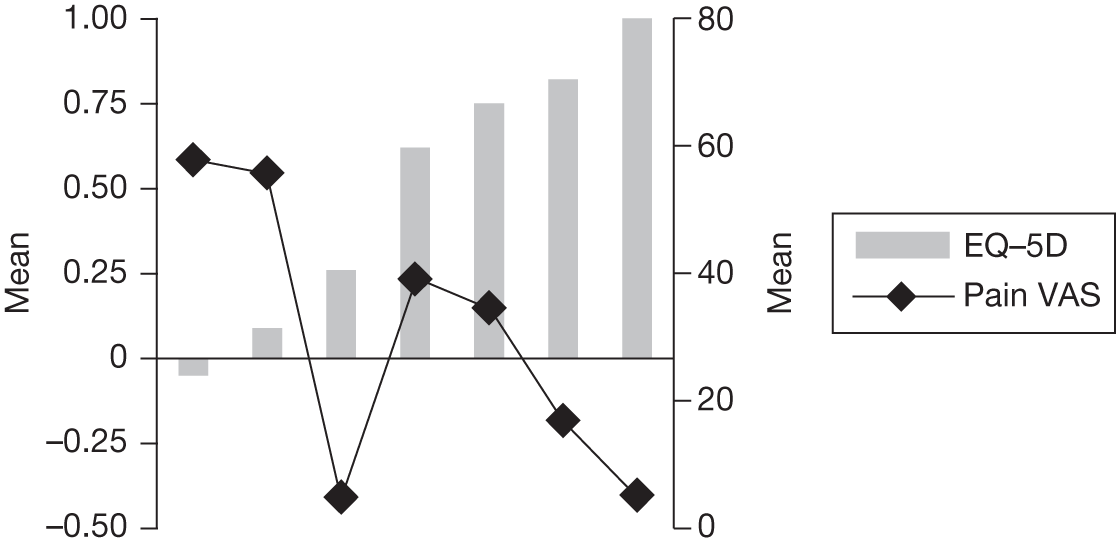
Regression models using pain visual analogue scale main effects with and without squared terms (using individual-level data at 1, 3 and 12 months)
| Model 1 | Model 2 | Model 3 | Model 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLS | OLS | TOBIT | CLAD | ||||
|
Independent variables |
Pain VAS night | Pain VAS night, squared terms | Pain VAS night, squared terms | Pain VAS night, squared terms | ||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | 0.7737689 | 0.0020501 | 0.735917 | 0.0860794 | 0.80135 | 0.80135 | 0.8 | 0.0242477 |
| pVASnight | –0.0031619 | 0.0825151 | –0.0033967 | 0.0024525 | –0.0054838 | –0.0054838 | –0.0042917 | 0.0016361 |
| pVASnight*pVASnight | 0.0000028 | 0.0000104 | 0.0000199 | 0.0000199 | 0.0000208 | 0.00002 | ||
| R 2 | 0.1009 | 0.1009 | – | – | ||||
| ME | 0.03051 | 0.03055 | 0.00183 | –0.04091 | ||||
| MAE | 0.20144 | 0.20167 | 0.19449 | 0.18155vw | ||||
| RMSE | 0.26497 | 0.26487 | 0.26403 | 0.26689 | ||||
| |Difference|a | ||||||||
| |Δ|≤ 0.10 | 69 | 35% | 70 | 36% | 73 | 37% | 90 | 46% |
| |Δ|≤ 0.05 | 39 | 20% | 40 | 20% | 39 | 20% | 50 | 26% |
| |Δ|≤ 0.01 | 5 | 3% | 5 | 3% | 8 | 4% | 2 | 1% |
| Actual | Predict | Predict | Predict | Predict | ||||
| Mean | 0.65250 | 0.62199 | 0.62195 | 0.65067 | 0.69341 | |||
| SD | 0.280635 | 0.096152 | 0.096383 | 0.118435 | 0.080537 | |||
| Min. | –0.240 | 0.418 | 0.424 | 0.452 | 0.579 | |||
| Max. | 1.00 | 0.734 | 0.736 | 0.801 | 0.800 | |||
| Rangeb | 1.240 | 0.316 | 0.312 | 0.349 | 0.221 | |||
Regression models using the main effects with and without squared terms and interaction term (using models estimated from 3-month data and used to predict EQ-5D scores at 12 months)
| Model 1 | Model 2 | Model 3 | Model 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Econometric estimation method | OLS | OLS | TOBIT | CLAD | ||||
|
Independent variables |
Pain VAS night | Pain VAS night, squared terms | Pain VAS night, squared terms | Pain VAS night, squared terms | ||||
| Coeff. | SE | Coeff. | SE | Coeff. | SE | Coeff. | SE | |
| Intercept | 0.5540896 | 0.0524505 | 0.5353785 | 0.0663995 | 0.5413921 | 0.0725511 | 0.6872609 | 0.0621687 |
| pVASnight | 0.0013853 | 0.0010672 | 0.0033283 | 0.0043293 | 0.0042145 | 0.0047531 | 0.0009522 | 0.0036473 |
| pVASnight*pVASnight | –0.000023 | 0.0000497 | –0.0000312 | 0.0000546 | –0.000013 | 0.0000385 | ||
| R 2 | 0.0081 | –0.0014 | – | – | ||||
| ME | 0.03958 | 0.0402 | 0.0206 | –0.0479 | ||||
| MAE | 0.21718 | 0.2218 | 0.2158 | 0.1877 | ||||
| RMSE | 0.276951 | 0.280038 | 0.280351 | 0.279724 | ||||
| |Difference|a | ||||||||
| |Δ|≤ 0.10 | 41 | 24% | 40 | 23% | 48 | 28% | 67 | 39% |
| |Δ|≤ 0.05 | 18 | 11% | 21 | 12% | 22 | 13% | 36 | 21% |
| |Δ|≤ 0.01 | 5 | 3% | 2 | 1% | 7 | 4% | 20 | 12% |
| Actual | Predict | Predict | Predict | Predict | ||||
| Mean | 0.66409 | 0.59563 | 0.59442 | 0.61247 | 0.69277 | |||
| SD | 0.304862 | 0.041060 | 0.048773 | 0.057819 | 0.009118 | |||
| Min. | –0.240 | 0.554 | 0.535 | 0.541 | 0.659 | |||
| Max. | 1.000 | 0.687 | 0.656 | 0.684 | 0.705 | |||
| Rangeb | 1.240 | 0.133 | 0.120 | 0.142 | 0.046 | |||
Actual versus predicted EQ-5D: mapping pain visual analogue scale to EQ-5D
FIGURE 41.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, OLS1 model.
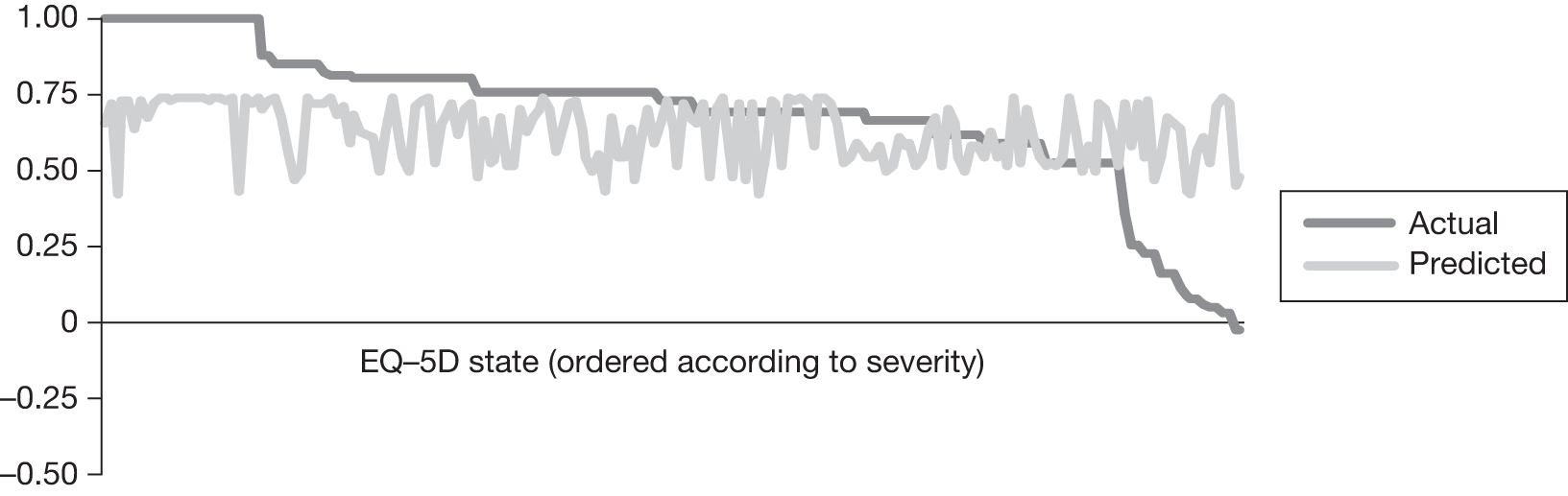
FIGURE 42.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, OLS2 model.
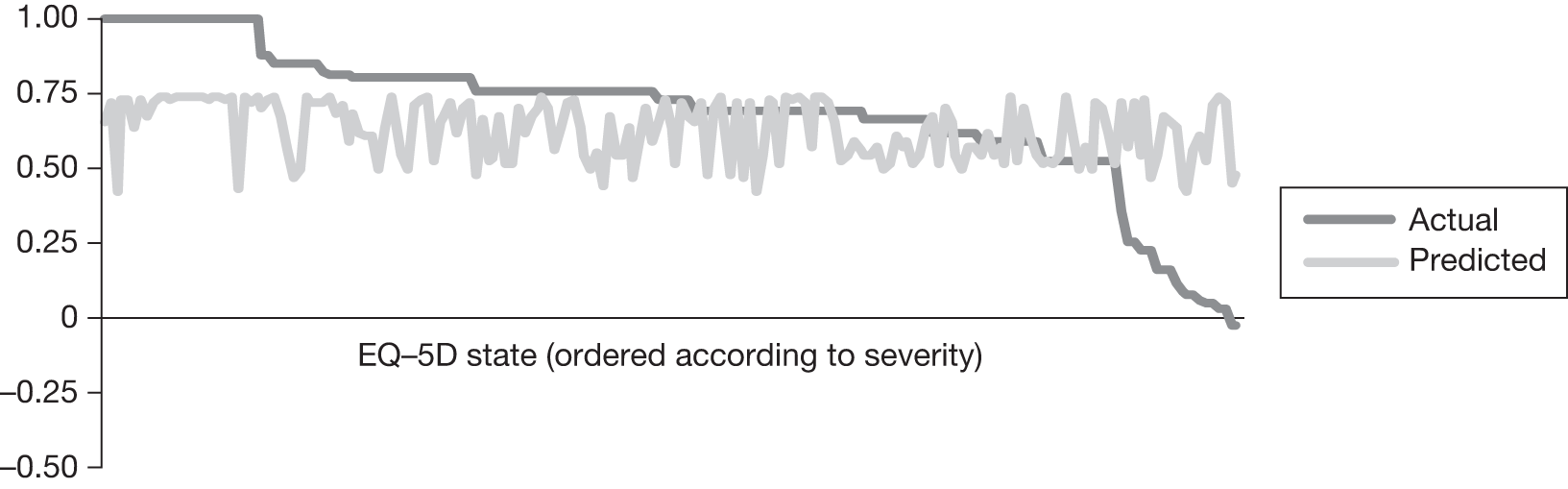
FIGURE 43.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, TOBIT model.
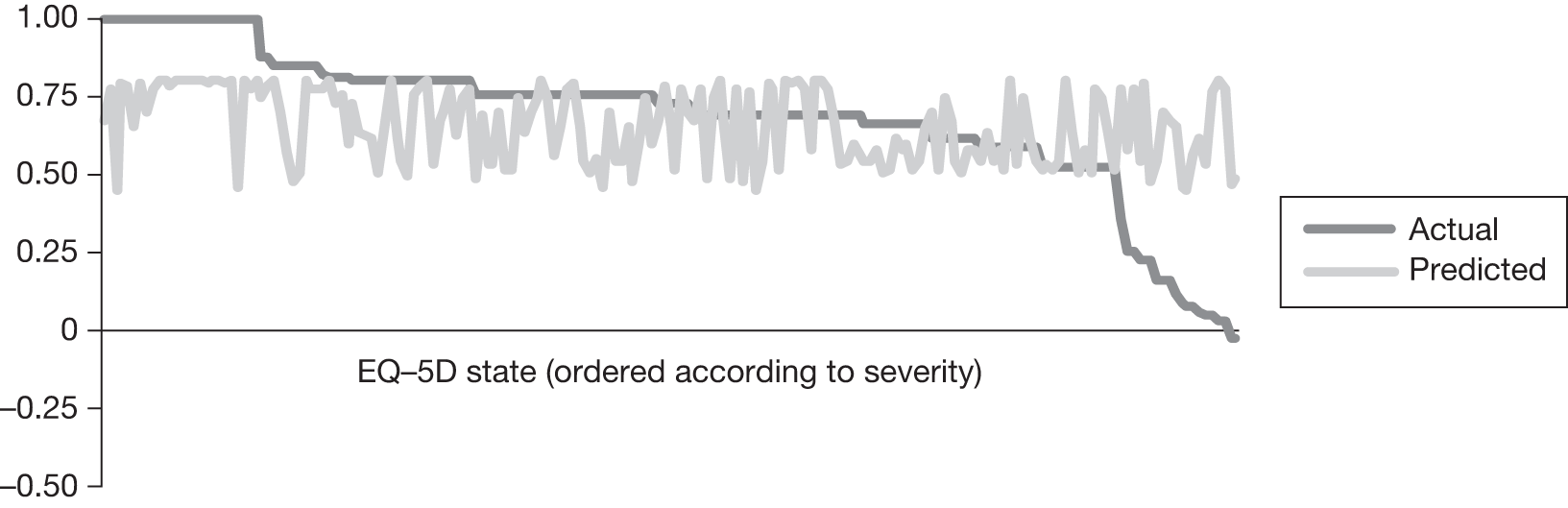
FIGURE 44.
Observed and predicted EQ-5D scores using 1, 3 and 12 months’ data, CLAD model.
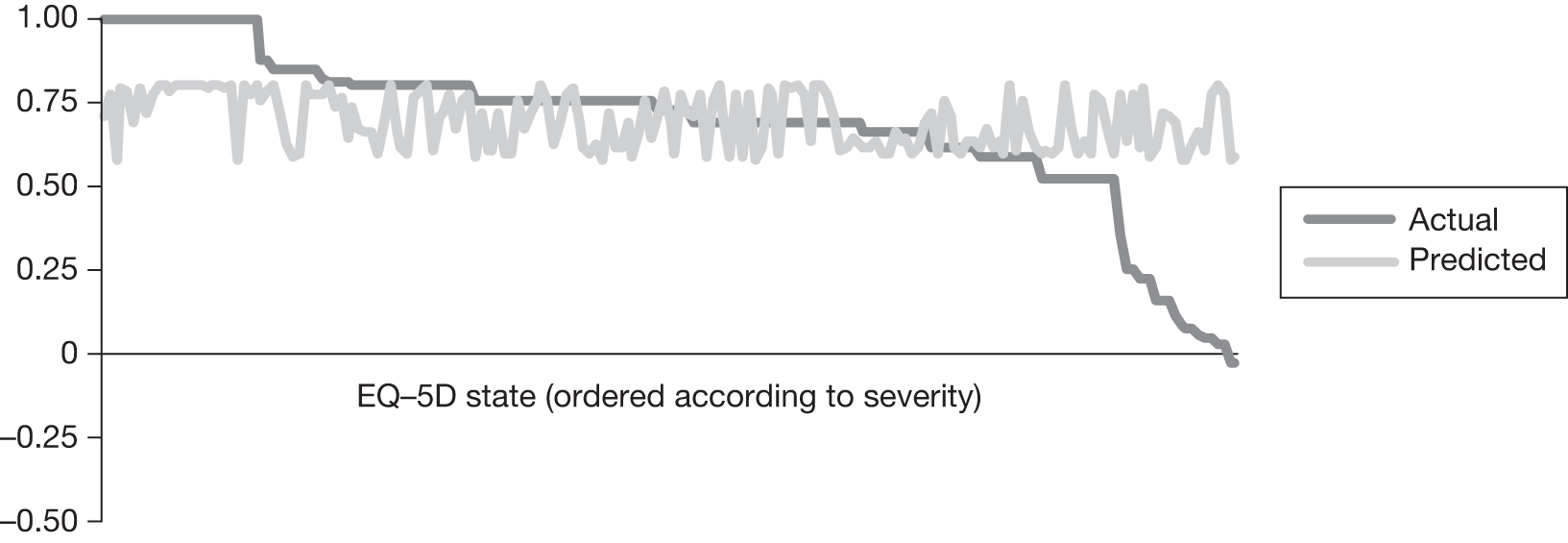
Appendix 15 Protocol: Management of frozen shoulder: a systematic review and decision analytic model (HTA No. 09/13)
Research protocol 1.1
1. Research objectives
The overall aim of the research project is to determine the clinical and cost effectiveness of different methods of managing frozen shoulder, with the following specific objectives:
-
to evaluate, via a systematic review, the clinical effectiveness (including adverse effects) of strategies currently used in the NHS for the management of frozen shoulder and identify the most appropriate intervention by stage of condition; specifically physical therapies, steroid and other shoulder injections, manipulation under anaesthesia, arthrographic distension, capsular release, watchful waiting and combinations of these interventions;
-
to evaluate, via a systematic review, the cost-effectiveness of the different interventions in order to inform the development of a decision model;
-
to develop a decision analytic model to estimate the cost-effectiveness of alternative treatment options for frozen shoulder;
-
to make recommendations for clinical practice; and
-
to identify any gaps in the evidence, undertake value of information (VoI) analysis to assess the potential value of future research on interventions for frozen shoulder and to make specific recommendations for further research.
2. Background
Frozen shoulder, also known as adhesive capsulitis, is a very painful condition of unknown aetiology, in which movements of the shoulder become severely restricted. The condition is thought to be the result of inflammation and swelling in the lining of the shoulder joint (capsule) and its associated ligaments, with resultant contracture of the shoulder joint capsule. Bunker describes pathology of fibrous contracture of the rotator interval and coracohumeral ligament of the shoulder joint. 1 The lining loses its normal characteristic of flexibilty and elasticity and becomes stiff and painful. The three key characteristics of frozen shoulder are gradual onset of shoulder stiffness, severe pain, especially at night, and near complete loss of passive and active external rotation of the shoulder. 2 Typically there are three overlapping phases of frozen shoulder:2
-
Phase 1 (painful freezing phase) – there is progressive stiffening and loss of motion in the shoulder with increasing pain on movement which may be worse at night (months 2 to 9);
-
Phase 2 (adhesive phase) – there is a gradual decrease in pain but stiffness remains and there is considerable restriction in the range of movement (months 4 to 12);
-
Phase 3 (resolution phase) – there is an improvement in range of movement (months 12 to 42).
Although the condition is classically described as having a resolution phase there may not be a complete resolution for all patients. There is variation across case series in the proportion of patients who do not regain full shoulder motion,2 possibly a reflection of variation in how outcome was assessed. Based on the largest series of patients with a mean follow-up of 4.4 years from onset of symptoms, 59% had normal or near normal shoulders, 35% had mild to moderate symptoms with pain being the most common complaint, and 6% had severe symptoms. 3 Recurrence is unusual though it is estimated that the other shoulder becomes affected in 6–17% of patients within 5 years. 2
The cumulative incidence of frozen shoulder is estimated at approximately 2.4/1000 per year based on a Dutch general practice sample. 4 It most commonly occurs in people in their mid-50’s and is slightly more common in women than men. In addition to primary or idiopathic frozen shoulder, there is an association between frozen shoulder and a number of other medical conditions, in particular diabetes. The incidence is reported to be 10% to 36% amongst people with diabetes, who tend not to respond as well to treatment. 2
Diagnosis and management
Diagnosis is based on clinical examination and medical history and a key alerting feature is restriction of shoulder movement in all directions. 5 Blood tests, X-rays and ultrasound are usually normal and not routinely required unless history or physical examination suggest the need to rule out other pathologies. 5
Frozen shoulder is commonly managed in the primary care setting. There are a number of management options, both surgical and non-surgical, but there is no consensus about management. The aims of treatment, depending on stage of condition, are pain relief, increasing arm movement, reducing the duration of symptoms and return to normal activities for the patient. Treatment options include:
-
Watchful waiting or ‘supervised neglect’, which involves explaining the condition to the patient and advising mobilisation within pain limits.
-
Oral medications such as non-steroidal anti-inflammatory drugs (NSAIDs) and oral steroids. Although the use of oral steroids is described in the literature they are not a commonly used intervention in the UK.
-
Gentle exercise supervised by a physiotherapist or as part of a home exercise program.
-
Physical therapies to help regain range of movement and prevent further stiffness. Several different regimes have been described in the literature including supervised exercises, mobilisation, acupuncture, and use of electrotherapeutic interventions such as laser therapy and ultrasound.
-
Intra-articular corticosteroid injections to reduce inflammation and provide pain relief. A range of different doses and number of injections are described in the literature. This intervention is usually delivered in the primary care setting but also in the secondary care setting, depending on how services are organized in a particular region.
-
Arthrographic distension (also called hydrodilation) which involves controlled dilation of the joint capsule with sterile saline or other solution such as local anaesthetic or steroid guided by radiological imaging (arthrography). This is thought to break the adhesions, which frees up the joint, improving the range of movement. The procedure lasts approximately 15 minutes and is performed under local anaesthetic.
-
Manipulation under anaesthesia (MUA) in which the shoulder is freed by rotation while the patient is under short general anaesthesia. This is usually a day procedure and generally lasts a maximum of 15 minutes including anaesthetic time.
-
Arthroscopic capsular release, a surgical procedure conducted under general or regional anaesthesia during which the contracted tissue is released. It can be undertaken as keyhole surgery (arthroscopic) or open procedure. This can be undertaken as a day procedure.
These interventions can be used individually or in combination depending on the disease stage. The optimal timing of the interventions is unclear though there is a suggestion that aggressive mobilisation should be avoided in the early, severely painful phase. 5 Surgical intervention is generally, though not exclusively, used where the condition is resistant to the other interventions. There are variations across the country in the order in which treatments are provided, though usually a step-up approach is adopted in terms of degree of invasiveness of the treatment, from primary to secondary care settings. The most commonly used or recommended interventions by G.P.’s, physiotherapists and orthopaedic surgeons in the NHS, based on a recent survey, were conservative treatment (watchful waiting, education, oral pain relief), physical therapy (mainly physiotherapy and mobisilation) and intra-articular injection during the early ‘painful’ phase and conservative treatment, physical therapy, intra-articular injection and surgery (mainly manipulation under anaesthesia and arthroscopic capsular release) for patients in the ‘resolution’ phase. 6
Existing research
We conducted scoping searches of the literature to inform the research proposal which involved searching key sources for clinical guidelines, systematic reviews and cost-effectiveness analyses (Appendix A). We identified only one guideline, from the New Zealand Guidelines Group, which was published five years ago and is therefore due for updating. 5 Clinical Evidence, last updated in February 2006, reviewed the evidence on interventions for shoulder pain in general. 7 Although several treatments were classified as likely to be beneficial, these were mainly in relation to other shoulder disorders. MUA plus intra-articular injection was identified as of likely benefit in people with frozen shoulder.
Systematic reviews were identified evaluating oral steroids,8 corticosteroid injections,9 physiotherapy,10,11 acupuncture12 and arthrographic distension,13 but not manipulation under anaesthesia or arthroscopic release (Table 1). Some of these reviews focused on shoulder pain in general, and included a range of conditions. None of the literature searches for the reviews identified are recent. The preliminary scoping searches also indicate that there may be limited evidence on the cost-effectiveness of these treatments for frozen shoulder. Two of the studies we identified were in relation to treatment of people with chronic shoulder complaints14 and new episodes of unilateral shoulder pain in primary care. 15 One study investigated the effectiveness and cost-effectiveness of physiotherapy following glenohumeral joint distension specifically in relation to patients with frozen shoulder. 16
| Author | Intervention | End date for literature search |
|---|---|---|
| Buchbinder et al.8 | Oral steroids | November 2005 |
| Buchbinder et al.13 | Arthrographic distension | November 2006 |
| Buchbinder et al.9 | Corticosteroids (for shoulder pain) | June 2002 |
| Cleland & Durall11 | Physical therapy | December 2000 |
| Green et al.10 | Physiotherapy (for shoulder pain) | June 2002 |
| Green et al.12 | Acupuncture (for shoulder pain) | December 2003 |
| Shah & Lewis18 | Corticosteroid injections | June 2006 |
It is apparent from previous reviews that there is variation in how frozen shoulder is defined across studies. A review of 21 randomised controlled trials (RCTs) of interventions for frozen shoulder could not derive a consistent description of the condition from the trials investigating this patient group. 17 The included RCTs required that participants had restricted shoulder movement but there was inconsistency across trials in the number of degrees of restriction, the type of restriction (active or passive) and the direction of the restriction (abduction or external rotation). 17 This highlights the difficulty of applying a strict definition for frozen shoulder within the context of a systematic review.
3. Research methods
We will undertake a systematic review of the literature on the effectiveness of different methods of managing frozen shoulder, with particular reference to the stage of the condition. The systematic review will inform the development of a decision analytic model. This will be a large and complex project which will involve undertaking a systematic review of six different interventions, one of which (physical therapy) encompasses several different types of therapy, as well as a decision model that reflects the complexity of management of the condition.
3.1 Systematic review of effectiveness of interventions
Search strategy
Both published and unpublished literature will be identified from systematic searches of electronic sources, hand searching, consultation with experts in the field, and reference checking.
The following databases will be searched: MEDLINE, MEDLINE In-Process, Cumulative Index to Nursing & Allied Health (CINAHL), EMBASE, Science Citation Index, BIOSIS Previews, PEDro, Cochrane Database of Systematic Reviews (CDSR), Database of Abstracts of Reviews of Effects (DARE), Health Technology Assessment (HTA) database, Cochrane Central Register of Controlled Trials (CENTRAL), PASCAL, Manual, Alternative and Natural Therapy (MANTIS) and Latin American and Caribbean Health Sciences (LILACS). Searches of electronic databases will not be restricted by language or study type.
In addition, information on studies in progress, unpublished research or research reported in the grey literature will be sought by searching a range of relevant databases including Conference Proceedings Citation Index, Science, Health Management Information Consortium (HMIC), ClinicalTrials.gov and NTIS.
Selected musculoskeletal disease websites will also be searched such as those of the National Institute of Arthritis & Musculoskeletal and Skin Diseases (NIAMS), the British Elbow & Shoulder Society (BESS), National Physiotherapy Research Network and Primary Care Rheumatology Society.
The MEDLINE search strategy is provided in Appendix B. This will be converted to run appropriately on other databases.
[Protocol addition: Where papers are not available from the British Library, extended searches will be undertaken only for papers published after 1965 and where it was in a language where we have identified a translator.]
Inclusion criteria
Systematic reviews and primary studies will be included if they meet the following criteria:
Population: patients with idiopathic (primary) frozen shoulder (adhesive capsulitis) will be included [Protocol addition: studies where at least 90% of the participants had primary frozen shoulder will be included]. Ideally, only patients with loss of active and passive external rotation of the involved shoulder with a normal x-ray would be included. This would allow for exclusion of patients with arthritis of the shoulder which can present as a similar clinical picture. However, based on a sample of the studies we have examined for the application, x-rays are not generally used to exclude joint arthritis. We will therefore take a pragmatic approach and include studies based on the authors’ definition of frozen shoulder to ensure we have identified all the relevant evidence. (The impact of how frozen shoulder is defined will then be explored in the synthesis). Studies of general shoulder conditions will only be included if outcome data are reported separately for participants with frozen shoulder. Frozen shoulder in people with diabetes is defined as primary in some classifications and in others as secondary frozen shoulder. In this review this group is defined as having primary frozen shoulder and will therefore be included in the review.
Intervention: The following interventions, either alone or in combination, will be included:
-
physical therapies including physiotherapy, acupuncture, chiropractic and osteopathy interventions). Physiotherapy encompasses a wide range of techniques including mobilisation, biofeedback, ultrasound and laser therapy and all therapies falling under the physiotherapy umbrella will be eligible for inclusion
-
arthrographic distension
-
steroid and other shoulder injections such as sodium hyaluronate
-
manipulation under anesthesia
-
capsular release (arthroscopic and open) and combinations of these treatments will be included
-
the approach of ‘watchful waiting’ will also be included.
There are a number of other treatments that have been researched that are not commonly used on the NHS such as radiotherapy, collagenase injection, salmon calcitonin and antibodies to tumour necrosis factor-α. These interventions will not be included in the synthesis, though information will be collated on the number of studies assessing uncommon treatments and their study design.
[Protocol addition: Studies of acupuncture will be included only where the comparator is one of the other treatments of interest in the review. This excludes studies comparing different forms of acupuncture and studies comparing acupuncture with alternative therapies such as moxibustion.]
Comparator: Any of the above treatments studies (including studies comparing different regimens of the same intervention), no treatment or placebo.
Outcomes: pain (at rest, on movement, at night); range of movement (e.g. internal and external rotation, elevation); function and disability; quality of life; time to recovery, return to work and recreation; and adverse events.
Study design: Only randomised controlled trials (RCTs) will be eligible for inclusion where this level of evidence is available on an intervention/management strategy. In the absence of randomised trials, quasi-experimental studies (i.e. with a control group) will be eligible for inclusion. If controlled trials are not available for MUA or capsular release, which is likely to be the case, case series will be included. Only case series of at least 50 participants will be included due to the problems of small case series being unrepresentative the clinical population. Where important adverse effects data may not be captured in RCTs, other study designs will also be considered to inform the economic model.
Systematic reviews will be included if (1) they fulfill all the relevant criteria, (2) have no significant sources of error and bias and (3) are reported in detail and the raw data are available from the report or authors to allow an update of the synthesis (if searches are more than 12 months out of date). If they do not meet all the criteria, systematic reviews will be used as sources of potentially relevant studies. It is anticipated that most of the systematic reviews available will be sources of relevant primary studies.
Screening and study selection
Two researchers will independently screen all titles and abstracts obtained through the searches for potentially relevant studies. Full manuscripts of potentially relevant studies will be ordered and two researchers will independently assess the relevance of each study using the criteria above. Discrepancies will be resolved by consensus or recourse to a third researcher if necessary.
Data extraction
A data extraction form will be developed, piloted on a small selection of studies and adjusted as necessary. Data extracted will include details of the study methods, setting, patient characteristics (including stage of condition), intervention, comparators, outcome measures and results. Data will be extracted into EPPI-Reviewer (a software package for managing systematic review production).
For continuous outcomes the post-intervention mean (and standard deviation) for each group will be extracted, where available. Otherwise the mean change from baseline for each group will be extracted.
Authors will be contacted where clarification of data is required for any of the primary outcomes (see synthesis below). Standard data imputation methods will be used, where necessary. 19
Data extraction will be undertaken by one researcher and checked by another, with discrepancies resolved by consensus or recourse to a third researcher if necessary.
Quality assessment
Quality assessment will also be undertaken by one researcher and checked by a second with discrepancies resolved by consensus or recourse to a third researcher if necessary. Studies will be quality assessed using the checklist in Appendix C. The criteria for assessing randomized and nonrandomised trials are based on recent CRD guidance;20 the criteria for case series are based on those used in recent systematic review including case series. 21
Data synthesis
The synthesis will focus on comparing the main treatment options (for example whether mobilization is more effective with or without steroid injection during the adhesive phase of the disease), rather than the effect of small variations in approach within the treatment classes. However, in reality there may be considerable variability within the different treatment options which will influence the type of analyses that are possible.
The primary outcomes will be patient-assessed pain intensity, quality of life (including disability measures such as the Oxford Shoulder Score and generic quality of life such as SF-36) and range of movement. Given that the symptoms of frozen shoulder change over time (with pain being the strongest characteristic of the early stages but not later) it is not appropriate to use a single primary outcome. Other outcomes such as time to return to work will be considered, evidence permitting. In addition to the proposed primary outcomes being the most clinically useful and patient-focused, it will also be more feasible to map these onto a utility measure for the decision model than the secondary physiological outcomes. Adverse effects of treatment will also be considered.
A narrative and tabular summary of key study characteristics, results and quality assessment will be provided. Where appropriate (based on clinical and statistical heterogeneity and the necessary data being available) individual study results will be combined in a series of pair-wise meta-analyses based on type of intervention and comparator, using a random effects model. As it is anticipated that the measures used to assess continuous outcome (for example pain) will vary between studies, standardized mean differences will be calculated, where appropriate, and combined using the generic inverse variance method. Heterogeneity will be assessed using χ2 tests20 and inconsistency will be quantified using the I2 statistic. 22
Given the range of interventions being considered, a mixed treatment comparison or network analysis could permit ranking of the benefits and harms of the different treatments options. 23 However, the appropriateness of such an approach depends on the principle of exchangeability, i.e. that there are no systematic differences between the trials that test particular types of intervention. From the information we have gathered so far, and our clinical experience of the condition, we anticipate that the exchangeability assumption is unlikely to be met by the studies available. The treatment that patients currently receive is at least partly determined by the severity of symptoms, stage of the condition and progress with a given treatment modality. If this is reflected in the trials then it is unlikely, for example, that the populations included in trials of arthroscopic capsular release are similar to those where the intervention being investigated is home exercise. However, the feasibility and appropriateness of a MTC will be explored and conducted if appropriate. 24 Current guidance on good practice will be followed. 25
Sub-group analyses will be restricted to a small number of potentially important characteristics that may reasonably be expected to modify the effect of the intervention. This will include sub-grouping studies based on how frozen shoulder was defined, stage of condition and/or severity (if such information is available), and whether study participants had diabetes.
Where meta-analysis is not appropriate a narrative synthesis will be undertaken. Where possible, results will be shown graphically. Studies will be grouped by type of intervention and comparator in the first instance and also the sub-groups identified above. Results will be interpreted in the context of the quality of the individual studies.
3.2 Systematic review of previous economic evaluations
A systematic review of economic evaluations will be undertaken to identify any models used previously and to inform the estimation of parameters for the decision model. Searches for economic evaluations of management strategies for frozen shoulder will be undertaken in the databases listed above (3.1). The search strategy will be adapted to focus on economic evaluations using search terms derived from the strategies used to identify studies for inclusion on the NHS Economic Evaluation Database (NHS EED) (see link for details http://www.crd.york.ac.uk/crdweb/html/helpdoc.htm#item17). In addition, searches of NHS EED and the Health Economic Evaluation Database (HEED) will be undertaken.
All full economic evaluations which meet the population and intervention inclusion criteria above will be eligible for inclusion.
A full economic evaluation will be defined as any study in which a comparison of two or more relevant alternatives was undertaken and with costs and outcomes examined separately for each alternative. This will include cost-effectiveness analysis (including cost–consequence analysis) where health outcomes are expressed in natural units; cost–utility analysis where benefits are measured in utility units or utility weighted life-years; and cost–benefit analyses, where benefits are measured in monetary form using approaches such as ‘willingness to pay’ or ‘human capital approach’. Based on our preliminary scoping of the evidence available, we believe that only a small number of economic evaluations of management strategies for frozen shoulder are likely to be available. The quality of economic evaluations will be assessed based on a modified version of the Drummond checklist26 and relevant data will be extracted.
3.3 Systematic review of service-users’ views of interventions for frozen shoulder
Time permitting, a systematic review of the research literature on patients’ views about interventions for frozen shoulder will also be undertaken.
Searches of MEDLINE, CINAHL and PsycINFO (from 1980 onwards) will be carried out. The search strategy used will be based upon the one used to identify studies for the effectiveness review (Appendix B) but will be adapted to include a qualitative design filter. 27
Studies investigating patients views about the treatments included in the main review will be eligible for inclusion. Only English language qualitative studies assessing patients’ views and experiences in relation to treatments for frozen shoulder will be eligible; expert opinion, letters containing no data on patient views, editorials and discussion papers will be excluded.
The processes for study selection, data extraction and quality assessment will follow those of the main review. Information extracted will include study aim, participant characteristics, methods of collecting data on patient views and experiences, method of analysis, results in the form of a summary of key themes arising from the analysis and authors’ conclusions. Study quality will be assessed using a tool developed by Hawker et al. 28 A narrative synthesis of the data will be undertaken.
3.4 Development of a decision model
A decision analytic model will be developed to estimate the cost-effectiveness of the different treatments for frozen shoulder. The specific objectives of the cost-effectiveness analysis will be to (1) assess the cost-effectiveness of the named interventions for frozen shoulder to inform clinical practice and (2) to identify the key uncertainties relating to the cost-effectiveness analysis and to use these to inform future research priorities.
In developing the model, NICE guidance on methods for technology appraisal will be followed. 25 The approach will be as follows:
-
A clinically relevant and appropriate decision model will be structured to map patients’ care pathways for the alternative therapies, in a way that is clinically appropriate and accounts for the phase of condition when treatment is received. The effect of treatment on short and longer-term costs and health related quality of life will be considered. The clinical experts on the team (from general practice, physiotherapy and orthopaedic surgery) will review the structure of the model to ensure it has good clinical face validity and only those pathways considered clinically meaningful will be modelled. In addition, the results of a current survey of a large sample of healthcare professionals will be used to inform the model.
-
Treatment order will be an important aspect to incorporate into the model. In the clinical setting there are variations in practice but, in general, a step up approach tends to be used in terms of treatment invasiveness, from primary to secondary care settings. The methods used to identify the optimum ordering of treatments will build on previous work undertaken by the CRD/CHE technology assessment group. 29
-
An appropriate time horizon will be chosen for the decision model that is long enough to capture the relevant costs and benefits. It is anticipated this will be at least 5 years duration.
-
The model will be populated using the most appropriate data identified systematically from the literature and routine sources. The parameter point estimates and distributions for the effectiveness of the different interventions will be taken directly from the results of the systematic review. For those parameters where estimates are not available directly from the systematic review, the health economists, information specialist and the researchers undertaking the systematic review will work closely to identify the best quality evidence available for that parameter. The information specialist will work in close liaison with the health economist to identify the model questions. Information to answer these questions will be provided by focused searching of appropriate databases, statistical sources and other relevant sources of information. The quality of all data used in the model will be explicitly discussed. The specific details of the data to be used to populate the model will await the development of the model structure and systematic review.
-
Health benefits will be expressed in terms of quality adjusted life years (QALYs).
-
The primary analysis will calculate the incremental cost-effectiveness of the different strategies based on an assessment of long-term NHS and Personal Social Service costs and quality adjusted utility.
-
The uncertainty in the data used to populate the model will be captured through the use of probabilistic modelling which requires that each input in the model is entered as a distribution rather than a fixed parameter. Using Monte Carlo simulation, this parameter uncertainty will be translated into uncertainty in the overall results. The results of this analysis will be presented graphically using cost-effectiveness acceptability curves which show the probability that each intervention is cost-effective conditional on a range of possible threshold values attached to an additional QALY.
-
To inform future research priorities, the model will be used to undertake a value of information (VoI) analysis. Decisions based on existing information will inherently be uncertain. We propose to conduct an expected value of information analysis to help estimate the cost of this uncertainty and identify whether it is of value to conduct further research in this area. If the expected value of perfect information for the population of interest exceeds the expect costs of such additional research, then potentially, it will be cost-effective for further research to be funded to better inform this decision in the future.
3.5 Dissemination
It will be important to ensure that those who need to know about the results of this review are informed and make sense of the findings. A detailed dissemination strategy will be produced to ensure that key groups are informed about the findings. Health professionals often differ in the amount of information they want to receive. CRD’s research into, and experience of disseminating the results of systematic reviews has repeatedly shown that providing a brief overview of the topic, results and implications is the best way to communicate important messages to time-poor health professionals. We will produce a short non-technical summary giving brief background details, information about the quality of evidence, the results and clinical implications. The summary report will be targeted to appropriate clinical groups throughout the UK, such as orthopedic surgeons, GPs and physiotherapists and via networks such as the National Physiotherapy Research Network, the British Elbow and Shoulder Society and the Primary Care Rheumatology Society. Publication of the findings will be press released and the potential for short articles in the relevant lay media explored.
Other dissemination activities will include the submission of papers for peer-reviewed publication and submission of abstracts to conferences. The results will also be made available on the CRD website. All dissemination activities will involve signposting those interested in further details to the full HTA report.
4. Advisory Group
The project Advisory Group will meet on three occasions and between meetings contact will be made with the group or individuals depending on the query. Three individuals who currently or previously have had frozen shoulder have also been invited to provide input in relation to: identifying the outcomes that have most significance for people with the condition and whether the care pathways underpinning the economic model reflect their experience. They will also be invited to comment on the non-technical summary of the final report.
5. Project timetable and milestones
The project will take place over a 12 month period (1 March 2010 to 14 March 2011). The key milestones are as follows:
| ■ Protocol development and peer review | Month 1–2 | April 2010 |
| ■ Literature searches (including economics) | Month 3–4 | April 2010 |
| ■ Screening and study selection | Month 4–5 | May-June 2010 |
| ■ Develop decision model structure | Month 3–4 | May-June 2010 |
| ■ Data extraction and checking | Month 5–6 | June-July 2010 |
| ■ Populate decision model with parameters not derived from systematic review | Month 5–6 | July-August 2010 |
| ■ Systematic review data analysis and synthesis | Month 7–9 | August-October 2010 |
| ■ De-bug decision model, analysis including sensitivity analysis | Month 7–9 | September to Nov 2010 |
| ■ Draft final report | Month 9–10 | Nov-December 2010 |
| ■ Draft report to advisory panel | Month 11 | January 2011 |
| ■ Address peer comments | Middle of month 12 | February 2011 |
| ■ Submit final report | End of Month 12 | 14 March 2011 |
| ■ Draft summary and papers for dissemination |
6. References
- Bunker TD. Frozen shoulder: unravelling the enigma. Ann R Coll Surg Engl 1997;79:210-13.
- Dias R, Cutts S, Massoud S. Frozen shoulder. BMJ 2005;331:1453-56.
- Hand C, Clipsham K, Rees JL, Carr AW. Long-term outcome of frozen shoulder. J Shoulder Elbow Surg 2008;17:231-36.
- van der Windt DA, Koes BW, de Jong BA, Bouter LM. Shoulder disorders in general practice: incidence, patient characteristics, and management. Ann Rheum Dis 1995;54:959-64.
- New Zealand Guidelines Group . The Diagnosis and Management of Soft Tissue Shoulder Injuries and Related Disorders 2004. URL: http://www.nzgg.org.nz.
- Dennis L, Brealey S, Rangan A, Rookmoneea M, Watson J. Managing idiopathic frozen shoulder: a survey of health professionals’ current practice and research priorities. Shoulder &Amp; Elbow 2010.
- Speed C. Shoulder pain. BMJ Clinical Evidence; 2006.
- Buchbinder R, Green S, Youd Joanne M, Johnston Renea V. Oral steroids for adhesive capsulitis. In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd Chichester, UK; 2006.
- Buchbinder R, Green S, Youd Joanne M. Corticosteroid injections for shoulder pain. In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd Chichester, UK; 2003.
- Green S, Buchbinder R, Hetrick Sarah E. Physiotherapy interventions for shoulder pain. In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd Chichester, UK; 2003.
- Cleland J, Durall CJ. Physical therapy for adhesive capsulitis: systematic review (Provisional abstract). Physiotherapy 2002;88:450-57. URL: http://www.mrw.interscience.wiley.com/cochrane/cldare/articles/DARE-12002008686/frame.html.
- Green S, Buchbinder R, Hetrick Sarah E. Acupuncture for shoulder pain. In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd Chichester, UK; 2005.
- Buchbinder R, Green S, Youd Joanne M, Johnston Renea V, Cumpston M. Arthrographic distension for adhesive capsulitis (frozen shoulder). In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd Chichester, UK; 2008.
- Geraets JJ, Goossens ME, de BCP, de GIJ, Koke AJ, Pelt RA, et al. Cost-effectiveness of a graded exercise therapy program for patients with chronic shoulder complaints (Structured abstract). Int J Technol Assess Health Care 2006;22:76-83. URL: http://www.mrw.interscience.wiley.com/cochrane/cleed/articles/NHSEED-22006008086/frame.html.
- James M, Stokes EA, Thomas E, Dziedzic K, Hay EM. A cost consequences analysis of local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in primary care (Structured abstract). Rheumatology 2005;44:1447-51. URL: http://www.mrw.interscience.wiley.com/cochrane/cleed/articles/NHSEED-22005001752/frame.html.
- Buchbinder R, Youd JM, Green S, Stein A, Forbes A, Harris A, et al. Efficacy and cost-effectiveness of physiotherapy following glenohumeral joint distension for adhesive capsulitis: a randomized trial (Provisional abstract). Arthritis Care and Research 2007;57:1027-37. URL: http://www.mrw.interscience.wiley.com/cochrane/cleed/articles/NHSEED-22007001854/frame.html.
- Schellingerhout JM, Verhagen AP, Thomas S, Koes BW. Lack of uniformity in diagnostic labeling of shoulder pain: Time for a different approach. Man Ther 2008;13:478-83.
- Shah N, Lewis M. Shoulder adhesive capsulitis: systematic review of randomised trials using multiple corticosteroid injections (Provisional abstract). Br J Gen Pract 2007;57:662-67. URL: http://www.mrw.interscience.wiley.com/cochrane/cldare/articles/DARE-12007003045/frame.html.
- Higgins J, Deeks JJ, Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Intervention. The Cochrane Collaboration; 2008.
- Centre for Reviews and Dissemination . Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care 2009.
- Rodgers M, McKenna C, Palmer S, Chambers D, Van Hout S, Golder S, et al. Curative catheter ablation in atrial fibrillation and typical atrial flutter: systematic review and economic evaluation. Health Technol Assess 2008;12.
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analysis. BMJ 2003;327:557-60.
- Caldwell DM, Ades AE, Higgins JP. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ 2005;2005:879-900.
- Ades AE. A chain of evidence with mixed comparisons: models for multi-parameter synthesis and consistency of evidence. Stat Med 2003;22:2995-3016.
- National Institute for Health and Clinical Excellence . Guide to the Methods of Technology Appraisal 2008.
- Drummond MF. Methods for the economic evaluation of healthcare programmes. Oxford: Oxford University Press; 2005.
- Flemming K, Briggs M. Electronic searching to locate qualitative research: evaluation of three strategies. J Adv Nurs 2006;57:95-100.
- Hawker S, Payne S, Kerr C, Hardey M, Powell J. Appraising the evidence: reviewing disparate data systematically. Qualitative Health Research 2002;12:1284-99.
- Woolacott N, Hawkins N, Mason A, Kainth A, Khadjesari Z, Bravo Vergel Y, et al. Etanercept and efalizumab for the treatment of psoriasis: a systematic review. Health Technol Assess 2006;10.
Appendix A Rapid appraisal search to identify systematic reviews, published and in progress, guidelines and ongoing primary research
| Completed and ongoing reviews | |
|---|---|
|
Cochrane Database of Systematic Reviews |
17 (11) |
|
DARE |
18 (14) |
|
HTA Database |
6 (5) |
|
SIGN Guidelines |
0 |
|
NICE (published appraisals) |
0 |
|
National Guideline Clearinghouse |
6 (3) |
|
HSTAT |
0 |
|
National Coordinating Centre for Health Technology Assessment |
0 |
|
TRIP |
423 (4) |
| Economic evaluations | |
|
NHS EED |
8 (7) |
| Indexes to and summaries of clinical effectiveness sources including reviews, appraisals of reviews, and evidence based guidelines | |
|
Clinical Evidence |
1 (1) |
|
Health Evidence Bulletins Wales |
0 |
| Supplementary MEDLINE search | |
|
MEDLINE |
2969 |
Appendix B Search strategy
Database: Ovid MEDLINE(R)
Search Strategy:
Appendix C Quality assessment
| Criteria | Score (‘Yes’, ‘No’, ‘Unclear’, ‘Not applicable (NA)’) | |
|---|---|---|
| 1 | Was the number of participants randomised stated? | |
| 2 | Was the method of randomisation adequate (e.g. use of random number table, computer random number generator, coin tossing, shuffling of cards or envelopes, throwing of dice)? | |
| 3 | Was allocation concealment adequate (e.g. central allocation, sequentially numbered opaque sealed envelopes)? | |
| 4 | Were the treatment groups comparable at baseline for important prognostic factors? | |
| 5 | If the above answer was no, was a suitable statistical method used to adjust for possible baseline imbalance? | |
| 6 | Was the study reported as being at least double blind? | |
| 7 | Were patients blinded? | |
| 8 | Were outcome assessors blinded? | |
| 9 | Were care givers blinded? | |
| 10 | Was intention-to treat analysis used (i.e. were all participants included in the analysis in the group to which they were allocated)? | |
| 11 | Were there any unexpected imbalances in drop outs between groups? If so, were they explained or adjusted for? | |
| 12 | Was selection/eligibility criteria adequately reported? | |
| 13 | Was the selected population representative of that seen in normal practice? | |
| 14 | Was an appropriate measure of variability reported? | |
| 15 | Was loss to follow-up reported or explained? | |
| 16 | Were at least 90% of those included at baseline followed up? | |
| 17 | Were patients recruited prospectively? | |
| 18 | Were patient recruited consecutively? | |
| 19 | Did the study report relevant prognostic factors? |
Case series quality rating
Good: the answer is ‘yes’ to criteria 12–19
Satisfactory: the answer is ‘yes’ to criteria 13 and 15–18
Poor: the answer is not ‘yes’ to one or more of the criteria listed for satisfactory
Glossary
- Case series
- A group of case reports of patients who were given similar treatments. There is no control group involved.
- Cost-effectiveness analysis
- An economic analysis that expresses the effects or consequences of interventions on a single dimension. This would normally be expressed in ‘natural’ units (e.g. cases cured, life-years gained, additional strokes prevented). The difference between interventions in terms of costs and effects is typically expressed as an incremental cost-effectiveness ratio (e.g. the incremental cost per life-year gained).
- Cost–utility analysis
- The same as a cost-effectiveness analysis but the effects or consequences of interventions are expressed in generic units of health gain, usually quality-adjusted life-years.
- Exercise therapy
- Gentle rhythmic active exercises, function-based exercises [to maintain/restore the range or quality (co-ordination and/or control) of movement] or both.
- Incremental cost-effectiveness ratio
- An expression of the additional cost of health gain associated with an intervention relative to an appropriate comparator. Expressed as the difference in mean costs (relative to the comparator) divided by the difference in mean health gain.
- Intention-to-treat analysis
- Analysis that compares participants in the groups to which they were originally assigned.
- Mixed-treatment comparison
- This is an extension of a traditional meta-analysis. Whereas a traditional meta-analysis includes only trials making direct comparisons between an intervention and a comparator, a mixed-treatment comparison also includes indirect evidence. This approach overcomes the limitations of the traditional approach in cases in which there are no or limited trials making the relevant head-to-head comparison.
- Mobilisations
- Therapist-applied passive movements of joints or other structures performed in such a way that they are always within the control of the patient. They may be performed by various techniques and may be combined with active movement on the part of the patient.
- Physical therapy
- This term is used to refer to the broad range of physical interventions such as physiotherapy (including mobilisation or other modalities such as electrotherapy) and chiropractic and osteopathic interventions.
- Quality-adjusted life-year
- An index of health gain in which survival duration is weighted or adjusted by the patient’s quality of life during the survival period. Quality-adjusted life-years have the advantage of incorporating changes in both quantity (mortality) and quality (morbidity) of life.
- Randomised controlled trial
- A trial in which the participants are randomly allocated to the control or treatment groups.
- WinBUGS
- Statistical software for Bayesian analysis using Markov chain Monte Carlo methods.
List of abbreviations
- 15D
- 15-dimension utility measure
- ASES
- American Shoulder and Elbow Surgeons
- CI
- confidence interval
- CLAD
- censored least absolute deviations
- CRD
- Centre for Reviews and Dissemination
- CrI
- credible interval
- DASH
- Disabilities of the Arm, Shoulder and Hand
- EPPI
- Evidence for Policy and Practice Information
- EQ-5D
- European Quality of Life-5 Dimensions
- ERM
- end-range mobilisation
- FLEX-SF
- Flexilevel Scale of Shoulder Function
- GP
- general practitioner
- HAQ
- Health Assessment Questionnaire
- HGMT
- high-grade mobilisation technique
- HRQoL
- health-related quality of life
- HUI
- Health Utilities Index
- ICER
- incremental cost-effectiveness ratio
- IQR
- interquartile range
- ITT
- intention to treat
- LGMT
- low-grade mobilisation technique
- MAE
- mean absolute error
- MCID
- minimum clinically important difference
- MCMC
- Markov chain Monte Carlo
- MCS
- mental component summary
- MD
- mean difference
- ME
- mean error
- MRM
- mid-range mobilisation
- MTC
- mixed-treatment comparison
- MUA
- manipulation under anaesthesia
- MWM
- mobilisation with movement
- NICE
- National Institute for Health and Clinical Excellence
- NSAID
- non-steroidal anti-inflammatory drug
- NTIS
- National Technical Information Service
- OLS
- ordinary least squares
- PCS
- physical component summary
- PSWD
- pulsed short-wave diathermy
- QALY
- quality-adjusted life-year
- QoL
- quality of life
- RCT
- randomised controlled trial
- RMSE
- root-mean-square error
- SD
- standard deviation
- SDQ
- Shoulder Disability Questionnaire
- SE
- standard error
- SF-6D
- Short Form questionnaire-6 Dimensions
- SF-36
- Short Form questionnaire-36 items
- SMD
- standardised mean difference
- SPADI
- Shoulder Pain and Disability Index
- SRQ
- Shoulder Rating Questionnaire
- SWD
- short-wave diathermy
- TENS
- transcutaneous electrical nerve stimulation
- UCLA
- University of California Los Angeles
- VAS
- visual analogue scale
All abbreviations that have been used in this report are listed here unless the abbreviation is well known (e.g. NHS), or it has been used only once, or it is a non-standard abbreviation used only in figures/tables/appendices, in which case the abbreviation is defined in the figure legend or in the notes at the end of the table.
Notes
Health Technology Assessment programme
-
Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Professor of Dermato-Epidemiology, Centre of Evidence-Based Dermatology, University of Nottingham
Prioritisation Group
-
Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Professor Imti Choonara, Professor in Child Health, Academic Division of Child Health, University of Nottingham
Chair – Pharmaceuticals Panel
-
Dr Bob Coates, Consultant Advisor – Disease Prevention Panel
-
Dr Andrew Cook, Consultant Advisor – Intervention Procedures Panel
-
Dr Peter Davidson, Director of NETSCC, Health Technology Assessment
-
Dr Nick Hicks, Consultant Adviser – Diagnostic Technologies and Screening Panel, Consultant Advisor–Psychological and Community Therapies Panel
-
Ms Susan Hird, Consultant Advisor, External Devices and Physical Therapies Panel
-
Professor Sallie Lamb, Director, Warwick Clinical Trials Unit, Warwick Medical School, University of Warwick
Chair – HTA Clinical Evaluation and Trials Board
-
Professor Jonathan Michaels, Professor of Vascular Surgery, Sheffield Vascular Institute, University of Sheffield
Chair – Interventional Procedures Panel
-
Professor Ruairidh Milne, Director – External Relations
-
Dr John Pounsford, Consultant Physician, Directorate of Medical Services, North Bristol NHS Trust
Chair – External Devices and Physical Therapies Panel
-
Dr Vaughan Thomas, Consultant Advisor – Pharmaceuticals Panel, Clinical
Lead – Clinical Evaluation Trials Prioritisation Group
-
Professor Margaret Thorogood, Professor of Epidemiology, Health Sciences Research Institute, University of Warwick
Chair – Disease Prevention Panel
-
Professor Lindsay Turnbull, Professor of Radiology, Centre for the MR Investigations, University of Hull
Chair – Diagnostic Technologies and Screening Panel
-
Professor Scott Weich, Professor of Psychiatry, Health Sciences Research Institute, University of Warwick
Chair – Psychological and Community Therapies Panel
-
Professor Hywel Williams, Director of Nottingham Clinical Trials Unit, Centre of Evidence-Based Dermatology, University of Nottingham
Chair – HTA Commissioning Board
Deputy HTA Programme Director
HTA Commissioning Board
-
Professor of Dermato-Epidemiology, Centre of Evidence-Based Dermatology, University of Nottingham
-
Department of Public Health and Epidemiology, University of Birmingham
-
Professor of Clinical Pharmacology, Director, NIHR HTA programme, University of Liverpool
-
Professor Ann Ashburn, Professor of Rehabilitation and Head of Research, Southampton General Hospital
-
Professor Judith Bliss, Director of ICR-Clinical Trials and Statistics Unit, The Institute of Cancer Research
-
Professor Peter Brocklehurst, Professor of Women’s Health, Institute for Women’s Health, University College London
-
Professor David Fitzmaurice, Professor of Primary Care Research, Department of Primary Care Clinical Sciences, University of Birmingham
-
Professor John W Gregory, Professor in Paediatric Endocrinology, Department of Child Health, Wales School of Medicine, Cardiff University
-
Professor Steve Halligan, Professor of Gastrointestinal Radiology, University College Hospital, London
-
Professor Angela Harden, Professor of Community and Family Health, Institute for Health and Human Development, University of East London
-
Dr Martin J Landray, Reader in Epidemiology, Honorary Consultant Physician, Clinical Trial Service Unit, University of Oxford
-
Dr Joanne Lord, Reader, Health Economics Research Group, Brunel University
-
Professor Stephen Morris, Professor of Health Economics, University College London, Research Department of Epidemiology and Public Health, University College London
-
Professor Dion Morton, Professor of Surgery, Academic Department of Surgery, University of Birmingham
-
Professor Gail Mountain, Professor of Health Services Research, Rehabilitation and Assistive Technologies Group, University of Sheffield
-
Professor Irwin Nazareth, Professor of Primary Care and Head of Department, Department of Primary Care and Population Sciences, University College London
-
Professor E Andrea Nelson, Professor of Wound Healing and Director of Research, School of Healthcare, University of Leeds
-
Professor John David Norrie, Chair in Clinical Trials and Biostatistics, Robertson Centre for Biostatistics, University of Glasgow
-
Dr Rafael Perera, Lecturer in Medical Statisitics, Department of Primary Health Care, University of Oxford
-
Professor Barney Reeves, Professorial Research Fellow in Health Services Research, Department of Clinical Science, University of Bristol
-
Professor Peter Tyrer, Professor of Community Psychiatry, Centre for Mental Health, Imperial College London
-
Professor Martin Underwood, Professor of Primary Care Research, Warwick Medical School, University of Warwick
-
Professor Caroline Watkins, Professor of Stroke and Older People’s Care, Chair of UK Forum for Stroke Training, Stroke Practice Research Unit, University of Central Lancashire
-
Dr Duncan Young, Senior Clinical Lecturer and Consultant, Nuffield Department of Anaesthetics, University of Oxford
-
Dr Tom Foulks, Medical Research Council
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
HTA Clinical Evaluation and Trials Board
-
Director, Warwick Clinical Trials Unit, Warwick Medical School, University of Warwick and Professor of Rehabilitation, Nuffield Department of Orthopaedic, Rheumatology and Musculoskeletal Sciences, University of Oxford
-
Professor of the Psychology of Health Care, Leeds Institute of Health Sciences, University of Leeds
-
Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Professor Keith Abrams, Professor of Medical Statistics, Department of Health Sciences, University of Leicester
-
Professor Martin Bland, Professor of Health Statistics, Department of Health Sciences, University of York
-
Professor Jane Blazeby, Professor of Surgery and Consultant Upper GI Surgeon, Department of Social Medicine, University of Bristol
-
Professor Julia M Brown, Director, Clinical Trials Research Unit, University of Leeds
-
Professor Alistair Burns, Professor of Old Age Psychiatry, Psychiatry Research Group, School of Community-Based Medicine, The University of Manchester & National Clinical Director for Dementia, Department of Health
-
Dr Jennifer Burr, Director, Centre for Healthcare Randomised trials (CHART), University of Aberdeen
-
Professor Linda Davies, Professor of Health Economics, Health Sciences Research Group, University of Manchester
-
Professor Simon Gilbody, Prof of Psych Medicine and Health Services Research, Department of Health Sciences, University of York
-
Professor Steven Goodacre, Professor and Consultant in Emergency Medicine, School of Health and Related Research, University of Sheffield
-
Professor Dyfrig Hughes, Professor of Pharmacoeconomics, Centre for Economics and Policy in Health, Institute of Medical and Social Care Research, Bangor University
-
Professor Paul Jones, Professor of Respiratory Medicine, Department of Cardiac and Vascular Science, St George‘s Hospital Medical School, University of London
-
Professor Khalid Khan, Professor of Women’s Health and Clinical Epidemiology, Barts and the London School of Medicine, Queen Mary, University of London
-
Professor Richard J McManus, Professor of Primary Care Cardiovascular Research, Primary Care Clinical Sciences Building, University of Birmingham
-
Professor Helen Rodgers, Professor of Stroke Care, Institute for Ageing and Health, Newcastle University
-
Professor Ken Stein, Professor of Public Health, Peninsula Technology Assessment Group, Peninsula College of Medicine and Dentistry, Universities of Exeter and Plymouth
-
Professor Jonathan Sterne, Professor of Medical Statistics and Epidemiology, Department of Social Medicine, University of Bristol
-
Mr Andy Vail, Senior Lecturer, Health Sciences Research Group, University of Manchester
-
Professor Clare Wilkinson, Professor of General Practice and Director of Research North Wales Clinical School, Department of Primary Care and Public Health, Cardiff University
-
Dr Ian B Wilkinson, Senior Lecturer and Honorary Consultant, Clinical Pharmacology Unit, Department of Medicine, University of Cambridge
-
Ms Kate Law, Director of Clinical Trials, Cancer Research UK
-
Dr Morven Roberts, Clinical Trials Manager, Health Services and Public Health Services Board, Medical Research Council
Diagnostic Technologies and Screening Panel
-
Scientific Director of the Centre for Magnetic Resonance Investigations and YCR Professor of Radiology, Hull Royal Infirmary
-
Professor Judith E Adams, Consultant Radiologist, Manchester Royal Infirmary, Central Manchester & Manchester Children’s University Hospitals NHS Trust, and Professor of Diagnostic Radiology, University of Manchester
-
Mr Angus S Arunkalaivanan, Honorary Senior Lecturer, University of Birmingham and Consultant Urogynaecologist and Obstetrician, City Hospital, Birmingham
-
Dr Diana Baralle, Consultant and Senior Lecturer in Clinical Genetics, University of Southampton
-
Dr Stephanie Dancer, Consultant Microbiologist, Hairmyres Hospital, East Kilbride
-
Dr Diane Eccles, Professor of Cancer Genetics, Wessex Clinical Genetics Service, Princess Anne Hospital
-
Dr Trevor Friedman, Consultant Liason Psychiatrist, Brandon Unit, Leicester General Hospital
-
Dr Ron Gray, Consultant, National Perinatal Epidemiology Unit, Institute of Health Sciences, University of Oxford
-
Professor Paul D Griffiths, Professor of Radiology, Academic Unit of Radiology, University of Sheffield
-
Mr Martin Hooper, Public contributor
-
Professor Anthony Robert Kendrick, Associate Dean for Clinical Research and Professor of Primary Medical Care, University of Southampton
-
Dr Nicola Lennard, Senior Medical Officer, MHRA
-
Dr Anne Mackie, Director of Programmes, UK National Screening Committee, London
-
Mr David Mathew, Public contributor
-
Dr Michael Millar, Consultant Senior Lecturer in Microbiology, Department of Pathology & Microbiology, Barts and The London NHS Trust, Royal London Hospital
-
Mrs Una Rennard, Public contributor
-
Dr Stuart Smellie, Consultant in Clinical Pathology, Bishop Auckland General Hospital
-
Ms Jane Smith, Consultant Ultrasound Practitioner, Leeds Teaching Hospital NHS Trust, Leeds
-
Dr Allison Streetly, Programme Director, NHS Sickle Cell and Thalassaemia Screening Programme, King’s College School of Medicine
-
Dr Matthew Thompson, Senior Clinical Scientist and GP, Department of Primary Health Care, University of Oxford
-
Dr Alan J Williams, Consultant Physician, General and Respiratory Medicine, The Royal Bournemouth Hospital
-
Dr Tim Elliott, Team Leader, Cancer Screening, Department of Health
-
Dr Joanna Jenkinson, Board Secretary, Neurosciences and Mental Health Board (NMHB), Medical Research Council
-
Professor Julietta Patrick, Director, NHS Cancer Screening Programme, Sheffield
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Dr Ursula Wells, Principal Research Officer, Policy Research Programme, Department of Health
Disease Prevention Panel
-
Professor of Epidemiology, University of Warwick Medical School, Coventry
-
Dr Robert Cook, Clinical Programmes Director, Bazian Ltd, London
-
Dr Colin Greaves, Senior Research Fellow, Peninsula Medical School (Primary Care)
-
Mr Michael Head, Public contributor
-
Professor Cathy Jackson, Professor of Primary Care Medicine, Bute Medical School, University of St Andrews
-
Dr Russell Jago, Senior Lecturer in Exercise, Nutrition and Health, Centre for Sport, Exercise and Health, University of Bristol
-
Dr Julie Mytton, Consultant in Child Public Health, NHS Bristol
-
Professor Irwin Nazareth, Professor of Primary Care and Director, Department of Primary Care and Population Sciences, University College London
-
Dr Richard Richards, Assistant Director of Public Health, Derbyshire County Primary Care Trust
-
Professor Ian Roberts, Professor of Epidemiology and Public Health, London School of Hygiene & Tropical Medicine
-
Dr Kenneth Robertson, Consultant Paediatrician, Royal Hospital for Sick Children, Glasgow
-
Dr Catherine Swann, Associate Director, Centre for Public Health Excellence, NICE
-
Mrs Jean Thurston, Public contributor
-
Professor David Weller, Head, School of Clinical Science and Community Health, University of Edinburgh
-
Ms Christine McGuire, Research & Development, Department of Health
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
External Devices and Physical Therapies Panel
-
Consultant Physician North Bristol NHS Trust
-
Reader in Wound Healing and Director of Research, University of Leeds
-
Professor Bipin Bhakta, Charterhouse Professor in Rehabilitation Medicine, University of Leeds
-
Mrs Penny Calder, Public contributor
-
Dr Dawn Carnes, Senior Research Fellow, Barts and the London School of Medicine and Dentistry
-
Dr Emma Clark, Clinician Scientist Fellow & Cons. Rheumatologist, University of Bristol
-
Mrs Anthea De Barton-Watson, Public contributor
-
Professor Nadine Foster, Professor of Musculoskeletal Health in Primary Care Arthritis Research, Keele University
-
Dr Shaheen Hamdy, Clinical Senior Lecturer and Consultant Physician, University of Manchester
-
Professor Christine Norton, Professor of Clinical Nursing Innovation, Bucks New University and Imperial College Healthcare NHS Trust
-
Dr Lorraine Pinnigton, Associate Professor in Rehabilitation, University of Nottingham
-
Dr Kate Radford, Senior Lecturer (Research), University of Central Lancashire
-
Mr Jim Reece, Public contributor
-
Professor Maria Stokes, Professor of Neuromusculoskeletal Rehabilitation, University of Southampton
-
Dr Pippa Tyrrell, Senior Lecturer/Consultant, Salford Royal Foundation Hospitals’ Trust and University of Manchester
-
Dr Nefyn Williams, Clinical Senior Lecturer, Cardiff University
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Dr Morven Roberts, Clinical Trials Manager, Health Services and Public Health Services Board, Medical Research Council
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Dr Ursula Wells, Principal Research Officer, Policy Research Programme, Department of Health
Interventional Procedures Panel
-
Professor of Vascular Surgery, University of Sheffield
-
Consultant Colorectal Surgeon, Bristol Royal Infirmary
-
Mrs Isabel Boyer, Public contributor
-
Mr Sankaran Chandra Sekharan, Consultant Surgeon, Breast Surgery, Colchester Hospital University NHS Foundation Trust
-
Professor Nicholas Clarke, Consultant Orthopaedic Surgeon, Southampton University Hospitals NHS Trust
-
Ms Leonie Cooke, Public contributor
-
Mr Seumas Eckford, Consultant in Obstetrics & Gynaecology, North Devon District Hospital
-
Professor Sam Eljamel, Consultant Neurosurgeon, Ninewells Hospital and Medical School, Dundee
-
Dr Adele Fielding, Senior Lecturer and Honorary Consultant in Haematology, University College London Medical School
-
Dr Matthew Hatton, Consultant in Clinical Oncology, Sheffield Teaching Hospital Foundation Trust
-
Dr John Holden, General Practitioner, Garswood Surgery, Wigan
-
Dr Fiona Lecky, Senior Lecturer/Honorary Consultant in Emergency Medicine, University of Manchester/Salford Royal Hospitals NHS Foundation Trust
-
Dr Nadim Malik, Consultant Cardiologist/Honorary Lecturer, University of Manchester
-
Mr Hisham Mehanna, Consultant & Honorary Associate Professor, University Hospitals Coventry & Warwickshire NHS Trust
-
Dr Jane Montgomery, Consultant in Anaesthetics and Critical Care, South Devon Healthcare NHS Foundation Trust
-
Professor Jon Moss, Consultant Interventional Radiologist, North Glasgow Hospitals University NHS Trust
-
Dr Simon Padley, Consultant Radiologist, Chelsea & Westminster Hospital
-
Dr Ashish Paul, Medical Director, Bedfordshire PCT
-
Dr Sarah Purdy, Consultant Senior Lecturer, University of Bristol
-
Dr Matthew Wilson, Consultant Anaesthetist, Sheffield Teaching Hospitals NHS Foundation Trust
-
Professor Yit Chiun Yang, Consultant Ophthalmologist, Royal Wolverhampton Hospitals NHS Trust
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Dr Morven Roberts, Clinical Trials Manager, Health Services and Public Health Services Board, Medical Research Council
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Dr Ursula Wells, Principal Research Officer, Policy Research Programme, Department of Health
Pharmaceuticals Panel
-
Professor in Child Health, University of Nottingham
-
Senior Lecturer in Clinical Pharmacology, University of East Anglia
-
Dr Martin Ashton-Key, Medical Advisor, National Commissioning Group, NHS London
-
Dr Peter Elton, Director of Public Health, Bury Primary Care Trust
-
Dr Ben Goldacre, Research Fellow, Division of Psychological Medicine and Psychiatry, King’s College London
-
Dr James Gray, Consultant Microbiologist, Department of Microbiology, Birmingham Children’s Hospital NHS Foundation Trust
-
Dr Jurjees Hasan, Consultant in Medical Oncology, The Christie, Manchester
-
Dr Carl Heneghan, Deputy Director Centre for Evidence-Based Medicine and Clinical Lecturer, Department of Primary Health Care, University of Oxford
-
Dr Dyfrig Hughes, Reader in Pharmacoeconomics and Deputy Director, Centre for Economics and Policy in Health, IMSCaR, Bangor University
-
Dr Maria Kouimtzi, Pharmacy and Informatics Director, Global Clinical Solutions, Wiley-Blackwell
-
Professor Femi Oyebode, Consultant Psychiatrist and Head of Department, University of Birmingham
-
Dr Andrew Prentice, Senior Lecturer and Consultant Obstetrician and Gynaecologist, The Rosie Hospital, University of Cambridge
-
Ms Amanda Roberts, Public contributor
-
Dr Gillian Shepherd, Director, Health and Clinical Excellence, Merck Serono Ltd
-
Mrs Katrina Simister, Assistant Director New Medicines, National Prescribing Centre, Liverpool
-
Professor Donald Singer, Professor of Clinical Pharmacology and Therapeutics, Clinical Sciences Research Institute, CSB, University of Warwick Medical School
-
Mr David Symes, Public contributor
-
Dr Arnold Zermansky, General Practitioner, Senior Research Fellow, Pharmacy Practice and Medicines Management Group, Leeds University
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Mr Simon Reeve, Head of Clinical and Cost-Effectiveness, Medicines, Pharmacy and Industry Group, Department of Health
-
Dr Heike Weber, Programme Manager, Medical Research Council
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Dr Ursula Wells, Principal Research Officer, Policy Research Programme, Department of Health
Psychological and Community Therapies Panel
-
Professor of Psychiatry, University of Warwick, Coventry
-
Consultant & University Lecturer in Psychiatry, University of Cambridge
-
Professor Jane Barlow, Professor of Public Health in the Early Years, Health Sciences Research Institute, Warwick Medical School
-
Dr Sabyasachi Bhaumik, Consultant Psychiatrist, Leicestershire Partnership NHS Trust
-
Mrs Val Carlill, Public contributor
-
Dr Steve Cunningham, Consultant Respiratory Paediatrician, Lothian Health Board
-
Dr Anne Hesketh, Senior Clinical Lecturer in Speech and Language Therapy, University of Manchester
-
Dr Peter Langdon, Senior Clinical Lecturer, School of Medicine, Health Policy and Practice, University of East Anglia
-
Dr Yann Lefeuvre, GP Partner, Burrage Road Surgery, London
-
Dr Jeremy J Murphy, Consultant Physician and Cardiologist, County Durham and Darlington Foundation Trust
-
Dr Richard Neal, Clinical Senior Lecturer in General Practice, Cardiff University
-
Mr John Needham, Public contributor
-
Ms Mary Nettle, Mental Health User Consultant
-
Professor John Potter, Professor of Ageing and Stroke Medicine, University of East Anglia
-
Dr Greta Rait, Senior Clinical Lecturer and General Practitioner, University College London
-
Dr Paul Ramchandani, Senior Research Fellow/Cons. Child Psychiatrist, University of Oxford
-
Dr Karen Roberts, Nurse/Consultant, Dunston Hill Hospital, Tyne and Wear
-
Dr Karim Saad, Consultant in Old Age Psychiatry, Coventry and Warwickshire Partnership Trust
-
Dr Lesley Stockton, Lecturer, School of Health Sciences, University of Liverpool
-
Dr Simon Wright, GP Partner, Walkden Medical Centre, Manchester
-
Dr Kay Pattison, Senior NIHR Programme Manager, Department of Health
-
Dr Morven Roberts, Clinical Trials Manager, Health Services and Public Health Services Board, Medical Research Council
-
Professor Tom Walley, CBE, Director, NIHR HTA programme, Professor of Clinical Pharmacology, University of Liverpool
-
Dr Ursula Wells, Principal Research Officer, Policy Research Programme, Department of Health